3. THE GUIDELINE
3.1. Classification
Urinary tract infections (UTIs) encompass a wide spectrum of clinical and pathological conditions affecting various parts of the urinary tract. Each condition has its own unique epidemiology, natural history and diagnostic considerations. A precise distinction is crucial as it significantly impacts treatment and prognosis. Therefore, standardised terminology is essential for effective communication on this subject. Various classification systems of UTI exist. The most widely used systems are from the Centres for Disease Control and Prevention (CDC) [5], the Infectious Diseases Society of America (IDSA) [6], the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) [7], and the United States Food and Drug Administration (FDA) [8, 9]. Current guidelines often differentiate between uncomplicated and complicated UTIs with some modifications. According to these definitions, uncomplicated UTIs occur in healthy, nonpregnant women, while all other UTIs fall into the category of complicated UTIs. This classification is straightforward, yet it carries inherent risks, as it can significantly affect initial patient management and treatment selection.
The Guidelines Panel propose a new classification scheme for UTIs aimed at enhancing consistency in clinical practice and providing a comprehensive framework for understanding various clinical presentations. The proposed classification no longer uses the terms ‘uncomplicated’ and ‘complicated.’ Instead, it emphasises the difference between localised and systemic UTIs identified by clinical signs and symptoms (see Figure 1 and Table 1).
The definitions are as follows (Figure 1):
- Localised UTI (i.e. cystitis): A cystitis without any signs and symptoms of systemic infection in either sex.
- Systemic UTI: An infection with signs and symptoms of systemic infection with or without localised symptoms originating from any site in the urinary tract in either sex.
According to the new definition, UTIs can manifest as either localised (i.e. cystitis) or systemic (e.g. pyelonephritis, prostatitis, etc.) infection. Both conditions may be accompanied by risk factors that increase the likelihood of a challenging clinical course and jeopardise treatment success. Clinicians must be aware of these risk factors and should address them accordingly.
This definition enables medical practitioners to distinguish between a localised UTI that can generally be treated on an outpatient basis and a more complex systemic UTI that may necessitate blood sampling, imaging (e.g. ultrasound, cross-sectional imaging), intravenous antimicrobial treatment and hospitalisation.
Figure 1: Classification of UTI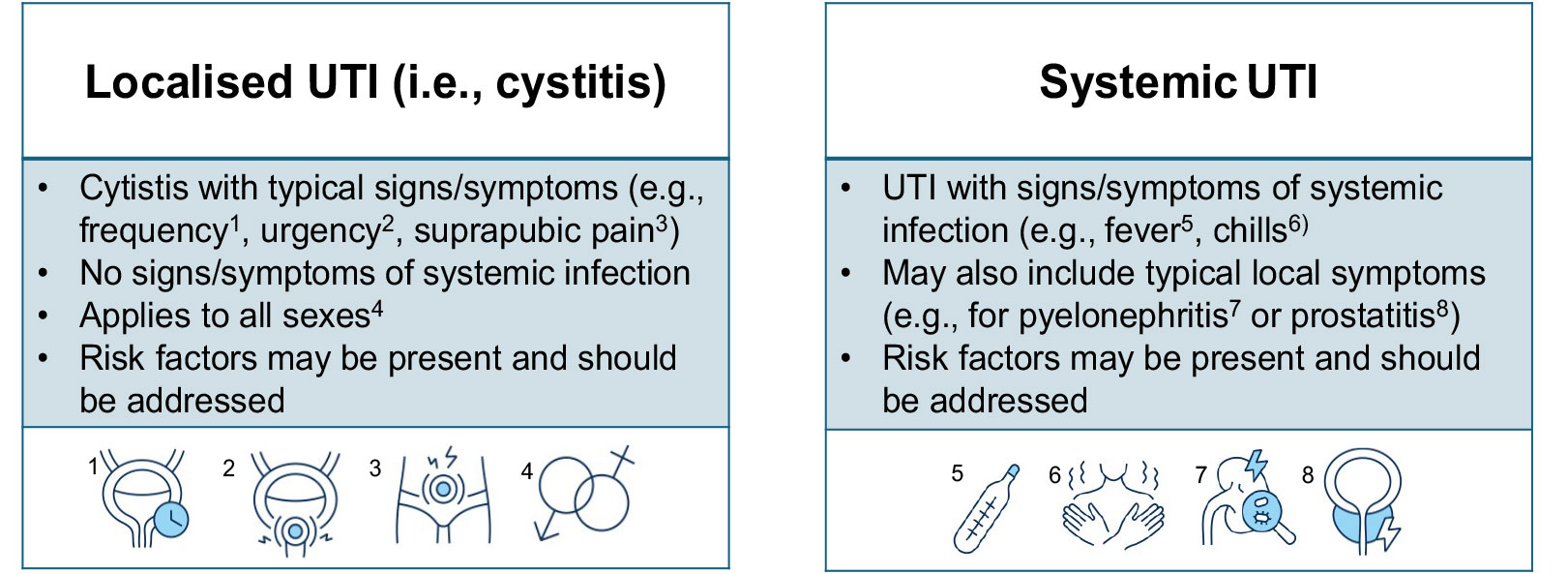
Table 1: Localised and systematic signs and symptoms of UTI
| Localised UTI1 | Systemic UTI1,2 |
| Dysuria (pain, burning, stinging) | Fever or hypothermia |
| Urgency | Rigors, shaking chills |
| Frequency | Delirium |
| Incontinence | Hypotension |
| Urethral purulence | Tachycardia |
| Pressure or cramping in the lower abdomen | Costovertebral angle pain/tenderness |
1. Recent onset of these localised and/or systemic signs and symptoms.
2. These signs and symptoms are possibly caused by a systemic UTI, but there may also be alternative explanations.
3.1.1. Risk factors
When managing UTIs, it is essential to consider risk factors that may predispose patients to a severe clinical course or treatment failure. Factors such as urinary tract abnormalities, the presence of urinary catheters, previous antibiotic treatment and underlying health conditions (e.g. diabetes, renal impairment and neurological disorders) can significantly influence the outcome of UTIs. Importantly, in the proposed classification, male sex is not considered as a risk factor as it lacks support from contemporary literature. Table 2 provides a clear overview of risk factors for clinicians. By identifying and addressing these risk factors early in the treatment process, clinicians can optimise patient care and improve treatment outcomes.
Table 2: UTI Risk Factors*
| UTI Risk Factor | ||
| Infants | Immunocompromised state | Male sex • Prostatic involvement (e.g. benign prostatic obstruction, chronic bacterial prostatitis) |
| Geriatric or frail patients | Significant post void residual volume | |
| Anatomic or functional abnormalities of the urinary tract | Neurourological disease | Female sex • Pregnancy • Pelvic organ prolapse |
| Previous antibiotic use | ||
| Indwelling urinary catheters | Resistant organisms | |
| Stones | Urinary tract obstruction | Recent instrumentation |
* Both localised and systemic UTIs may be accompanied by risk factors that increase the likelihood of a challenging clinical course and jeopardise treatment success. Clinicians must be aware of these risk factors to adjust treatment if necessary.
3.2. Antimicrobial stewardship
Although the benefits to patients of antibiotic use are clear, overuse and misuse have contributed to the growing problem of resistance amongst uropathogenic bacteria, which is a serious threat to public health [10, 11]. In acute care hospitals, 20–50% of prescribed antibiotics are either unnecessary or inappropriate [12]. In response, a worldwide initiative seeks to incorporate antimicrobial stewardship programmes in healthcare [13]. Antimicrobial stewardship aims to reduce unnecessary antibiotic use, optimise clinical outcomes and ensure cost-effective therapy, whilst minimising unintended consequences of antimicrobial use, such as healthcare-associated infections including Clostridioides difficile, toxicity, selection of virulent organisms and emergence of resistant bacterial strains [14].
Stewardship programmes have two main sets of actions. The first set mandates the use of recommended care at the patient level conforming to guidelines. The second set describes strategies to achieve adherence to the mandated guidance. These include persuasive actions such as education and feedback, together with restricting availability linked to local formularies. A Cochrane review of effectiveness of interventions to improve antibiotic prescribing practices for hospital inpatients, updated in 2017, found high-certainty evidence that such interventions are effective in increasing adherence with antibiotic policy leading to reduced antibiotic treatment duration and that it may also reduce hospital stay. The review found no evidence that reduced antibiotic usage increased mortality [15].
The important components of antimicrobial stewardship programmes are [16]:
- regular training of staff in best use of antimicrobial agents;
- adherence to local, national or international guidelines;
- regular ward visits and consultation with infectious diseases physicians and clinical microbiologists;
- audit of adherence and treatment outcomes; and
- regular monitoring and feedback to prescribers of their performance and local pathogen resistance profiles.
A 2016 systematic review of evidence for effectiveness of various antimicrobial stewardship interventions in healthcare institutions identified 145 studies of nine stewardship objectives. Guideline-driven empirical therapy using a restricted choice of antibiotics and including de-escalation, intravenous to oral switch, therapeutic drug monitoring and bedside consultation resulted in a 35% (95% CI: 20-46%) relative risk reduction (RRR) in mortality. Use of de-escalation (tailoring to a narrower spectrum agent) showed an RRR of 56% (95% CI: 34–70%) in mortality [17].
To facilitate local initiatives and audit, a set of valid, reliable and applicable indicators of the quality of antibiotic use in the treatment of hospitalised patients with complicated UTI was developed [18]. The use of this set of indicators in the Netherlands appeared to result in shortened hospital stay [19]. A literature search of PubMed from April 2014 [17] to February 2017 identified no further randomised controlled trials (RCTs) relating to stewardship programmes for UTIs. Studies to provide high-quality evidence of effectiveness of stewardship programmes in urology patients are urgently needed.
3.3. Asymptomatic bacteriuria in adults
3.3.1. Background
Urinary growth of bacteria in an asymptomatic individual (asymptomatic bacteriuria [ABU]) is common and corresponds to a commensal colonisation [20]. Clinical studies have shown that ABU may protect against superinfecting UTI. Therefore, treatment of ABU should only be performed in cases of proven benefit for the patient to avoid the risk of selecting antimicrobial resistance and eradicating a potentially protective ABU strain [21, 22]. The aim of this section is to support the clinician in deciding when ABU should or should not be treated.
3.3.2. Epidemiology, aetiology and pathophysiology
Asymptomatic bacteriuria occurs in an estimated 1–5% of healthy premenopausal females. The prevalence increases to 4–19% in otherwise healthy elderly women and men, 0.7–27% in diabetic patients, 2–10% in pregnant women, 15–50% in institutionalised elderly populations and 23–89% in patients with spinal cord injuries [23]. Asymptomatic bacteriuria in younger men is uncommon, but when detected, chronic bacterial prostatitis must be considered. The spectrum of bacteria in ABU is similar to species found in localised or systemic UTIs, depending on the presence of risk factors (see Sections 3.4 and 3.7).
3.3.3. Diagnostic evaluation
Asymptomatic bacteriuria is defined as bacterial growth of ≥ 10⁵ CFU/mL in a midstream urine sample – confirmed in two consecutive samples in women [24] and in one single sample in men [25] in an individual without urinary tract symptoms. Cystoscopy and/or imaging of the upper urinary tract is not mandatory if the medical history is otherwise without remark. If persistent growth of urease-producing bacteria, such as Proteus mirabilis, is detected, stone formation in the urinary tract must be excluded [26-28]. In men, a digital rectal examination (DRE) must be performed to investigate the possibility of prostate diseases (see Section 3.11).
3.3.4. Evidence summary
A systematic search of the literature from November 2016 to January 2000 identified 3,582 titles, of which 224 were selected for full text review and 50 were included [29]. For the subgroups of pregnancy, prior to urologic surgeries, postmenopausal women and institutionalised elderly patients, only data from RCTs were included, on which a meta-analysis was performed [29]. For the other subgroups, non-RCTs were also included in the narrative analysis [29]. An update systematic literature search from 1 December 2016 to 1 June 2023 identified 1,503 titles, of which 36 were selected for full text review and 18 were included. The following patient populations were not covered by the systematic review: immunocompromised patients and patients with indwelling catheters. For these groups, the Guideline was updated using a structured PubMed search. The evidence question addressed was: What is the most effective management for people with asymptomatic bacteriuria?
3.3.5. Disease management
3.3.5.a. Patients without identified risk factors
Asymptomatic bacteriuria does not cause renal disease or damage [30]. Only one prospective, nonrandomised study investigated the effect of treatment of ABU in adult, nondiabetic, nonpregnant women [31], and found no difference in the rate of symptomatic UTIs. Moreover, as the treatment of ABU has been proven to be unnecessary in most high-risk patient subgroups, there is panel consensus that the results of these subgroups can also be applied to patients without identified risk factors. Therefore, screening and treatment of ABU is not recommended in patients without risk factors.
3.3.5.b. Patients with ABU and recurrent UTI, otherwise healthy
One RCT investigated the effect of ABU treatment in female patients with recurrent symptomatic UTI without identified risk factors [22] and demonstrated that treatment of ABU increases the risk for a subsequent symptomatic UTI episode compared to untreated patients (RR 0.28, 95% CI: 0.21–0.38; n = 673). This protective effect of spontaneously developed ABU can be used as part of prevention in female patients with recurrent symptomatic UTI, therefore, treatment of ABU is not recommended.
3.3.5.c. Pregnant women
3.3.5.c.1. Is treatment of ABU beneficial in pregnant women?
Twelve RCTs comparing antibiotic treatments of ABU with placebo controls or no treatment [32-43], with various antibiotic doses and regimens were identified: ten published before 1988 and one in 2015. Eleven RCTs (n = 2,002) reported on the rate of symptomatic UTIs [32, 34-42, 44]. Antibiotic treatment significantly reduced the number of symptomatic UTIs compared to placebo or no treatment (average RR 0.22, 95% CI: 0.12–0.40).
Six RCTs reported on the resolution of bacteriuria [32-34, 36, 39, 41]. Antibiotic treatment was effective in the resolution of bacteriuria compared to placebo (average RR 2.99, 95% CI: 1.65–5.39; n = 716). Eight RCTs reported on the rate of low birthweights [32, 34-37, 40, 43, 44]. Antibiotic treatment was associated with lower rates of low birthweight compared to placebo or no treatment (average RR 0.58, 95% CI: 0.36–0.94; n = 1689). Four RCTs reported on the rate of preterm deliveries [40, 41, 43, 44]. Antibiotic treatment was associated with lower rates of preterm delivery compared to placebo or no treatment (average RR 0.34, 95% CI: 0.18–0.66; n = 854). Three additional systematic reviews and meta-analyses have reported that treatment of ABU in pregnancy may be associated with a decreased rate of pyelonephritis, low birthweight or preterm delivery [45-47]. However, they also emphasised the low to very low quality of the evidence of the identified studies.
Based on the beneficial maternal and foetal effects of antibiotic treatment, pregnant women should be screened and treated for ABU. However, the panel would like to emphasise that most available studies have low methodological quality and are from the 1960s to 1980s. Diagnostic and treatment protocols and accessibility to medical services have dramatically changed since then. Therefore, the quality of evidence for this recommendation is low. In a newer study of higher methodological quality, the beneficial effects of antibiotic treatment are not as evident [44]. Therefore, it is advisable to consult national recommendations for pregnant women.
3.3.5.c.2. Which treatment duration should be applied to treat ABU in pregnancy?
Sixteen RCTs comparing the efficacy of various antibiotic treatments in pregnant women with ABU were identified [48-63]. Significant heterogeneity was found amongst the studies. Studies compared various antibiotic regimens or the same antibiotic regimens with different durations. The duration of treatment ranged from single-dose to continuous treatment (until delivery). For practical purposes, the grouping strategy used by the previously published Cochrane Review was adopted with some modifications [64]. The following treatment groups were used for comparison:
- single dose (single day)
- short course (2–7 days)
- long course (8–14 days)
- continuous (until delivery)
Nine studies compared single-dose to short-course treatment [49, 53, 54, 58-63], one study compared single dose to long course treatment [57] and one study compared long course to continuous treatment [50]. As long-term and continuous antibiotic treatment is not used in current practice, only studies comparing single dose to standard short course treatment are presented.
3.3.5.c.2.a. Single dose vs. short course treatment
Three RCTs reported on the rate of symptomatic UTIs [53, 62, 63], with no significant difference between the two durations (average RR 1.07, 95% CI: 0.47–2.47; n = 891). Nine RCTs reported on the rate of ABU resolution [49, 53, 54, 58-63], with no significant difference between the two durations (average RR 0.97, 95% CI: 0.89–1.07; n = 1,268). Six RCTs reported on the rate of side effects [49, 53, 58, 59, 61, 62]. Single-dose treatment was associated with significantly less side effects compared to short course treatment (average RR 0.40, 95% CI: 0.22–0.72; n = 458). Three RCTs reported on the rate of preterm deliveries [53, 55, 63], with no significant difference between the two durations (average RR 1.16, 95% CI: 0.75–1.78; n = 814). One RCT reported on the rate of low birth weights [63]. There were significantly more babies with low birth weight in the single-dose duration compared to short-course treatment (average RR 1.65, 95% CI: 1.06–2.57; n = 714).
According to the data analysis, single-dose treatment was associated with a significantly lower rate of side effects but a significantly higher rate of low birth weight. A meta-analysis on the use of single-dose fosfomycin trometamol in women with cystitis or ABU reported on a subgroup analysis of pregnant women with ABU [65]. The study identified five RCTs involving 577 patients. The resolution rate of ABU in pregnant women treated with single-dose fosfomycin trometamol was not significantly different from those who received other antibiotics (OR 1.32, 95% CI: 0.78–2.22, p = 0.30). Therefore, standard short-course treatment or single-dose fosfomycin trometamol should be applied to treat ABU in pregnancy. However, it should be emphasised that the overall quality of the scientific evidence backing this recommendation is low.
3.3.5.d. Patients with identified risk factors
3.3.5.d.1. Diabetes mellitus
Diabetes mellitus, even when well regulated, is reported to correlate to a higher frequency of ABU [66]. One RCT demonstrated that eradicating ABU did not reduce the risk of symptomatic UTI and infectious complications in patients with diabetes mellitus. The time to first symptomatic episode was also similar in both groups. Moreover, untreated ABU did not correlate to diabetic nephropathy [67]. Screening and treatment of ABU in well-controlled diabetes mellitus is therefore not recommended. However, poorly regulated diabetes is a risk factor for symptomatic UTI and infectious complications.
3.3.5.d.2. ABU in postmenopausal women
Elderly women have an increased incidence of ABU [68]. Four RCTs compared antibiotic treatment of ABU with placebo controls or no treatment, in a postmenopausal female population, with various antibiotic doses and regimens [69-72]. Women in these studies were mostly nursing home residents, which may bias the results of this analysis. Three RCTs reported on the rate of symptomatic UTIs (average RR 0.71, 95% CI: 0.49–1.05; 208 women) and the resolution of bacteriuria (average RR 1.28, 95% CI: 0.50–3.24; 203 women) [53, 62, 63], with no significant benefit of antibiotic treatment. Therefore, ABU in postmenopausal women does not require treatment, and should be managed as for premenopausal women.
3.3.5.d.3. Elderly institutionalised patients
The rate of ABU is 15–50% in elderly institutionalised patients [73]. Differential diagnosis of ABU from symptomatic UTI is difficult in the multidiseased and mentally deteriorated patients and is probably a cause of unnecessary antibiotic treatment [74, 75]. Seven RCTs compared antibiotic treatment of ABU with placebo controls or no treatment in elderly patients with various antibiotic doses and regimens [69-72, 76-78].
Three RCTs reported on the rate of symptomatic UTIs [69, 71, 76]. Antibiotic treatment was not significantly beneficial in reducing the rate of symptomatic UTIs compared to placebo or no treatment (average RR 0.68, 95% CI: 0.46–1.00; n = 210). Six RCTs reported on the resolution of bacteriuria [69, 71, 72, 76-78]. There was no benefit of antibiotic treatment compared to placebo in the resolution of ABU (average RR 1.33, 95% CI: 0.63–2.79; n = 328). One RCT compared the rates of incontinence in this patient group before and after the eradication of ABU and found no effect of antibiotic treatment [79]. A subsequent systematic review and meta-analysis of nine RCTs found that antibiotic treatment of ABU in this group was associated with significantly more adverse effects with no clinical benefit [80]. Screening and treatment of ABU is therefore not recommended in this patient group.
3.3.5.d.4. Patients with renal transplants
Two RCTs and two retrospective studies compared the effect of antibiotic treatment to no treatment in renal transplant patients [81-84]. Meta-analysis of the two RCTs did not find antibiotic treatment beneficial in terms of reducing symptomatic UTIs between 12 and 22 months after renal transplantation (RR 0.86, 95% CI: 0.51–1.45; n = 200). The two retrospective studies reached the same conclusion. Moreover, no significant differences were found in the rate of ABU clearance, graft loss or change in renal function during long-term follow-up of up to 24 months [81-84].
A further two RCTs [85,86], one observational study [87] and two systematic reviews and meta-analyses [88,89] were identified. The first RCT reported that, during the first two months following renal transplantation, the incidence of and risk for UTIs (25% vs. 10%, HR 2.8, 95% CI: 0.8–9.1, p = 0.07) and pyelonephritis (15% vs. 2.5%, HR 6.5, 95% CI: 0.8–54.7, p = 0.08) was higher in patients receiving antibiotic treatment for ABU versus no treatment [85]. In the second RCT, no difference in acute graft pyelonephritis was found between the treatment and no-treatment group (12.2% vs. 8.7%, RR 1.40, 95% CI: 0.40–4.87) in the first year after renal transplantation. However, rates of antimicrobial resistance were higher in the treatment group [86]. The first of the two additional meta-analyses reported the same results as the original study [88]. The second meta-analysis of n = 1,353 patients reported ABU incidence rates of 22% in the first month and 32% during the first year after renal transplantation [89]. The analysis did not find a correlation between ABU and acute graft pyelonephritis (OR 1.8, 95% CI: 0.78–1.79), a benefit of ABU antibiotic treatment on the risk of UTI (OR 1.08, 95% CI: 0.63–1.84) or a change of renal function (mean difference in serum creatinine concentration – 0.03mg/dL [95% CI: 0.15–0.10]) [89].
Therefore, treatment of ABU is not recommended in renal transplant recipients.
3.3.5.d.5. Patients with dysfunctional and/or reconstructed lower urinary tracts
Patients with lower urinary tract dysfunction (LUTD) (e.g. neurogenic lower urinary tract dysfunction [NLUTD]) secondary to multiple sclerosis; spinal cord injury patients; patients with incomplete bladder emptying; patients using clean intermittent catheterisation [CIC]; or patients with reconstructed lower urinary tract including ileal conduits, orthotopic bladder replacement or continent reservoirs frequently become colonised [90,91]. A systematic review reported ABU prevalence rates ranging from 25 to 86% for intestinal conduits in four studies and from 9.1 to 85% for orthotopic neobladders in nine studies [92]. Studies have shown no long-term benefit in ABU treatment in these patient groups [84,85,92]. Moreover, in LUTD patients who do not spontaneously develop ABU, deliberate colonisation with an ABU strain (Escherichia coli 83972) has shown a protective effect against symptomatic recurrences [93,94]. Screening and treatment of ABU in these patient groups is therefore not recommended. If these patient groups develop recurrent symptomatic UTI (see Section 3.5) the potential protective effect of a spontaneously developed ABU against UTI must be considered before any treatment.
3.3.5.d.6. Patients with catheters in the urinary tract
Patients with indwelling or suprapubic catheters and nephrostomy tubes invariably become carriers of ABU, with antibiotic treatment showing no benefit [95]. This is also applicable for patients with ABU and indwelling ureteral stents [96]. Routine treatment of catheter-associated ABU is therefore not recommended. For detailed recommendations see Section 3.8.
3.3.5.d.7. Patients with ABU subjected to catheter placements/exchanges
In patients subjected to uncomplicated placement/exchanges of indwelling urethral catheters, ABU is not considered a risk factor and should not be screened or treated [97]. In patients subjected to placement/exchanges of nephrostomy tubes and indwelling ureteral stents, ABU is considered a risk factor for infectious complications [98].
3.3.5.d.8. Immunocompromised and severely diseased patients, patients with candiduria
These patient groups must be considered individually, and the benefit of screening and treatment of ABU should be reviewed in each case.
3.3.5.e. Prior to urological surgery
In diagnostic and therapeutic procedures not entering the urinary tract, ABU is generally not considered a risk factor, and screening and treatment are not considered necessary. On the other hand, in procedures entering the urinary tract and breaching the mucosa, particularly in endourological surgery, bacteriuria is a definite risk factor for postoperative UTI.
Two RCTs [99, 100] and two prospective nonrandomised studies [101, 102] compared the effect of antibiotic treatment to no treatment before transurethral prostate or bladder tumour resections. Antibiotic treatment significantly reduced the number of postoperative symptomatic UTIs compared to no treatment in the meta-analysis of the two RCTs (average RR 0.20, 95% CI: 0.05–0.86; n = 167). The rates of postoperative fever and septicaemia were also significantly lower in case of antibiotic treatment compared to no treatment in the two RCTs. One RCT including patients with spinal cord injury undergoing elective endoscopic urological surgeries found no significant difference in the rate of postoperative UTIs between single-dose or three to five days’ short-term preoperative antibiotic treatment of ABU [103].
A urine culture must therefore be taken prior to such interventions and in case of ABU, preoperative treatment is recommended.
3.3.5.f. Prior to orthopaedic surgery
One RCT (n = 471) and one multicentre cohort study (n = 303) comparing the treatment of ABU with no treatment prior to orthopaedic surgery (hip arthroplasty/hemiarthroplasty or total knee arthroplasty) were identified [104, 105]. Neither of the studies showed a beneficial effect of antibiotic treatment in terms of prosthetic joint infection (3.8% vs. 0% and 3.9% vs. 4.7%, respectively). The cohort study reported no significant difference in the rate of postoperative symptomatic UTI (0.65% vs. 2.7%) [105]. One further RCT investigated the efficacy of preoperative ABU treatment with fosfomycin trometamol for prevention of early periprosthetic joint infections (PJI) after hip hemiarthroplasty for fractures. Asymptomatic bacteriuria was not predictive of early PJI (OR: 1.06, 95% CI 0.33-3.38), and its treatment did not modify early PJI incidence (OR: 1.03, 95%CI 0.15-7.10) [106]. Furthermore, four additional meta-analyses did not find a benefit for preoperative screening or treatment of ABU prior to orthopaedic surgery [107-110]. Therefore, treatment of bacteriuria is not recommended prior to arthroplasty surgery.
3.3.5.g. Prior to cardiovascular surgery
One systematic review and meta-analysis including three retrospective nonrandomised studies involving a total of 1,116 patients was identified [111]. The procedures performed were nonvalvular coronary artery bypass grafting (42%), valvular replacements (51%) and thoracic aortic surgeries (7%). Preoperative treatment of ABU in 116 patients did not result in significant benefit regarding the rate of SSI compared to no treatment (12.9% vs. 8.2%, p = 0.086). A moderate heterogeneity was observed in the meta-analysis and preoperative treatment of ABU had no significant effect on the rate of infectious complications (OR: 1.38, 95% CI 0.56–3.39). Due to the very low number, retrospective and nonrandomised design of the included studies, only limited conclusions can be drawn from this. Further studies with appropriate design and sample size are needed to confirm these findings.
3.3.5.h. Pharmacological management
If the decision is taken to eradicate ABU, the treatment should be tailored according to the drug sensibility pattern of the pathogen identified. Treatment should be tailored and not empirical.
3.3.6. Follow-up
There are no studies focusing on follow-up after treatment of ABU.
3.3.7. Summary of evidence and recommendations for the management of ABU
| Summary of evidence | LE |
| Treatment of asymptomatic bacteriuria is not beneficial in the following conditions: |
|
| - women without risk factors; | 3b |
| - patients with well-regulated diabetes mellitus; | 1b |
| - postmenopausal women; | 1a |
| - elderly institutionalised patients; | 1a |
| - patients with dysfunctional and/or reconstructed lower urinary tracts; | 2b |
| - patients with renal transplants; | 1a |
| - patients prior to arthroplasty surgeries; | 1a |
| - patients prior to cardiovascular surgeries. | 1b |
| Treatment of asymptomatic bacteriuria is harmful in patients with recurrent urinary tract infections. | 1b |
| Treatment of asymptomatic bacteriuria is beneficial prior to urological procedures breaching the mucosa. | 1a |
| Treatment of asymptomatic bacteriuria in pregnant women was found to be beneficial by meta-analysis of the available evidence, however, most studies are old. A more-recent study reported that untreated or placebo-treated asymptomatic bacteriuria-positive women developed pyelonephritis more frequently than asymptomatic bacteriuria-negative women (2.4% vs. 0.6%). | 1a |
| Recommendations | Strength rating |
Do not screen or treat asymptomatic bacteriuria in the following conditions:
| Strong |
| Do not screen or treat asymptomatic bacteriuria in patients prior to cardiovascular surgeries. | Weak |
| Screen for and treat asymptomatic bacteriuria prior to urological procedures breaching the mucosa. | Strong |
| Screen for and treat asymptomatic bacteriuria in pregnant women with standard short-course treatment or single dose fosfomycin trometamol*. | Weak |
* The panel wishes to emphasise that most available studies have low methodological quality and are from the 1960s to 1980s. Diagnostic and treatment protocols and accessibility to medical services have dramatically changed since then. The quality of evidence for this recommendation is therefore low. In a newer study of higher methodological quality, the beneficial effects of antibiotic treatment are not as evident . Therefore, it is advisable to consult national recommendations for pregnant women.
3.4. Cystitis in women
3.4.1. Introduction
Cystitis in women is defined as a symptomatic lower urinary tract infection limited to the bladder, in the absence of systemic symptoms or signs such as fever, chills, flank pain, or malaise.
3.4.2. Epidemiology, aetiology and pathophysiology
Nearly half of all women will experience at least one episode of cystitis during their lifetime. Nearly one in three women will have had at least one episode of cystitis by the age of 24 [112]. Risk factors include sexual intercourse, use of spermicides, a new sexual partner, a mother with a history of cystitis, and a history of cystitis during childhood. The majority of cases of cystitis are caused by E. coli.
3.4.3. Diagnostic evaluation
3.4.3.a. Clinical diagnosis
The diagnosis of cystitis can be made with a high probability based on a focused history of lower urinary tract symptoms (dysuria, frequency and urgency) and the absence of vaginal discharge [113, 114]. In elderly women, genitourinary symptoms are not necessarily related to cystitis [115, 116].
3.4.3.b. Differential diagnosis
Cystitis should be differentiated from ABU, which is not considered to be infection, but rather a commensal colonisation, which should not be treated and therefore not screened for, unless it is considered a risk factor in clearly defined situations (see Section 3.3).
3.4.3.c. Laboratory diagnosis
In patients presenting with typical symptoms of cystitis urine analysis (i.e. urine culture, dip stick testing, etc.) leads only to a minimal increase in diagnostic accuracy [117]. However, if the diagnosis is unclear, dipstick analysis can increase the likelihood of a cystitis diagnosis [118, 119]. Taking a urine culture is recommended in patients with atypical symptoms, as well as those who fail to respond to appropriate antimicrobial therapy [120, 121].
3.4.3.d. Summary of evidence and recommendations for the diagnostic evaluation of cystitis
| Summary of evidence | LE |
| An accurate diagnosis of cystitis in women can be based on a focused history of lower urinary tract symptoms and the absence of vaginal discharge or irritation. | 2b |
| Recommendations | Strength rating |
Diagnose cystitis in women who have no other risk factors for systemic urinary tract infections based on:
| Strong |
| Use urine dipstick testing for diagnosis of acute cystitis. | Weak |
Urine cultures should be done in the following situations:
| Strong |
3.4.4. Disease management
When deciding on the management of cystitis, several therapeutic options are available. These can be broadly divided into two groups: non-antibiotic treatments and antibiotic treatments. Patient preferences should always be appropriately considered. This is particularly relevant for nonantibiotic strategies, which may involve accepting a higher symptom burden and a slightly increased risk of complications such as pyelonephritis.
3.4.4.a. Nonantibiotic treatments for the management of cystitis
3.4.4.a.1. Evidence Summary
In July 2024, an update on the role of nutraceuticals in managing cystitis was conducted, in which 995 abstracts were identified, retrieved and screened for relevance, with 44 selected for full-text review. This review resulted in the inclusion of 20 systematic reviews or guidelines based on systematic literature searches and 24 original publications for further analysis in the Guidelines. Among the clinical trials, nine studies evaluated the efficacy of nutraceuticals in managing acute episodes of cystitis, while 16 focused on recurrence prevention. Cranberry extract, either alone or in combination with other compounds, was the most extensively studied nutraceutical. The primary focus was determining which nutraceutical compounds effectively alleviate symptoms during the acute phase of cystitis and reduce recurrence rates in women with acute or recurrent symptomatic cystitis.
3.4.4.a.2. Management of acute cystitis
Treatment with cranberry products
The role of cranberry products in managing acute cystitis was evaluated in a systematic review of three RCTs [122]. In two of those RCTs, cranberry juice was used [123, 124], while the third trial utilised encapsulated cranberry powder [125]. In all three RCTs, the proanthocyanidin content varied significantly (from 7.5 to 224mg), and the primary objective of the studies was not assessment of cranberry extract as an acute cystitis treatment. The risk of bias of the included studies was judged as moderate. Hence, the current evidence supporting or opposing the use of cranberry products for managing acute cystitis remains insufficient.
Treatment with D-mannose
The role of D-mannose in managing cystitis symptoms was investigated in a systematic review [126] that included seven studies, two of which were RCTs [127, 128]. One RCT compared the use of D-mannose combined with cranberry and antibiotics to antibiotics alone [127], while the other compared D-mannose alone to antibiotics [128]. Among the non-RCTs, only one study evaluated D-mannose alone, with the remainder testing D-mannose in combination with other compounds [129]. Although this review suggests potential benefits of D-mannose in treating acute cystitis, current evidence remains insufficient to recommend it in this setting, due to the limited data for a detailed analysis of the effects of various doses of D-mannose or its impact when used alone versus in combination with other compounds. Moreover, most of the data originates from uncontrolled studies. Overall, the findings from this systematic review and meta-analysis are based on a very limited number of studies with small sample sizes.
A recent single-centre, randomised, double-blind, placebo-controlled trial examined the efficacy of D-mannose-based dietary supplement (D-mannose, citric acid, prebiotic fibres, Astragalus and dandelion [DAPAD] complex) for subjective (clinical resolution/response) and objective (midstream bacteriuria) outcomes in women with acute E. coli cystitis [130]. This study reported higher clinical and bacteriological resolution rates in the treatment group compared to placebo, with both groups receiving standard care and advice on increased fluid intake. However, despite these findings, due to study limitations and limited evidence, D-mannose cannot yet be recommended in this context.
Treatment with phytotherapeutics
A double-blind, parallel group, randomised, multicentre, noninferiority phase III trial aimed to determine whether herbal therapy with Centaurii herba, Levistici radix and Rosmarini folium (BNO 1045) is noninferior to fosfomycin trometamol (FT) in treating acute lower cystitis [131]. Women aged 18 to 70 with typical symptoms of newly diagnosed acute lower cystitis were randomised to BNO 1045 (n = 325) or FT (n = 334) with a corresponding matched placebo. The primary endpoint was the proportion of patients who received additional antibiotics to treat cystitis between days one and 38 ± 3. Between days one and 38, 238 (83.5%) patients in the BNO 1045 group and 272 (89.8%) patients in the FT group received no additional antibiotics. At a 15% noninferiority margin, BNO 1045 was noninferior to FT in treating uncomplicated UTIs (uUTIs) (non-AB rate difference: -6.26%; 95% CI: -11.99 to -0.53%; 2-sided p = 0.0014). This study demonstrated the noninferiority of this herbal combination compared to fosfomycin trometamol.
Another RCT evaluated the efficacy of a phytotherapeutical combination of L-Methionine accompanied by Hibiscus sabdariffa and Boswellia serrata for the treatment of acute episodes of cystitis in women affected by recurrent cystitis [132]. Forty-six patients were enrolled in the phytotherapeutics combination group (Group A), and 47 in the short-term antibiotic treatment (Group B). At the first follow-up (30 days), both groups showed a statistically significant improvement in quality-of-life scores as compared with baseline assessment [Group A: (QoL 94.3 vs. 98.5 p < 0.001); Group B: (QoL 94.5 vs. 98.7 p < 0.001)]. An improvement from baseline was also seen at the second follow-up evaluation after three months [Group A: (QoL 94.3 vs. 99.1; p < 0.001); Group B: (QoL 94.5 vs. 98.1 p < 0.001)]. At the second follow-up visit, a statistically significant difference in QoL was reported between the two groups (99.1 vs. 98.1; p < 0.003) and a transition from cystitis to ABU was observed 12 of 46 (26%) patients in Group A, while no patients in Group B demonstrated ABU (p = 0.007). It has been demonstrated that phytotherapeutic combinations are able, in comparison to antibiotic treatment, to improve patients’ QoL, reducing symptoms in the acute setting and preventing recurrences. Interestingly, a significantly higher proportion of patients in the phytotherapy group had ABU after three months.
A recent systematic review on herbal medicine for cystitis symptom control highlighted that combinations containing Centaurii herba, Levistici radix, Rosmarini folium, L-methionine, Hibiscus sabdariffa and Boswellia serrata show promise for treating women with recurrent cystitis [133].
Treatment with xyloglucan, gelose, hibiscus and propolis
In a multicentre, randomised, parallel group, double-blind, phase IV study, the safety and efficacy of a combination containing xyloglucan-gelose-hibiscus-propolis were investigated as adjuvant therapy to first-line antimicrobials for treatment of cystitis in adults [134]. In this study, the medical device (n = 20) or placebo (n = 20) were administered orally in combination with an antimicrobial agent (e.g. ciprofloxacin) for five days, then alone for five days and then, beginning on day 30 of the study, for 15 days per month for two months. Xyloglucan/gelose reduced positive urine cultures (defined as a bacterial count ≥ 103 CFU/mL) from 100% of patients at baseline to 0% at day 11, with recurrence in three patients (15%) by day 76. Corresponding results with placebo were all patients had positive urine cultures at baseline reduced to 45% at day 11, with recurrence in 14 patients (70%) by day 76. Xyloglucan/gelose significantly reduced the frequency of urinary incontinence and urgency of micturition compared with placebo (both p < 0.05), with symptom resolution in all patients by day 90. These findings were supported by a systematic review and meta-analysis of studies testing this xyloglucan-containing preparation [135]. The primary endpoint was clinical or microbiological success – defined as the complete (cure) and/or non-complete (improvement) resolution of symptoms at the end of treatment – or microbiological resolutions. Three studies were included, recruiting a total of 178 patients. All three studies used placebo as comparator. A statistically significant difference was found in terms of clinical and microbiological resolution between the medical device and the comparator (three RCTs, 178 patients, OR: 0.13; 95% CI: 0.05–0.33; p < 0.0001). No clinically significant adverse effects have been reported.
Nonsteroidal anti-inflammatory drugs
The indication for antibiotic therapy should be critically assessed [136]. A number of systematic reviews have compared the effects of primarily symptomatic treatment with nonsteroidal anti-inflammatory drugs (e.g. diclofenac, ibuprofen) [137], D-mannose [126] and phytotherapeutic preparations (e.g. uva ursi, BNO 1045) [136] with immediate antibiotic treatment. Nonantibiotic therapies showed good cure rates, however, in most studies, their success rates were lower than those achieved with antibiotic treatment when outcomes such as incomplete resolution, additional antibiotic therapy or the development of pyelonephritis, were taken into account [126, 136, 137]. The use of nonsteroidal anti-inflammatory drugs (ibuprofen and diclofenac) and phytotherapy (uva ursi and BNO 1045) have all been shown to result in a reduction in antibiotic therapy [138-142]. Overall, across all studies, nonantibiotic therapy led to a 63% reduction in antibiotic use [136]. For geriatric patients, there is no evidence regarding symptomatic treatment with nonsteroidal anti-inflammatory drugs. Moreover, nonsteroidal anti-inflammatory drugs are considered potentially inappropriate medications with associated risks in this age group [143].
3.4.4.a.3. Summary of evidence and recommendations for nonantibiotic management of cystitis
| Summary of evidence | LE |
| An RCT showed that Centaurii herba, Levistici radix and Rosmarini folium alone are effective in relieving acute cystitis symptoms compared to fosfomycin trometamol. | 1b |
| An RCT demonstrated the efficacy of L-methionine combined with Hibiscus sabdariffa and Boswellia serrata in relieving acute cystitis symptoms, as compared to fosfomycin trometamol. | 1b |
| A combination of xyloglucan, hibiscus and propolis is effective in relieving acute cystitis symptoms and preventing recurrence. | 1a |
| There is contradictory evidence on the efficacy of D-mannose to reduce the number of cystitis episodes. | 2 |
| Recommendations | Strength rating |
| Advise female patients on the possibility of antibiotic-sparing approaches for the treatment and prevention of acute cystitis. Patients should be fully informed on the level of evidence for the various approaches. | Strong |
| Use non-antibiotic therapy options as an alternative to antibiotic treatment in nongeriatric patients. Shared decision-making with the patients is essential. | Strong |
3.4.4.b. Antibiotic treatment
Antimicrobial therapy may be considered in women with cystitis, as clinical success is significantly more likely compared to placebo [144]. In nongeriatric patients, nonantibiotic therapy alone should be considered as an alternative to antibiotic treatment. A shared decision-making process with the patients is essential [145].
The choice of antimicrobial therapy should be guided by [113]:
- spectrum and susceptibility patterns of the aetiological pathogens;
- efficacy for the particular indication in clinical studies;
- tolerability and adverse reactions;
- adverse ecological effects;
- costs; and
- availability.
According to these principles and the available susceptibility patterns in Europe, oral treatment with fosfomycin trometamol 3g single-dose, pivmecillinam 400mg three times daily for three to five days, and nitrofurantoin (e.g. nitrofurantoin monohydrate/macrocrystals 100mg twice daily for five days) should be considered for first-line treatment when available [146-149].
Alternative antimicrobials include trimethoprim alone or combined with a sulphonamide. Co-trimoxazole (160/800mg twice daily for three days) or trimethoprim (200mg twice daily for five days) should only be considered as drugs of first choice in areas with known resistance rates for E. coli of < 20% [150, 151].
Aminopenicillins are no longer suitable for empirical therapy due to worldwide high E. coli resistance. Aminopenicillins in combination with a beta-lactamase inhibitor such as ampicillin/sulbactam or amoxicillin/clavulanic acid and oral cephalosporins are not recommended for empirical therapy due to ecological collateral damage, but may be used in selected cases [152, 153].
Important note:
On 11 March 2019, the European Commission implemented stringent regulatory conditions regarding the use of fluoroquinolones due to their disabling and potentially long-lasting side effects [154]. This legally binding decision is applicable in all EU countries. National authorities have been urged to enforce this ruling and to take all appropriate measures to promote the correct use of this class of antibiotics. In cystitis, a fluoroquinolone should only be used when it is considered inappropriate to use other antibacterial agents that are commonly recommended for the treatment of these infections [154].
3.4.4.c. Cystitis with risk factors
3.4.4.c.1. Cystitis in pregnancy
Short courses of antimicrobial therapy can also be considered for treatment of cystitis in pregnancy [155], but not all antimicrobials are suitable during pregnancy. In general, penicillins, cephalosporins, fosfomycin, nitrofurantoin (not in case of glucose-6-phosphate dehydrogenase deficiency and during the end of pregnancy), trimethoprim (not in the first trimester), and sulphonamides (not in the last trimester) can be considered.
3.4.4.c.2. Renal insufficiency
In patients with renal insufficiency, the choice of antimicrobials may be influenced by decreased renal excretion. However, most antimicrobials have a wide therapeutic index. No adjustment of dose is necessary until the glomerular filtration rate (GFR) is < 20mL/min, with the exception of antimicrobials with nephrotoxic potential, e.g. aminoglycosides. The combination of loop diuretics (e.g. furosemide) and a cephalosporin is nephrotoxic. Nitrofurantoin is contraindicated in patients with an estimated GFR of less than 30mL/min/1.73m2, as accumulation of the drug leads to increased side effects, as well as reduced urinary tract recovery, with the risk of treatment failure [156].
3.4.4.d. Cystitis in men
As localised UTIs are rare in men, robust comparative trials are lacking. For empirical oral treatment of acute uncomplicated cystitis in younger men, pivmecillinam and nitrofurantoin are recommended. For nitrofurantoin, evidence-based data on the optimal treatment duration are lacking, and it should only be used if prostatic involvement has been reliably excluded [157].
3.4.4.e. Summary of evidence and recommendations for antimicrobial therapy for cystitis
| Summary of evidence | LE |
| Clinical success for the treatment of cystitis is significantly more likely in women treated with antimicrobials than placebo. | 1b |
| The use of nonsteroidal anti-inflammatory drugs and phytotherapy has resulted in a reduction of antibiotic therapy. Overall, across all studies, nonantibiotic therapy led to a 63% reduction in antibiotic use. | 1a |
| Aminopenicillins are no longer suitable for antimicrobial therapy in cystitis due to negative ecological effects, high resistance rates and their increased selection for extended spectrum beta-lactamase (ESBL)-producing bacteria. | 3 |
| Recommendations | Strength rating |
| Prescribe fosfomycin trometamol, pivmecillinam, nitrofurantoin or nitroxoline as first-line treatment for cystitis in women. | Strong |
| Do not use aminopenicillins or fluoroquinolones to treat cystitis. | Strong |
Table 3: Suggested regimens for antimicrobial therapy in cystitis
| Antimicrobial | Daily dose | Duration of therapy | Comments |
| First-line women | |||
| Fosfomycin trometamol | 3g SD | 1 day | |
| Nitrofurantoin microcrystal* | 50–100mg four times a day | 5 days | |
| Nitrofurantoin monohydrate/macrocrystals* | 100mg b.i.d | 5 days | |
| Nitrofurantoin macrocrystal prolonged release* | 100mg b.i.d | 5 days | |
| Pivmecillinam | 400mg t.i.d | 3–5 days | |
| Nitroxoline | 250mg t.i.d | 5 days | |
| Alternatives | |||
| Cephalosporins (e.g. cefadroxil) | 500mg b.i.d | 3 days | Or comparable |
| If the local resistance pattern for E. coli is < 20% | |||
| Trimethoprim | 200mg b.i.d | 5 days | Not in the first trimester of pregnancy |
| Trimethoprim-sulfamethoxazole | 160/800mg b.i.d | 3 days | Not in the last trimester of pregnancy |
| Treatment in men | |||
| Trimethoprim-sulfamethoxazole | 160/800mg b.i.d | 7 days | |
SD = single dose; b.i.d = twice daily; t.i.d = three times daily.
* Recommended in young men without involvement of the prostate (e.g. acute bacterial prostatitis), regarding the duration, there are no evidence-based data for nitrofurantoin.
3.4.5. Follow-up
Routine post-treatment urinalysis or urine cultures in asymptomatic patients are not indicated [23]. In women whose symptoms do not resolve by end of treatment and in those whose symptoms resolve but recur within two weeks, urine culture and antimicrobial susceptibility testing should be performed [158]. For therapy in this situation, one should assume that the infecting organism is not susceptible to the agent originally used. Retreatment with a seven-day regimen using another agent should be considered [158].
3.5. Recurrent cystitis
3.5.1. Introduction
Recurrent cystitis is defined by at least three episodes of cystitis/year or two episodes of cystitis in the last six months. Recurrent cystitis negatively impacts patient quality of life leading to a reduction in the quality of social and sexual relationships, self-esteem and capacity for work [159].
3.5.2. Diagnostic evaluation
Recurrent cystitis is common. Table 4 outlines the risk factors. Initial diagnosis of recurrent cystitis should be confirmed by urine culture. An extensive routine workup including cystoscopy, imaging and so on is not routinely recommended, because the diagnostic yield is low [160]. However, an extensive routing workup should be performed without delay in atypical cases, for example, if renal calculi, outflow obstruction, interstitial cystitis or urothelial cancer is suspected.
Table 4: Age-related associations of recurrent cystitis in women
| Young and premenopausal women | Postmenopausal and elderly women |
|
|
3.5.3. Disease management and follow-up
Prevention of recurrent cystitis includes counselling regarding avoidance of risk factors, nonantimicrobial measures and antimicrobial prophylaxis [158, 162]. These interventions should be attempted in this order. Any urological risk factor must be identified and treated. Significant residual urine should be treated optimally, including by CIC when judged to be appropriate.
3.5.3.a. Evidence Summary
A broad literature search with cut-off of 31 May 2021 identified 3,604 abstracts, of which 361 were selected for full text review. In a total of 114 systematic reviews or guidelines based on systematic literature searches and 131 original publications were selected for further analysis. A further 18 relevant publications were identified from the references of the reviewed studies. Selected studies were assigned to one of nine subgroups based on the method of prevention. An updated search with cutoff date of 1 June 2022 identified a further 316 abstracts, of which 25 were selected for further analysis. The evidence question addressed was: In women with recurrent symptomatic lower urinary tract infection, what interventions reduce the rate of recurrence?
3.5.3.b. Behavioural modifications
Women with recurrent cystitis should be counselled on avoidance of risks (e.g. insufficient hydration, habitual and post-coital delayed urination, wiping from back to front after defecation, douching and wearing occlusive underwear) before initiation of long-term prophylactic drug treatment, although there is limited evidence available regarding these approaches [163, 164]. An open-label RCT found that additional fluid intake of 1.5L per day in premenopausal women with recurrent cystitis who were low-volume drinkers (< 1.5L a day) reduced the number of cystitis episodes and antibiotic usage over a 12-month period [165].
3.5.3.c. Nonantimicrobial prophylaxis
3.5.3.c.1. Hormonal replacement
Based on the results of four meta-analyses, topical oestrogen therapy (either as a cream or a pessary) shows a trend towards recurrent cystitis prevention [166-169]. All studies reported that application was superior compared to placebo but was inferior compared to antibiotics. Due to its pharmacokinetics, vaginal admission has no systematic side effects. However, local irritation and minor bleeding can occur. The use of oral oestrogens was not effective for recurrent cystitis prophylaxis compared to placebo. Moreover, the use of oral oestrogens was associated with an unfavourable systematic side-effect profile. A single prospective, noncomparative study of 30 premenopausal women with recurrent cystitis on oral contraceptives reported a beneficial effect for additional topical oestrogen therapy [170].
3.5.3.c.2. Immunomodulation
A systematic review and meta-analysis evaluated the effectiveness of immunomodulation in preventing recurrent cystitis [171]. Fourteen comparative studies were included, with a total of 2,822 patients across five substance types (StroVac, OM-89, ExPEC4V, MV140, and Solco-Urovac). The pooled risk ratio from eight placebo-controlled studies on the percentage of patients remaining cystitis-free in the short term (6–12 months) was 1.52 (95% CI: 1.05–2.20), indicating that patients treated with immunomodulation were approximately 50% more likely to remain cystitis-free in the short term in comparison to placebo. The number needed to treat (NNT) was 6.45 (95% CI: 2.80–64.80). However, there was substantial heterogeneity and a risk of publication bias. A subgroup analysis by substance type suggests that short-term effectiveness in reducing recurrent cystitis compared to placebo varies depending on the agent. Current evidence indicates no significant difference between OM-89, Solco-Urovac and ExPEC4V for short-term cystitis prevention compared to placebo, because the risk ratios favoured the immunomodulatory agents but were not statistically significant. Data for MV140 compared to placebo show a risk ratio of 2.23 (95% CI: 1.43–3.47), the largest difference among all included studies, however, this finding is from a single RCT of 146 participants. Three additional cohort studies comparing MV140 with antibiotic therapy demonstrated a significantly higher percentage of cystitis-free patients in the immunomodulatory group.
The review also concluded that cystitis immunomodulatory agents are generally safe, with serious adverse events (AEs) being rare and no reports of AEs leading to death. A pooled analysis found no significant difference in AE incidence rates between immunomodulatory agents and placebo. The most frequently reported AEs were headaches, gastrointestinal disturbances and vaginitis. Immunomodulatory-specific side effects appear to be related to the agent used and method of administration.
Given the low certainty of the available data, the previous strong recommendation has been downgraded to weak. The composition and mechanism of action of individual immunomodulatory agents are sufficiently different, such that pooled analyses of results from these disparate trials is likely to result in excessive noise. The panel would therefore recommend further intensive study of the most promising agents in larger high-quality randomised clinical trials using standardised definitions of recurrent cystitis and other outcome measure.
3.5.3.c.3. Prophylaxis with probiotics (Lactobacillus spp.)
Five meta-analyses with differing results and eleven relevant systematic reviews were identified [167, 172-185]. Two meta-analyses reported significant positive effects for recurrent cystitis prevention with effective probiotics compared to placebo [176, 178]. The contradictory results of the four meta-analyses are a result of the analysis of various Lactobacillus strains and various administration regimes, treatment durations and patient populations. Most studies concluded that not all Lactobacillus strains are effective for vaginal flora restoration and recurrent cystitis prevention. The highest efficacy was shown with L. rhamnosus GR-1, L. reuteri B-54, L. reuteri RC-14, L. casei shirota and L. crispatus CTV-05 [167,174,176,178]. Although meta-analyses including all known Lactobacilli strains did not show a significant treatment benefit [167,174,176,178], sensitivity analysis excluding studies using ineffective strains resulted in a positive treatment effect [176].
Of the eleven systematic reviews, seven concluded that prophylaxis with vaginal probiotics has a beneficial clinical impact for the prevention of recurrent cystitis [168, 169, 172, 175, 177, 179-182, 184]. The available data is too minimal or of too low quality to allow the panel to make recommendations on the route of admission, optimal dosage and treatment duration for probiotic prophylaxis.
3.5.3.c.4. Prophylaxis with xyloglucan, hibiscus and propolis
Preclinical evidence supports the use of xyloglucan in managing recurrent urinary tract infections (rUTIs) by creating a barrier that prevents uropathogenic E. coli from adhering to epithelial cells, as demonstrated in models of intestinal and uroepithelial cells [135]. A systematic review and meta-analysis including three RCTs with data on 178 patients found that the combination of xyloglucan, hibiscus and propolis was effective in preventing recurrent cystitis compared to placebo, showing high patient-compliance and a reduction in antibiotic use [135]. A statistically significant difference was found in terms of clinical or microbiological resolution between the combination of xyloglucan, hibiscus and propolis and placebo (OR: 0.13; 95% CI: 0.05–0.33; p < 0.0001). Additionally, another review also highlighted the efficacy of this approach in managing recurrent cystitis [186].
3.5.3.c.5. Prophylaxis with Centaurii herba, Levistici radix and Rosmarini folium
An RCT involving 90 patients with cystitis compared antimicrobial therapy alone (control group) with antimicrobial therapy, plus a combination of Centaurii herba, Levistici radix and Rosmarini folium for three months (two tablets, three times daily). The frequency rate of recurrent episodes of cystitis in the test group was always lower than in the control group, with a statistically significant difference at six months (8.9% vs. 17.8%) and at 12 months (15.5% vs. 35.5%) [187]. This combination has also been tested in pregnant women, showing no teratogenic, embryotoxic effects or developmental defects in infants [188].
3.5.3.c.6. Prophylaxis with cranberry
Seven meta-analyses and several systematic reviews were identified [167, 189-194]. A Cochrane systematic review and meta-analysis found that, when compared with placebo, water or no treatment, cranberry products did not significantly reduce the occurrence of symptomatic cystitis overall or in women with recurrent cystitis [189]. However, six subsequent meta-analyses concluded that consumption of cranberry-containing products may protect against cystitis in certain patient populations [167, 190-194]. The differing outcomes across the meta-analyses can be contributed to the clinical and methodological heterogeneity of the included studies [195]. An RCT of 145 women randomised to high-dose versus low-dose cranberry proanthocyanidin extract reported no significant reduction in the number of symptomatic cystitis episodes between the groups [196]. In another RCT of 46 diabetic postmenopausal women, those randomised to receive 120mg of a highly standardised cranberry extract phytosome showed a significant reduction in cystitis recurrence compared to placebo [197]. Another RCT with 172 adult women with a history of recurrent cystitis comparing high-dose proanthocyanidins (240mg) to placebo reported that cranberry extract was associated with reduced cystitis and prolonged cystitis-free survival [198].
Regarding cranberry formulation, a systematic review indicated that cranberry juice combined with increased fluid intake may reduce cystitis rates and antibiotic use [199]. However, optimal dosing and treatment duration remain unclear. Nevertheless, another systematic review and meta-analysis of 18 RCTs and two non-RCTs with moderate to low certainty supports the use of cranberry products for symptom relief and reducing antibiotic usage [199]. This study also highlights the potential benefits of increased fluid intake in reducing cystitis rates, emphasising the advantage of cranberry in liquid form [199].
Although the efficacy of cranberry products remains unclear, clinicians may recommend them for recurrent cystitis prevention in women who are informed of the weak evidence base, due to their favourable benefit to harm ratio. However, there is no clear clinical evidence regarding the appropriate dose and treatment duration.
3.5.3.c.7. Prophylaxis with D-mannose
A meta-analysis including one RCT, one randomised cross-over trial and one prospective cohort study analysed data on 390 patients and found that D-mannose was effective for recurrent cystitis prevention compared to placebo with comparable efficacy to antibiotic prophylaxis [200]. Another systematic review concluded that D-mannose had a significant effect on cystitis, but that further studies were needed to confirm these findings [172]. A further systematic review including 695 patients reported that D-mannose improved quality of life and significantly reduced recurrent cystitis in both catheter and non-catheter users and was effective in reducing the incidence of recurrent cystitis and prolonging cystitis-free periods [201]. However, a Cochrane systematic review including 719 patients was unable to determine whether D-mannose significantly reduced the number of recurrent cystitis episodes when compared to no treatment, other supplements or antibiotics [202]. The overall evidence quality was low.
To determine whether D-mannose taken for six months reduces the proportion of women with recurrent cystitis experiencing a medically attended cystitis, a two-group (2g daily of D-mannose powder or matched volume of placebo powder), double-blind, randomised, placebo-controlled trial was performed [203]. The primary outcome measure was the proportion of women experiencing at least one further episode of clinically suspected cystitis for which they sought ambulatory care within six months of study entry. Secondary outcomes included symptom duration, antibiotic use, time to next medically attended cystitis, number of hospital admissions for suspected cystitis and cystitis-related issues. The proportion of participants contacting ambulatory care with a clinically suspected cystitis was 150 of 294 (51.0%) in the D-mannose group and 161 of 289 (55.7%) in the placebo group (risk difference: -5%; 95% CI: -13% to 3%; p = 0.26). Estimates were similar in per-protocol analyses, imputation analyses and preplanned subgroups. No statistically significant differences were observed in any secondary outcome measures. In this RCT, daily D-mannose did not reduce the proportion of women with recurrent cystitis in primary care who experienced a subsequent clinically suspected cystitis.
In the context of rising antimicrobial resistance, antibiotic-sparing strategies are increasingly essential for therapeutic and environmental benefits. Patients should be informed about these strategies, and urologists are encouraged to provide accurate information on the use of nutraceuticals in clinical practice, as supported by a broad body of research and data.
3.5.3.c.8. Endovesical instillation
Endovesical instillations of hyaluronic acid (HA) and chondroitin sulphate (CS) have been used for glycosaminoglycan (GAG) layer replenishment in the treatment of interstitial cystitis, overactive bladder, radiation cystitis and for prevention of recurrent cystitis [204]. A meta-analysis (n = 143) based on two RCTs and two non-RCTs found significantly decreased cystitis rates per patient/year (pooled mean difference [MD] -2.56; 95% CI: -3.86, -1.26; p < 0.001) and significantly longer mean cystitis recurrence times for HA and HA-CS therapy compared to control treatment (pooled MD 130.05 days; 95% CI: 5.84, 254.26; p = 0.04) [205]. The duration of treatment intervention ranged from two to six months and the duration of total follow-up ranged from 12 to 18 months. In addition, subgroup analysis of the two RCTs using HA-CS reported a significantly decreased cystitis rate per patient-year, significantly longer mean cystitis recurrence time and a significantly better pelvic pain and urgency/frequency (PUF) total score. However, 24-hour urinary frequency measured as number of voids in three days were not significantly improved after therapy [205].
Another meta-analysis (n = 800) including two RCTs and six non-RCTs found that, when compared to control treatment, HA, with or without CS, was associated with a significantly lower mean cystitis rate per patient-year and a significantly longer time to cystitis recurrence [206]. Furthermore, HA-CS therapy was associated with significantly greater mean reductions in PUF total and symptom scores, and the percentage of patients with cystitis recurrence during follow-up was also lower [206].
As randomised controlled studies are available only for HA plus CS, the quality of evidence is higher for the combination than for HA alone.
3.5.3.c.9. Methenamine hippurate
A Cochrane review from 2012 based on thirteen studies with high levels of heterogeneity concluded that methenamine hippurate may be effective for preventing cystitis in patients without renal tract abnormalities, particularly when used for short-term prophylaxis [207]. A meta-analysis from 2021 based on six studies found that, although studies showed a trend towards a benefit for methenamine hippurate in prevention of recurrent cystitis, no statistically significant difference was observed between the efficacy of methenamine hippurate and any comparators [208]. A subsequent RCT including 240 women randomised (1:1) to receive once-daily, low-dose antibiotic prophylaxis or twice-daily methenamine hippurate for twelve months reported that the incident rate of patient-reported symptomatic cystitis decreased to 1.38 episodes per person per year for the methenamine hippurate group versus 0.89 episodes per person per year for the antibiotic group. The absolute difference was 0.49, thus confirming that methenamine hippurate was not inferior to antibiotic prophylaxis. The rate of adverse events was similar in both groups and a sustained benefit for both treatment arms was observed at six-months follow-up [209, 210].
3.5.3.d. Antimicrobials for preventing recurrent cystitis
3.5.3.d.1. Continuous low-dose antimicrobial prophylaxis and postcoital prophylaxis
Four meta-analyses and numerous systematic reviews and guidelines were identified [169, 211-221]. All available meta-analyses conclude that antibiotic prophylaxis is the most effective approach against cystitis recurrences compared with placebo or no treatment [211-213]. Antimicrobials may be given as continuous low-dose prophylaxis for longer periods or as post-coital prophylaxis. No significant difference was observed in the efficacy of the two approaches. There is no available evidence about the optimal duration of continuous antimicrobial prophylaxis, with studies reporting treatment duration of three to twelve months. After discontinuation of the drug, cystitis tends to recur, especially among those who have had three or more infections annually. After counselling, it is mandatory to offer either continuous low-dose antimicrobial prophylaxis or postcoital prophylaxis when behavioural modifications and nonantimicrobial measures have failed. The choice of strategy should be individualised based on whether episodes are temporally related to intercourse and on patient preference.
Differences in outcomes between antibiotics did not reach statistical significance. The choice of agent should be based on the local resistance patterns. Regimens include nitrofurantoin 50mg or 100mg once daily, fosfomycin trometamol 3g once weekly, trimethoprim 100mg once daily and, during pregnancy, cephalexin 125mg or 250mg or cefaclor 250mg once daily [158, 222, 223]. Postcoital prophylaxis should be considered in pregnant women with a history of frequent cystitis before onset of pregnancy to reduce their risk of cystitis [224].
3.5.3.d.2. Self-diagnosis and self-treatment
In patients with good compliance, self-diagnosis and self-treatment with a short-course regimen of an antimicrobial agent should be considered [225]. The choice of antimicrobials is the same as for sporadic acute cystitis (Section 3.4.4.4).
3.5.4. Summary of evidence and recommendations for the diagnostic evaluation and treatment of recurrent cystitis
| Summary of evidence | LE |
| Extensive routine workup (e.g. cystoscopy, imaging) has a low diagnostic yield for the diagnosis of recurrent cystitis. | 3 |
| Increased water intake is an effective antimicrobial-sparing strategy to prevent recurrent cystitis in premenopausal women at high risk for recurrence who drink low volumes (< 1.5L) of fluids daily. | 3 |
| Vaginal oestrogen replacement has shown a trend towards preventing recurrent cystitis in postmenopausal women. | 1b |
| There is limited evidence to suggest that immunomodulatory agents are effective at reducing cystitis recurrence in adult female patients in the short term. | 1a |
| Of the currently available immunomodulatory agents, OM-89 and MV140 are the most widely studied, with MV140 showing the most promising results. | 1a |
| Probiotics containing L. rhamnosus GR-1, L. reuteri B-54 and RC-14, L. casei shirota or L. crispatus CTV-05 are effective for vaginal flora restoration and have shown a trend towards prevention of recurrent cystitis. | 1b |
| An RCT showed that Centaurii herba, Levistici radix and Rosmarini folium alone are effective in relieving acute cystitis symptoms compared to fosfomycin trometamol. | 1b |
| An RCT demonstrated the efficacy of L-methionine combined with Hibiscus sabdariffa and Boswellia serrata in relieving acute cystitis symptoms, as compared to fosfomycin trometamol. | 1b |
| A combination of xyloglucan, hibiscus and propolis is effective in relieving acute cystitis symptoms and preventing recurrence. | 1a |
| Centaurii herba, Levistici radix and Rosmarini folium are effective in preventing recurrence and reducing antibiotic use. | 1b |
| Highly standardised cranberry extract phytosome and high-dose proanthocyanidins appear effective in preventing recurrent cystitis. | 1b |
| Current scientific evidence regarding the efficacy of cranberry remedies, in combination with increased water intake, supports their use for the treatment of acute cystitis symptoms and the prevention of recurrence. | 1a |
| An RCT demonstrated the efficacy of D-mannose alone to relieve acute symptoms in acute cystitis compared to cotrimoxazole. | 1b |
| Based on limited evidence intravesical GAG therapy can reduce the number of cases of cystitis per patient per year and prolong the time interval between recurrent cystitis episodes. | 2 |
| An RCT demonstrated the noninferiority of twice daily methenamine hippurate to daily antibiotic prophylaxis. | 1b |
| Both continuous low-dose antimicrobial prophylaxis and postcoital antimicrobial prophylaxis have been shown to reduce the rate of recurrent cystitis. | 1b |
| A prospective cohort study showed that intermittent self-start therapy is effective, safe and economical in women with recurrent cystitis. | 2b |
| Recommendations | Strength rating |
| Diagnose recurrent cystitis by urine culture. | Strong |
| Do not perform an extensive routine workup (e.g. cystoscopy, full-abdominal ultrasound) in women younger than 40 years of age with recurrent cystitis and no risk factors. | Weak |
| Advise premenopausal women regarding increased fluid intake, as it might reduce the risk of recurrent cystitis. | Weak |
| Use vaginal oestrogen replacement in postmenopausal women to prevent recurrent cystitis. | Strong |
| Use immunomodulatory prophylaxis to reduce recurrent cystitis in women in the context of a well-regulated clinical trial. | Weak |
| Advise patients on the use of local or oral probiotics containing strains of proven efficacy for vaginal flora regeneration to prevent cystitis. | Weak |
| Advise patients on the use of a combination of xyloglucan, hibiscus and propolis, or Centaurii herba, Levistici radix and Rosmarini folium to reduce recurrent cystitis episodes and reduce antibiotic use. | Weak |
| Advise patients on the use of cranberry products, favouring juice, for symptom relief in acute cystitis and to prevent recurrence; however, patients should be informed that the quality of evidence underpinning this is low with contradictory findings. | Strong |
| Use D-mannose to reduce recurrent cystitis episodes, but patients should be informed of the overall weak and contradictory evidence of its effectiveness. | Weak |
| Use methenamine hippurate to reduce recurrent cystitis episodes in women without abnormalities of the urinary tract. | Strong |
| Use endovesical instillations of hyaluronic acid or a combination of hyaluronic acid and chondroitin sulphate to prevent recurrent cystitis in patients for which less-invasive preventive approaches have been unsuccessful. Patients should be informed that further studies are needed to confirm the results of initial trials. | Weak |
| Use continuous or postcoital antimicrobial prophylaxis to prevent recurrent cystitis when non-antimicrobial interventions have failed. Counsel patients regarding possible side effects. | Strong |
| Consider self-administered short-term antimicrobial therapy for patients with good compliance. | Strong |
3.6. Pyelonephritis
3.6.1. Diagnostic evaluation
3.6.1.a. Clinical diagnosis
Pyelonephritis is suggested in case of fever (> 38°C), chills, flank pain, nausea, vomiting or costovertebral angle tenderness, with or without the typical symptoms of cystitis [226]. Pregnant women with acute pyelonephritis require special attention, as this type of infection may not only have an adverse effect on the mother with anaemia, renal and respiratory insufficiency, but also on the unborn child with more frequent preterm labour and birth [227].
3.6.1.b. Differential diagnosis
It is vital to differentiate as soon as possible between pyelonephritis with or without risk factors, such as obstructive pyelonephritis, as the latter can rapidly lead to urosepsis. This differential diagnosis should be made using the appropriate imaging technique (see Section 3.6.1.d).
3.6.1.c. Laboratory diagnosis
Urinalysis, including the assessment of white and red blood cells and nitrite, is recommended for routine diagnosis [228]. In addition, urine culture and antimicrobial susceptibility testing should be performed in all cases of pyelonephritis.
3.6.1.d. Imaging diagnosis
Evaluation of the upper urinary tract with ultrasound (US) should be performed to rule out urinary tract obstruction or renal stone disease in patients with a history of urolithiasis, renal function disturbances or a high urine pH [229]. Additional investigations, such as a contrast-enhanced computed tomography (CT) scan or excretory urography, should be considered if the patient remains febrile after 72 hours of treatment, or immediately if there is deterioration in clinical status [229]. For diagnosis of complicating factors in pregnant women, US or magnetic resonance imaging (MRI) should be used preferentially to avoid radiation risk to the foetus [229].
3.6.2. Summary of evidence and recommendations for the diagnostic evaluation of pyelonephritis
| Summary of evidence | LE |
| Urine culture and antimicrobial susceptibility testing should be performed in all cases of pyelonephritis in addition to urinalysis. | 4 |
| A prospective observational cohort study found that radiologic imaging can be applied selectively in adults with systemic UTI without loss of clinically relevant information by using a simple clinical prediction rule. | 2b |
| Additional imaging investigations, such as an unenhanced helical computed tomography, should be carried out if the patient remains febrile after 48–72 hours of treatment or in patients with suspected complications, such as sepsis. | 4 |
| Recommendations | Strength rating |
| Perform urinalysis (e.g. using the dipstick method), including the assessment of white and red blood cells and nitrite, for routine diagnosis. | Strong |
| Perform urine culture and antimicrobial susceptibility testing in patients with pyelonephritis. | Strong |
| Perform imaging of the urinary tract to exclude urgent urological disorders. | Strong |
3.6.3. Disease management
3.6.3.a. Outpatient treatment
Fluoroquinolones and cephalosporines are the only antimicrobial agents that can be recommended for oral empirical treatment of pyelonephritis [230]. However, oral cephalosporines achieve significantly lower blood and urinary concentrations than intravenous cephalosporines. Other agents, such as nitrofurantoin, oral fosfomycin and pivmecillinam, should be avoided as there is insufficient data regarding their efficacy [231]. In the setting of fluoroquinolone hypersensitivity or known resistance, other acceptable choices include trimethoprim-sulfamethoxazole (160/800mg) or an oral beta-lactam, if the uropathogen is known to be susceptible. If such agents are used in the absence of antimicrobial susceptibility results, an initial intravenous dose of a long-acting parenteral antimicrobial (e.g. ceftriaxone) should be administered. For acute pyelonephritis, a short outpatient antibiotic course of treatment has been shown to be equivalent to longer durations of therapy in terms of clinical and microbiological success. However, this is associated with a higher recurrence rate of infection within four to six weeks and needs to be tailored to local policies and resistance patterns [232].
3.6.3.b. Inpatient treatment
Patients with pyelonephritis requiring hospitalisation should be treated initially with an intravenous antimicrobial regimen, e.g. a fluoroquinolone, an aminoglycoside (with or without ampicillin) or an extended-spectrum cephalosporin or penicillin/tazobactam [233]. Ceftolozane/tazobactam achieved a clinical response rate of over 90% in patients with pyelonephritis [234, 235]. Ceftolozane/tazobactam also demonstrated significantly higher composite cure rates than levofloxacin among levofloxacin-resistant pathogens [236]. Ceftazidime-avibactam combination therapy has been shown to be effective for treating ceftazidime-resistant Enterobacterales and Pseudomonas aeruginosa UTIs [237].
Novel antimicrobial agents include imipenem/cilastatin, cefiderocol, meropenem-vaborbactam and plazomicin. Imipenem/cilastatin has been investigated in a phase II randomised trial and showed good clinical response rates [238]. Cefatazidime-avibactam and doripenem showed similar efficacy against ceftazidime nonsusceptible pathogens and may offer an alternative to carbapenems in this setting [239]. Meropenem-vaborbactam has been shown to be noninferior to piperacillin-tazobactam in a phase III RCT [240]. Meropenem-vaborbactam was also effective for treating carbapenem-resistant Enterobacterales, with cure rates of 65% compared to best available treatment [241]. Once-daily plazomicin was noninferior to meropenem for the treatment of complicated UTIs and acute pyelonephritis caused by Enterobacterales, including multidrug-resistant strains [242]. Cefiderocol was noninferior to imipenem/cilastatin for the treatment of complicated UTI in people with multidrug-resistant, Gram-negative infections in a phase II RCT [243].
Carbapenems and novel broad spectrum antimicrobial agents should only be considered in patients with early culture results indicating the presence of multidrug-resistant organisms. The choice between these agents should be based on local resistance patterns and optimised on the basis of drug susceptibility results. In patients presenting with signs of urosepsis, empiric antimicrobial coverage for ESBL-producing organisms is warranted [244]. Patients initially treated with parenteral therapy who improve clinically and can tolerate oral fluids may transition to oral antimicrobial therapy [245].
3.6.3.c. Summary of evidence and recommendations for the treatment of pyelonephritis
| Summary of evidence | LE |
| Fluoroquinolones and cephalosporines are the only microbial agents that can be recommended for oral empirical treatment of pyelonephritis. | 1b |
| Intravenous antimicrobial regimens for pyelonephritis may include a fluoroquinolone, an aminoglycoside (with or without ampicillin) or an extended-spectrum cephalosporin or penicillin. | 1b |
| Carbapenems should only be considered in patients with early culture results indicating the presence of multidrug-resistant organisms. | 4 |
| The appropriate antimicrobial should be chosen based on local resistance patterns and optimised on the basis of drug susceptibility results. | 3 |
| Recommendations | Strength rating |
| Treat patients with pyelonephritis not requiring hospitalisation with short-course fluoroquinolones as first-line treatment. | Strong |
| Treat patients with pyelonephritis requiring hospitalisation with an intravenous antimicrobial regimen initially. | Strong |
| Switch patients initially treated with parenteral therapy who improve clinically and can tolerate oral fluids to oral antimicrobial therapy. | Strong |
| Do not use nitrofurantoin, oral fosfomycin and pivmecillinam to treat pyelonephritis. | Strong |
Table 5: Suggested regimens for empirical oral antimicrobial therapy in pyelonephritis
| Antimicrobial | Daily dose | Duration of therapy | Comments |
| Ciprofloxacin | 500–750mg b.i.d | 7 days | Fluoroquinolone resistance should be less than 10%. |
| Levofloxacin | Standard dosage: 500mg oral q.d High dosage: 500mg oral b.i.d | 5 days | |
| Trimethoprim sulfamethoxazole | 160/800mg b.i.d | 14 days | If such agents are used empirically, an initial intravenous dose of a long-acting parenteral antimicrobial (e.g. ceftriaxone) should be administered. |
| Cefpodoxime | 200mg b.i.d | 10 days | |
| Ceftibuten | 400mg q.d | 10 days |
b.i.d = twice daily; q.d = every day.
Table 6: Suggested regimens for empirical parenteral antimicrobial therapy in pyelonephritis
| Antimicrobials | Daily dose | Comments |
| First-line treatment | ||
| Ciprofloxacin | 400 mg b.i.d | |
| Levofloxacin | Standard dosage: 500mg oral q.d High dosage: 500mg oral b.i.d | |
| Cefotaxime | 2g t.i.d | Not studied as monotherapy in acute pyelonephritis. |
| Ceftriaxone | Standard dosage: 2g IV q.d High dosage: 2g IV b.i.d | Lower dose studied, but higher dose recommended. |
| Second-line treatment | ||
| Cefepime | Standard dosage: 1g IV t.i.d or 2g IV b.i.d High dosage: 2g IV t.i.d | Lower dose studied, but higher dose recommended. |
| Piperacillin/tazobactam | Standard dosage: 4.5g t.i.d High dosage: 4.5g q.i.d prolonged infusion | |
| Gentamicin | 6–7mg/kg q.d | Not studied as monotherapy in acute pyelonephritis. |
| Amikacin | 25–30mg/kg q.d | |
| Last-line alternatives | ||
| Imipenem/cilastatin | Standard dosage: 0.5g IV q.i.d over 30 minutes High dosage: 1g IV q.i.d over 30 minutes | Consider only in patients with early culture results indicating the presence of multidrug-resistant organisms. |
| Meropenem | 1g t.i.d | |
| Ceftolozane/tazobactam | 1.5g t.i.d | |
| Ceftazidime/avibactam | 2.5g t.i.d | |
| Cefiderocol | 2g t.i.d | |
| Meropenem-vaborbactam | 2g t.i.d | |
| Plazomicin | 15mg/kg o.d | |
b.i.d = twice daily; IV = intravenous; t.i.d = three times daily; q.d = every day; q.i.d = four times daily; o.d = once daily.
In pregnant women with pyelonephritis, outpatient management with appropriate parenteral antimicrobials may also be considered, provided symptoms are mild and close follow-up is feasible [246, 247]. In more severe cases of pyelonephritis, hospitalisation and supportive care are usually required. After clinical improvement parenteral therapy can also be switched to oral therapy for a total treatment duration of seven to ten days. In men with febrile UTI, pyelonephritis or recurrent infection, or whenever a complicating factor is suspected, a minimum treatment duration of two weeks is recommended, preferably with a fluoroquinolone, since prostatic involvement is frequent [248].
3.6.4. Follow-up
Post-treatment urinalysis or urine cultures in asymptomatic patients post-therapy are not indicated.
3.7. Systemic urinary tract infections
3.7.1. Introduction
Systemic UTIs are infections that originate from various organs of the urinary tract. In contrast to localised UTIs (i.e. cystitis), these infections typically present with systemic signs and symptoms of infection (see Table 1). Local signs and symptoms of infection may also be present. Examples of systemic UTI include pyelonephritis, acute prostatitis and urosepsis. Catheter-associated UTIs might present as localised as well as systemic infections.
3.7.2. Diagnostic evaluation
3.7.2.a. Clinical presentation
Systemic UTI should be suspected in patients presenting with dysuria, urinary frequency, urgency or suprapubic pain, accompanied by fever, chills, flank pain or pelvic/perineal pain, or when patients appear clinically unwell. Systemic UTI may also be considered in patients with unexplained fever or sepsis. Evaluation should include a thorough clinical assessment to rule out other potential causes of illness. Physical examination should assess for costovertebral angle tenderness and abdominal or suprapubic tenderness. In females, a pelvic examination may be necessary – particularly when symptoms do not clearly suggest a UTI – to evaluate for cervical motion tenderness or uterine tenderness, which could indicate pelvic inflammatory or sexually transmitted disease. In biological men presenting with pelvic or perineal pain, a digital rectal examination is essential to assess for tenderness or swelling of the prostate, which may suggest acute prostatitis.
3.7.2.b. Urinalysis and urine culture
For patients with suspected systemic UTI, urine should be collected for both urinalysis and culture with antimicrobial susceptibility testing.
3.7.2.c. Pregnancy testing
Pregnancy testing is recommended for women of childbearing potential when pregnancy cannot be reasonably ruled out based solely on medical history.
3.7.2.d. Routine blood tests and blood cultures
Routine blood tests should always be considered in patients presenting with systemic signs and symptoms of infection. Blood cultures are indicated for patients presenting with sepsis or severe illness or when Staphylococcus aureus is isolated from the urine. S. aureus in urine is a ‘marker’ of S. aureus blood stream infections (SABSI). Patients whose urine tests positive for SABSI have a higher mortality than those without bacteriuria [249] as well as higher rates of recurrence after antibiotic cessation [250].
3.7.2.e. Imaging
In patients with systemic UTIs, the primary goal of imaging is to identify underlying conditions that may delay therapeutic response or require intervention (e.g. urinary retention, hydronephrosis and so on) as well as to diagnose potential complications of infection (e.g. renal or perinephric abscess or prostatic abscess). Initial imaging should be performed by ultrasound. In case of hydronephrosis or if obstruction of the upper urinary tract is suspected, cross-sectional imaging (CT, MRI) should be carried out. In severely ill patients and in patients exhibiting persistent clinical symptoms despite 48 to 72 hours of appropriate antimicrobial therapy, cross-sectional imaging (CT, MRI) should be performed as well [251, 252].
3.7.3. General principles of systemic UTI treatment
Empiric antimicrobial therapy should be initiated without delay, with subsequent modifications based on antimicrobial susceptibility testing results. Identified or suspected anatomical abnormalities should be thoroughly evaluated and managed as indicated.
3.7.3.a. Choice of antimicrobials
3.7.3.a.1. Empiric antimicrobial therapy
Empiric therapy for systemic UTIs should consider illness severity, resistance risk factors and patient-specific factors [231]. Treatment selection depends on prior urinary isolate susceptibility, patient characteristics (e.g. allergies, tolerability, prior antimicrobial use), local resistance patterns and drug-related factors such as toxicity, interactions, availability and costs. Upon identifying the pathogen’s susceptibility profile, the empiric regimen should be adjusted accordingly. Evidence for various systemic UTI regimens is limited, with few options formally evaluated [253, 254].
3.7.3.a.2. Critical illness and/or urinary tract obstruction
In critically ill patients with systemic UTIs, patients worsening on current therapy or patients with suspected urinary tract obstruction, broad-spectrum antimicrobials are recommended. A carbapenem, for example, imipenem (500mg IV every 6 hours) or meropenem (1g IV every 8 hours), is advised to cover ESBL-producing organisms and Pseudomonas aeruginosa. Vancomycin or alternatives (daptomycin, linezolid) should be added for MRSA coverage. Broad-spectrum coverage is crucial in critically ill or obstructed cases due to high adverse outcome risks and increasing multidrug-resistant pathogens. If cultures confirm susceptibility, regimens should be narrowed. Advanced options such as beta-lactamase-inhibitor combinations - cefiderocol, plazomicin and parenteral fosfomycin, where available - target some ESBL-producing Enterobacterales and certain multidrug-resistant P. aeruginosa strains [237, 240, 255-257]. However, given the cost, antibiotic stewardship concerns and limited data, the use of these advanced options is reserved for highly resistant cases. Urine culture and susceptibility results should guide both confirmation and refinement of therapy, prioritising narrow-spectrum agents when possible.
3.7.3.a.3. Outpatients
Outpatient treatment may be appropriate for patients with acute systemic UTIs of mild to moderate severity who can reliably take oral medications. The choice of empiric antimicrobials should consider the risk of multidrug-resistant organisms, particularly ESBL producers. Fluoroquinolones are a key option, offering broad activity, including against P. aeruginosa, and high urinary concentrations. Despite rising resistance rates, fluoroquinolones, when suitable, remain effective; ciprofloxacin and levofloxacin are preferred.
3.7.3.a.4. Directed antimicrobial therapy
Regimen selection
Urine culture and antimicrobial susceptibility results should guide tailoring of the antimicrobial regimen. Broad-spectrum empiric treatments can often be replaced with narrower-spectrum agents. Patients initially on parenteral therapy may transition to oral agents once symptoms improve, provided culture results support the switch.
Duration
Antimicrobial therapy duration generally ranges from five to ten days, based on clinical response and selected agent. In general, among hospitalised patients with bloodstream infection, antibiotic treatment for seven days is noninferior to treatment for 14 days [258]. For patients with symptomatic improvement within 48–72 hours, recommended durations are five to seven days for fluoroquinolones, seven to ten days for trimethoprim-sulfamethoxazole [233] and seven to ten days for beta-lactams. If clinical response is slow (no improvement within 48–72 hours), further evaluation for complications or anatomical abnormalities is advised [245, 259]. Extended therapy may be needed for persistent infection sources, such as a nonremovable stone. Routine extension of therapy duration is unnecessary in cases of uncomplicated bacteremia without complicating factors, because bacteraemia alone does not worsen prognosis, except for S. aureus bacteraemia, addressed separately. Systematic reviews and meta-analyses show similar cure rates with shorter courses (≤ 7 days) for systemic UTIs, mostly based on fluoroquinolone trials comparing five- or seven-day regimens to longer treatments [260-262]. However, in males with systemic UTIs, seven days of antibiotic treatment is inferior to 14 days [262, 263]. Limited data exist for other antibiotics, but beta-lactam studies, primarily with older agents, showed no clear benefit of extending treatment beyond ten days [259, 264].
3.7.3.a.5. Addressing risk factors
In addition to antimicrobial therapy, urinary obstruction should be assessed and managed if present. Patients with anatomical or functional urinary tract abnormalities (e.g. neurogenic lower urinary tract dysfunction, indwelling transurethral catheters, nephrostomy tubes, ureteral stents) may need additional interventions. Effective treatment may require addressing these underlying conditions, because antimicrobials alone may be insufficient [265].
3.7.3.a.6. Follow-up
Symptoms should improve quickly with effective antimicrobial therapy. Outpatients with pyelonephritis require follow-up, either in person or by phone, within 48–72 hours. Further evaluation is advised for patients with worsening symptoms post-initiation of antimicrobial therapy, persistent symptoms after 48–72 hours of appropriate therapy, or recurrence within weeks. This may include abdominal or pelvic imaging (typically cross-sectional imaging) to identify factors delaying recovery. Urine culture and antimicrobial susceptibility testing should also be repeated and therapy adjusted as needed. For those presenting with haematuria, a follow-up urinalysis is recommended several weeks post-treatment to check for persistence.
3.7.4. Summary of evidence and recommendations for the treatment of systemic UTIs
| Summary of evidence | LE |
| Patients with a UTI with systemic symptoms requiring hospitalisation should be initially treated with an intravenous antimicrobial regimen chosen based on local resistance data and previous urine culture results from the patient. The regimen should be tailored on the basis of susceptibility result. | 1b |
| If the prevalence of fluoroquinolone resistance is thought to be < 10% and the patient has contraindications for third-generation cephalosporins or an aminoglycoside, ciprofloxacin can be prescribed as an empirical treatment. | 2 |
| In the event of hypersensitivity to penicillin a cephalosporins can still be prescribed, unless the patient has had systemic anaphylaxis in the past. | 2 |
| In patients with a systemic UTI, empirical treatment should cover ESBL if there is an increased likelihood of ESBL infection based on prevalence in the community, earlier urine cultures and prior antimicrobial exposure of the patient. | 2 |
| Recommendations | Strength rating |
Use the following antimicrobials as empirical intravenous treatment for systemic UTI:
| Strong |
Use ciprofloxacin, provided that:
| Strong |
| Do not use ciprofloxacin or other fluoroquinolones for the empirical treatment of systemic UTI in patients from urology departments or when patients have used fluoroquinolones in the last six months. | Strong |
| Manage any urological abnormality and/or underlying complicating factors. | Strong |
3.8. Catheter-associated UTIs
3.8.1. Introduction
Catheter-associated UTI refers to UTIs occurring in a person whose urinary tract is currently catheterised or has been catheterised within the past 48 hours. The urinary catheter literature is problematic, as many published studies use the term CA-bacteriuria without providing information on what proportion are CA-ABU and CA-UTI, and some studies use the term CA-UTI when referring to CA-ABU or CA-bacteriuria [28].
3.8.2. Epidemiology, aetiology and pathophysiology
Catheter-associated UTIs are the leading cause of secondary healthcare-associated bacteraemia. Approximately 20% of hospital-acquired bacteraemias arise from the urinary tract, and the mortality associated with this condition is approximately 10% [266]. A multistate point-prevalence survey of 11,282 patients across 183 hospitals reported that UTI accounted for 12.9% of healthcare-acquired infections [267]. The incidence of bacteriuria associated with indwelling catheterisation is 3–8% per day [268-272]. The duration of catheterisation is the most important risk factor for the development of a CA-UTI [273, 274]. A systematic review and meta-analysis reported an average CA-UTI incidence of 13.79/1,000 hospitalised patients with a prevalence of 9.33% [275]. This study also demonstrated that patients at high risk for CA-UTI were female, had a prolonged duration of catheterisation, had diabetes and had longer hospital and intensive care unit (ICU) stays [275].
Urinary catheterisation perturbs host defence mechanisms and provides easier access of uropathogens to the bladder. Indwelling urinary catheters facilitate colonisation with uropathogens by providing a surface for the attachment of host-cell-binding receptors recognised by bacterial adhesins, thus enhancing microbial adhesion. In addition, the uroepithelial mucosa is damaged, exposing new binding sites for bacterial adhesins, and residual urine in the bladder is increased through pooling below the catheter bulb [276]. Catheter-associated UTIs are often polymicrobial and caused by multidrug-resistant uropathogens.
3.8.3. Diagnostic evaluation
3.8.3.a. Clinical diagnosis
Signs and systemic symptoms compatible with CA-UTI include new onset or worsening of fever, rigors, altered mental status, malaise or lethargy with no other identified cause, as well as flank pain, costovertebral angle tenderness, acute haematuria, pelvic discomfort, and in those whose catheters have been removed, dysuria, urgent or frequent urination, and suprapubic pain or tenderness [277]. In the catheterised patient, the presence or absence of odorous or cloudy urine alone should not be used to differentiate CA-ABU from CA-UTI [28, 277].
3.8.3.b. Laboratory diagnosis
Microbiologically, CA-UTI is defined by microbial growth of ≥ 103 CFU/mL of one or more bacterial species in a single catheter urine specimen or in a midstream voided urine specimen from a patient whose urethral, suprapubic or condom catheter has been removed within the previous 48 hours [28]. In catheterised patients, pyuria is not diagnostic for CA-UTI. The presence, absence or degree of pyuria should not be used to differentiate CA-ABU from CA-UTI. Pyuria accompanying CA-ABU should not be interpreted as an indication for antimicrobial treatment. The absence of pyuria in a symptomatic patient suggests a diagnosis other than CA-UTI [28].
3.8.3.c. Summary of evidence table and recommendations for diagnostic evaluation of CA-UTI
| Summary of evidence | LE |
| Patients with indwelling or suprapubic catheters become carriers of ABU, with antibiotic treatment showing no benefit. | 1a |
| In the catheterised patient, the presence or absence of odorous or cloudy urine alone should not be used to differentiate CA-ABU from CA-UTI. | 2 |
| Microbiologically, CA-UTI is defined by microbial growth of ≥ 103 CFU/mL of one or more bacterial species in a single catheter urine specimen or in a mid-stream voided urine specimen from a patient whose catheter has been removed within the previous 48 hours. | 3 |
| Recommendations | Strength rating |
| Do not carry out routine urine culture in asymptomatic catheterised patients. | Strong |
| Do not use pyuria as sole indicator for catheter-associated UTI. | Strong |
| Do not use the presence or absence of odorous or cloudy urine alone to differentiate catheter-associated asymptomatic bacteriuria from catheter-associated UTI. | Strong |
3.8.4. Disease management
3.8.4.a. Limiting catheterisation and appropriate catheter discontinuation
Indwelling catheters should be placed only when they are clinically indicated, for example, for management of urinary retention or where strict monitoring of fluid balance is required. Catheter restriction protocols are an important part of multimodal interventions to reduce CA-UTI rates. Nurse-driven protocols in hospitals, as well as community-based multimodal targeted infection programmes, have been proven to reduce CA-UTI rates [278, 279]. Adjunctive devices such as electronic reminder systems have also been shown to assist in prompt catheter removal in hospital settings (including non-ICU). A systematic review of nineteen different interventions to reduce UTI in nursing home patients (including catheter discontinuation and limiting catheterisation) reported successful CA-UTI reduction and reduced catheter usage [280]. Another report of over 2,800 patients on a surgical oncology unit found that increasing catheter bundle compliance resulted in a significant reduction in CA-UTI rates [281].
3.8.4.b. Urethral cleaning and chlorhexidine bathing
A network meta-analysis of 33 studies (6,490 patients) found no difference in the incidence of CA-UTI comparing the various urethral cleaning methods versus disinfection [282]. The efficacy of chlorhexidine baths (either using 2% chlorhexidine-impregnated cloths or 4% chlorhexidine-based soap) in reducing CA-UTI is debatable. In an RCT of 10,783 ICU patients, no difference in CA-UTI rates were reported between chlorhexidine and control bathing groups [283]. However, a systematic review of fifteen studies involving only ICU patients reported that daily chlorhexidine bathing was associated with a significant reduction in CA-UTI (RR 0.68) [284].
3.8.4.c. Alternatives to indwelling urethral catheterisation
Alternatives include intermittent urethral catheterisation (IC) or suprapubic catheterisation. In a systematic review of patients undergoing gynaecological surgery, indwelling catheters were associated with higher rates of symptomatic UTIs compared to IC [285]. A further meta-analysis of postpartum women reported no difference in the incidence of UTI after labour between continuous catheterisation and IC [285]. A prospective cohort study of nursing home residents found that residents with a suprapubic catheter had fewer CA-UTIs and were hospitalised less, but were more likely to be colonised with multidrug-resistant organisms [286].
A Cochrane review found insufficient evidence to assess the value of various policies for replacing long-term urinary catheters on patient outcomes [97]. Another Cochrane review investigating the role of urethral (indwelling or intermittent) versus suprapubic catheterisation in the short-term found inconclusive evidence of an effect on UTI rates [287]. For patients with NLUTD, a further systematic review found no randomised or quasi-randomised controlled trials and therefore no conclusions regarding the use of the various types of catheters could be made [288]. Therefore, based on the available literature, while some limited studies show a benefit of IC or suprapubic catheterisation over urethral catheterisation for CA-UTI rates, insufficient evidence is available to recommend those approaches routinely [289].
3.8.4.d. Impregnated or coated catheters
Hydrophilic coated catheters have been found to be beneficial for reducing CA-UTI rates. A meta-analysis of seven studies investigating RCTs comparing hydrophilic coated to PVC (standard) catheters for IC found a statistically lower risk ratio (0.84) for the frequency of UTI in the hydrophilic catheter group [290]. A systematic review and practice policy statements on UTI prevention in patients with spina bifida recommended the use of single-use and hydrophilic catheters for IC [291].
Silver-alloy-impregnated catheters have not been associated with reduced CA-UTI rates. A small RCT of 54 ICU patients showed no significant difference in UTI rates between the silver-alloy-impregnated group and the standard silicone-foley-catheter group [292]. In a cohort study of patients undergoing suprapubic catheter placement at the time of pelvic organ prolapsed surgery, a 5% difference in UTI rate at six weeks was noted, although this was not significant [293]. A systematic review of 26 trials (12,422 patients) reported that silver-alloy-coated catheters were not associated with a statistically significant reduction in CA-UTI and were considerably more expensive [294]. However, the same study found that nitrofurazone-impregnated catheters reduce the risk of symptomatic CA-UTI, although this was borderline significant (RR 0.84, 95% CI: 0.71–0.99) [294]. A more recent RCT (214 patients) evaluating the use of nitrofurazone-infused catheters post-renal transplant found no benefit for their use [295]. Additionally, another RCT showed no benefit for the use of silver-alloy-coated indwelling catheters for reduction of UTI in 489 patients with spinal cord injury [296].
From a microbiological perspective, there may be a difference in organisms causing CA-UTI from urethral and suprapubic catheters and therefore urine culture results are important to guide therapy [289].
3.8.4.e. Antibiotic prophylaxis for catheter removal or insertion
The issue of whether antibiotic prophylaxis reduce the rate of symptomatic UTI in adults following indwelling bladder catheter removal has been the subject of multiple RCTs. A review and meta-analysis identified seven RCTs with 1,520 participants. Meta-analysis showed overall benefit for use of prophylaxis RR (95% CI) = 0.45 (0.28–0.72); ARR 5.8% (10.5–4.7%) with a number needed to treat (NNT) of 17 [214]. Results for individual trials were inconsistent, with five trials including the possibility of no benefit [214]. In an affectional RCT with 172 participants undergoing laparoscopic radical prostatectomy randomised to seven days of ciprofloxacin (n = 80) or no treatment (n = 80) at the time of catheter removal, which occurred at a mean of nine days postoperatively, there was no difference in infective complications recorded at up to four weeks after catheter removal. More isolates obtained from the prophylaxis group (11) were resistant to ciprofloxacin compared to the no-treatment group (3) [215]. With regards to catheter insertion, a systematic review and meta-analysis showed that prophylactic antibiotics reduced the rate of bacteriuria and other signs of infection, such as pyuria, fever and gram‐negative isolates in patients’ urine in surgical patients who undergo bladder drainage for at least 24 hours postoperatively [297].
3.8.4.f. Antibiotic prophylaxis for intermittent self-catheterisation (ISC)
An RCT investigating the effect of antibiotic prophylaxis in patients performing ISC showed that the frequency of symptomatic antibiotic-treated UTI was reduced by 48% using prophylaxis in a cohort of 404 patients performing ISC [298]. However, resistance against the antibiotics used for UTI treatment was more frequent in urinary isolates from the prophylaxis group than in those from the control group at 9–12 months.
While the literature shows some benefit for reduction of CA-UTI by utilising antibiotics, the routine use of antibiotics for such a common procedure in the healthcare setting would result in an increased usage of antimicrobials. As highlighted in some of the RCTs, this strategy is associated with increased antimicrobial resistance. Antibiotic use is the main driving force in the development of antimicrobial resistance. Current antimicrobial stewardship principles would not favour the routine use of antibiotic prophylaxis for either catheter changes or ISC, even when UTIs could be prevented [289].
3.8.4.g. Antimicrobial treatment for suspected CAUTI
A urine specimen for culture should be obtained prior to initiating antimicrobial therapy for presumed CA-UTI due to the wide spectrum of potential infecting organisms and the increased likelihood of antimicrobial resistance. The urine culture should be obtained from the freshly placed catheter prior to the initiation of antimicrobial therapy [28]. Based on the global prevalence on infections in urology (GPIU) study, the causative microorganisms in CA-UTI are comparable with the causative microorganisms in other cUTIs. Symptomatic CA-UTIs should therefore be treated according to the recommendations for localised and systemic UTIs [299].
Seven days is the recommended duration of antimicrobial treatment for patients with CA-UTI who have prompt resolution of symptoms, and fourteen days of treatment is recommended for those with a delayed response, regardless of whether or not the patient remains catheterised [28]. A five-day regimen of levofloxacin may be considered in patients with CA-UTI who are not severely ill. Data are insufficient to make such a recommendation regarding other fluoroquinolones. With the rise in fluoroquinolone resistance, alternative antimicrobial agents should be selected where possible to start empirical therapy based on local microbiological information. A five-day antibiotic regimen with catheter exchange has been shown in one study to be non-inferior to a 10-day regimen with catheter retention based on clinical cure [300].
A three-day antimicrobial regimen may be considered for women aged ≤ 65 years who develop CA-UTI without upper urinary tract symptoms after an indwelling catheter has been removed. If an indwelling catheter has been in place for two weeks at the onset of CA-UTI and is still indicated, the catheter should be replaced to hasten resolution of symptoms and to reduce the risk of subsequent CA-bacteriuria and CA-UTI. If use of the catheter can be discontinued, a culture of a voided midstream urine specimen should be obtained prior to the initiation of antimicrobial therapy to help guide treatment [28]. Long-term indwelling catheters should not be changed routinely. Follow appropriate practices for catheter insertion and care [301].
3.8.4.h. Recommendations for disease management and prevention of CA-UTI
| Summary of evidence | LE |
| A systematic review of nineteen different interventions to reduce UTI including catheter discontinuation and limiting catheterisation in nursing home patients reported successful CA-UTI reduction and reduced catheter usage. | 1b |
| A meta-analysis of seven studies investigating RCTs comparing hydrophilic coated to PVC (standard) catheters for IC found a statistically lower risk ratio (0.84) for the frequency of UTI in the hydrophilic catheter group. | 1a |
| A meta-analysis showed overall benefit for use of prophylaxis for reduction of infective complications after catheter removal. However, results from individual trials were inconsistent with five out of seven trials, including the possibility of no benefit. | 1a |
| A subsequent RCT found no benefit of antibiotic prophylaxis for reduction of infective complications at up to four weeks after catheter removal. | 1b |
| Recommendations | Strength rating |
| Treat symptomatic catheter-associated UTI according to the recommendations for localised and systemic UTI. | Strong |
| Take a urine culture prior to initiating antimicrobial therapy in catheterised patients in whom the catheter has been removed. | Strong |
| Do not treat catheter-associated asymptomatic bacteriuria in general. | Strong |
| Treat catheter-associated asymptomatic bacteriuria prior to traumatic urinary tract interventions (e.g. transurethral resection of the prostate). | Strong |
| Replace or remove the indwelling catheter before starting antimicrobial therapy. | Strong |
| Do not apply topical antiseptics or antimicrobials to the catheter, urethra or meatus. | Strong |
| Do not use prophylactic antimicrobials to prevent catheter-associated UTIs. | Strong |
| Do not routinely use antibiotic prophylaxis to prevent clinical UTI after urethral catheter removal. | Weak |
| The duration of catheterisation should be minimal. | Strong |
| Use hydrophilic-coated catheters to reduce catheter-associated UTIs. | Strong |
| Do not routinely use antibiotic prophylaxis to prevent clinical UTI after urethral catheter removal or in patients performing intermittent self-catheterisation. | Weak |
3.9. Urosepsis
Section 3.9 is currently under review, and an updated version will be published in the 2027 edition of the Guidelines. In the interim, users are advised to refer to the Surviving Sepsis Campaign Guidelines 2021 from the Society of Critical Care Medicine [302].
3.10. Urethritis
3.10.1. Introduction
Urethritis can be of either infectious or non-infectious origin. Inflammation of the urethra usually presents with subjective symptoms (dysuria, alguria, burning, itching and pain around the distal urethra and external urethral meatus) and clinical signs (urethral discharge, erythema around the external urethral meatus and inguinal lymphadenopathy). These symptoms and signs must be distinguished from those of other infections of the lower urinary tract. Urethral infection is typically spread by sexual contact. The following recommendations are based on the consultation draft of the S3 guideline on urethritis, ‘Management of Urethritis in Male Adolescents and Adults’ (AWMF registry number 013-099) [303].
3.10.2. Epidemiology, aetiology and pathophysiology
In most cases, urethritis is caused by sexually transmitted bacteria, the most common of which are C. trachomatis, N. gonorrhoeae and M. genitalium. It should be noted that pathogens whose relevance as a cause of urethritis symptoms is questionable or must be determined on a case-by-case basis, such as U. urealyticum, U. parvum and M. hominis, are also frequently detected. Urethral infections with more than one pathogen are common. Empirical antibiotic treatment must therefore take into account the potential effects of targeting the primary pathogen on any coexisting infections. From a therapeutic and clinical point of view, gonorrhoeal urethritis (GU) caused by Neisseria gonorrhoeae must be differentiated from non-gonococcal urethritis (NGU). Non-gonococcal urethritis is a non-specific diagnosis that can have many infectious aetiologies. Causative pathogens include C. trachomatis, M. genitalium, U. urealyticum and Trichomonas vaginalis. The role of Ureaplasma spp. as urethritis causative pathogens is controversial. Recent data suggests that U. urealyticum, but not U. parvum is an aetiological agent in NGU [304]. The prevalence of isolated causative pathogens are: C. trachomatis 11–50%; M. genitalium 6–50%; Ureaplasmas 5–26%; T. vaginalis 1–20%; and adenoviruses 2–4% [305].
The causative pathogens either remain extracellularly on the epithelial layer or penetrate into the epithelium (N. gonorrhoeae and C. trachomatis) and cause pyogenic infection. Although arising from urethritis, chlamydiae and gonococci can spread further through the urogenital tract to cause epididymitis in males or cervicitis, endometritis and salpingitis in females [306].
3.10.3. Diagnostic evaluation
In symptomatic patients the diagnosis of urethritis can be made on the basis of the presence of any of the following criteria [305, 306]:
- Mucoid (watery-clear), mucopurulent (whitish-opaque urethral discharge), or purulent (yellow-green) urethral discharge.
- Gram- or methylene-blue staining of urethral discharge showing inflammation. Five or more polymorphonuclear leucocytes (PMNL) per high-power field (HPF) is the historical cut-off for the diagnosis of urethritis. A cut-off of ≥ 2 PMNL/HPF has recently been proposed based on improved diagnostic accuracy [307-310], but this was not supported by other studies [311]. Therefore, in line with the 2016 European Guideline on the management of NGU [305], the use of ≥ 5 PMNL/HPF cut-off is recommended until the benefit of alternative cut-off levels is confirmed.
- The presence of ≥ 10 PMNL/HPF in the sediment from a spun first-void urine sample or a positive leukocyte esterase test in first-void urine.
Evidence of urethral inflammation on the Gram stain of urethral secretions with intracellular gonococci as Gram-negative diplococci indicates GU. Nongonococcal urethritis is confirmed when staining of urethral secretions indicates inflammation in the absence of intracellular diplococci. Clinicians should always perform point-of-care diagnostics (e.g. Gram staining, first-void urine with microscopy, leukocyte esterase test) when available to obtain objective evidence of urethral inflammation and to guide treatment [305, 306, 312]. Recent studies have shown that processing time of point-of-care diagnostics is highly relevant in terms of patient compliance and real-life applicability [313, 314].
3.10.4. Urethral swab, urinalysis, NAAT
In cases of urethral discharge and suspected GU, a meatal/urethral swab should be taken for microbiological culture and N. gonorrhoeae resistance testing. Regardless of the initial classification of urethritis as GU or NGU, a nucleic acid amplification test (NAAT) should be performed to detect N. gonorrhoeae, M. genitalium and C. trachomatis [305, 315]. For molecular genetic diagnosis of N. gonorrhoeae, C. trachomatis and M. genitalium in cases of suspected urethral infection, both meatal/urethral swabs and first-void urine can be used. The sensitivity and specificity of NAAT are better than the other tests available for the diagnosis of chlamydial and gonococcal infections [316, 317]. The performance of first-catch urine is noninferior to urethral swabs [316]. In case of delayed treatment, if a NAAT is positive for gonorrhoea, a urethral swab culture should be performed prior to treatment to assess the antimicrobial resistance profile of the infecting strain [306]. N. gonorrhoeae and C. trachomatis cultures are mainly used to assess treatment failures and to monitor the development of resistance to current treatment. Trichomonas spp. can usually be identified microscopically [306] or by NAATs [318].
Nongonococcal urethritis is considered persistent if symptoms do not resolve within three to four weeks following treatment. In this case, NAAT for urethritis pathogens including T. vaginalis should be performed four weeks after completion of therapy [305, 319].
3.10.5. Disease management
In the context of increasing antimicrobial resistance among potentially relevant pathogens, the question is increasingly being raised of whether empirical antibiotic therapy for urethritis, initiated before pathogen identification, remains appropriate. However, current international guidelines and reviews on urethritis in males generally recommend immediate empirical antibiotic therapy based on distinct objective findings (e.g. purulent discharge), severe patient distress, clear sexual exposure or a high risk of transmission combined with limited follow-up availability. Conversely, empirical treatment should be deferred in favour of a diagnostic-first approach in cases of chronic symptoms, low subjective distress, lack of objective signs or recent antibiotic pretreatment. All sexual partners at risk should be assessed and treated whilst maintaining patient confidentiality [305, 320].
3.10.5.a. Suspected gonococcal urethritis
If GU is suspected, a combination treatment using two antimicrobials is recommended [306]. Ceftriaxone 1–2g intramuscularly or intravenously with doxycycline 100mg orally twice a day for seven days should be used as first-line treatment. In cases of suspected GU with contraindications to doxycycline, and if follow-up cannot be ensured, an alternative approach is a combined empirical treatment with ceftriaxone as outlined above and azithromycin administered orally according to a four-day regimen (day 1: 1g; days 2–4: 500mg orally).
3.10.5.b. Suspected non-gonococcal urethritis
For NGU without an identified pathogen, oral doxycycline 100mg twice daily for seven days should be used as first-line treatment. In cases of suspected NGU with contraindications to doxycycline, and if follow-up cannot be ensured, an alternative empirical treatment with oral azithromycin following the four-day regimen (day 1: 1g; days 2–4: 500mg orally) should be considered.
3.10.5.c. Gonococcal urethritis
In cases in which N. gonorrhoeae is detected, first-line therapy should consist of a single dose of ceftriaxone 1–2g given intravenously or intramuscularly. In cases of contraindications to cephalosporins and detection of N. gonorrhoeae with culture-confirmed sensitivity to azithromycin, treatment with oral azithromycin should be administered as follows: if coinfection with M. genitalium has been excluded: a single dose of 1g; if coinfection with M. genitalium cannot be excluded: four-day regimen (day 1: 1g; days 2–4: 500mg daily). In cases of contraindications to cephalosporins and detection of N. gonorrhoeae with molecularly or culture-confirmed sensitivity to ciprofloxacin, a single oral dose of ciprofloxacin 500mg may be considered. Other alternative regimens for the treatment of GU have been studied and are shown in Table 8 [306, 321-328].
3.10.5.d. Nongonococcal urethritis
3.10.5.d.1. Chlamydia trachomatis
When C. trachomatis is detected, first-line therapy should consist of doxycycline 100mg, administered orally twice daily for seven days. If C. trachomatis is detected and doxycycline is contraindicated, treatment with oral azithromycin should be considered. If coinfection with M. genitalium has been excluded, a single dose of 1g is recommended. If coinfection with M. genitalium cannot be ruled out, azithromycin should be administered according to the four-day regimen, with 1g on the first day followed by 500mg daily on days two through four. Fluoroquinolones, such as ofloxacin or levofloxacin, may be used as second-line treatment only in selected cases where other agents cannot be used [329].
3.10.5.d.2. Mycoplasma genitalium
In cases of M. genitalium detection without molecular resistance testing or without the detection of macrolide resistance-associated mutations (MRAM), first-line therapy should consist of azithromycin administered according to the four-day regimen: 1g on day one, followed by 500mg daily on days two through four. In cases of M. genitalium detection, treatment with moxifloxacin 400mg, taken orally once daily for seven days, is recommended if contraindications to azithromycin are present; if molecular genetic testing confirms the presence of macrolide resistance-associated mutations (MRAM); if the transmission setting suggests a high likelihood of azithromycin resistance; or if there was no response to prior treatment with azithromycin [305, 306, 330]. In case of a failure to, or contraindications to, azithromycin and moxifloxacin, treatment with sitafloxacin 100mg, taken orally twice daily for seven days, may be considered if M. genitalium is detected.
3.10.5.d.3. Mycoplasma hominis and Ureaplasma spp.
In cases of urethral detection of U. urealyticum, it is often a colonisation without clinical relevance. The indication for treatment should be evaluated on an individual basis in the absence of other pathogens. Urethral detection of M. hominis and U. parvum generally indicates colonisation without clinical relevance and does not warrant treatment. If U. urealyticum is identified as the sole causative agent of symptomatic urethritis, treatment with doxycycline 100mg, administered orally twice daily for seven days, is recommended. If U. urealyticum is identified as the sole cause of symptomatic urethritis and contraindications to doxycycline are present, treatment with a single oral dose of azithromycin 1g is recommended [305, 331].
3.10.5.d.4. Trichomonas vaginalis
If T. vaginalis is detected, first-line therapy should consist of a single oral dose of metronidazole 1.5–2g. In cases in which contraindications to metronidazole exist, an alternative treatment with tinidazole 2g orally may be considered. For treatment options for persistent or recurrent T. vaginalis infection, refer to the review of Sena et al. [318].
3.10.6. Follow-up
Individuals with a clinical diagnosis of acute urethritis should be advised to abstain from sexual activity for at least one week after completion of antibiotic therapy, or longer if symptoms persist. Sexual partners from the weeks prior to the onset of symptoms should be informed of the diagnosis and the need for appropriate testing and, if necessary, treatment. If symptoms persist for more than two weeks after completion of antibiotic therapy, or if symptoms recur, follow-up medical consultation is recommended. For the initial diagnosis of N. gonorrhoeae, C. trachomatis or M. genitalium, a follow-up cure test should be scheduled six to twelve weeks after completion of antibiotic therapy. In addition, when urethritis is diagnosed, testing for HIV and other sexually transmitted infections should be offered in addition to the urethritis diagnostic procedures recommended in this Guideline [332].
3.10.7. Summary of evidence and recommendations for the diagnostic evaluation and antimicrobial treatment of urethritis
| Summary of evidence | LE |
| A Gram stain of urethral discharge or a urethral smear that shows ≥ 5 leukocytes per high-power field (× 1,000) and gonococci located intracellularly as Gram-negative diplococci indicates gonococcal urethritis. | 3b |
| Validated NAATs of first-void urine samples have better sensitivity and specificity than any of the other tests available for the diagnosis of chlamydial and gonococcal infections. | 2a |
| For GU dual treatment with ceftriaxone and azithromycin is the most effective combination. | 2a |
| In case of urogenital C. trachomatis infection in men, azithromycin is likely less effective than doxycycline for microbiological failure. | 1a |
| In case of U. urealyticum infection, the efficacy of doxycycline 100mg twice for seven days is similar to azithromycin 1g single-dose treatment. | 2a |
| Recommendations | Strength rating |
| Perform a Gram stain of urethral discharge or a urethral smear to preliminarily diagnose gonococcal urethritis. | Strong |
| Perform a validated nucleic acid amplification test (NAAT) on a first-void urine sample or urethral smear prior to empirical treatment to diagnose chlamydial and gonococcal infections. | Strong |
| If possible, delay treatment until the results of the NAATs are available to guide treatment choice in patients with mild symptoms. | Strong |
| Perform a urethral swab culture, prior to initiation of treatment, in patients with a positive NAAT for gonorrhoea to assess the antimicrobial resistance profile of the infective strain. | Strong |
| Use a pathogen directed treatment based on local resistance data. | Strong |
| Sexual partners should be treated whilst maintaining patient confidentiality. | Strong |
Table 9: Suggested regimens for antimicrobial therapy for urethritis
| Suspected | Antimicrobial | Dosage & duration of therapy | Alternative regimens |
| Gonococcal infection | Ceftriaxone Doxycycline | 1–2g IM or IV*, SD 100mg b.i.d, p.o 7 days | In case of doxycycline allergy, in combination with ceftriaxone: Azithromycin four-day regimen: |
| Non-Gonococcal infection | Doxycycline | 100mg b.i.d, p.o 7 days | Azithromycin four-day regimen: Day 1: 1g; Days 2–4: 500mg p.o |
* Despite the lack of RCTs, there is increasing evidence that intravenous treatment with ceftriaxone is safe and effective for the treatment of gonorrhoeal infections and avoids the discomfort of an intramuscular injection for patients [333].
Table 10: Regimens for antimicrobial therapy for urethritis with causing pathogen detected
| Pathogen | Antimicrobial | Dosage & duration of therapy | Alternative regimens |
| Neisseria gonorrhoeae | Ceftriaxone Doxycycline | 1–2g IM or IV*, SD 100mg b.i.d, p.o 7 days |
In case of doxycycline allergy, in combination with ceftriaxone: |
| Chlamydia trachomatis | Doxycycline | 100mg b.i.d, p.o for 7 days |
|
| Mycoplasma genitalium | Azithromycin | Four-day regimen: Day 1 1g; Days 2–4: 500mg p.o | In case of macrolide resistance:
|
| Ureaplasma urealyticum | Doxycycline | 100mg b.i.d, p.o 7 days | Azithromycin 1g p.o, SD |
| Trichomonas vaginalis | Metronidazole | 1.5-2 g p.o, SD | Tinidazole 2g p.o, SD |
SD = single dose; b.i.d = twice daily; q.d = every day; p.o = orally; IM = intramuscular; IV = intravenously.
* Despite the lack of RCTs, there is increasing evidence that intravenous treatment with ceftriaxone is safe and effective for the treatment of gonorrhoeal infections and avoids the discomfort of an intramuscular injection for patients [333].
3.11. Bacterial Prostatitis
3.11.1. Introduction
Bacterial prostatitis is a clinical condition caused by bacterial pathogens. Urologists are recommended to use the classification suggested by the National Institute of Diabetes, Digestive and Kidney Diseases (NIDDK) of the U.K. National Institutes of Health (NIH), in which bacterial prostatitis, with confirmed or suspected infection, is distinguished from chronic pelvic pain syndrome (CPPS) (Table 9) [334-336].
Table 9: Classification of prostatitis and CPPS according to NIDDK/NIH
| Type | Name and description |
| I | Acute bacterial prostatitis (ABP) |
| II | Chronic bacterial prostatitis (CBP) |
| III | Chronic non-bacterial prostatitis - CPPS |
| IIIA | Inflammatory CPPS (white cells in semen/EPS/VB3) |
| IIIB | Non-inflammatory CPPS (no white cells in semen/EPS/VB3) |
| IV | Asymptomatic inflammatory prostatitis (histological prostatitis) |
CPPS = chronic pelvic pain syndrome; EPS = expressed prostatic secretion; VB3 = voided bladder urine specimen 3 (urine following prostatic massage).
3.11.2. Evidence summary
A systematic literature search from 1980 through June 2017 was carried out. One systematic review [337], six RCTs [338-343], two narrative reviews [344, 345], one prospective cohort study [346], two prospective cross-sectional studies [347,348] and one retrospective cohort study [340] were selected from among 856 references.
A retrospective study [349] investigated the potential role of unusual pathogens in prostatitis syndrome in 1,442 patients over a four-year period. An infectious aetiology was determined in 74.2% of patients; C. trachomatis, T. vaginalis and U. urealyticum infections were found in 37.2%, 10.5% and 5% of patients, respectively; whilst E. coli infection was found in only 6.6% of cases. Cross-sectional studies confirmed the validity of the Meares and Stamey test to determine the bacterial strain and targeted antibiotic therapies [347, 348]. The evidence levels were good, in particular those relating to information on atypical strains, epidemiology and antibiotic treatments.
A systematic review on antimicrobial therapy for CBP [337] compared multiple antibiotic regimens from eighteen selected studies enrolling a total of 2,196 patients. The role of fluoroquinolones as first-line agents was confirmed with no significant differences between levofloxacin, ciprofloxacin and prulifloxacin in terms of microbiological eradication, clinical efficacy and adverse events. The efficacy of macrolides and tetracyclines on atypical pathogens was confirmed.
Randomised controlled trials on combined treatments [342,343] indicated that the combination of plants/herbal extracts or PDE5Is with antibiotics may improve quality of life and symptoms in patients with CBP. However, the number of enrolled patients was inadequate to obtain definitive conclusions.
A review of treatment of bacterial prostatitis [344] indicated that the treatment of CBP is hampered by the lack of an active antibiotic transport mechanism into infected prostate tissue and fluids. The review underlined the potential effect of various compounds in the treatment of ABP and CBP on the basis of over 40 studies on the topic.
One RCT compared the effects of two different metronidazole regimens for the treatment of CBP caused by T. vaginalis [341]. Metronidazole 500mg three times daily for 14 days was found to be efficient for pathogen eradication in 93.3% of patients with clinical failure in 3.33% of cases.
3.11.3. Epidemiology, aetiology and pathogenesis
Prostatitis is a common diagnosis, but fewer than 10% of cases have proven bacterial infection [228]. Enterobacterales, particularly E. coli, are the predominant pathogens in ABP [350]. In CBP, the spectrum of species is wider and may include atypical microorganisms [344]. In patients with immune deficiency or HIV infection, prostatitis may be caused by fastidious pathogens, such as M. tuberculosis, Candida spp., and other rare pathogens, such as Coccidioides immitis, Blastomyces dermatitidis and Histoplasma capsulatum [351]. The significance of identified intracellular bacteria, such as C. trachomatis, is uncertain [352]. However, two studies have highlighted its possible role as a pathogen in CBP [353,354].
3.11.4. Diagnostic evaluation
3.11.4.a. History and symptoms
Acute bacterial prostatitis usually presents abruptly with voiding symptoms and distressing but poorly localised pain, and is often associated with malaise and fever. Transrectal prostate biopsy increases the risk of ABP despite antibiotic prophylaxis and antiseptic prevention procedures [338]. Chronic bacterial prostatitis is defined by symptoms that persist for at least three months [355-357]. The predominant symptoms are pain at various locations, including the perineum, scrotum, penis and inner part of the leg, as well as LUTS [334-336].
3.11.4.b. Symptom questionnaires
In CBP, symptoms appear to have a strong basis for use as a classification parameter [358]. Prostatitis symptom questionnaires have therefore been developed to assess severity and response to therapy [358, 359]. These questionnaires include the validated Chronic Prostatitis Symptom Index (CPSI), however, the usefulness of the CPSI in clinical practice is uncertain [346].
3.11.4.c. Clinical findings
In ABP, the prostate may be swollen and tender on DRE. Prostatic massage should be avoided, as it can induce bacteraemia and sepsis. Urine dipstick testing for nitrite and leukocytes has a positive predictive value of 95% and a negative predictive value of 70% [360]. Blood culture and complete blood count are useful in ABP. Imaging studies can detect a suspected prostatic abscess [344].
In case of longer-lasting symptoms, CPPS, as well as other urogenital and anorectal disorders, must be taken into consideration. Symptoms of CBP or CPPS can mask prostate tuberculosis. Pyospermia and hematospermia in males in endemic regions or with a history of tuberculosis should trigger investigation for urogenital tuberculosis.
3.11.4.d. Urine cultures and expressed prostatic secretion
The most important investigation in the evaluation of a patient with ABP is midstream urine culture [344]. In CBP, quantitative bacteriological localisation cultures and microscopy of the segmented urine and expressed prostatic secretion (EPS), as described by Meares and Stamey [361], are still important investigations to categorise clinical prostatitis [347, 348]. Accurate microbiological analysis of samples from the Meares and Stamey test may also provide useful information on the presence of atypical pathogens such as C. trachomatis, T. vaginalis and U. urealiticum [349]. The two-glass test has been shown to offer similar diagnostic sensitivity to the four-glass test [362].
3.11.4.e. Prostate biopsy
Prostate biopsies cannot be recommended as routine workup and are not advisable in patients with untreated bacterial prostatitis, due to the increased risk of sepsis.
3.11.4.f. Other tests
Transrectal ultrasound may reveal endoprostatic abscesses, calcification in the prostate and dilatation of the seminal vesicles. However, ultrasound is unreliable as a diagnostic tool for prostatitis [363].
3.11.4.g. Additional investigations
3.11.4.g.1. Ejaculate analysis
Performing an ejaculated semen culture improves the diagnostic utility of the four-glass test [347], however, semen cultures are more often positive than EPS cultures in males with non-bacterial prostatitis [348]. Bladder outflow and urethral obstruction should always be considered and ruled out by uroflowmetry, retrograde urethrography or endoscopy.
3.11.4.g.2. First-void urine sample
First-void urine is the preferred specimen for the diagnosis of urogenital C. trachomatis infection in men by NAATs, because it is noninvasive and yet allows the detection of infected epithelial cells and associated C. trachomatis particles [364].
3.11.4.g.3. Prostate-specific antigen (PSA)
Prostate-specific antigen (PSA) level is increased in approximately 60% and 20% of males with ABP and CBP, respectively [345]. The PSA level decreases after antibiotic therapy (which occurs in approximately 40% of patients) and correlates with clinical and microbiological improvement [339]. Measurement of free and total PSA adds no practical diagnostic information in prostatitis [365].
3.11.4.h. Summary of evidence and recommendations for the diagnosis of bacterial prostatitis
| Summary of evidence | LE |
| Urine dipstick testing for nitrite and leukocytes has a positive predictive value of 95% and a negative predictive value of 70% in patients with ABP. | 3 |
| The four-glass Meares and Stamey test is the optimum test for diagnosis of CBP. The two-glass test has been shown to offer similar diagnostic sensitivity in a comparison study. | 2b |
| First-void urine is the preferred specimen for the diagnosis of urogenital C. trachomatis infection in men by NAATs. | 2b |
| Transrectal ultrasound is unreliable and cannot be used as a diagnostic tool in prostatitis. | 3 |
| Semen culture sensitivity is reported to be approximately 50% and is, therefore, not routinely part of the diagnostic assessment of CBP. | 3 |
| Prostate-specific antigen levels may be elevated during active prostatitis. Therefore, PSA testing should be avoided, as it offers no practical diagnostic information for prostatitis. | 3 |
| Recommendations | Strength rating |
| Do not perform prostatic massage in acute bacterial prostatitis (ABP). | Strong |
| Take a midstream urine dipstick to check nitrite and leukocytes in patients with clinical suspicion of ABP. | Weak |
| Take a midstream urine culture in patients with ABP symptoms to guide diagnosis and tailor antibiotic treatment. | Weak |
| Take a blood culture and a total blood count in patients presenting with ABP. | Weak |
| Perform accurate microbiological evaluation for atypical pathogens such as Chlamydia trachomatis or Mycoplasma spp. in patients with chronic bacterial prostatitis (CBP). | Weak |
| Perform the Meares and Stamey 2- or 4-glass test in patients with CBP. | Strong |
| Perform transrectal ultrasound in selected cases to rule out the presence of prostatic abscess. | Weak |
| Do not routinely perform microbiological analysis of the ejaculate alone to diagnose CBP. | Weak |
3.11.5. Disease management
3.11.5.a. Antimicrobials
Antimicrobials are life-saving in ABP and recommended in CBP. Culture-guided antibiotic treatments are the optimum standard, however, empirical therapies should be considered in all patients with ABP.
In ABP, parenteral administration of high doses of bactericidal antimicrobials, such as broad-spectrum penicillins, a third-generation cephalosporin or fluoroquinolones, is recommended [366]. For initial therapy, any of these antimicrobials can be combined with an aminoglycoside [350-359, 366-370]. Ancillary measures include adequate fluid intake and urine drainage [228]. After normalisation of infection parameters, oral therapy can be substituted and continued for a total of two to four weeks [371].
Fluoroquinolones, despite the high resistance rates of uropathogens, are recommended as first-line agents in the empirical treatment of CBP, due to their favourable pharmacokinetic properties [372], their generally good safety profile, and antibacterial activity against Gram-negative pathogens, including P. aeruginosa and C. trachomatis [337, 373]. However, increasing bacterial resistance is a concern. Azithromycin and doxycycline are active against atypical pathogens such as C. trachomatis and genital Mycoplasma spp. [340,349]. Levofloxacin did not demonstrate significant clearance of C. trachomatis in patients with CBP [374]. Metronidazole treatment is indicated in patients with T. vaginalis infections [341].
Duration of fluoroquinolone treatment must be at least fourteen days, while azithromycin and doxycycline treatments should be extended to at least three to four weeks [340, 349]. In CBP, antimicrobials should be given for four to six weeks after initial diagnosis [344]. If intracellular bacteria have been detected, macrolides or tetracyclines should be given [337, 372, 375].
3.11.5.b. Intraprostatic injection of antimicrobials
This treatment has not been evaluated in controlled trials and should not be considered [376, 377].
3.11.5.c. Combined treatments
A combination of fluoroquinolones with various herbal extracts may attenuate clinical symptoms without increasing the rate of adverse events [342]. However, a combination of fluoroquinolones with vardenafil did not improve microbiological eradication rates or attenuated pain or voiding symptoms in comparison with fluoroquinolone treatment alone [343].
3.11.5.d. Drainage and surgery
Approximately 10% of males with ABP will experience urinary retention [378] that can be managed by urethral or suprapubic catheterisation. However, recent evidence suggests that suprapubic catheterisation can reduce the risk of development of CBP [379].
In case of prostatic abscess, both drainage and conservative treatment strategies appear feasible [380], however, the abscess size may matter. In one study, conservative treatment was successful if the abscess cavities were < 1cm in diameter, while larger abscesses were better treated by single aspiration or continuous drainage [381].
3.11.5.e. Summary of evidence and recommendations for the disease management of bacterial prostatitis
| Summary of evidence | LE |
| The treatment regimen for ABP is based on clinical experience and a number of uncontrolled clinical studies. For systemically ill patients with ABP, parenteral antibiotic therapy is preferable. After normalisation of infection parameters, oral therapy can be substituted and continued for a total of two to four weeks. | 3 |
| The role of fluoroquinolones as first-line agents for antimicrobial therapy for CBP was confirmed in a systematic review, with no significant differences between levofloxacin, ciprofloxacin and prulifloxacin in terms of microbiological eradication, clinical efficacy and adverse events. | 1a |
| Metronidazole 500mg three times daily for fourteen days was found to be efficient for eradication in 93.3% of patients with T. vaginalis CBP. | 1b |
| In patients with CBP caused by obligate intracellular pathogens, macrolides showed higher microbiological and clinical cure rates compared to fluoroquinolones. | 1a |
| Clinicians should consider local drug-resistance patterns when choosing antibiotics. | 3 |
| Recommendations | Strength rating |
| Acute bacterial prostatitis (ABP) | |
| Treat ABP according to the recommendations for systemic UTIs (see Section 3.7.4). | Strong |
| Chronic bacterial prostatitis (CBP) | |
| Prescribe a fluoroquinolone (e.g. ciprofloxacin, levofloxacin) as first-line treatment for CBP. | Strong |
| Prescribe a macrolide (e.g. azithromycin) or a tetracycline (e.g. doxycycline) if intracellular bacteria have been identified as the causative agent of CBP. | Strong |
| Prescribe metronidazole in patients with T. vaginalis CBP. | Strong |
Table 10: Suggested regimens for antimicrobial therapy for chronic bacterial prostatitis
| Antimicrobial | Daily dose | Duration of therapy | Comments |
| Fluoroquinolone | Optimal oral daily dose | 4–6 weeks | |
| Doxycycline | 100mg b.i.d | 10 days | Only for C. trachomatis or mycoplasma infections |
| Azithromycin | 500mg o.d | up to 3 weeks | Only for C. trachomatis infections |
| Metronidazole | 500mg t.i.d | 14 days | Only for T. vaginalis infections |
b.i.d = twice daily; o.d = once daily; t.i.d = three times daily.
3.11.6. Follow-up
In asymptomatic post-treatment patients, routine urinalysis and/or urine culture is not mandatory, as there are no validated tests of cure for bacterial prostatitis except for cessation of symptoms [344]. In patients with persistent symptoms and repeated positive microbiological results for sexually transmitted infectious pathogens, microbiological screening of the patient’s partner(s) is recommended. Antibiotic treatments may be repeated with a more prolonged course, higher dosage and/or different compounds [344].
3.12. Acute epididymitis
3.12.1. Epidemiology, aetiology and pathophysiology
Epididymitis is a common condition with incidence ranging from 25 to 65 cases per 10,000 adult males per year and can be acute, chronic or recurrent [382]. Acute epididymitis is clinically characterised by unilateral pain, palpable swelling and increased temperature of the epididymis, which worsen over several days. The testes and scrotal skin may be involved. The pain is typically localised to the posterior aspect of the testis affected. Acute epididymitis is generally caused by sexually transmitted infections (STIs) or Enterobacterales. Pathogens migrate from the urethra or bladder and can be identified by appropriate diagnostics in up to 90% of patients [383].
The predominant pathogens isolated in sexually transmitted epididymitis in otherwise healthy young and sexually active biological men are N. gonorrhoeae, C. trachomatis, U. urealyticum and M. genitalium. Acute epididymitis caused by an STI is usually accompanied by urethritis and discharge, which in case of gonorrhoea is thick, green, white or yellow and very productive. Non-sexually transmitted epididymitis is typically caused by Enterobacterales (e.g. E. coli) [384]. Older patients with a history of urinary tract infection are most commonly affected. Risk factors include benign prostatic hyperplasia, neurogenic bladder and recent instrumentation. Males who have unprotected anal intercourse are at higher risk of epididymitis caused by Enterobacterales [385]. The mumps virus should be considered in case of bilateral scrotal involvement, viral prodromal symptoms and salivary gland enlargement. Tuberculous epididymitis may occur, typically as chronic epididymitis, in high-risk groups such as males with immunodeficiency and those from countries with a high prevalence of tuberculous. Tuberculous epididymitis frequently results in a discharging scrotal sinus. Brucella or Candida spp. are rare potential pathogens. Torsion of the spermatic cord (testicular torsion) is the most important differential diagnosis in boys and young males.
3.12.2. Diagnostic evaluation
Culture of a midstream specimen of urine should be performed, and any previous urine culture results should be checked. Sexually transmitted infections including C. trachomatis or N. gonorrhoeae should be detected by means of NAAT on first-voided urine or urethral swab. A urethral swab or smear might be performed for Gram staining and culture of N. gonorrhoeae, when available [382, 386, 387]. Detection of these pathogens should be reported according to local procedures. All patients with probable STIs should be screened for other STIs. Patients with Enterobacterales may require investigation for lower urinary tract abnormalities. If tuberculous epididymitis is suspected, three sequential early morning urine samples should be cultured for acid-fast bacilli (AFB) and sent for screening by NAAT for M. tuberculosis DNA [388]. If appropriate, prostate secretion, ejaculate, discharge from a draining scrotal fistula, as well as fine-needle aspiration and biopsy specimens should be investigated using microscopy, AFB culture and NAAT. Scrotal ultrasound is more accurate for the diagnose of acute epididymitis than urinalysis alone [389] and may also be beneficial for the exclusion of other pathologies [390].
3.12.3. Disease management
Empirical antimicrobial therapy must be chosen with consideration of the most probable pathogen and degree of penetration into the inflamed epididymis and may need to be varied according to local pathogen sensitivities and guidance. Generally, both C. trachomatis and Enterobacterales should be covered initially and the regimen modified according to pathogen identification. Doxycycline and fluoroquinolones have good clinical and microbiological cure rates in patients with suspected C. trachomatis or M. genitalium, and both achieve adequate levels in inflamed male genital tissues with oral dosing. Macrolide antibiotics such as azithromycin are effective against C. trachomatis but have not been tested in epididymitis. However, initial pharmacokinetic studies suggest that azithromycin may effectively penetrate epididymal tissue when given in multiple doses [391]. Fluoroquinolones remain effective for oral treatment of Enterobacterales, although resistance is increasing and local advice should be sought. Fluoroquinolones should not be considered for gonorrhoea. Single parenteral dose of ceftriaxone is effective against N. gonorrhoeae. However, current resistance patterns and local public health recommendations should guide choice of agent. Patients with likely or proven STI should be assessed fourteen days after end of treatment to ensure that the patient has been cured and ensure tracing and treatment of contacts according to local public health recommendations. Nonsteroidal anti-inflammatory drugs and local cooling can be used to manage pain and swelling. Elevating the scrotum may also help reduce discomfort. Men with suspected STI should be informed of the risks to others and advised not to have sex until they are free of infection. In exceptional cases, individuals with epididymitis may experience severe pain, fever, a testicular abscess or signs of systemic illness. These patients should be admitted to the hospital for intravenous antibiotic treatment and fluid replacement.
3.12.4. Evidence summary
Relating to this chapter, four guidelines based on systematic reviews were identified [306,386,392,393]. No evidence quality assessments were detailed. A high-quality RCT demonstrated that a ten-day course of ciprofloxacin was superior to pivampicillin for clinical cure (80% vs. 60%) in men aged > 40 years [394]. Data from a large comparative case series suggested that young age and history of sexual activity are not sufficiently predictive of a sexually transmitted pathogen to guide antibiotic treatment in acute epididymitis [383].
Empiric antibiotic regimens from existing guidelines [306, 386, 392, 393] and panel consensus:
For males with acute epididymitis at low risk of gonorrhoea (e.g. no urethritis, no urethral discharge), a single agent active against Enterobacterales and C. trachomatis (e.g. levofloxacin) or a combination of two agents (e.g. doxycycline and trimethoprim-sulfamethoxazol) to eradicate C. trachomatis and Enterobacterales should be used. Options include:
A. Levofloxacin 500mg orally once daily for ten to fourteen days*OR
B. Doxycycline 200mg orally initial dose and then 100mg twice daily for ten to fourteen days* plus trimethoprim-sulfamethoxazol 160/800mg twice daily for ten to fourteen days**
- For biological males with likely gonorrhoeal acute epididymitis, a combination regimen active against N. gonorrhoeae and C. trachomatis must be used, such as:
A. Ceftriaxone 1,000mg intramuscularly or i.v. single dose plus doxycycline 200mg orally initial dose and then 100mg twice daily for ten to fourteen days*
- For non-sexually active males with acute epididymitis, a single agent of sufficient dose and duration to eradicate Enterobacterales should be used. Appropriate option is a fluoroquinolone once daily (e.g. levofloxacin 500mg) or trimethoprim-sulfamethoxazol 160/800mg orally twice daily for ten to fourteen days*.
* Depending upon pathogen identification and clinical response.
** A parenteral option will be required for men with severe infection requiring hospitalisation.
Surgical exploration may be required to drain abscesses or debride tissue. A comparative cohort study found that lack of separation of epididymis and testis on palpation and the presence of abscess on ultrasound may predict requirement for surgery following initial antibiotic treatment [395].
A cohort study found that semen parameters may be impaired during epididymitis but recovered following successful treatment [396]. Comparative clinical cohort studies suggest that adherence to guidelines for assessment and treatment of epididymitis is low, particularly by urologists compared to sexual health specialists [397] and by primary care physicians [398].
3.12.5. Screening
A large cohort screening study for carriage of C. trachomatis including a randomly selected group of 5,000 males of whom 1,033 were tested, showed no benefit in terms of reduction in risk of epididymitis over nine years of observation [399].
3.12.6. Summary of evidence and recommendations for the diagnosis and treatment of acute infective epididymitis
| Summary of evidence | LE |
| In young, sexually active patients, both STIs and Enterobacterales must be considered as aetiological agents. | 3 |
| In patients > 40 years, antibiotic therapy with ciprofloxacin is superior to pivmecillinam. | 1b |
| A negative sexual risk history does not exclude STIs in sexually active men. | 3 |
| Recommendations | Strength rating |
| Obtain a midstream urine and a first-voided urine for pathogen identification by culture and nucleic acid amplification test. | Strong |
| Initially prescribe a single antibiotic or a combination of two antibiotics active against Chlamydia trachomatis and Enterobacterales in young, sexually active men. In older men without sexual risk factors, only Enterobacterales have to be considered. | Strong |
| If gonorrhoeal infection is likely, give single-dose ceftriaxone 1,000mg intramuscularly or intravenously* in addition to a course of an antibiotic active against Chlamydia trachomatis. | Strong |
| Adjust antibiotic agent when pathogen has been identified and adjust duration according to clinical response. | Weak |
| Follow national policies on reporting and tracing/treatment of contacts for sexually transmitted infections. | Strong |
* Despite the lack of RCTs there is increasing evidence that intravenous treatment with ceftriaxone is safe and effective for the treatment of gonorrhoeal infections and avoids the discomfort of an intramuscular injection for patients .
Figure 2: Diagnostic and treatment algorithm for men with acute epididymitis i.m. = intramuscular; i.v. = intravenously.
i.m. = intramuscular; i.v. = intravenously.
* Despite the lack of RCTs, there is increasing evidence that intravenous treatment with ceftriaxone is safe and effective for the treatment of gonorrhoeal infections and avoids the discomfort of an intramuscular injection for patients .
3.13. Fournier’s gangrene (necrotising fasciitis of the perineum and external genitalia)
3.13.1. Epidemiology, aetiology and pathophysiology
Fournier’s gangrene is an aggressive and frequently fatal polymicrobial soft tissue infection of the perineum, perianal region and external genitalia [400]. It is an anatomical subcategory of necrotising fasciitis with which it shares a common aetiology and management pathway.
3.13.2. Diagnostic evaluation
Fournier’s gangrene is typically accompanied by painful swelling of the scrotum or perineum and characterised by severe inflammation and infection spreading along fascial planes, often leading to rapid tissue destruction and sepsis [400]. Examination shows small necrotic areas of skin with surrounding erythema and oedema. Crepitus on palpation and a foul-smelling exudate occurs with more advanced disease. Patient risk factors for occurrence and mortality include being immunocompromised – most commonly diabetes or malnutrition – as well as recent urethral or perineal surgery and high body mass index (BMI). In up to 40% of cases, the onset is more insidious, with undiagnosed pain often resulting in delayed treatment [401]. A high index of suspicion and careful examination, particularly of obese patients, is required. Computed tomography or MRI can help define pararectal involvement, suggesting the need for bowel diversion [400].
3.13.3. Disease management
The degree of internal necrosis is usually vastly greater than suggested by external signs, and consequently, adequate, repeated surgical debridement with urinary diversion by suprapubic catheter is required to reduce mortality [400]. Consensus from case series suggests that surgical debridement should be early (< 24 hours) and complete, as delayed and/or inadequate surgery may result in higher mortality [400]. Immediate empiric parenteral antibiotic treatment should be given that covers all probable causative organisms and can penetrate inflammatory tissue. A suggested regimen would comprise a broad-spectrum penicillin or third-generation cephalosporin, gentamicin and metronidazole or clindamycin [400]. This can then be refined, guided by microbiological culture.
3.13.4. Evidence summary
A systematic literature search from 1980 to July 2017 was performed. From among 640 references, one RCT [402], two systematic reviews [403,404], one narrative review [400], three registry studies [405-407], one prospective cohort study [408] and two retrospective comparative cohort studies with at least 25 patients [409,410] were selected. The three registry studies from the United States [405-407] found mortality rates of 10%, 7.5% and 5% from 650, 1,641 and 9,249 cases, respectively. Older age, diabetes and high BMI were associated with higher risk. A prospective cohort study showed that disease-specific severity scores did predict outcome but were not superior to generic scoring systems for critical care [408]. The evidence questions addressed were:
- What is the best antimicrobial treatment strategy to reduce mortality?
- What is the best debridement and reconstruction strategy to reduce mortality and aid recovery?
- Are any effective adjuvant treatments available that improve outcome?
Concerning the evidence questions:
- A low-quality retrospective case series [409] with 168 patients found no significant difference in mortality between patients given ≤ 10 days of parenteral antibiotics (80 patients) and those given > 10 days (88 patients).
- A systematic review of wound closure techniques [404] found low-quality evidence from 16 case series involving 425 male patients. These case series recommended primary or secondary wound closure for scrotal defects ≤ 50%, and the use of flaps or skin grafts for defects involving > 50% of the scrotum or with extension outside the scrotum.
- A systematic review on the use of hyperbaric oxygen therapy [403] included three comparative case series and four other case series. All were retrospective and published prior to 2000. No consistent evidence of benefit was found; an RCT was advised. A more recent comparative case series [410] suggested benefit for use of hyperbaric oxygen therapy in 16 patients compared to 12 cases without use of such therapy in terms of reduced mortality and fewer debridements (low quality evidence). A low-quality RCT [402] with 30 patients found that the use of honey-soaked dressings resulted in a shorter hospital stay (28 vs. 32 days) than dressing soaked with Edinburgh solution of lime (EUSOL). We found no evidence of benefit for use of negative-pressure (vacuum) wound therapy in Fournier’s gangrene.
3.13.5. Summary of evidence and recommendations for the disease management of Fournier’s gangrene
| Summary of evidence | LE |
| Immediate empiric parenteral antibiotic treatment should be given that covers all probable causative organisms and can penetrate inflammatory tissue. | 3 |
| A systematic review of wound closure techniques recommended primary or secondary wound closure for scrotal defects ≤ 50%, and the use of flaps or skin grafts for defects involving > 50% of the scrotum or with extension outside the scrotum. | 3 |
| No consistent evidence of benefit for hyperbaric oxygen therapy was found. | 3 |
| A low-quality RCT found that dressings soaked in honey resulted in a shorter hospital stay than dressings soaked in EUSOL. | 3 |
| No evidence of benefit for use of negative-pressure (vacuum) wound therapy in Fournier’s gangrene was found. | 4 |
| Recommendations | Strength rating |
| Start treatment for Fournier’s gangrene with broad-spectrum antibiotics on presentation, with subsequent refinement according to culture results and clinical response. | Strong |
| Commence repeated surgical debridement for Fournier’s gangrene within 24 hours of presentation. | Strong |
| Do not use adjunctive treatments for Fournier’s gangrene except in the context of clinical trials. | Weak |
Table 11: Suggested regimens for antimicrobial therapy for Fournier’s gangrene of mixed microbiological aetiology adapted from [411]
| Antimicrobial | Dosage |
Piperacillin-tazobactam plus Vancomycin | 4.5g q.i.d. or t.i.d, IV 15mg/kg b.i.d |
| Imipenem-cilastatin | Standard dosage: 0.5g IV q.i.d over 30 minutes High dosage: 1g IV q.i.d over 30 minutes |
| Meropenem | 1g t.i.d. IV |
| Ertapenem | 1g o.d. |
| Gentamicin | 6–7 mg/kg IV q.d |
Cefotaxime plus metronidazole or clindamycin | 2g q.i.d IV 500mg q.i.d IV 600–900mg t.i.d IV |
Cefotaxime plus fosfomycin plus metronidazole | 2g q.i.d IV 5g t.i.d IV 500mg q.i.d IV |
b.i.d = twice daily; IV = intravenous; o.d = once daily; q.i.d = four times daily; q.d = every day; t.i.d = three timesdaily.
3.14. Management of human papillomavirus in men
3.14.1. Epidemiology
Human papilloma virus (HPV) is one of the most frequently sexually transmitted viruses, encompassing both oncogenic (low- and high-risk variants) and non-oncogenic viruses. Urologists primarily encounter male patients presenting with visible external genital lesions, most notably condylomata acuminata, or obstructive voiding symptoms caused by meatal involvement. Additionally, consultations are frequently driven by the need for screening and counselling following a confirmed high-risk HPV diagnosis in a sexual partner. HPV16 is the most common oncogenic variant, detected in 20% of all HPV cases [412]. A recent meta-analysis revealed a prevalence of 49% of any type of HPV and 35% of high-risk HPV in males [413]. Similar to the female genital tract, half of all HPV infections in the male genital tract are co-infections (≥ 2 HPV strains) [414].
HPV presence is dependent on the clinical setting. In men attending urological clinics, HPV was detected in 6% of urine samples [415]. A meta-analysis reported seminal HPV in 4.5–15.2% of patients resulting in seminal HPV being associated with decreased male fertility [412]. A cross-sectional study of 430 males presenting for fertility treatment detected HPV in 14.9% of semen samples [416]. The presence of HPV in semen was not associated with impaired semen quality [416]. However, another systematic review reported a possible association between HPV and altered semen parameters and, in women, possible miscarriage or premature rupture of the membrane during pregnancy [417]. HPV6 and/or 11 were the most common genotypes detected in an observational study of anogenital warts, whilst HPV16 is correlated with severity of anal cytology [418]. The incidence of non-oncogenic HPV infection has been shown to be higher in males than females [419]. In males, approximately 33% of penile cancers and up to 90% of anal cancers are attributed to high-risk HPV infections, primarily with HPV16 [420]. Oral HPV is associated with oropharyngeal carcinomas. Approximately 22.4%, 4.4% and 3.5% of oral cavity, oropharynx and larynx cancers, respectively, are attributed to HPV [420]. Systematic reviews have reported prevalence rates of oral HPV from 5.5-7.7%, with HPV16 present in 1–1.4% of patients [421, 422].
3.14.2. Risk factors
Risk factors for HPV infection include early age of first sexual intercourse, sexual promiscuity, higher frequency of sexual intercourse, smoking and poor immune function [423-427]. Incidence and prevalence of overall HPV was considerably higher in men who have sex with men (MSM) compared to heterosexuals [421, 424]. Overall, the prevalence of HPV in different sites appears to be higher in young, sexually active adults compared to other population groups [423]. Stable sexual habits, circumcision and condom use are protective factors against HPV [413,427-431]. Added risk factors of oral HPV infection are alcohol consumption, poor oral hygiene and sexual behaviours (oral and vaginal) [421,423]. Positive HIV status, phimosis and HPV status of the partner have also been associated with anogenital HPV status and decreased clearance [428].
3.14.3. Transmission
HPV typically spreads by sustained direct skin-to-skin or mucosal contact, with vaginal, oral and anal sex being the most common transmission routes [425]. In addition, HPV has been found on surfaces in medical settings and public environments, thus raising the possibility of object-to-skin/mucosa transmission [432]. Further studies on non-sexual and non-penetrative sexual transmission are needed to understand the complexity of HPV transmission. HPV transmission may also be influenced by genotype, with a higher incidence of HPV51 and HPV52 and a high prevalence of HPV16 and HPV18 in the general and high-risk male population [425].
3.14.4. Clearance
HPV time-to-clearance ranges from 1.3 to 42.1 months [433]. Clearance may be influenced by HPV genotype, patients’ characteristics and affected body site [424, 428, 433]. HPV16 has the highest incidence of high-risk HPV variants and has the lowest clearance across sites [428].
3.14.5. Diagnosis
There is currently no approved test for HPV in males. Routine testing to check for HPV or HPV-related disease in males are not recommended. A physical examination to identify HPV lesions should be carried out. An acetic acid test to diagnose subclinical HPV lesions may be performed. If the diagnosis is uncertain or if there is a suspicion of cancer, a biopsy should be carried out. Intraurethral condylomas are relatively uncommon and are usually limited to the distal urethral meatus [434, 435]. Urethrocystoscopy may be used to diagnose the presence of intraurethral or bladder warts [435], however, there is no high-level evidence for the use of invasive diagnostic tools for localisation of intraurethral HPV. For detailed recommendations on the diagnosis of anogenital warts, please refer to the IUSTI‐European guideline for the management of anogenital warts [436].
3.14.6. Treatment of HPV-related diseases
Approximately 90% of HPV infections do not cause any problems and are cleared by the body within two years. However, treatment is required when HPV infection manifests as anogenital warts to prevent the transmission of HPV-associated anogenital infection and to minimise the discomfort caused to patients [436]. Of the treatment options available, only surgical treatment has a primary clearance rate approaching 100%.
3.14.6.a. Treatments suitable for self-application
Patient-applied treatments include podophyllotoxin, salicylic acid, imiquimod, polyphenon E, 5-fluoracil and potassium hydroxide [436]. Imiquimod 5% cream showed a total clearance of external genital or perianal warts in 50% of immunocompetent patients [437], as well as in HIV positive patients successfully treated with highly active antiretroviral therapy [438]. A Cochrane review of published RCTs found imiquimod to be superior to placebo in achieving complete clearance of warts (RR: 4.03, 95% CI: 2.03–7.99) [439]. The recommended treatment schedule is imiquimod 5% cream applied to all external warts overnight three times each week for 16 weeks [436]. In an RCT involving 502 patients with genital and/or perianal warts, sinecatechins 15% and 10% showed a complete clearance of all baseline and newly occurring warts in 57.2% and 56.3% of patients, respectively, versus 33.7% for placebo [440]. In addition, sinecatechins 10% has been shown to be associated with lower short-term recurrence rates when used as sequential therapy after laser CO2 ablative therapy [441]. Sinecatechins is applied three times daily until complete clearance, or for up to 16 weeks. Clearance rates of 36–83% for podophyllotoxin solution and 43–70% for podophyllotoxin cream have been reported [436]. A systematic review and meta‐analysis confirmed the effectiveness of podophyllotoxin 0.5% solution relative to placebo (RR: 19.86, 95% CI: 3.88–101.65) [442]. Podophyllotoxin is self‐applied to lesions twice daily for three days, followed by four rest days, for up to four or five weeks. An RCT has also shown potassium hydroxide 5% to be an effective, safe and low-cost treatment modality for genital warts in men [443].
3.14.6.b. Physician-administered treatment
Physician-administered treatments included cryotherapy (79-88% clearance rate; 25–39% recurrence rate), surgical treatment (61–94% clearance rate), including excision, electrosurgery, electrocautery and laser therapy (75% clearance rate) [444, 445]. Physician-administered therapies are associated with close to 100% clearance rates, but they are also associated with high rates of recurrence, as they often fail to eliminate invisible HPV-infected lesions [444,445]. No data on the superiority of one treatment over another are available. However, among all interventions evaluated in a recent systematic review and network meta-analysis, surgical excision appeared to be the most effective treatment at minimising risk of recurrence [446].
3.14.6.c. Summary of evidence and recommendations for the treatment of anogenital warts
| Summary of evidence | LE |
| A Cochrane review of published RCTs found imiquimod to be superior to placebo in achieving complete clearance of warts. | 1b |
| In an RCT, sinecatechins 15% and 10% showed a complete clearance of all baseline and newly occurring warts in 57.2% and 56.3% of patients, respectively, versus 33.7% for placebo. | 1b |
| A systematic review and meta‐analysis confirmed the effectiveness of podophyllotoxin 0.5% solution relative to placebo. | 1b |
| A systematic review and meta-analysis reported that among all physician-applied therapies, surgical excision seemed to be the most effective at minimising risk of recurrence. | 1a |
| Recommendations | Strength rating |
| Use self-administered imiquimod 5% cream applied to all external warts overnight three times each week for 16 weeks for the treatment of anogenital warts. | Strong |
| Use self-administered sinecatechins 15% or 10% applied to all external warts three times daily until complete clearance, or for up to 16 weeks for the treatment of anogenital warts. | Strong |
| Use self-administered podophyllotoxin 0.5% self‐applied to lesions twice daily for three days followed by four rest days for up to four or five weeks for the treatment of anogenital warts. | Strong |
| Use cryotherapy or surgical treatment (excision, electrosurgery, electrocautery and laser therapy) to treat anogenital warts based on an informed discussion with the patient. | Strong |
3.14.7. Circumcision for reduction of HPV prevalence
Male circumcision is a simple surgical procedure that has been shown to reduce the incidence of STIs, including HIV, syphilis and HSV-2 [447]. Two systematic reviews and meta-analyses showed an inverse association between male circumcision and genital HPV prevalence in males [431,433]. It has been suggested that male circumcision could be considered as an additional one-time preventative intervention likely to reduce the burden of HPV-related diseases in both males and females, particularly among those countries in which HPV vaccination programmes and cervical screening are not available [433].
| Summary of evidence | LE |
| Two systematic reviews and meta-analyses showed an inverse association between male circumcision and genital HPV prevalence in males. | 1a |
| Recommendation | Strength rating |
| Discuss male circumcision with patients as an additional one-time preventative intervention for HPV-related diseases. | Strong |
3.14.8. Therapeutic vaccination
To date, three different vaccines against HPV have been licensed, but routine vaccination of males is currently implemented in only a few countries, including Australia, Canada, the United States and Austria. The aim of male vaccination is to reduce the rate of anal and penile cancers as well as head and neck cancers [420, 448].
A systematic review including a total of 5,294 patients reported vaccine efficacy against persisting (at least six months) anogenital HPV16 infections of 46.9% (28.6–60.8%) and against persisting oral infections of 88% (2–98%). A vaccine efficacy of 61.9% (21.4–82.8%) and 46.8% (20–77.9%) was observed against anal intraepithelial neoplasia grade 2 and 3 lesions, respectively [420]. The systematic review reported no meaningful estimates on vaccine efficacy against penile intraepithelial neoplasia grade 2 or 3, and no data were identified for anal, penile or head and neck squamous cell cancers [420].
A phase III clinical trial including 180 male patients evaluated the potential of MVA E2 recombinant vaccinia virus to treat intraepithelial lesions associated with papillomavirus infection [449]. The study showed promising results in terms of immune system stimulation against HPV lesions, as well as regression in intraepithelial lesions.
| Summary of evidence | LE |
| The role of therapeutic HPV vaccination in males in terms of effectiveness and safety is limited by the small number of relevant studies. | 2 |
| Therapeutic HPV vaccination in males is moderately effective against persistent anogenital HPV16 infection [(46.9% (28.6–60.8%)] and high-grade anal intraepithelial lesions [grade 2: 61.9% (21.4–82.8%); grade 3: 46.8% (20–77.9%)]. | 1b |
| Recommendation | Strength rating |
| Offer HPV vaccine to males after surgical removal of high-grade anal intraepithelial neoplasia. | Weak |
3.14.9. Prophylactic vaccination
A systematic review and meta-analysis reported that vaccination is moderately effective against genital HPV-related diseases, irrespective of an individual’s HPV status. However, higher vaccine efficacy was observed in HPV-naïve males, thus supporting the early vaccination of boys with the goal of establishing optimal vaccine-induced protection before the onset of sexual activity [420]. An RCT including 1,124 patients demonstrated high efficacy of the quadrivalent HPV vaccine versus placebo against HPV6/11/16/18-related persistent infections [450]. Moreover, the vaccine elicited a robust immune response and was well tolerated with mild vaccination-related adverse events, such as injection-site pain and swelling [450]. In addition, a Cochrane review demonstrated that the quadrivalent HPV vaccine appears to be effective in the prevention of external genital lesions and genital warts in males [451].
Despite the fact that quadrivalent HPV vaccines were approved for use in young adult males in 2010, vaccination rates have remained low at 10–15% [452]. Barriers to uptake in this patient group include lack of awareness about HPV vaccines and HPV-related diseases, concerns about vaccine safety and efficacy, economic/cost issues related to vaccine uptake, underestimation of HPV infection risks, and sexual activity [452]. Health care professionals should provide easily understood and accessible communication resources regarding these issues to educate young adult males and their families on the importance of HPV vaccination to reduce the incidence of certain cancers later in life [452, 453].
| Summary of evidence | LE |
| HPV vaccine is effective in the prevention of external genital lesions and genital warts in males. | 1a |
| HPV vaccination is moderately effective against genital HPV-related diseases irrespective of an individual’s HPV status, however, higher vaccine efficacy was observed in HPV-naïve males. | 1a |
| A systematic review of HPV vaccination barriers among adolescent and young adult males identified a number of barriers to vaccine uptake, including fear of side effects, limited HPV awareness, financial costs and changes in sexual activity. | 1b |
| An intervention study to evaluate whether electronic messaging can increase human papillomavirus vaccine completion and knowledge among college students concluded that intervention increased knowledge but not vaccine completion. | 2b |
| Recommendations | Strength rating |
| Offer early HPV vaccination to boys with the goal of establishing optimal vaccine-induced protection before the onset of sexual activity. | Strong |
| Apply diverse communication strategies to improve HPV vaccination knowledge in young adult males. | Strong |
Figure 3: Diagnostic and treatment algorithm for the management of HPV in males
3.15. Herpes simplex virus
3.15.1. Epidemiology, aetiology and pathogenesis
Genital herpes (GH) is a highly prevalent sexually transmitted, lifelong viral infection caused by the herpes simplex virus (HSV), which exists in two types: HSV-1 and HSV-2. Genital herpes accounts for up to 60% of genital ulcers, with approximately one-third caused by HSV-1 and two-thirds by HSV-2 [454]. Coinfection with both virus types occurs in up to 44.7% of patients [455]. The estimated seroprevalence of HSV-1 among healthy adults is 84.8%, while for HSV-2 it ranges from 10 to 12.44% [456-459]. Both HSV-1 and 2 result in chronic infection, with frequent reactivations and subclinical shedding, leading to a high risk of transmission to sexual partners.
Risk factors for GH include high sexual promiscuity, a large number of lifetime sexual partners, rural residence and unprotected sex [460, 461]. Increased prevalence is seen in intermediate- and high-risk subgroups, such as MSM, HIV-infected patients, those living with HIV-infected partners, STDI clinic attendees and transgender individuals [457-459].
3.15.2. Diagnosis
Clinical diagnosis of genital herpes can be challenging, as ulcerative lesions may be absent during evaluation. When present, the lesions are typically painful, erythematous, vesicular and recurrent. Less-common presentations include nodular, hypertrophic, verrucous, vegetative or exophytic lesions [462]. Whenever possible, GH diagnosis should be confirmed by laboratory testing [463]. Swabs should be taken from the lesion for type-specific virus identification using PCR or culture [462]. PCR detection of HSV DNA is rapid and more sensitive than traditional culture and immunoassay methods [464-466]. While PCR accuracy may vary, most tests available exhibit sensitivity and specificity > 90% [467, 468]. HSV serological assays, ideally using the Western blot technique, can detect antibodies against multiple HSV-1 and HSV-2 antigens, including glycoprotein G [469-471]. However, the Panel does not recommend serological testing for diagnosing localised genital HSV infections. During the last few years, rapid point-of-care tests for serological HSV diagnosis have become available [472,473]. Although these methods show high sensitivity and specificity, they do not provide quantitative results, making them less useful than laboratory tests. Insufficient evidence is available currently to recommend these rapid tests as the diagnostic standard in clinical practice. Serological testing is primarily indicated for detecting past HSV infections and is not recommended for diagnosing acute or recent infections, where virus detection from active lesions remains the preferred method.
3.15.3. Disease management
3.15.3.a. Systemic treatment
Antiviral drugs used to treat HSV include acyclovir, valacyclovir, pritelivir [474], amenamevir [475,476] and inosine pranobex [477]. Short-term recurrence rates are high, ranging from 27 to 48% [477]. Antiviral therapies reduce both the frequency and viral load during shedding [474, 478, 479]. With treatment, the rate of viral shedding decreases from 16% to approximately 5% [474], and the presence of visible lesions is reduced from 9% to approximately 1.2% [474]. While the optimal duration of treatment is not clearly defined, beneficial effects such as the reduction of ulcerative lesions and pain can be observed even after a single dose [476].
3.15.3.b. Topical treatment
Topical treatments play a limited role in managing GH, and their use is not recommended. Immunomodulation with resiquimod showed no improvement in outcomes and increased the incidence of local side effects [480]. CS21 (oxygenated glycerol triesters-based barrier genital gel) improved symptoms and slowed disease progression compared to both placebo and topical acyclovir [481]. Topical acyclovir did not outperform placebo in terms of disease progression [481].
3.15.3.c. Vaccination
Numerous studies have evaluated the efficacy of therapeutic vaccines for HSV treatment and transmission prevention [482-487]. However, there is currently insufficient evidence to support their use in clinical practice.
3.15.3.d. Surgical management
The impact of surgical interventions in patients with HSV remains unclear. Following circumcision, there may be an increase in viral shedding in the initial weeks due to inflammation [488]. Photodynamic therapy using a photosensitizer has shown potential in a small trial for treating recurrent lesions, however, further research is needed before it can be recommended in clinical practice [489].
3.15.4. Prevention, screening and contact tracing, follow-up
Glandular exposure through circumcision has been reported to prevent HSV infections [490]. A study on the pericoital application of tenofovir gel showed a reduced HSV-2 acquisition in women [491]. However, there is insufficient data to recommend any form of pre-exposure prophylaxis with HSV antiviral medications for prevention, and it should not be offered as a preventive strategy.
In accordance with most national and international guidelines, the Panel does not recommend routine herpes testing for asymptomatic individuals. Screening is recommended only in the presence of genital symptoms potentially related to HSV or if a sexual partner has GH.
Follow-up testing is not indicated, except when there are recurrent signs and symptoms. Counselling of individuals with GH and their sexual partners is of mandatory importance to prevent transmission and cope with the infection.
| Level of evidence | LE |
| Genital herpes accounts for up to 60% of genital ulcers with approximately one-third caused by HSV-1 and two-thirds by HSV-2. | 2b |
| Detection of HSV DNA via PCR is rapid and more sensitive than traditional culture and immunoassay methods. | 2a |
| Antiviral therapies reduce both the frequency and viral load during viral shedding. | 1b |
| Viral shedding may temporarily increase in the weeks following circumcision due to inflammation. | 3 |
| Topical antiviral therapy does not improve clinical outcomes of HSV patients or increase local side effects, but can reduce the local viral load. | 1b |
| Recommendations | Strength rating |
| Obtain a comprehensive medical history, including history of previous sexual contacts, from all patients presenting with genital ulcers potentially related to HSV. | Strong |
| Confirm the diagnosis with a clinical swab and type-specific virologic testing, such as PCR or culture, from the lesion. | Strong |
| Treat the first clinical episode of genital HSV infection. | Strong |
Table 12: Treatment regimens for genital HSV infection
| Antimicrobials | Dosage |
| Recommended therapy and dose for first clinical episode HSV | |
| Aciclovir | 400mg orally t.i.d for 10 days OR 200mg orally five times daily for 10 days. |
| Valaciclovir | 500mg orally b.i.d for 10 days. |
| Recommended therapy and dose for recurrent genital HSV | |
| Aciclovir | 400mg orally t.i.d for five days OR 800mg b.i.d for five days OR 800mg t.i.d for two days. |
| Valaciclovir | 500mg orally b.i.d for 3 days. |
t.i.d = three times daily; b.i.d = twice daily.
3.16. Genitourinary tuberculosis
3.16.1. Epidemiology, aetiology and pathophysiology
An estimated 246,000 new and relapse tuberculosis (TB) cases occurred in the WHO European Region in 2019, with 49,752 of these cases occurring within the 31 countries comprising the European Union (EU)/European Economic Area (EEA) region [492]. An estimated 12.0% of incident TB cases in 2019 were coinfected with HIV. Extrapulmonary TB was notified on average for 16.6% of all incident TB cases in the region. Eleven countries reported more than 30% of their TB cases having extrapulmonary localisation. The proportion of TB that is extrapulmonary is significantly greater among migrants from, for example, Southeast Asia, Sub-Saharan Africa and Eastern Europe and Central Asia than non-migrants across the European Union (EU)/European Free Trade Association (EFTA). Genitourinary tuberculosis (GUTB) accounted for 4.6% of extrapulmonary TB cases in the EU between 1997 and 2017 [493]. Tuberculosis is an infectious disease caused by a group of Mycobacterium species called the Mycobacterium tuberculosis complex (MBTC) [494]. Genitourinary TB can affect all genitourinary organs and is almost always secondary due to the hematogenous spread of chronic latent TB infection (LTBI) [495]. Risk factors include primary and latent TB infection, diabetes, old age, low BMI, oncological comorbidities, immune suppression (including HIV), renal failure and poor socioeconomic living conditions. The risk of reactivation is estimated to be up to 15% during one’s lifetime [496]. The WHO recommend either a tuberculin skin test (TST) or interferon-gamma release assay (IGRA) for the diagnosis LTBI [497].
3.16.2. Diagnosis
Patients generally present with non-specific urological complaints for which no obvious cause is identified, including haematuria; increased urinary frequency; difficulty voiding; abdominal, lumbar and suprapubic pain; and, in female patients, menstrual irregularities and pelvic pain. Patients may also present for infertility issues, however, infertility and TB will not be addressed in detail in this text. The diagnosis of GUTB is challenging, as no single diagnostic test exists. Diagnosis relies on a high suspicion of infection based on patient history; microbiological, molecular and histological testing; and imaging findings.
3.16.2.a. Smear microscopy
Smear microscopy is a simple and cost-effective way of detecting the presence of acid-fast bacilli (AFB) in urine samples, semen, tissue specimens, pus or discharged or prostatic massage fluid, through microscopic examination using Ziehl-Neelsen or auramine staining [498,499]. A major limitation of smear microscopy is its low sensitivity (ranging from 0 to 25%) in urine [500, 501].
3.16.2.b. Culture
The culture-based method (both solid and liquid media) for biological specimens is the reference standard for M. tuberculosis isolation from biological samples. Three midstream, first-void urine samples on consecutive days are recommended for TB culture [499]. A disadvantage of culture-based methods is the long incubation period needed for results at least nine to ten days for positive results and six weeks to be considered negative, as well as the need for highly equipped laboratories. In addition, studies have reported high specificities of 92–100%, but low sensitivities 23.3–30% for urine culture in renal TB specimens [502, 503].
3.16.2.c. Nucleic acid amplification tests
In recent years, nucleic acid amplification tests (NAATs) have been introduced in the diagnostic pathway of TB to shorten diagnostic work-up. In 2021, the WHO issued an update to its guidelines for the rapid diagnosis of TB, in which they made a conditional recommendation that, in patients with signs and symptoms of extrapulmonary TB, Xpert MTB/RIF may be used as an initial diagnostic test [504]. Xpert MTB/RIF pooled sensitivity and specificity were 84.7% (70.8–93.1) and 97.3% (91.0–99.2) for the diagnosis of genitourinary TB [505]. The 2021 WHO guidelines also contain recommendations for additional PCR testing systems, as well as moderate complexity automated NAATs [504].
Note: Several other diagnostic tests are currently under investigation by the WHO but cannot be recommended for the diagnosis of GUTB at this time.
3.16.2.d. Imaging
Imaging modalities aid in the localisation of the foci of infection in GUTB and in the assessment of the extent of the damage to the genitourinary system. Imaging techniques for the diagnosis of GUTB have a sensitivity of approximately 90% [506]. However, the quality of the evidence available for diagnostic imaging of TB is low to very low, and further studies are required to enable the Panel to make recommendations on this topic.
Ultrasound is a cost-effective and noninvasive imaging modality that has been shown to be effective for the diagnosis of testicular, epididymal and vas deferens TB [507-511]. Ultrasound examination may also allow for the identification of parenchymal masses, cavities, mucosal thickening of the collecting system and bladder, stenosis and consecutive obstruction of the collecting system, vesicoureteral reflux, and calcifications [512]. In female GUTB patients, ultrasound may identify ovarian masses, intrauterine thickening and calcifications [513].
Intravenous urography aids in the identification of renal and ureteral TB but lacks specificity. Approximately 10–15% of patients may have normal findings on intravenous urogram (IVU) [514, 515]. The most common findings on IVU are hydrocalycosis, hydronephrosis or hydroureter due to stricture, autonephrectomy and urinary calcifications [516-518].
In recent years, CT and MRI have largely replaced IUV. The most common findings on CT are parenchymal scarring, hydrocalycosis, hydronephrosis or hydroureter due to stricture, and thickening of the renal pelvis, ureter and bladder walls [516-518]. In TB of the seminal vesicles and vas deferens, CT imaging can show enlarged heterogeneously enhancing seminal vesicles with possible wall thickening, contraction and intraluminal or wall calcifications [519, 520]. Prostate TB appears as a low attenuating and marginally enhancing cystic mass that is indistinguishable from a non-TB prostatic abscess [521]. In female GUTB, the fallopian tubes are most frequently affected area and present with enlargement, hydrosalpinx, pyosalpinx and wall-thickening, with calcification on CT [522].
Magnetic resonance imaging has low sensitivity for the diagnosis of GUTB in the early stages of the infection [523]. As an imaging modality, MRI is useful in patients in whom CT is contraindicated, including patients with renal failure or contrast hypersensitivity reactions or those who wish to avoid exposure to radiation. Renal and ureteral abnormalities are comparable to those described for CT findings and must be distinguished from acute pyelonephritis [513, 524]. Epididymitis and testicular TB appears as a diffusely enlarged epididymis or testis with heterogeneous high T2 signal due to fibrosis and calcification [520]. Multiparametric MRI of the prostate distinguishes between the nodular or diffuse patterns of prostate TB [525].
Female GUTB has a wide range of appearances on hysterosalpingogram (HSG) affecting the fallopian tubes, endometrium and uterus [526, 527]. Tubal obstruction is the most common finding with HSG [527]. In addition, deformity of the uterine cavity can be observed, such as a T-shaped and dwarfed uterus, resulting from abnormal scarring and fibrosis [526]. As the disease progresses this process can potentially lead to a complete obliteration of the uterine cavity referred to as Netter syndrome [528].
3.16.3. Medical treatment
The WHO recommends a six-month daily regimen for treatment of newly diagnosed extrapulmonary TB, including an intensive phase of two months with isoniazid, rifampicin, pyrazinamide and ethambutol, followed by a continuation phase of four months with isoniazid and rifampicin [529]. For the treatment of multidrug-resistant (MDR) TB (i.e. resistance to rifampicin and isoniazid), an individualised treatment regimen should be applied with at least five effective tuberculosis medicines during the intensive phase, including pyrazinamide and four core second-line tuberculosis medicines [530].
3.16.4. Surgical treatment
Combination drug therapy is the first-line treatment for GUTB. However, in more than 50% of patients, ablative, endoscopic or reconstructive surgery is required, due to the destructive nature of the infection coupled with a delay in initial diagnosis [531-533]. A total of 26.9% of cases of diagnosed GUTB involve a non-functioning unilateral kidney and 7.4% involve renal failure [533].
In the largest observational study of 4,288 GUTB patients, a total of 2,364 different surgical procedures were carried out, 948 of which were reconstructive [534]. In a retrospective series of 241 patients who underwent surgery for GUTB, a total of 128 reconstructive procedures were performed, in which 30.29% of patients had bladder augmentation [535]. A retrospective single-centre study of 128 patients reported that kidneys affected by TB in the reconstruction group had a 5.44-times longer survival than the permanent diversion group, suggesting that, when feasible, renal reconstruction may be better for renal function preservation [536]. Reconstructive surgery may include augmentation cystoplasty, ureteroureterostomy, ureteroneocystostomy, ureteral reimplant, pyeloplasty, ureterocalicostomy and ileal ureter or external diversion, where indicated [537].
Limited evidence is available with regard to the optimum surgical approach. Minimally invasive options have been reported as feasible and safe strategies as compared to open surgery [538-542]. In addition, the optimal timing for surgery is controversial. A delay of two to six weeks up to nine months after the initiation of medical treatment has been proposed to allow for a reduction in active inflammation and stabilisation of the TB lesions [523].
Due to a lack of high-quality evidence for surgical treatment of GUTB, the Panel is unable to currently give a recommendation on surgical treatment. Patients with GUTB should be assessed on an individualised basis and the decision to operate taken depending on the location, extent of disease progression and damage to the genitourinary system.
3.16.5. Summary of evidence and recommendations for the diagnosis and treatment of GUTB
| Summary of evidence | LE |
| The risk of reactivation of latent TB is estimated to be 15% in an individual’s lifetime. | 2a |
| Smear microscopy for acid-fast bacilli has a low sensitivity in urine ranging from 0 to 25%. | 2a |
| Studies have reported high specificities of 92–100% but low sensitivities of 23.3–30% for urine culture in renal TB specimens. | 2a |
| Xpert MTB/RIF pooled sensitivity and specificity were 84.7% (70.8–93.1) and 97.3% (91.0–99.2) for the diagnosis of GUTB. | 1b |
| Standard six-month antituberculous drug regimens are effective in all forms of TB (pulmonary and extrapulmonary). | 1a |
| There is limited evidence with regard to the optimum surgical approach and timing of surgery in GUTB patients. | 3 |
| Recommendations | Strength rating |
| Diagnosis | |
| Take a full medical history, including history of previous tuberculosis (TB) infection (pulmonary and extrapulmonary), from all patients presenting with persistent non-specific genitourinary symptoms and no identifiable cause. | Strong |
| Perform smear microscopy on urine, semen, tissue specimens and discharged or prostatic massage fluid using Ziehl-Neelsen (ZN) or auramine staining in patients with suspected genitourinary tuberculosis (GUTB). | Weak |
| Perform an acid-fast bacilli culture on three midstream first-void urine samples, on three consecutive days for M. tuberculosis isolation in patients with suspected GUTB. | Strong |
| Use a recommended PCR test system in addition to the microbiological reference standard (MRS) in urine specimens as a diagnostic test in patients with signs and symptoms of GUTB. | Weak |
| Use imaging modalities in combination with culture and/or PCR to aid in the diagnosis of GUTB and to assess the location and extent of damage to the genitourinary system. | Weak |
| Treatment | |
| Use medical treatment as first-line treatment for GUTB. | Strong |
| Use a daily six-month regimen for treatment of newly diagnosed GUTB. This should include an intensive phase of two months with isoniazid, rifampicin, pyrazinamide and ethambutol, followed by a continuation phase of four months with isoniazid and rifampicin. | Strong |
| Treat multidrug-resistant TB with an individualised treatment regimen including at least five effective tuberculosis medicines during the intensive phase, including pyrazinamide and four core, second-line tuberculosis medicines. | Strong |
Table 13: Treatment regimens for newly diagnosed GUTB and MDR-TB [530]
| Antimicrobials | Dosage |
| Six-month regimen for treatment of newly diagnosed GUTB | |
| Intensive two-month phase | |
| Isoniazid | 5mg/kg q.d; max. daily dosage 300mg |
| Rifampicin | 10mg/kg q.d; max. daily dosage 600mg |
| Pyrazinamide | 25mg/kg q.d; max. daily dosage 2,000mg |
| Ethambutol | 15–20mg/kg q.d; max. daily dosage ranging from 800mg to 1,600mg, depending on body weight |
| Continuation four-month phase | |
| Isoniazid | 5mg/kg q.d; max. daily dosage 300mg |
| Rifampicin | 10mg/kg q.d; max. daily dosage 600mg |
| Treatment regimen for multidrug-resistant TB | |
| Treat multidrug-resistant TB with an individualised treatment regimen including at least five effective tuberculosis medicines during the intensive phase, including pyrazinamide and four core, second-line tuberculosis medicines*. | |
Group A Fluoroquinolones | Levofloxacin, moxifloxacin and gatifloxacin |
Group B Second-line injectables | Amikacin, capreomycin, kanamycin and streptomycin** |
Group C Other second-line agents | Ethionamide/ prothionamide, cycloserine/terizidone, linezolid and clofazimine |
Group D Add-on agents (not part of the core MDR-TB regimen) | D1: Pyrazinamide, ethambutol and high-dose isoniazid D2: Bedaquiline and delamamid D3: p-aminosalicylic acid, imipenem-cilastatin, meropenem, amoxicillin-clavulanate and thioacetazone*** |
b.i.d. = twice daily; t.i.d. = three times daily; q.d. = every day; o.d. = once daily.
* Drugs should be chosen as follows: one from group A, one from group B and at least two from group C. If the minimum number of five TB medicines cannot be composed from drugs included in Groups A to C, an agent from group D2 and other agents from group D3 can be added to bring the total to five [530].
** Streptomycin can substitute other injectable drugs if none of these agents can be used and if the strain is shown not to be resistant [530].
** Thioacetazone should not be used if the patient is HIV seropositive .
3.17. Fungal urinary tract infection
3.17.1. Epidemiology and risk factors
Only 2–11% of patients with funguria present with symptoms of a UTI [543, 544]. The prevalence of funguria is considerably higher in hospital-acquired settings than in outpatient environments, ranging from < 1% in outpatients to 5–10% of positive urine cultures in hospitalised patients [545,546]. In a large multicentre European study evaluating urine cultures of UTIs, Candida species were the third most common pathogen, accounting for 9.4% of specimens [547]. The incidence of funguria and fungal UTI is particularly high among ICU patients but is subject to regional variations [548, 549].
Candida albicans is the predominant fungal uropathogen accounting for 50–70% of all urinary fungal isolates, while Candida glabrata and Candida tropicalis together account for 10–35%. However, their prevalence varies depending on geographic region and patient population [543, 550]. Less common species include Candida parapsilosis and Candida krusei [543]. Candidozyma auris, an emerging multidrug-resistant pathogen associated with high mortality, is only rarely detected in urine [551-553]. Isolation of non-Candida fungal species from urine cultures is uncommon.
Well-established risk factors for fungal UTIs include female sex, advanced age and comorbidities, such as diabetes mellitus, urinary tract abnormalities, chronic liver disease, renal insufficiency, malignancy, immunosuppression and malnutrition [554-557]. Fungal UTIs are strongly associated with urinary tract instrumentation and indwelling catheters or drains [543, 558-560]. After radical cystectomy, Candida spp. rank among the second to third most common pathogens in postoperative UTIs [561,562]. One further risk factor is prior exposure to broad-spectrum antibiotics or antifungals [554, 563].
3.17.2. Clinical presentation and diagnosis
3.17.2.a. Clinical presentation
The presence of fungi in urine represents a spectrum of clinical conditions from asymptomatic colonisation to systemic infection such as life-threatening urosepsis. Most fungal UTIs present as localised UTIs (i.e. cystitis). Since Candida spp. adhere poorly to the bladder urothelium, cystitis usually requires predisposing factors, such as urinary obstruction, immunosuppression or biofilm formation on indwelling devices. Infection may extend to the upper urinary tract either by ascending spread from the bladder, particularly in case of reflux or obstruction (including fungal balls), resulting in fungal pyelonephritis, or by hematogenous dissemination with direct invasion of the renal tubules [545].
3.17.2.b. Symptoms
In patients with funguria, only 2–11% present with typical UTI symptoms [543, 544]. Fungal UTI should only be considered in symptomatic cases. However, many patients with funguria are in intensive care or long-term care settings, where symptom assessment is unreliable. These patients are frequently unable to report symptoms, and long-term catheters obscure typical signs such as frequency or dysuria [564].
3.17.2.c. Urine culture and urinalysis
The diagnostic standard is urine culture, although colony count thresholds distinguishing colonisation from infection remain undefined [564,565]. Similar to bacterial UTIs, a cut-off of ≥ 105 CFU/mL is frequently reported in studies [554, 566-570]. Susceptibility testing should be performed, particularly in non-albicans Candida spp.
It should be noted that some Candida spp., such as Candida glabrata, may grow poorly on routine media or require prolonged incubation, and additional diagnostic measures (e.g. subculturing on selective fungal media or molecular identification) may therefore be warranted when infection is suspected but cultures remain negative [571].
Repeating cultures using a freshly catheterised specimen or after device removal can help to distinguish contamination from funguria, while the presence of symptoms confirms infection [567].
Pyuria may suggest infection but lacks specificity, particularly in catheterised or elderly patients. The greatest value of pyuria therefore lies in its negative predictive ability, as the absence of pyuria makes a UTI unlikely, and instead suggests that a positive culture result is due to contamination [572, 573].
3.17.2.d. Alternative markers
Several studies have assessed additional diagnostic markers for fungal UTIs, but none have demonstrated sufficient clinical utility. Elevated Interleukin-17 levels correlate with funguria, but fails to distinguish colonisation and infection and therefore has no diagnostic role [544].
3.17.2.e. Imaging
Imaging is reserved for symptomatic patients to identify infection site and complications. Ultrasound can detect pyelonephritis, abscesses, fungal balls and hydronephrosis. Typical findings may include calyceal wall thickening, increased renal vascularity or loss of corticomedullary differentiation. Fungal balls appear as echogenic debris without Doppler flow, while hydronephrosis suggests obstruction [574]. Computed tomography urography provides greater sensitivity for pyelonephritis, abscesses, emphysematous infections and obstruction, while MRI with contrast or renal cortical scintigraphy (Tc-99m DMSA) can be used in selected cases [565,575].
3.17.3. Treatment
3.17.3.a. Asymptomatic funguria
Asymptomatic funguria generally requires no antifungal treatment, but should be considered in neutropenic patients, infants with birth weights < 1,500g or before urological intervention breaching the mucosa [576, 577]. Patients with asymptomatic candiduria may have an underlying disorder or defect.
3.17.3.b. Treatment of fungal urinary tract infections
Nonantimicrobial measures should be implemented whenever necessary. For example, catheter replacement alone can lead to a significant clearance of funguria [578]. In cases of urinary tract obstruction, prompt drainage with ureteral stent placement or percutaneous nephrostomy should be performed [579].
Treatment depends on the isolated species and its susceptibility pattern recommended agents include fluconazole, amphotericin B deoxycholate and oral flucytosine, with weight-based dosing critical for efficacy [576, 580]. Species-specific resistance must be considered. C. krusei and C. glabrata are > 90% fluconazole-resistant and a species-specific resistance can be assumed. Clinical data indicate that C. glabrata UTI can only be treated with fluconazole if the isolate is confirmed to be fluconazole-susceptible, as resistance rates are high and vary by region and patient population. Large surveillance studies have shown that C. glabrata isolates from urine often have higher fluconazole minimum inhibitory concentrations (MICs) than those from other sites, with resistance rates in some cohorts exceeding 15% and a significant proportion of isolates falling into the susceptible-dose dependent (SDD) category, requiring higher dosing for efficacy [581].
Moreover, substance specifics must be considered. For example, echinocandins (e.g. caspofungin) have poor urinary penetration and are unsuitable for fungal cystitis but achieve high renal tissue levels, making them useful for pyelonephritis. However, the echinocandin micafungin can reach therapeutic levels in bladder tissue despite low urinary excretion and could be considered for treatment of fungal cystitis in exceptional cases such as infections by fluconazole-resistant species such as C. glabrata, C. krusei and C. auris [582-584].
Empirical therapy usually consists of a two-week course of oral fluconazole, starting with a one-time double loading dose on the first day, followed by a daily maintenance dose [576]. In a retrospective single-centre study of hospitalised adults with fungal UTI (n = 103), clinical success rates were similar for 14-day (93.3%, 42/45) and shorter fluconazole courses (< 14 days) (93.1%, 54/58), suggesting shorter antifungal regimens might be sufficient. However, subgroup analysis distinguishing local from systemic UTI was not performed [585]. Other azoles, such as ketoconazole and voriconazole, achieved urine clearance of candiduria in small case series only [586, 587]. Rising fluconazole resistance and non-fluconazole-susceptible Candida spp. are being detected more frequently [566, 588]. One laboratory-based study reported a significant increase in the rate of fluconazole resistance among Candida isolates obtained from urine specimens, increasing from 6.8% in 2010–2011 to 29.5% in 2012–2013, with persistently elevated rates thereafter [581]. This highlights the need for alternative treatments.
Bladder irrigation with amphotericin B deoxycholate is described as an alternative option for fungal cystitis. A meta-analysis published in 2009 found 80–90% fungal clearance within 24 hours and superiority to fluconazole (OR 0.57, 95% CI: 0.32–1.00) but included only asymptomatic funguria. [589]. In one study of 95 patients with fungal cystitis, amphotericin B deoxycholate bladder instillations (25mg in 500mL dextrose in water at 42mL/hour) for a median of five days (range 2–11) achieved 80% eradication [568]. In an RCT of fungal UTI, amphotericin B deoxycholate bladder instillation achieved early clearance in 96% of patients compared with 73% for fluconazole. However, one-month mortality was higher in the amphotericin group (41% vs. 22%), suggesting local therapy may be linked to poorer survival [568]. Continuous irrigation with 50mg/L amphotericin B deoxycholate for five days may be considered in localised fluconazole-resistant cases. Careful patient selection is necessary to exclude upper tract or systemic spread before use.
In a retrospective study (n = 33), micafungin showed a high rate of microbiological eradication for C. albicans, C. glabrata and C. krusei in candiduria and UTI, though no UTI-specific analysis was performed [570].
3.17.4. Sodium-glucose cotransporter 2 inhibitors and fungal cystitis
Sodium-glucose cotransporter 2 (SGLT2) inhibitors may increase the risk of urogenital infections, particularly genital mycotic infections [590], due primarily to their mechanism of action, which increases urinary glucose excretion (glucosuria), creating a nutrient-rich environment that can promote bacterial growth in the urinary tract [591]. However, the overall increase in UTI risk with SGLT2 inhibitors is modest and not consistently observed across all agents or patient populations. Most meta-analyses and large cohort studies show no significant difference in UTI rates compared to other antidiabetic drugs, except for a dose-dependent increase with dapagliflozin [590, 592-594]. Fungal cystitis has been rarely reported. One retrospective observational cohort study of type 2 diabetic patients with advanced chronic kidney disease under either SGLT2 inhibitors or control treatment found a UTI rate of 19.9% in the SGLT2 inhibitor arm versus 24.0% in the control arm. Overall, the majority of UTIs were caused by Gram-negative organisms (n = 752), followed by mixed growth (n = 663), Gram-positive (n = 162) and fungal organisms (n = 160). The SGLT2 inhibitors group developed a lower rate of Gram-negative organisms related UTI (p < 0.001) but a higher rate of fungal UTI, which was predominantly caused by Candida spp. infection (p < 0.001) [595]. In an insurance-based study comparing SGLT2 inhibitors with glucagon-like peptide-1 receptor agonists, Candida and non-Candida UTI rates were 2.2% and 6.3% among patients on SGLT2 inhibitors, versus 0.9% and 8.0% among those on glucagon-like peptide-1 receptor agonists [596].
| Summary of evidence | LE |
| Only 2–11% of patients with funguria present with symptoms of a UTI. The incidence of funguria and fungal UTI is particularly high among ICU patients. | 2b |
| Evaluation of classical UTI symptoms combined with urine culture remains the primary diagnostic approach. | 2a |
| Asymptomatic funguria usually does not require treatment, except in neutropenic patients, infants with very low birth weight or those undergoing mucosa breaching urological procedures. | 2b |
| Empirical therapy typically consists of oral fluconazole for two weeks, starting with a double loading dose on day one, followed by daily maintenance. Rising fluconazole resistance and non-fluconazole-susceptible Candida spp. are being detected more frequently. | 1b |
| Recommendations | Strength rating |
| Take a full medical history, including UTI symptoms and risk factors for fungal UTI. | Strong |
| Perform urine culture for species identification and susceptibility testing, particularly in non-albicans Candida UTI. | Strong |
| Replace indwelling catheters or stents for significant clearance of funguria. | Weak |
| Treat localised fungal UTI with a two-week course of oral fluconazole. | Strong |
| A treatment duration of less than two weeks can be used in localised fungal UTI (i.e. cystitis) without risk factors. | Weak |
| Treat fluconazole-resistant Candida spp. in localised UTI with amphotericin B deoxycholate instillations, amphotericin B deoxycholate instillations IV, flucytosine or capsofungin, taking in account comorbidities and kidney function. | Strong |
| Treat fluconazole-resistant Candida spp. in systemic UTI with amphotericin B deoxycholate instillations IV, flucytosine or capsofungin, taking in account comorbidities and kidney function. | Strong |
| Localised fungal UTI | |||
| Pathogens | Antimicrobial | Dose & Duration of therapy | Renal function correction |
| Fluconazole-susceptible species | Fluconazole |
|
|
Fluconazole-resistant species | Amphotericin B deoxycholate | Bladder instillations: 200ml/L in a 100ml infusion bag of 5% glucose, three times daily for seven days. | No dosage adjustment required |
| Fluconazole-susceptible species | Flucytosine |
|
|
| Fluconazole-susceptible species | Amphotericin B deoxycholate |
| No dosage adjustment required |
| Caspofungin | 70mg IV day 1, then 50mg daily for two to three weeks. | No dosage adjustment required | |
| Systemic fungal UTI | |||
| Fluconazole-susceptible species | Fluconazole |
|
|
| Fluconazole-resistant species | Amphotericin B deoxycholate |
| No dosage adjustment required |
| Flucytosine |
|
| |
| Caspofungin | 70mg IV day 1, then 50mg daily for two to three weeks | No dosage adjustment required | |
b.i.d = twice daily; CrCI = creatinine clearance; IV = intravenous; p.o. = orally.
3.18. Periprocedural antibiotic prophylaxis
3.18.1. General principles
3.18.1.a. Definition of infectious complications
The European Centre for Disease Prevention and Control (ECDC) and the United States Centers for Disease Control and Prevention (CDC) have both presented similar definitions recommended for the evaluation of infectious complications [597, 598].
3.18.1.b. Nonantibiotic measures for asepsis
A number of nonantibiotic measures have been designed to reduce the risk of surgical site infection (SSI), many of which are historically part of the routine of surgery. The effectiveness of measures tested by RCTs are summarised in systematic reviews conducted by the Cochrane Wounds Group (http://wounds.cochrane.org/news/reviews). Urological surgeons and the institutions in which they work should consider and monitor maintenance of an aseptic environment to reduce risk of infection from pathogens within patients (microbiome) and from outside the patient (nosocomial/healthcare-associated). This should include the use of correct methods of instrument cleaning and sterilisation, frequent and thorough cleaning of operating rooms and recovery areas, and thorough disinfection of any contamination. The surgical team should prepare to perform surgery by effective handwashing [599], donning of appropriate protective clothing and maintenance of asepsis. These measures should continue as required in recovery and ward areas.
Patients should be encouraged to shower preoperatively, but the use of chlorhexidine soap does not appear to be beneficial [600]. Although evidence quality is low, any required hair removal appears best done by clipping – as opposed to shaving – just prior to incision [601]. Mechanical bowel preparation should not be used, because evidence review suggests harm rather than benefit [602, 603]. Some weak evidence is available showing that skin preparation using alcoholic solutions or chlorhexidine result in a lower rate of SSI than iodine solutions [604]. Studies on the use of plastic adherent drapes showed no evidence of benefit in reducing SSI [605].
3.18.1.c. Detection of bacteriuria prior to urological procedures
Identifying bacteriuria prior to diagnostic and therapeutic procedures aims to reduce the risk of infectious complications by controlling any preoperatively detected bacteriuria and to optimise antimicrobial coverage in conjunction with the procedure. A systematic review of the evidence identified eighteen studies comparing the diagnostic accuracy of various index tests (dipstick, automated microscopy, dipslide culture and flow cytometry) with urine culture as the reference standard [606]. The systematic review concluded that none of the alternative urinary investigations for the diagnosis of bacteriuria in adult patients prior to urological interventions can currently be recommended as an alternative to urine culture [606].
3.18.1.d. Choice of agent
Urologists should have knowledge of local pathogen prevalence for each type of procedure, their antibiotic susceptibility profiles and virulence to establish written local guidelines. These Guidelines should cover the five modalities identified by the ECDC following a systematic review of the literature [607]. The agent should ideally not be one that may be required for treatment of infection. When risk of skin wound infection is low or absent, an aminoglycoside (gentamicin) should provide cover against likely uropathogens, provided the eGFR is > 20mL/min; second generation cephalosporins are an alternative [608]. Recent urine culture results, including presence of any multidrug-resistant organisms, drug allergy, history of C. difficile-associated diarrhoea, recent antibiotic exposure, evidence of symptomatic infection pre-procedure, and serum creatinine should be checked. The Panel have decided not to make recommendations for specific agents for particular procedures, because there is considerable variation in Europe and worldwide regarding bacterial pathogens, their susceptibility and availability of antibiotic agents.
3.18.2. Specific procedures and evidence question
An updated literature search from February 2017 (cut-off of last update) to June 2021 identified RCTs, systematic reviews and meta-analyses that investigated the benefits and harms of using antibiotic prophylaxis prior to specific urological procedures. The available evidence enabled the panel to make recommendations concerning urodynamics, cystoscopy, stone procedures (extracorporeal shockwave lithotripsy [ESWL], ureteroscopy and percutaneous nephrolithotomy [PCNL]), transurethral resection of the prostate (TURP) and transurethral resection of the bladder (TURB). For nephrectomy and prostatectomy, the scientific evidence was too weak to allow the Panel to make recommendations either for or against antibiotic prophylaxis. The general evidence question was: Does antibiotic prophylaxis reduce the rate of postoperative symptomatic UTI in patients undergoing each named procedure?
3.18.2.a. Urodynamics
The literature search identified one systematic review for antibiotic prophylaxis in females only [609]. This included three RCTs (n = 325 patients) with the authors reporting that prophylactic antibiotics reduced the risk of bacteriuria but not clinical UTI after urodynamics [609]. A previous Cochrane review identified nine RCTs enrolling 973 patients with overall low quality and high or unclear risks of bias [610]. The outcome of clinical UTI was reported in four trials, with no benefit found for antibiotic prophylaxis versus placebo [RR (95% CI) 0.73 (0.52–1.03)]. A meta-analysis of nine trials showed that the use of antibiotics reduced the rate of post-procedural bacteriuria [RR (95% CI) 0.35 (0.22–0.56)] [610].
3.18.2.b. Cystoscopy
Three systematic reviews and meta-analyses [611-613] and one additional RCT [614] on cystoscopy for stent removal were identified. Garcia-Perdomo et al. included seven RCTs with a total of 3,038 participants. The outcome of symptomatic UTI was measured by five trials of moderate overall quality and meta-analysis showed a benefit for using antibiotic prophylaxis [RR (95% CI) 0.53 (0.31–0.90)]; ARR 1.3% (from 2.8% to 1.5%) with a NNT of 74 [612]. This benefit was not seen if only the two trials with low risk of bias were used in the meta-analysis. Carey et al. included seven RCTs with 5,107 participants. Six trials were included in meta-analysis of the outcome of symptomatic bacteriuria, which found benefit for use of antibiotic prophylaxis [RR (95% CI) 0.34 (0.27–0.47)]; ARR 3.4% (from 6% to 2.6%) with NNT of 28 [611]. Zeng et al. included twenty RCTs and two quasi-RCTs with a total of 7,711 participants. The outcome of symptomatic UTI was measured by eleven RCTs of low overall quality and meta-analysis showed a possible benefit for using antibiotic prophylaxis [RR 95% CI 0.49 (0.28–0.86)] [613]. For systemic UTI, antibiotic prophylaxis showed no effect compared with placebo or no treatment in five RCTs [RR (95% CI) 1.12 (0.38–3.32)]. However, prophylactic antibiotics may increase bacterial resistance [RR (95% CI) 1.73 (1.04–2.87)].
Given the low absolute risk of postprocedural UTI in well-resourced countries, the high number of procedures being performed, and the high risk of contributing to increasing antimicrobial resistance, the Panel consensus was to strongly recommend not to use antibiotic prophylaxis in patients undergoing urethrocystoscopy (flexible or rigid).
3.18.2.c. Interventions for urinary stone treatment
3.18.2.c.1. Extracorporeal shockwave lithotripsy
For patients without bacteriuria undergoing ESWL, two systematic reviews and meta-analyses were identified with latest search dates of November 2011 and October 2012, respectively [615, 616], and two further trials [617]. Lu et al. included nine RCTs with a total of 1,364 patients and found no evidence of benefit in terms of reducing the rate of postprocedural fever or bacteriuria [615]. Mrkobrada et al., included eight RCTs with a total of 940 participants and found no evidence of benefit for antibiotic prophylaxis to reduce rate of fever or trial-defined infection [616]. An RCT with 274 patients and severe risk of bias found no reduction in fever at up to one week post-procedure using a single dose of levofloxacin 500mg and no difference in the rate of bacteriuria [617]. Another RCT (n = 600), again with severe risk of bias, found no difference in UTI and positive urine culture rates at two weeks post-procedure using 200mg ofloxacin postoperatively for three days versus placebo [618].
For patients with bacteriuria or deemed at high risk of complications, one RCT comparing the use of ofloxacin or trimethoprim-sulfamethoxazole for three days prior and four days subsequent to ESWL in 56 patients with ureteric stents was identified [619]. The RCT found no difference in rate of clinical UTI at seven days (no events) and no difference in post-ESWL bacteriuria.
A large RCT (n = 1,694) evaluated single-dose ciprofloxacin for preventing postoperative infectious complications [620]. The composite primary outcome occurred in 2.7% in the ciprofloxacin group versus 3.9% in the placebo group (RR 0.68, 95% CI: 0.41–1.15), indicating no statistically significant difference. Symptomatic UTI was observed in 1.3% versus 2.7% (RR 0.49, 95% CI: 0.19–1.23), again without statistical significance and with wide confidence intervals. Pyelonephritis occurred in none of the ciprofloxacin patients and in nine placebo patients (1.2%; RR 0.05, 95% CI: 0.003–0.93), suggesting a possible effect but with a very small absolute risk reduction. No urosepsis or deaths occurred. Overall, the absolute benefit of single-dose ciprofloxacin is limited, and its use must be interpreted in the context of the legally binding EU restrictions on fluoroquinolones (see Section 3.4.4.b and EMA Article-31 referral [154]) and antimicrobial stewardship.
3.18.2.c.2. Ureteroscopy
One updated systematic review and meta-analysis with last search date of June 2017 was identified and included eleven RCTs with 4,591 patients [621]. The meta-analysis found that postoperative pyuria and bacteriuria rates were significantly lower in patients who received preoperative antibiotic prophylaxis (OR: 0.42, 95% CI: 0.25–0.69 and OR: 0.25, 95% CI 0.11–0.58, respectively). Five studies assessed postoperative febrile UTI (fUTI) and found no difference in the rate of fUTIs among patients who did or did not receive antibiotic prophylaxis (OR: 0.82, 95% CI: 0.40–1.67; p = 0.59). However, a significantly higher risk of postoperative fever in the preoperative antibiotic prophylaxis group (OR: 1.75, 95% CI: 1.22–2.50; p = 0.002) was reported. A subgroup analysis on the type of preoperative antibiotic prophylaxis found no difference between a single dose of oral versus intravenous antibiotics [621].
An RCT comparing various ciprofloxacin-based antibiotic prophylaxis regimens on the incidence of systemic inflammatory response syndrome (SIRS) after URS found no difference in the incidences of SIRS among the regimens, including the zero-dose regimen [622]. There was, however, a greater risk of SIRS in patients who did not receive antibiotic prophylaxis when the stone size was > 200mm2 [622]. Another RCT comparing the use of two oral doses of 3g fosfomycin tromethamine before surgery to standard of care did not find any difference in the incidence of infections, bacteriuria or fever [623].
Panel discussion considered that, despite low-quality evidence suggesting no benefit in reducing risk of clinical UTI, clinicians and patients would prefer to use prophylaxis to prevent kidney infection or sepsis. Ideally this should be examined in a robustly designed clinical study.
3.18.2.c.3. Percutaneous nephrolithotomy (PNL)
The largest systematic review and meta-analysis performed, with latest search date April 2019, included 1,549 patients in thirteen comparative studies on antibiotic prophylaxis strategies for PNL [624]. Compared with a single dose before surgery, preoperative antibiotic prophylaxis significantly reduced postoperative sepsis and fever (OR 0.31, 95% CI: 0.20–0.50 and OR 0.26, 95% CI 0.14-0.48, respectively) [624]. Similarly, the rate of positive pelvic urine and positive stones culture were reduced when preoperative prophylaxis was given. No difference in sepsis rates was observed among patients receiving or not receiving postoperative prophylaxis. However, patients who received postoperative antibiotic prophylaxis experienced fever more often [624].
Four RCTs with overall low risk of bias comparing various antibiotic regimes in PNL were identified [625-628]. Seyrek et al. compared the rate of SIRS following PNL in 191 patients receiving either a combination of sulbactam/ampicillin or cefuroxime. No difference was observed in SIRS or urosepsis rates [625]. Tuzel et al. investigated single-dose ceftriaxone versus ceftriaxone and subsequently an oral third-generation cephalosporin until after nephrostomy catheter withdrawal at mean (SD) of three (1) days in 73 participants undergoing PNL. The researchers found no difference in rate of infectious complications between the two antibiotic regimens [626]. Taken et al. compared the administration of 1g ceftriaxone and 1g cefazoline both administered 30 minutes before surgery and continued until nephrostomy removal. They found no difference in terms of SRIS or sepsis between the groups [628]. Omar et al. compared ciprofloxacin 200mg IV versus 2mg cefotaxime 30 minutes before and 12 hours after surgery and found a higher rate of fever in the cefotaxime group [627]. However, these results remain limited by the high risk of bias and the lack of data regarding postoperative infection. These studies give moderate evidence that a single dose of a suitable agent was adequate for prophylaxis against clinical infection after PNL.
3.18.2.d. Transurethral resection of the prostate
A systematic review of 39 RCTs with a search date cut-off in 2009 was identified [629]. A subsequent updated search to February 2017 did not reveal any further relevant studies. Of the 39 RCTs reviewed by Dahm et al., six trials involving 1,666 males addressed the risk of septic episodes, 17 trials reported procedure-related fever and 39 investigated bacteriuria. The use of prophylactic antibiotics compared to placebo showed a relative risk reduction (95% CI) for septic episode of 0.51 (0.27–0.96) with ARR of 2% (3.4–1.4%) and an NNT of 50. The risk reduction (95% CI) for fever was 0.64 (0.55–0.75) and 0.37 (0.32–0.41) for bacteriuria.
3.18.2.e. Transurethral resection of the bladder
One systematic review that included seven trials with a total of 1,725 participants was identified [630]. Antimicrobial prophylaxis showed no significant effect on postoperative UTIs [OR 95% CI: 1.55 (0.73–3.31)] and asymptomatic bacteriuria [OR (95% CI) 0.43 (0.18–1.04)] [630]. The review did not attempt subgroup analysis according to presence of risk factors for postoperative infection such as tumour size. Risk factors for development of postoperative UTIs were evaluated only by three of the included studies, and most of the parameters were analysed by no more than one study.
An RCT (n = 100) comparing oral fosfomycin 3g (the night before surgery) versus intravenous cefoxitin 2g (30 min. pre-surgery and 24 hours post-surgery) on postoperative UTIs found that a single oral administration of fosfomycin was noninferior to intravenous administration of cefoxitin in the prevention of post-TURB UTI, even in patients considered to be at higher risk [631].
Panel discussion concluded that a weak recommendation to use antibiotic prophylaxis for patients undergoing TURB who had a high risk of suffering postoperative sepsis would be appropriate.
3.18.2.f. Midurethral slings
One systematic review and meta-analysis identified one study assessing the role of preoperative antibiotics for midurethral sling surgery alone [632]. The study was halted due to low rate of infectious outcomes seen at the first scheduled interim analysis. The study enrolled 29 females in the antibiotic prophylaxis (cefazolin) group and 30 in the placebo group with a total follow-up of six months. No statistically significant difference between the cefazolin and placebo groups, with respect to wound infections [1 (3.3%) and 0 (0%)] or bacteriuria [3 (10%) and 1 (3.5%)] was found [632].
3.18.2.g. Renal tumour ablation (radiofrequency ablation, cryoablation, and microwave ablation of renal masses)
One systematic review publication dated 2018 included 6,952 patients across 51 studies [633]. Infectious complications were reported in 74 patients, including fever (60.8%), abscess (21.6%) and UTI (8.1%). Prophylactic antibiotic use was reported in 5.4% of patients, but it was not possible to study the association of prophylactic antibiotic use to infectious complications due to lack of reporting.
3.18.2.h. Prostate biopsy
3.18.2.h.1. Transperineal prostate biopsy
An updated meta-analysis of five RCTs [634-639] was performed by the Panel [640]. A total of thirteen randomised studies including 4,516 patients compared the impact of biopsy route on infectious complications. Infectious complications were significantly higher following transrectal biopsy (76 events among 2,243 men) compared to transperineal biopsy (35 events among 2,273 men) [RR 95% CIs: 2.00 (1.23–3.27)] [634-636,638-641]. Another systematic review and meta-analysis of 10 RCTs (4,188 biopsies) compared infectious outcomes and antibiotic use between transrectal and transperineal prostate biopsy. Transperineal biopsy was associated with significantly lower rates of postprocedural infection-related hospitalisation (OR 0.23 95% CI: 0.10–0.54) and fever (OR 0.68 95% CI: 0.52–0.89) than transrectal biopsy [642]. In addition, a systematic review including 165 studies with 162,577 patients described sepsis rates of 0.1% and 0.9% for transperineal and transrectal biopsies, respectively [643]. Finally, a population-based study from the UK (n = 73,630) showed lower readmission rates for sepsis in patients who had transperineal versus transrectal biopsies (1.0% vs. 1.4%, respectively) [644]. The available evidence demonstrates that the transrectal approach should be abandoned in favour of the transperineal approach despite any possible logistical challenges.
It should be noted that three of the current RCTs comparing transrectal biopsy with transperineal biopsy did not use any antibiotics at all in the transperineal groups [638, 645, 646]. A systematic review and meta-analysis of 23 studies (only two RCTs) including 12,324 patients reported no significant differences between patients receiving or not receiving antibiotic prophylaxis in terms of post-biopsy sepsis (0.16% vs. 0.13%) and hospitalisation due to infectious complications (0.35% vs. 0.29%) for the transperineal approach [647]. This is in line with another systematic review and meta-analysis of 112 individual patient cohorts, which also showed no significant difference in the number of patients experiencing post-transperineal biopsy infection with 1.35% of 29,880 patients receiving antibiotic prophylaxis and 1.22% of 4,772 not receiving antibiotic prophylaxis (p = 0.8) [648]. In addition, two recently published RCTs have reported comparably low post-biopsy infection rates for transperineal biopsy, regardless of whether or not antibiotic prophylaxis was administered [649, 650].
There is, therefore, a growing body of evidence to suggest that antibiotic prophylaxis may not be required for transperineal biopsy for patients at low risk of infectious complications.
3.18.2.h.2. Transrectal prostate biopsy
An updated meta-analysis of twelve RCTs including 2,437 males showed that the use of a rectal povidone-iodine preparation before biopsy, in addition to antimicrobial prophylaxis, resulted in a significantly lower rate of infectious complications [RR (95% CI) 0.47 (0.36–0.61) [640, 651-654]. A meta-analysis of two RCTs with 350 patients demonstrated no significant advantage of using chlorhexidine for rectal preparation compared to saline/no rectal preparation [RR (95% CI) 0.33 (0.11–1.00)] [652, 654]. Single RCTs showed no evidence of benefit for perineal skin disinfection [655], but reported an advantage for rectal povidone-iodine preparation before biopsy compared to after biopsy [656]. Routine surgical disinfection of the perineal skin before performing a transperineal biopsy is therefore recommended.
A meta-analysis of four RCTs including 671 males evaluated the use of rectal preparation by enema before transrectal biopsy. No significant advantage was found regarding infectious complications [RR (95% CI) 0.96 (0.64–1.54) [640].
An updated meta-analysis of 29 RCTs with 4,127 patients found no evidence that the use of periprostatic injection of local anaesthesia resulted in more infectious complications than no injection [RR (95% CIs) 1.08 (0.80–1.49)] [640, 641, 657, 658]. An updated meta-analysis of 10 RCTs including 2,342 patients found that extended biopsy templates showed comparable infectious complications to standard templates [RR (95% CIs) 0.82 (0.55–1.24)] [640, 659]. Additional meta-analyses found no difference in infection complications relating to needle guide type (disposable vs. reusable), needle type (coaxial vs. noncoaxial), needle size (large vs. small) and number of injections for periprostatic nerve block (standard vs. extended) [640].
A meta-analysis of eleven studies with 1,753 patients showed significantly reduced infections after transrectal prostate biopsy when using antimicrobial prophylaxis as compared to placebo/control [RR (95% CI) 0.56 (0.40–0.77)] [660].
Fluoroquinolones have traditionally been used for antibiotic prophylaxis in this setting, however, overuse and misuse of fluoroquinolones have resulted in an increase in fluoroquinolone resistance. In addition, the European Commission has implemented stringent regulatory conditions regarding the use of fluoroquinolones, resulting in the suspension of the indication for perioperative antibiotic prophylaxis, including prostate biopsy [154].
A systematic review and meta-analysis on antibiotic prophylaxis for the prevention of infectious complications following prostate biopsy concluded that, in countries in which fluoroquinolones are permitted as antibiotic prophylaxis, a minimum of a full one-day administration is recommended [660]. An updated meta-analysis of ten RCTs with 3,469 patients confirmed that targeted therapy (antibiotic guidance based on rectal swab microbiology) in case of fluoroquinolone resistance is associated with reduced infectious complications [RR (95% CI) 0.54 (0.41–0.71)] [660-662]. In addition, an updated meta-analysis of ten RCTs with 2,787 patients comparing augmented prophylaxis (a combination of two or more different classes of antibiotics) to standard prophylaxis showed augmented prophylaxis to be superior [RR (95% CI) 0.44 (0.32–0.59)] [660, 663]. In countries in which the use of fluoroquinolones has been suspended, cephalosporins or aminoglycosides can be used as individual agents with comparable infectious complications based on meta-analysis of two RCTs [660]. An updated meta-analysis of four RCTs compared fosfomycin trometamol to fluoroquinolones [RR (95% CI) 0.62 (0.37–1.06)] [660, 664]. Although initial RCTs suggested fosfomycin trometamol to be superior the latest Swedish study, which aimed to recruit 3,448 patients, was discontinued after 42 patients due to the unusually high number of hospitalisations in the fosfomycin trometamol group [664]. Another RCT showed that two doses of fosfomycin trometamol (second dose after 48 hours) were not superior to a single dose [665]. Therefore, routine general use should be critically assessed due to the relevant infectious complications also reported in nonrandomised studies [666]. Of note, the indication of fosfomycin trometamol for prostate biopsy has been withdrawn in Germany, because the manufacturers did not submit the necessary pharmacokinetic data in support of this indication. Urologists are advised checking their local guidance in relation to the use of fosfomycin trometamol for prostate biopsy. Another possibility is the use of augmented prophylaxis without fluoroquinolones, although no standard combination has been established to date. Finally, targeted prophylaxis based on rectal swap/stool culture is plausible, but no RCTs are available on non-fluoroquinolones. See Figure 4 for the prostate biopsy workflow to reduce infections complications.
3.18.3. Summary of evidence and recommendations for peri-procedural antibiotic prophylaxis
| Summary of evidence | LE |
| The outcome of clinical UTI was reported in four out of eleven RCTs, with no benefit found for antibiotic prophylaxis versus placebo in patients following filling and voiding cystometry. | 1b |
| A meta-analysis of five trials of moderate quality showed a benefit of using antibiotic prophylaxis for the reduction of symptomatic UTI in patients undergoing cystoscopy. However, this benefit was not seen if only the two trials with low risk of bias were used in the meta-analysis. | 1a |
| An RCT found only small, mostly non-significant reductions in postoperative infections with a modest absolute decrease in pyelonephritis when using antibiotic prophylaxis for ESWL. The clinical relevance of which is limited by current EU fluoroquinolone restrictions. | 1a |
| Two meta-analyses found no evidence of benefit for antibiotic prophylaxis prior to ureteroscopy in reducing the rate of clinical UTI, however, the rate of bacteriuria was reduced. | 1a |
| A meta-analysis of five RCTs demonstrated a moderate level of evidence that antibiotic prophylaxis was associated with a statistically significant reduction in the risk of post-procedural UTI following PNL. | 1a |
| Two RCTs concluded that a single dose of a suitable agent was adequate for prophylaxis against clinical infection after PNL. | 1b |
| A systematic review of 39 RCTs concluded that antibiotic prophylaxis reduced the rate of infectious complications in men undergoing TURP. | 1b |
| A systematic review of two RCTs found no benefit for antibiotic prophylaxis in patients undergoing TURB. | 1b |
| A meta-analysis of ten RCTs involving 4,188 patients demonstrated significantly lower rates of infectious complications, including hospital admissions and UTIs, after transperineal biopsy compared with transrectal biopsy. | 1a |
| A meta-analysis of 23 studies (including two RCTs) with 12,324 patients reported comparable rates of post-biopsy infections in patients undergoing transperineal biopsy, irrespective of whether or not antibiotic prophylaxis was given. | 2 |
| A meta-analysis of twelve RCTs including 2,437 men showed that the use of a rectal povidone-iodine preparation before transrectal biopsy, in addition to antimicrobial prophylaxis, resulted in a significantly lower rate of infectious complications. | 1a |
| An updated meta-analysis of ten RCTs with 3,469 patients confirmed that targeted prophylaxis (antibiotic guidance based on rectal swab microbiology) in case of fluoroquin-olone resistance is associated with reduced infectious complications. | 1a |
| A meta-analysis of ten RCTs with 2,787 patients comparing augmented prophylaxis (combination of two or more different classes of antibiotics) to standard prophylaxis showed augmented prophylaxis to be superior. | 1a |
| Recommendations | Strength rating |
Do not use antibiotic prophylaxis to reduce the rate of symptomatic urinary infection following:
| Strong |
| Do not use antibiotic prophylaxis for extracorporeal shockwave lithotripsy in patients with sterile urine. Prescribe antibiotic prophylaxis only in the case of suspected or diagnosed infected stones or bacteriuria. | Strong |
| Use antibiotic prophylaxis to reduce the rate of symptomatic urinary infection following ureteroscopy. | Weak |
| Use single-dose antibiotic prophylaxis to reduce the rate of clinical urinary infection following percutaneous nephrolithotomy. | Strong |
| Use antibiotic prophylaxis to reduce infectious complications in men undergoing transurethral resection of the prostate. | Strong |
| Use antibiotic prophylaxis to reduce infectious complications in high-risk patients undergoing transurethral resection of the bladder. | Weak |
| Perform prostate biopsy using the transperineal approach due to the lower risk of infectious complications and better antibiotic stewardship. | Strong |
| Omit perioperative antibiotic prophylaxis in transperineal biopsy in patients without risk factors for infectious complications. | Weak |
| Use rectal cleansing with povidone-iodine in men prior to transrectal prostate biopsy. | Strong |
| Do not use fluoroquinolones for prostate biopsy. This recommendation is in line with the European Commission final decision on EMEA/H/A-31/1452. | Strong |
For antibiotic prophylaxis in transrectal biopsy*, and from an antimicrobial-stewardship perspective, the following options are recommended**:
| Strong |
* The indication of fosfomycin trometamol for prostate biopsy has been withdrawn in Germany, as the manufacturers did not submit the necessary pharmacokinetic data in support of this indication. Urologists are advised to check their local guidance in relation to the use of fosfomycin trometamol for prostate biopsy.
** While most studies have been performed using fluoroquinolones, the applicability of these findings to nonfluoroquinolone antibiotics remains unclear.
Table 15: Suggested regimens for antimicrobial prophylaxis prior to urological procedures
As stated in Section 3.18.1.d, the panel has decided not to make recommendations for specific agents for particular procedures; the agents listed below represent possible choices only. Urologists should choose a specific antimicrobial based on their knowledge of local pathogen prevalence for each type of procedure, their antibiotic susceptibility profiles and virulence.
| Procedure | Prophylaxis recommended | Antimicrobial |
| Urodynamics | No | N/A |
| Cystoscopy | No | |
| Extracorporeal shock-wave lithotripsy | No | |
| Ureteroscopy | Yes | Trimethoprim Trimethoprim-sulfamethoxazole Cephalosporin group 2 or 3 Aminopenicillin plus a beta-lactamase inhibitor |
| Percutaneous nephro-lithotomy | Yes (single dose) | |
| Transurethral resec-tion of the prostate | Yes | |
| Transurethral resec-tion of the bladder | Yes, in patients who have a high risk of suffering postopera-tive sepsis. | |
| Transrectal prostate biopsy | Yes |
|
i.m.=intramuscular; IV = intravenous; p.o.=orally; q.d = every day.
* Note: option 2 contradicts antibiotic stewardship programmes
** The indication of fosfomycin trometamol for prostate biopsy has been withdrawn in Germany, as the manufacturers did not submit the necessary pharmacokinetic data in support of this indication. Urologists are advised to check their local guidance in relation to the use of fosfomycin trometamol for prostate biopsy.
Figure 4: Prostate biopsy workflow to reduce infectious complications 1. Two systematic reviews, including non-RCTs and two RCTs, describe comparable rates of post-biopsy infection in patients with and without antibiotic prophylaxis.
1. Two systematic reviews, including non-RCTs and two RCTs, describe comparable rates of post-biopsy infection in patients with and without antibiotic prophylaxis.
2. Be informed about local antimicrobial resistance.
3. Banned by European Commission due to side effects.
4. Contradicts principles of antimicrobial stewardship.
5. Fosfomycin trometamol (4 RCTs), cephalosporins (2 RCTs), aminoglycosides (2 RCTs).
6. Only one RCT comparing targeted and augmented prophylaxis.
7. Originally introduced to use alternative antibiotics in case of fluoroquinolone resistance.
8. Various schemes: fluoroquinolone plus aminoglycoside (4 RCTs), and fluoroquinolone plus cephalosporin(1 RCT).
Levels of evidence:
- High certainty: (⊕⊕⊕⊕) Very confident that the true effect lies close to that of the estimate ofthe effect.
- Moderate certainty: (⊕⊕⊕○) Moderately confident in the effect estimate. The true effect is likely tobe close to the estimate of the effect, but there is a possibility that it is substantially different.
- Low certainty: (⊕⊕○○) Confidence in the effect estimate is limited. The true effect may be substantially different from the estimate of the effect.
- Very low certainty: (⊕○○○) Very little confidence in the effect estimate. The true effect is likely tobe substantially different from the estimate of effect.
Figure reproduced from Pilatz et al. with permission from Elsevier.
* The indication of fosfomycin trometamol for prostate biopsy has been withdrawn in Germany, as the manufacturers did not submit the necessary pharmacokinetic data in support of this indication. Urologists are advised to check their local guidance in relation to the use of fosfomycin trometamol for prostate biopsy.