6. TREATMENT
This chapter reviews the available treatment modalities, followed by individual sections addressing treatment for the various disease stages.
6.1. Estimating life expectancy and health status
6.1.1. Introduction
Evaluation of life expectancy and health status is important in clinical decision-making for early detection, diagnosis and treatment of PCa. Prostate cancer is common in older men (median age 68 years) and diagnoses in men > 65 years will result in a continued increase in annual diagnosis in Europe and the USA associated with an aging population [557,558].
Active treatment mostly benefits patients with intermediate- or high-risk PCa and longest expected survival. In localised disease, over ten years’ life expectancy is considered mandatory for any benefit from local treatment, and an improvement in CSS may take longer to become apparent. Older age and worse baseline health status have been associated with a smaller benefit in PCSM and life expectancy of surgery versus AS [559]. Although in an RCT the benefit of surgery with respect to death from PCa was largest in men < 65 years of age (RR: 0.45), RP was associated with a reduced risk of metastases and use of androgen deprivation therapy (ADT) also among older men (RR: 0.68 and 0.60, respectively) [560]. External beam RT shows similar cancer control regardless of age, assuming a dose of > 72Gy when using intensity-modulated or image-guided RT [561].
Older men have a higher incidence of PCa and may be undertreated despite the high overall mortality rates [562,563]. Of all PCa-related deaths, 71% occur in men aged > 75 years [564], probably due to the higher incidence of advanced disease and death from PCa despite higher death rates from competing causes [565-567]. In the United States, only 41% of patients aged > 75 years with intermediate- and high-risk disease received curative treatment compared to 88% aged 65–74 [568].
6.1.2. Life expectancy
Life expectancy tables for European men are available online: https://ec.europa.eu/eurostat/. Survival may be variable, therefore estimates of survival must be individualised. Gait speed is a good single predictive method of life expectancy (from a standing start, at usual pace, generally over six meters). For men at age 75, ten-year survival ranged from 19% < 0.4 m/s to 87%, for ≥ 1.4 m/s [569].
Figure 6.1: Predicted median life expectancy by age and gait speed for males* [569]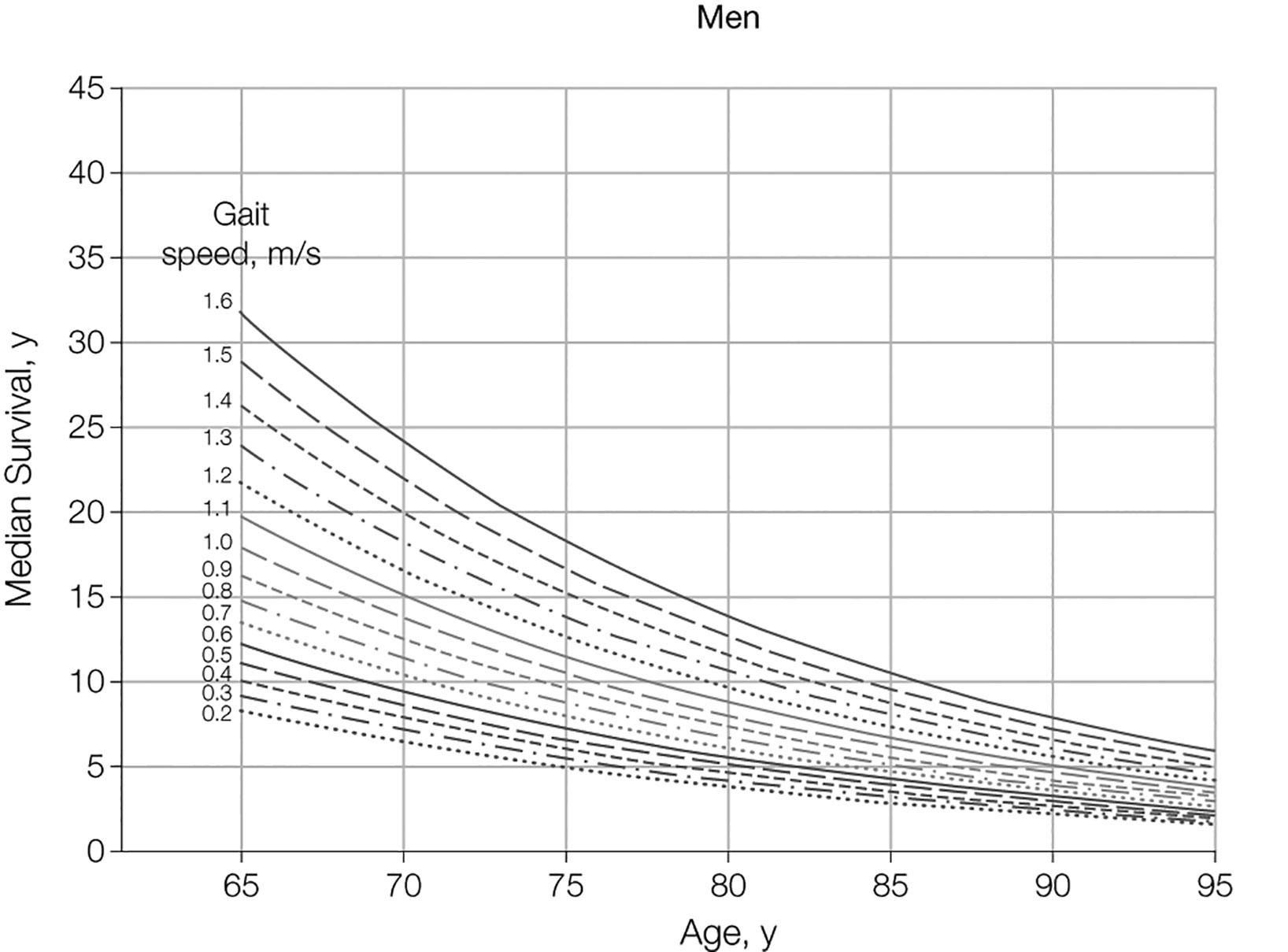 *From Studenski S. et al. JAMA 2011 305(1)50, figure reproduced with permission of the publisher.
*From Studenski S. et al. JAMA 2011 305(1)50, figure reproduced with permission of the publisher.
6.1.3. Health status screening
Heterogeneity in performance increases with advancing age, therefore, it is important to use measures other than age or performance status (PS) alone when considering treatment options. The International SIOG PCa Working Group recommends that treatment for adults over 70 years of age should be based on a systematic evaluation of health status using the Geriatric 8 (G8) screening tool (Table 6.1.1) [159]. This tool helps to discriminate between those who are fit and those with frailty - a syndrome of reduced ability to respond to stressors. Patients with frailty have a higher risk of mortality and negative side effects of cancer treatment [570]. Healthy patients with a G8 score > 14 or vulnerable patients with reversible impairment after resolution of their geriatric problems should receive the same treatment as younger patients. Frail patients with irreversible impairment should receive adapted treatment. Patients who are too ill should receive only palliative treatment (see Figure 5.3) [159]. Patients with a G8 score ≤ 14 should undergo a comprehensive geriatric assessment (CGA), because this score is associated with three-year mortality. A CGA is a multi-domain assessment that includes comorbidity, nutritional status, cognitive and physical function and social supports to determine if impairments are reversible [571]. An SR of the effect of geriatric evaluation for older cancer patients showed improved treatment tolerance and completion [572].
The Clinical Frailty Scale (CFS) is another screening tool for frailty (see Figure 5.4) [573]. Although not frequently used in the cancer setting, the CFS is considered a common language for expressing degree of frailty. The scale runs from one to nine, with higher scores indicating increasing frailty. Patients with a higher CFS score have a higher 30-day mortality after surgery and are less likely to be discharged home [574].
It is important to use a validated tool to identify frailty, such as the G8 or CFS, as clinical judgement has been shown to be poorly predictive of frailty in older patients with cancer [575].
6.1.3.a. Co-morbidity
Co-morbidity is a major predictor of non-cancer-specific death in localised PCa treated with RP and is more important than age [576,577]. Ten years after watchful waiting for PCa, most men with a high co-morbidity score had died from competing causes, irrespective of age or tumour aggressiveness [576]. Measures for co-morbidity include: Cumulative Illness Score Rating-Geriatrics (CISR-G) [578,579] (Table 6.1.2) and Charlson Co-morbidity Index (CCI) [580].
6.1.3.b. Nutritional status
Malnutrition can be estimated from body weight during the previous three months (good nutritional status < 5% weight loss; risk of malnutrition: 5–10% weight loss; severe malnutrition: > 10% weight loss) [581].
6.1.3.c. Cognitive function
Cognitive impairment can be screened for using the mini-COG (https://mini-cog.com/), which consists of three-word recall and a clock-drawing test and can be completed within five minutes. A score of ≤ 3/5 indicates the need to refer the patient for full cognitive assessment. Patients with any form of cognitive impairment (e.g. Alzheimer’s or vascular dementia) may need a capacity assessment of their ability to make an informed decision, which is an increasingly important factor in health status assessment [582-584]. Cognitive impairment also predicts risk of delirium, which is important for patients undergoing surgery [585].
6.1.3.d. Physical function
Measures for overall physical functioning include: Karnofsky score and ECOG scores [586]. Measures for dependence in daily activities include Activities of Daily Living (ADL; basic activities) and Instrumental Activities of Daily Living (IADL; activities requiring higher cognition and judgement) [587-589].
6.1.3.e. Shared decision-making
The patient’s own values and preferences should be considered as well as the above factors. A shared decision-making process also involves anticipated changes to QoL, functional ability and a patient’s hopes, worries and expectations about the future [590]. Particularly in older and frail patients, these aspects should be given equal importance to disease characteristics during the decision-making process [591]. Older patients may also wish to involve family members, and this is particularly important where cognitive impairment exists.
6.1.4. Conclusion
Individual life expectancy, health status, frailty and co-morbidity, not age alone, should be central in clinical decisions on screening, diagnostics, and treatment for PCa. A life expectancy of ten years is most commonly used as a threshold for benefit of local treatment. Older men may be undertreated. Patients aged 70 years or older who have frailty should receive a comprehensive geriatric assessment. Resolution of impairments in vulnerable men allows a similar urological approach as in fit patients.
Table 6.1.1: G8 screening tool (adapted from [592])
| Items | Possible responses (score) | |
| A | Has food intake declined over the past three months due to loss of appetite, digestive problems, chewing or swallowing difficulties? | 0 = severe decrease in food intake |
| 1 = moderate decrease in food intake | ||
| 2 = no decrease in food intake | ||
| B | Weight loss during the last three months? | 0 = weight loss > 3 kg |
| 1 = does not know | ||
| 2 = weight loss between 1 and 3 kg | ||
| 3 = no weight loss | ||
| C | Mobility? | 0 = bed- or chair-bound |
| 1 = able to get out of bed/chair but does not go out | ||
| 2 = goes out | ||
| D | Neuropsychological problems? | 0 = severe dementia or depression |
| 1 = mild dementia | ||
| 2 = no psychological problems | ||
| E | BMI? (weight in kg)/(height in m) | 0 = BMI < 19 |
| 1 = BMI 19 to < 21 | ||
| 2 = BMI 21 to < 23 | ||
| 3 = BMI > 23 | ||
| F | Takes more than three prescription drugs per day? | 0 = yes |
| 1 = no | ||
| G | In comparison with other people of the same age, how does the patient consider his/her health status? | 0.0 = not as good |
| 0.5 = does not know | ||
| 1.0 = as good | ||
| 2.0 = better | ||
| H | Age | 0 = > 85 |
| 1 = 80-85 | ||
| 2 = < 80 | ||
| Total score | 0-17 |
Figure 6.2: Decision tree for health status screening (men > 70 years)* [159]
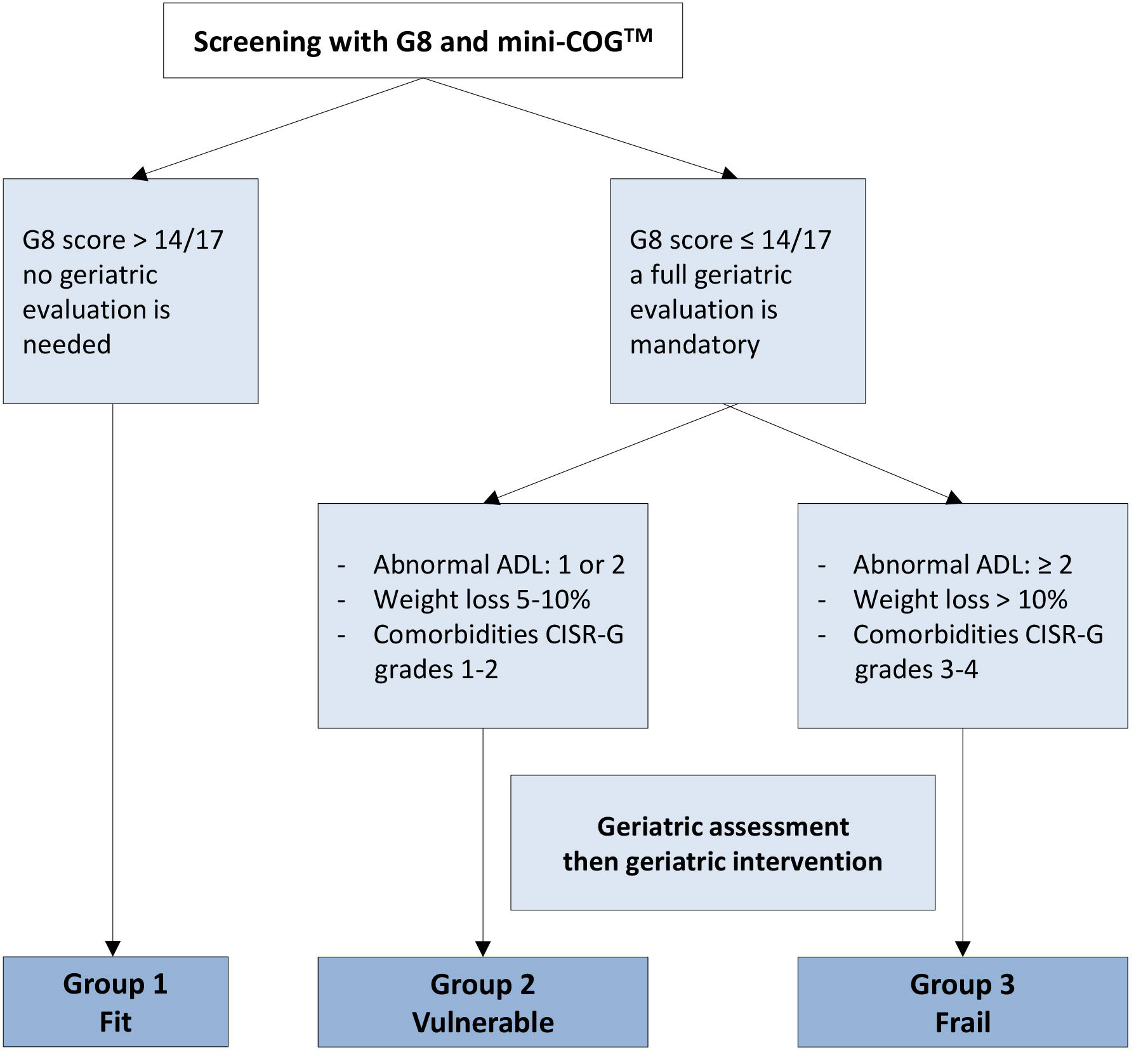 ADLs = activities of daily living; CGA = comprehensive geriatric assessment; CIRS-G = Cumulative Illness Rating Score - Geriatrics; Mini-COGTM = Mini-COGTM cognitive test.
ADLs = activities of daily living; CGA = comprehensive geriatric assessment; CIRS-G = Cumulative Illness Rating Score - Geriatrics; Mini-COGTM = Mini-COGTM cognitive test.
* For Mini-COGTM, a cut-off point of ≤ 3/5 indicates a need to refer the patient for full evaluation of potential dementia. From Boyle H. J., et al. Eur J Cancer 2019:116; 116 [159], reproduced with permission of Elsevier.
Figure 6.3: The Clinical Frailty Scale version 2.0 [573]*
 *Permission to reproduce the CFS was granted by the copyright holder.
*Permission to reproduce the CFS was granted by the copyright holder.
Table 6.1.2: Cumulative Illness Score Rating-Geriatrics (CISR-G)
| CISR-G | |
| 1 | Cardiac (heart only) |
| 2 | Hypertension (rating is based on severity; affected systems are rated separately) |
| 3 | Vascular (blood, blood vessels and cells, marrow, spleen, lymphatics) |
| 4 | Respiratory (lungs, bronchi, trachea below the larynx) |
| 5 | ENT (eye, ear, nose, throat, larynx) |
| 6 | Upper GI (oesophagus, stomach, duodenum. Biliar and pancreatic trees; do not include diabetes) |
| 7 | Lower GI (intestines, hernias) |
| 8 | Hepatic (liver only) |
| 9 | Renal (kidneys only) |
| 10 | Other GU (ureters, bladder, urethra, prostate, genitals) |
| 11 | Musculoskeletal-Integumentary (muscles, bone, skin) |
| 12 | Neurological (brain, spinal cord, nerves; do not include dementia) |
| 13 | Endocrine-Metabolic (includes diabetes, diffuse infections, infections, toxicity) |
| 14 | Psychiatric/Behavioural (includes dementia, depression, anxiety, agitation, psychosis) |
All body systems are scores on a 0 - 4 scale. 0: No problem affecting that system. 1: Current mild problem or past significant problem. 2: Moderate disability or morbidity and/or requires first line therapy. 3: Severe problem and/or constant and significant disability and/or hard to control chronic problems. 4: Extremely severe problem and/or immediate treatment required and/or organ failure and/or severe functional impairment. | |
| Total score 0-56 | |
6.1.5. Recommendations for evaluating health status and life expectancy
| Recommendations | Strength rating |
| Use individual life expectancy, health status and comorbidity in PCa management. | Strong |
| Use the Geriatric 8 (G8), mini-COG and Clinical Frailty Scale tools for health status screening. | Strong |
| Perform a full specialist geriatric evaluation in patients with a G8 score ≤ 14. | Strong |
| Consider standard treatment in vulnerable patients with reversible impairments (after resolution of geriatric problems), similar to fit patients if life expectancy is > 10 years. | Weak |
| Offer adapted treatment or watchful waiting to patients with irreversible impairment. | Weak |
| Offer palliative symptom-directed therapy alone to frail patients. | Strong |
6.2. Treatment modalities
6.2.1. Expectant management strategies
Two different strategies of expectant management are available watchful waiting (WW) and active surveillance (AS). The differences between WW and AS are presented in Table 6.2.1.
In patients with asymptomatic PCa in which curative therapy is not indicated due to a limited life expectancy based upon co-morbidities or age (<10 years) PCa may be managed conservatively and the patient followed until local or metastatic symptomatic progression occurs or is thought to be imminent. This approach is referred to as watchful waiting (WW). When predicting life expectancy co-morbidity is as important as age as it greatly increases the risk of dying from non-PCa-related causes. In an analysis of 19,639 patients aged > 65 years who were not given curative treatment, most men with a CCI (Charlson Comorbidity Index) score ≥ 2 had died from competing causes at ten years follow-up regardless of their age at time of diagnosis [576]. Tumour grade had little impact on OS suggesting that patients could have been spared biopsy and diagnosis of PCa. The oncological advantages of active treatment are unlikely to be relevant to them. This strategy maintains quality of life by delaying the side effects of palliative androgen deprivation therapy (ADT).
In patients with low- to intermediate-risk PCa, the natural course is so favourable that even in men with a long life-expectancy, curative local therapy may be postponed, or avoided altogether, by using active surveillance. Death from other causes is significantly more likely. In the ProtecT trial (see section 6.2.1.a), prostate cancer-related death was 3% at 15 years compared to death from any cause in 21.7% of patients - numbers that have been further validated in two large population-based studies from Canada and Sweden [593-595]. This occurs because the prevalence of cancer cells in the prostate is so much higher than the risk of developing clinical disease or dying from PCa. With the previous introduction of PSA, and now MRI, increased early detection of these small tumours there is a distinct risk of overdiagnosis and subsequent overtreatment of low- to intermediate-risk disease (Section 3.1 Epidemiology) [7,596,597]. Data from studies conducted on patients who did not undergo local treatment with long-term outcomes (up to 25 years) are available. The prognosis of low grade PCa is extremely favourable. Several series have shown a consistent CSS rate of 82–87% at ten years [598,599], and 80–95% for T1/T2 and ISUP GG ≤ 2 PCa [600]. In three studies with data beyond 15 years, the reported CSS rates were 80%, 79% and 58% [598,599,601]. Two studies reported 20-year CSS rates of 57% and 32% [598,601]. The observed heterogeneity in outcomes is due to different inclusion criteria, with some older studies from the pre-PSA era showing worse outcomes [601].
When managed with non-curative intent, intermediate-risk PCa is associated with 10-year and 15-year PCSM rates of 13.0% and 19.6%, respectively [602]. Cancer survival rates are even higher. Patients with well-, moderately and poorly differentiated tumours had 10-year CSS rates of 91%, 90% and 74%, respectively, correlating with data from a pooled analysis [600]. In addition, many patients classified as ISUP GG 1 would now be classified as ISUP GG 2–3 based on the 2005 Gleason classification, and accurate biopsy targeting following the introduction of pre-biopsy MRI suggesting that the above-mentioned results should be considered as minimal and current outcomes would be more favourable.
In screen-detected localised PCa, there is also a lead-time bias, resulting in a higher rate of early detected PCa, but also an even higher risk of detecting clinically insignificant PCa that never would have caused any symptoms [597]. Cancer-specific survival from untreated screen-detected PCa in patients with ISUP grade groups 1–2 is therefore likely to be even more favourable than for PCa detected of other reasons. Consequently, a high proportion of men with PSA-detected PCa are suitable for conservative management.
This highlights the importance of assessing co-morbidity even before considering a biopsy, but also before advising a patient with a PCa diagnosis on the optimal treatment for them. Estimation of competing benefits of active vs. conservative treatment and death from any cause at ten and fifteen years can be estimated using the PREDICT Prostate tool (https://prostate.predict.nhs.uk/), which is endorsed by the National Institute for Health and Care Excellence in the UK [603].
Table 6.2.1: Differences between active surveillance and watchful waiting [478]
| Active surveillance | Watchful waiting | |
| Treatment intent | Curative | Palliative |
| Follow-up | Predefined schedule | Patient-specific |
| Assessment/markers* used | DRE, PSA, rebiopsy, imaging (MRI) |
|
| Life expectancy | > 10 years | < 10 years |
| Aim | Minimise curative treatment-related toxicity without compromising survival, as the PCa is so indolent that it is unlikely to cause symptoms even with long life expectancy | Minimise palliative treatment-related (ADT) toxicity without compromising survival, PCa is unlikely to affect lifespan. |
| Eligible patients | Low-risk and selected intermediate-risk patients | Can apply to patients in all risk groups |
ADT - androgen deprivation therapy; DRE = digital rectal examination; MRI = magnetic resonance imaging; PSA = prostate-specific antigen.
*Molecular markers and/or PSMA-PET/CT (MRI) may be used.
6.2.1.a. Watchful waiting
Traditionally, WW has meant waiting for symptoms of PCa to develop without any specific follow-up schedule. However, for patients with locally advanced disease, a PSA doubling time (PSA-DT) < 12 months, and PSA-values over 30-50ng/mL early hormonal treatment might prolong survival in a clinically relevant time frame [604,605]. A more active follow-up of men on WW could therefore be beneficial for the higher risk groups (often associated with a higher ISUP GG), so that progression of local tumour or metastases can be detected and hormonal therapy initiated before they present with significant symptoms. The WW strategy should therefore be individualized. Biannual PSA, or annual PSA after a period of stable disease, followed by DRE or bone scan if PSA rises significantly, could then be of value.
In a Swedish registry study of men with nonmetastatic PCa on WW, after five years, 66.2% of patients with low-risk and 36.1% with high-risk disease, and 25.5% and 10.4% after ten years, were still alive and not receiving ADT [606]. At ten years, 4.1% and 10.8% had transitioned to castration-resistant disease, respectively. Importantly, 92.3% of low-risk patients and 84.1% of high-risk patients died due to causes other than PCa after ten years [606].
Watchful waiting vs. radical prostatectomy
Two RCTs and one Cochrane review have been published comparing the outcomes of WW to radical prostatectomy (RP). The SPCG-4 study was a RCT from the pre-PSA era, randomising patients into either WW or RP in 695 men (24% with nonpalpable disease) [607]. The study found RP to provide superior CSS, OS and progression-free survival (PFS) compared to WW at a median follow-up of 23.6 years (range 3 weeks to 28 years). However, the benefit in favour of RP over WW was only apparent after ten years.
The PIVOT trial, an RCT conducted in the early PSA era, made a similar comparison between RP versus WW in 731 men (50% with nonpalpable, 42% low-risk disease), but in contrast to the SPCG-4, it found little to no benefit of RP (cumulative incidence of all-cause death, RP vs. observation: 68% vs. 73%; RR: 0.92, 95% CI: 0.84–1.01) within a median follow-up period of 18.6 years (interquartile range, 16.6 to 20 years) [608]. Exploratory subgroup analysis showed that the borderline benefit from RP was most marked for intermediate-risk disease (RR: 0.84, 95% CI: 0.73–0.98), but there was no benefit in patients with low- or high-risk disease. Overall, no adverse effects on health related QoL (HRQoL) and psychological well-being was apparent in the first five years [609]. However, one of the criticisms of the PIVOT trial is the relatively high overall mortality rate in the WW group compared with more contemporary series, suggesting a selection bias.
A Cochrane review performed a pooled analysis of RCTs comparing RP versus WW [610]. Three studies were included: the previously mentioned SPCG-4 [607] and PIVOT [608], and the Veteran’s Administration Cooperative Urological Research Group (VACURG) study, which was conducted in the pre-PSA era [611]. The authors found that RP compared with WW reduced time to death by any cause (HR: 0.79, 95% CI: 0.70–0.90), time to death by PCa (HR: 0.57, 95% CI: 0.44–0.73), and time to metastatic progression (HR: 0.56, 95% CI: 0.46–0.70) at 29-years follow-up. However, RP was associated with higher rates of urinary incontinence (RR: 3.97, 95% CI: 2.34–6.74) and ED (RR: 2.67, 95% CI: 1.63–4.38).
ProtecT study
ProtecT, randomised 1,643 patients into one of three arms: active treatment with either RP or EBRT or active monitoring (AM) with outcomes reported at ten years and 15 years [593,612]. ProtecT trial did not apply a formal AS strategy. Active monitoring (AM) was a significantly less stringent surveillance strategy, using PSA only, with relaxed criteria to define progression. No repeat biopsies were performed as in AS.
At enrolment, 66% of the patients had low-risk disease, with 90% having a PSA < 10ng/mL, 77% ISUP GG 1 (20% ISUP GG 2–3) and 76% had T1c disease. The remaining patients had mainly intermediate-risk disease (approximately 40%).
The key finding was that AM was as effective as active treatment at 15 years (CSS = 96.9% in the AM-group vs. 97.8% in the RP group and 97.1% in the EBRT-group, p = 0.53), but has an increased metastatic progression risk (9.4% vs. 4.7% and 5.0%, respectively), as well as clinical progression at 15 years (25.9% for AM vs. 10.7% for RP/RT). Death from any cause occurred in 21.7% of the cohort, with similar numbers across treatment groups. Metastases, although rare, were more frequent than seen with comparable AS protocols [613]. A comprehensive characterisation of the ProtecT study cohort was performed after ten years, stratifying patients at baseline according to risk of progression using clinical stage, grade at diagnosis and PSA level [614]. Additionally, detailed clinicopathological information on participants who received RP were analysed.
The fifteen-year paper reported updated contemporary risk stratification according to D’Amico (24.1% Intermediate risk, 9.6% high risk), CAPRA (26.4% Score 3-5, 2.5% Score 6-10) and Cambridge Prognostic Group (20.5% Group 2, 8.8% Groups 3-5). Among patients who underwent RP, 50.5% were ISUP GG ≥2, while 28.5% had an increase in pathological stage, and 32% had an increase in tumour grade. Additionally, 51% of patients who developed metastases displayed ISUP GG 1 and 47.6% were low CAPRA risk. Over time, 61.1% of patients in the AM group received radical treatment (from 54.8% at ten years). Based on the ten-year report, the authors aimed to identify prognostic markers. The results showed that the following were associated with increased risk of disease progression (p < 0.001 for each):
- treatment received;
- age (65–69 vs. 50–64 years);
- PSA;
- ISUP GG at diagnosis;
- cT stage;
- risk group;
- number of PCa-involved biopsy cores;
- maximum length of tumour (median 5.0 vs. 3.0mm);
- aggregate length of tumour (median 8.0 vs. 4.0mm);
- presence of perineural invasion.
However, these factors could not reliably predict progression in individuals. Notably, 53% (n = 105) of patients who progressed had biopsy ISUP GG 1 disease, although, conversely, none of the participants who received RP and subsequently progressed had pathological ISUP GG 1 tumours. This discrepancy in progression and metastases rate between the AM arm of the ProtecT study and comparable AS protocols can most likely be explained by differences in intensity of surveillance, inadequate sampling by PSA testing and 10-core TRUS-guided biopsies.
Nevertheless, the ProtecT study has reinforced the role of deferred active treatment (i.e., either AS or some form of initial AM) as a feasible alternative to active curative interventions in all patients with low-grade and low-stage disease, as well as for many patients with favourable intermediate risk disease. Beyond 15 years, no RCT data are available yet, although AS is likely to give greater reassurance - particularly in younger men - based on more accurate risk stratification at recruitment and more stringent criteria regarding follow-up, imaging, repeat biopsy and reclassification. Individual life expectancy must continuously be evaluated before considering any active treatment in low-risk patients and in those with up to ten to 15 years’ individual life expectancy [614].
6.2.1.b. Active surveillance
Active surveillance (AS) aims to avoid over-treatment in men with PCa, whilst allowing identification and appropriate intervention for those who show reclassification during follow-up [615]. Patients remain under close surveillance through structured surveillance programmes with regular follow-up consisting of PSA testing, clinical examination, repeat prostate biopsies and an increasing role of imaging (usually MRI). Curative treatment is prompted by predefined thresholds indicative of development to potentially significant disease that is still curable while considering individual life expectancy.
No formal RCT is available comparing AS to curative treatment. Several cohorts have investigated AS in organ-confined disease, the findings of which were summarised in a SR [613,616]. Long term oncological outcomes of patients on AS are very favourable, with ten-year CSS rates of 98.1-100% and OS rates of 81-100% [612,617-622]. Although many men remain treatment-free during extended follow-up, more than one- third of patients are reclassified during follow-up, most of whom undergo curative treatment due to disease upgrading, upstaging, or increase in disease extent, or patient preference.
6.2.1.b.1. Active surveillance - inclusion criteria
Active surveillance inclusion criteria aim to select cases with a favourable natural course of disease, and in whom, in case of reclassification, the delay caused by the initial expectant management strategy does not lead to additional unfavourable outcomes. There is variation and heterogeneity between studies regarding exact patient selection, eligibility criteria, and follow-up policies (including frequency of clinical follow-up, use of PSA kinetics, PSA-density, frequency of standard repeat prostate biopsies, frequency and type of imaging such as MRI, and type of biopsy strategy (systematic, MRI- lesion targeted biopsies, combinations, or template biopsies). For men diagnosed with EAU risk group low-risk PCa who have an adequate life expectancy, AS is the first management option. In classic AS cohorts, additional selection criteria were incorporated such as PSA-density thresholds or biopsy core involvement parameters. With developments in the diagnostic algorithm regarding prostate biopsy indication and biopsy core strategy (i.e. use of pre-biopsy risk calculators, MRI, and targeted biopsies), the case-mix of patients considered for AS is changing, as well as the availability of parameters used for risk stratification and AS eligibility and follow-up.
Guidance regarding selection and follow-up criteria for AS is limited by the lack of data from prospective RCTs. As a consequence, the international collaborative DETECTIVE study involving healthcare practitioners and patients developed consensus statements for deferred treatment with curative intent for localised PCa, covering all domains of AS [623], as well as a formal SR on the various AS protocols [624].
The most frequently applied criteria include: ISUP GG 1 (on systematic biopsy), clinical stage cT1c or cT2a, PSA < 10ng/mL and PSA-D < 0.15ng/mL/cc [613,617]. The latter threshold remains controversial [617,618]. These criteria were supported by the DETECTIVE study consensus. MRI index lesion diameter may provide additional guidance, as thresholds of > 10mm and > 20mm have been used to predict BCR after RP, but are not yet used in AS criteria [619]. The Movember consensus group, consisting of 27 healthcare professional and 12 lived-experience participants from across the world, agreed that ISUP GG and MRI were the most important criteria for determining eligibility to AS [620].
A SR and meta-analysis found three clinicopathological variables that were significantly associated with reclassification: high PSA-D, > 2 positive cores (on systematic biopsies), and African-American descent [621]. A review of the risk of progression for African-American men on AS also indicated a potential increased risk of progression, but the association was not strong enough to discourage African-American men from undergoing AS, but thorough confirmatory testing is important [625]. Another review reported an association between family history and upgrading (adjusted disease progression HR 1.31, p<0.0001), although the certainty of evidence was low, and had no significant impact on adverse pathology at prostatectomy, indicating this factor alone is not a contraindication for AS [626].
In addition, a previous pathology consensus group suggested excluding men from AS when any of the following features were present: cribriform histology, predominant ductal carcinoma (including pure IDC), sarcomatoid carcinoma, small cell carcinoma, EPE or LVI in needle biopsy [627], or perineural invasion [628].
In men eligible for AS based upon systematic biopsy findings alone who did not have a prebiopsy MRI, a re-biopsy within six to twelve months (usually referred to as ‘confirmatory biopsy’) is mandatory to exclude sampling error.
6.2.1.b.2. Active surveillance – inclusion of intermediate risk disease
The outcomes of AS in intermediate-risk PCa have been analysed in three SRs and meta-analyses, summarising available data on its oncological outcomes and comparing patients with intermediate-risk PCa to patients with low-risk disease [629-631]. The definition of AS was not strictly defined in either of the reviews. Instead, the search strategies included ‘active surveillance’ as a search term, and no a priori study protocol was available. There was significant clinical heterogeneity in terms of inclusion criteria for intermediate-risk disease and use of MRI.
The results showed the proportion of patients who remained on AS was comparable between the low- and intermediate-risk groups after ten- and fifteen-years’ follow-up. Metastases-free survival did not significantly differ after five years (OR: 0.55, 95% CI: 0.2–1.53) but was worse in the intermediate-risk group after ten years (OR: 0.46, 95% CI: 0.28–0.77 and RR: 5.79, 95% CI: 4.61-7.29, respectively) [631]. Cancer-specific survival was worse in the intermediate-risk group after ten years (OR: 0.47, 95% CI: 0.31–0.69) and fifteen years (OR: 0.34; 95% CI: 0.2–0.58; RR: 3.93, 95% CI: 2.93-5.27, RR: 0.92, 95% CI: 0.89-0.96, respectively), although this is most likely due to less favourable baseline characteristics and not due to the delay caused by the initial period of AS. Overall survival was not statistically significantly different at five years’ follow-up (OR: 0.84, 95% CI: 0.45–1.57) but was significantly worse in the intermediate-risk group after ten years (OR: 0.43; 95% CI: 0.35–0.53, RR: 1.44, 95% CI: 1.11-1.86, RR: 0.87, 95% CI: 0.82-0.93, respectively). In a subgroup analysis of four studies comparing outcomes of patients with low- and intermediate-risk PCa of ISUP GG ≤ 2 (n = 1,900) no statistically significant difference could be found in terms of treatment free survival or risk of developing metastases (RR: 1.03, 95% CI: 0.62-1.71 and RR: 2.09, 95% CI: 0.75-5.82, respectively). It should be noted that many of the studies included patients with ISUP GG 3 disease. When these studies were excluded no difference in treatment free, CSS or OS was observed [630].
These reviews indicate that AS, in unselected intermediate-risk patients, implies a higher risk of progression over time. It remains unclear whether this difference only reflects the baseline difference in outcome, that can also be seen when comparing immediate treatment of low- and intermediate-risk PCa, or if the delay in treatment caused any worsening of the outcomes in the intermediate-risk group in any way. All three reviews conclude that AS could be offered to patients with intermediate-risk disease, but they should be informed of a higher risk of progression and whilst one of the reviews specifically suggest limiting the inclusion of intermediate-risk patients to those with low-volume ISUP GG 2 disease [629].
A Canadian consensus group proposes that low volume ISUP GG 2 (< 10% Gleason pattern 4 on systematic biopsies) may also be considered for AS. These recommendations have been endorsed by the ASCO [306] and the DETECTIVE study consensus [623] for those patients with a PSA < 10ng/mL and low core positivity. The DETECTIVE study concluded that men with favourable ISUP GG 2 PCa (PSA < 10ng/mL, low PSA density, clinical stage ≤ cT2a and a low number of positive systematic cores) should also be considered for deferred treatment [623]. In this setting, re-biopsy within six to twelve months to exclude sampling error is even more relevant than in low-risk disease [617,632]. The DETECTIVE study-related qualitative SR aimed to determine appropriate criteria for inclusion of intermediate-risk disease into AS protocols [624]. Out of 371 AS protocols included in the review, more than 50% included patients with intermediate-risk disease on the basis of PSA up to 20ng/mL (25.3%), ISUP GG 2 or 3 (27.7%), clinical stage cT2b/c (41.6%) and/or direct use of D’Amico risk grouping of intermediate risk or above (51.1%). The DETECTIVE study reached consensus that patients with ISUP GG 3, or patients with intraductal or cribriform histology, should not be considered for AS. The presence of any grade 4 pattern is associated with a threefold increased risk of metastases compared to ISUP GG 1, while a PSA up to 20ng/mL might be an acceptable threshold [632-634], especially in the context of low PSA-D.
The indicator of the tumour volume may be either the number of positive cores and the length of cancer in each core based on systematic biopsies, or the volume of the dominant lesion seen on mpMRI [623]. If targeted biopsies based upon mpMRI images are performed, the number of positive cores of the targeted biopsies are not an indicator of the extent of disease or tumour volume when considering a patient for AS due to the altered biopsy protocol.
MRI-targeted biopsies have been associated with upgrading of tumours, but improved outcomes [113].
The large prospective PRIAS study on AS expanded inclusion criteria when MRI and targeted systematic biopsies are used at inclusion (https://www.prias-project.org/):
- cT ≤ 2
- ISUP: GG 1 or GG 2 without invasive cribriform growth and intraductal carcinoma
- PSA: ≤ 20ng/mL
- PSA density: < 0.25ng/mL/cc
- Number of positive cores:
- - For ISUP GG 1: No limit.
- - For ISUP GG 2 (without invasive cribriform growth and intraductal carcinoma): ≤ 50% systematic cores (where multiple positive cores from the same lesion on MRI count for one positive core).
During follow-up, upgrading is the only criterium for discontinuation, defined as ISUP GG ≥ 3 or ISUP GG ≥ 2 with cribriform growth or intraductal carcinoma, or ISUP GG ≥ 2 with > 50% positive cores.
A multidisciplinary consensus conference on germline testing has suggested a genetic implementation framework for the management of PCa [185]. Based on consensus, BRCA2 gene testing was recommended for AS discussions and could be performed in men with a family history of prostate, breast or ovarian cancers. However, the nature of such discussions and how a positive result influences management were beyond the scope of the project. Currently, BRCA2 mutation does not exclude a patient from AS if tumour factors are otherwise favourable. Moreover, if included in AS programmes, patients with a known BRCA2 mutation should be cautiously monitored until such time that more robust data are available.
6.2.1.b.3. Tissue-based prognostic biomarker testing for selection for AS
Biomarkers, including Oncotype Dx®, Prolaris®, Decipher®, PORTOS and ProMark®, are promising. However, further data and comparisons with other parameters (including MRI) will be needed before such markers can be used in standard clinical practice [635].
6.2.1.b.4. Magnetic resonance imaging for selection for active surveillance
Two RCTs and an SR showed that adding MRI-targeted biopsy to systematic sampling at confirmatory biopsy increased the number of cancers labelled ISUP GG ≥ 2 and thus may aid patient selection for AS, although the impact of MRI and targeted biopsies with corresponding stage shift on long-term oncological outcomes of AS is lacking [135,636-641]. Adding MRI-targeted biopsy to systematic sampling at confirmatory biopsy improved upgrade detection by increments of 0-7.9 per 100 men depending on the series [636]. In a meta-analysis of six studies, the rate of upgrading to ISUP GG ≥ 2 cancer increased from 20% (95% CI: 16–25%) to 27% (95% CI: 22–34%) when MRI-targeted biopsy was added to systematic biopsy [641]. The Active Surveillance MRI Study (ASIST) randomised men on AS scheduled for confirmatory biopsy to either 12-core systematic biopsy or to MRI with targeted biopsy (when indicated), combined with systematic biopsy (up to 12 cores in total). After two years of follow-up, the use of MRI before confirmatory biopsy resulted in fewer failures of surveillance (19% vs. 35%, p = 0.017) and in fewer patients progressing to ISUP GG ≥ 2 cancer (9.9% vs. 23%, p = 0.048) [639]. However, systematic biopsy retains its added value, which argues for a combined biopsy approach [636,641]. The DETECTIVE study agreed that men eligible for AS after combined systematic and MRI-targeted biopsy do not require a confirmatory biopsy; a recommendation further supported by the results of the MRIAS trial [623,642].
If the PCa diagnosis is made on MRI-targeted biopsy alone to lower the risk of over detection of insignificant (see Section 5.4.1, and 5.4.2), and the number of positive systematic cores used as an indication for tumour volume during AS is not available, MRI lesion diameter can be used as a surrogate, although specific definitions have not yet been tested in an AS setting (e.g. for ISUP GG 2 tumours no PIRADS 5 or < 20mm lesion size) [619].
Several studies indicate that PSMA-PET-CT or PSMA-PET-MRI may have additional value to the above-mentioned clinicopathological variables for risk stratification before AS [136,643]. Thus far, however, the studies are too small, the follow-up too short and association with long-term oncological outcomes is lacking to draw any hard conclusions and for this modality to be recommended outside of clinical trials.
6.2.1.b.5. Active surveillance follow-up
Based on the DETECTIVE consensus study, the surveillance strategy should be based on serial DRE (at least once yearly), PSA (at least once every six months), and repeated biopsy (no consensus on frequency, but 1-4-7 years is a commonly applied schedule).
A panel SR incorporating 263 surveillance protocols showed that 78.7% of protocols mandated per-protocol repeat biopsies within the first two years, and that 57.7% of the protocols performed repeat biopsy at least every three years for ten years after the start of AS [624].
There was clear agreement in the DETECTIVE consensus meeting, as well as in the Movember consensus group, that a PSA change alone, including PSA-doubling time (PSA-DT, < 3 years) should not change management based on its weak link with grade progression [644,645], but rather trigger further investigation such as biopsy or repeat MRI. It was also agreed that changes on repeat MRI during AS needed a repeat biopsy before considering continuing to active treatment [620,623].
The Movember consensus group made several recommendations that in some ways differ from the DETECTIVE consensus study, such as:
- routine DRE was not supported if MRI or other imaging was carried out routinely during AS;
- routine biopsy can be omitted if MRI combined with other parameters (PSA kinetics and density) is stable; and
- change in clinical parameters should prompt MRI with possible biopsy rather than immediate biopsy [620].
STRATCANS (STRATified CANcer Surveillance) stratifies patients into three groups based on the combination of Cambridge Prognostic Group (CPG; CPG 1 - ISUP GG1 and PSA <10 and cT1-2, CPG 2 – ISUP GG 2 or PSA 10-20 and cT1-2), PSAD (<0.15 vs. ≥0.15), and MRI visibility based on risk of progression [646]. This may be used to individualise follow-up intensities (18-12-6 months follow-up and MRI every 36-18-12 months with increasing risk, respectively, and no standard repeat biopsy in the lowest risk-tier).
6.2.1.b.6. Magnetic resonance imaging for follow-up during active surveillance
The Prostate Cancer Radiological Estimation of Change in Sequential Evaluation (PRECISE) criteria were established to standardise the assessment of tumour progression on serial MRI [647]. PRECISE is a strong predictor of histological upgrading [648,649]. Two independent meta-analyses assessed the value of MRI progression criteria for predicting histological progression (mostly defined as progression to ISUP GG ≥ 2). The pooled histological progression rate was 27% in both reviews. If biopsies were triggered only by MRI progression findings, approximately two-thirds of the biopsies would be avoided, at the cost of missing 40% of men with histological progression. In addition, at least half of biopsied men would have had negative findings for histological progression and thus would have undergone unnecessary biopsies. If histological progression were restricted to progression to ISUP GG > 3, approximately 30% of histological progression would be missed and approximately 80% of the biopsies performed would be unnecessary. The use of the PRECISE criteria did not appear to change these results [650,651].
Another study analysed a prospectively maintained AS cohort of 369 patients (272 with ISUP GG 1 cancer and 97 with ISUP GG 2 cancer) who had been selected for AS after combined systematic and MRI-targeted sampling during confirmatory biopsy [652]. At two years, systematic biopsy, MRI-targeted biopsy and combined biopsy detected grade progression in 44 (15.9%), 73 (26.4%) and 90 patients (32.5%), respectively. This suggests that both biopsy approaches retain added value, not only for confirmatory biopsy, but also during AS [652]. Systematic biopsy cores can therefore be considered to be added to follow-up biopsy to rule out more widespread disease [226,264,375]. The disadvantage of overdiagnosis due to systematic cores is not present in the AS follow-up setting. Conversely, extra biopsy cores may cause discomfort and, as in the primary diagnostic setting, the risk of leaving significant PCa undetected is small and of limited relevance in a surveillance setting. As in the primary setting, the strategy of targeted/perilesional cores is therefore also recommended during AS repeat biopsy.
6.2.1.b.7. Individualised repeat biopsy during active surveillance
The basis for AS protocols includes standard repeat biopsy. However, several factors have been found to be associated with low reclassification rates and long PFS and can be used to individualise the need and frequency of AS biopsy schedules: low PSA-D [642,653-655], low PSA velocity (PSAV) [656,657], negative biopsy (i.e., no cancer at all) at confirmatory or repeat biopsy during AS [521,658], and negative baseline or repeat MRI during AS [642,653-655,659-662]. Negative repeat biopsy during AS was associated with a 50% decrease in the risk of future reclassification and upgrading [663]. In a single-centre AS cohort of 514 patients who underwent at least three protocol-mandated biopsies after diagnosis (the confirmatory biopsy and at least two additional surveillance biopsies), men with one negative biopsy (i.e., no cancer at all) at confirmatory or second biopsy, or men with two consecutive negative biopsies had a lower likelihood of a positive third biopsy and significantly better 10-year treatment free survival [664]. Patients with stable results (PRECISE 3) on repeat MRI during AS combined with a low PSA-D (< 0.15) have a very low rate of progression and may be a group in whom standard repeat biopsy may be omitted [665].
6.2.1.b.8. Active surveillance - change in treatment
Patients may remain on AS whilst they have a life expectancy of > 10 years and the disease remains insignificant. A transition from AS to WW due to rising age or new comorbidity should be incorporated within conservative management strategies for PCa and in discussion with patients [666].
Histopathology criteria are the strongest reason to trigger a change in management, including. reclassification to ISUP GG ≥ 3 or detection of cribriform or intraductal growth patterns, based on systematic biopsy. The exact criteria in the targeted biopsy era remain debated. MRI-targeted biopsy induces a grade shift and ISUP GG 2–3 cancers detected by MRI-targeted biopsy have, on average, a better prognosis than those detected by means of systematic sampling. Additionally, men upgraded during AS have more favourable outcomes than men with the same ISUP GG detected at first biopsy [667]. As an increasing number of patients with favourable intermediate-risk disease are managed with AS (see Section 6.2.1.b), progression to ISUP GG 2 should not be deemed as a definitive reason to stop AS, especially when found on targeted biopsy. In addition, as acknowledged in the DETECTIVE consensus meeting, the number of positive cores is no longer an indicator of tumour volume if targeted biopsies are performed [623,668]. Based on the findings of an SR incorporating 271 reclassification protocols, patients with low-volume ISUP GG 2 disease at recruitment and with increased systematic core positivity (> 3 cores involvement [> 50% per core]) on repeat systematic biopsies not using MRI should be reclassified [624]. As for inclusion, MRI tumour volume may be used during follow-up as a surrogate for tumour volume estimation based on systematic biopsies, though specific definitions are lacking.
6.2.1.b.9. Psychological factors during active surveillance
Active surveillance is the first management option in low and favourable intermediate risk PCa. A review of patient-reported considerations identified several factors influencing decision-making when considering AS. Among the identified factors were personal risk assessment, influence of family and friends, beliefs about treatment as well as doctor and system factors; underscoring the importance of individualised, relevant and clear information to support decision-making [669]. The feeling of loss of control may lead to uncertainty during AS, while re-establishing agency may restore confidence [670]. Anxiety regarding continued surveillance occurs in approximately 10% of patients on AS [671]. and was recognised as a valid reason for active treatment [231]. An alternative for patients suitable for continuing AS would be to offer psychological support to reduce the level of anxiety, as the Movember consensus group also indicated [620]. A population-based cohort study from Sweden on regional differences in AS uptake and subsequent transition to radical treatment concluded that a regional tradition of a high uptake of AS was associated with a lower probability of transition to radical treatment, but not with AS failure [672].
6.2.1.b.10. Interventions during active surveillance
A review of potential interventions during AS found that the use of 5-ARIs was associated with improved progression-free survival (PFS; hazard ratio: 0.59; 95% confidence interval 0.48-0.72) with limited increased toxicity [673].
A phase II RCT randomised patients to AS plus enzalutamide or AS alone. This study indicated that PSA progression could be delayed and the odds of a negative biopsy increased during the median follow-up time of 1.3 years, but patients had more side effects from the treatment without showing any long-term benefits from the treatment [674]. Evidence in support of other interventions is weak.
6.2.1.c. Summary of evidence and recommendations for active surveillance strategy
| Summary of evidence | LE |
| The oncological outcomes of low and favourable-intermediate risk PCa are very favourable, even when untreated, especially when detected during screening. | 1 |
| No modality has shown superiority over any other active management options or deferred active treatment in terms of overall and PCa-specific survival for clinically localised low/intermediate-risk disease. | 1 |
| The long-term oncological outcomes of active surveillance cohorts based on systematic prostate biopsy are very favourable. | 2 |
| The standard AS strategy should be based on PSA (at least once every six months), serial DRE (at least once yearly), and repeated biopsy. | 3 |
| Magnetic resonance imaging detects more cancers labelled with higher ISUP GG and can be used before starting AS (if not performed previously), although the impact on long-term oncological endpoints is lacking. | 3 |
| Patients with stable MRI findings have a lower risk of upgrading on repeat biopsy, allowing for de-escalation of standard repeat DRE. | 2 |
| A progression on MRI mandates a repeat biopsy to confirm histological progression before a change in treatment strategy. | 3 |
| A stable MRI (PRECISE 1-3) does not make repeat biopsy superfluous but it might be excluded in patients with low-risk tumour and a stable low PSA-D < 0.15. | 2 |
| Recommendations | Strength rating |
| Offer active surveillance (AS) as standard of care to all suitable patients (all low-risk disease and selected patients with favourable intermediate-risk disease). | Strong |
| Exclude patients with cribriform or intraductal histology on biopsy from AS. | Strong |
| Do not perform confirmatory biopsies if a patient has had upfront magnetic resonance imaging (MRI) and targeted and perilesional biopsies. | Weak |
| Perform MRI before a confirmatory biopsy if it hasn’t been performed before the initial biopsy. | Strong |
| Take targeted and perilesional biopsy cores (of any PI-RADS ≥ 3 lesion) if a confirmatory or repeat biopsy is performed. | Strong |
| Perform per-protocol confirmatory prostate biopsies if MRI is not available. | Weak |
Base the strategy of AS on a strict follow-up protocol including prostate-specific antigen (PSA) (at least once every six months), digital rectal examination (DRE), and repeated biopsy with or without MRI (every 2-3 years) until life expectancy falls below ten years. | Strong |
| Exclude patients with a low-risk PCa, a stable MRI (PRECISE 3) and a stable low PSA density (< 0.15) from repeat biopsy when MRI is repeated before repeat biopsy. | Weak |
| Perform MRI and repeat biopsy if PSA is rising (PSA doubling time < 3 years). | Strong |
| Base change in treatment on biopsy progression, not on progression on MRI, PSA, and/or DRE. | Weak |
6.2.2. Radical prostatectomy
6.2.2.a. Introduction
The goal of RP by any approach is the eradication of cancer while preserving pelvic organ function whenever possible [675]. The procedure involves removing the entire prostate with its capsule intact and SVs, followed by vesico-urethral anastomosis. The main results from multicentre RCTs involving RP are summarised in Table 6.2.3.
Table 6.2.3: Oncological results of radical prostatectomy in organ-confined disease in RCTs
| Study | Acronym | Population | Treatment period | Median FU (mo.) | Risk category | CSS (%) |
| Bill-Axelson, et al. 2018 [607] | SPCG-4 | Pre-PSA era | 1989-1999 | 283 | Low risk &intermediate risk | 80.4 (at 23 yr.) |
| Wilt, et al. 2017 [608 | PIVOT | Early years of PSA testing | 1994-2002 | 152 | Low risk & intermediate risk | 95.9 91.5 (at 19.5 yr.) |
| Hamdy, et al. 2023 [593] | ProtecT | Screened population | 1999-2009 | 180 | Mainly low & intermediate risk | 97 (at 15 yr.) |
CSS = cancer-specific survival; FU = follow-up; mo. = months; PSA = prostate-specific antigen; yr. = years.
6.2.2.b. Preoperative preparation
6.2.2.b.1. Patient education
Perioperative education has been shown to improve long-term patient satisfaction following RP [676]. Augmentation of standard verbal and written educational materials such as the use of interactive multimedia tools [677,678] and preoperative patient-specific 3D-printed prostate models should be considered to optimise patient-centred care [679].
6.2.2.b.2. Surgical planning
Preoperative surgical planning is expected to assist surgeons in achieving an optimal surgical outcome. Contemporary planning involves diagnostic information combined into nomograms, in addition to imaging (e.g. mpMRI). A SR that including eight studies (one RCT, seven prospective non-randomised studies) showed that preoperative surgical planning reduced positive surgical margins regardless of whether a nomogram (RR 0.56, p = 0.009) or MRI (RR 0.72, p = 0.02) was used [680]. Incorporation of membranous urethral length according to preoperative MRI may also be useful for counselling on relative likelihood of early post-operative continence return [681].
Awareness of predisposing factors that may complicate surgery is also pivotal. In particular, higher prostate volume can increase operative complexity (but not positive surgical margins) [682], while obesity is associated with worse perioperative, oncological (positive surgical margins, RR 1.2, p < 0.01) and functional outcomes (continence, RR 1.17, p = 0.01; impotence, RR 1.08, p < 0.01) [683,684]. Similarly, prior TURP can result in worse perioperative, oncological (positive margin rate, OR 1.25, p = 0.03) and functional (continence recovery, OR 0.60, p = 0.007; erectile function, RR 0.8, p < 0.001) outcomes [684,685].
6.2.2.c. Intraoperative considerations
6.2.2.c.1. Nerve-sparing surgery
Age and preoperative function are important predictors for postoperative erectile function. During RP, preservation of the neurovascular bundles (NVB) with parasympathetic nerve branches of the pelvic plexus can spare erectile function [686,687].
A large SR and meta-analysis reported that bilateral NS resulted in improved urinary continence recovery (RR 1.08 at 12 months, p < 0.0001) across all time points with heterogeneous pooled estimates [688]. Technical factors including dissection technique, fascial dissection plane (closer the better), antegrade versus retrograde, use of thermal energy and traction and clips or low bipolar energy can be considered [689].
Patient selection for NS surgery remains challenging for clinicians, with a reliance on clinical and radiological factors, that are generally poor at predicting EPE, and consequently, the appropriateness of NS [690]. High-risk disease is not necessarily a contraindication for NS [691].
A reasonable concern is the oncological compromise and positive surgical margin rate. A 2022 SR of 18 comparative studies (no RCTs) of NS versus non-nerve-sparing RP showed a RR of side-specific positive margins of 1.5, but none of them included patients with high-risk PCa [692]. No effect of NS on BCR was seen. Follow-up was short, however, and studies were subject to selection bias with mainly low-risk patients. For those patients with high-risk PCa, side-specific NS was avoided if disease was palpable or EPE was present on MRI. Indeed, a 2019 SR showed that MRI affected the decision on whether or not to perform NS in 35% of cases without any negative impact on surgical margin rate [693].
Use of intraoperative surgical margin assessment, such as the frozen section examination (NeuroSAFE) technique, enables a systematic evaluation of surgical margins intra-operatively, allowing for adjustment if positive margins are detected to reduce positive surgical margin rates (OR 0.68; 95% 0.51-0.91 without oncological compromise according to retrospective data [694].
The multicentre prospective NeuroSAFE PROOF trial randomised 381 patients for RP with or without intraoperative margin assessment, the majority of whom were stage pT2 (64-66%) and ISUP GG 2 (76%). Higher rates of bilateral NS (82% vs. 56%) and intravesical NS (76% vs. 52%) were reported in the NeuroSAFE group. Significant improvement in patient-reported IIEF-5 scores was noted with the NeuroSAFE technique (12.7 vs. 9.7) among 344 patients at twelve months. Regarding continence, a higher ICIQ score was noted at three months (MD -1.41, p = 0.006) but not six months (MD -0.37, p = 0.46) [695]. Regarding positive surgical margins, small (≤ 3 mm) margins were higher in the NeuroSAFE group (21% vs. 13%) but large and multifocal margins were similar (14% vs. 16%). NeuroSAFE was positive in 37% of frozen sections, while tumour was present in the secondary resection in 44% of NeuroSAFE patients. At twelve months, PSA persistence or biochemical recurrence occurred in 9% of the NeuroSAFE and 6% of the control group, while freedom from recurrence or treatment at twelve months was 86% for NeuroSAFE versus 93% for standard RARP.
In summary, NS is likely to improve functional outcomes after RP such as early continence and erectile function. Nerve sparing is also more likely to result in positive surgical margins, which may influence use of salvage therapies and their associated toxicity. Surgeons should consider judicious preoperative planning and use of available resources (e.g. NeuroSAFE) to best balance optimal postoperative oncological and functional outcomes.
6.2.2.c.2. Bladder neck preservation
Whilst the majority of urinary continence is maintained by the external urethral sphincter at the membranous urethra a minor component is contributed by the internal lissosphincter at the bladder neck [696]. Preservation of the bladder neck has therefore been proposed to improve continence recovery post-RP. A RCT assessing continence recovery at twelve months and four years showed improved objective and subjective urinary continence in both the short and long term without any adverse effect on oncological outcome [697]. These findings have been confirmed by a SR [698]. However, concern remains regarding margin status for cancers located at the prostate base and caution is advised if a nearby tumour is suspected or in the presence of a large median lobe or prior TURP given their contribution to poor oncological and functional outcomes.
6.2.2.c.3. Urethral length preservation
The membranous urethra sits immediately distal to the prostatic apex and is chiefly responsible, along with its surrounding pelvic floor support structures, for urinary continence. It consists of the external rhabdosphincter, which surrounds an inner layer of smooth muscle. Preoperative MRI studies have indicated that the length of membranous urethra is variable, but very important, as every extra millimetre of membranous urethral length seen on MRI preoperatively improves early return to continence post-RP [699-701], and is a prognostic factor for regaining continence at all points until twelve months [701]. Surgeons should attempt to preserve as much urethral length as possible during RP to maximise the chance of early return to continence.
6.2.2.c.4. Vesicourethral anastomosis and reconstruction
Following prostate removal, the bladder neck is anastomosed to the membranous urethra. The objective is to create a precisely aligned, watertight, tension-free and stricture-free anastomosis that preserves the integrity of the intrinsic sphincter mechanism. Several methods have been described, based on the direct or indirect approach, the type of suture (i.e. barbed vs. non-barbed/monofilament) and variation in suturing technique (e.g. continuous vs. interrupted, or single-needle vs. double-needle running suture). Overall, although a variety of approaches, methods, and techniques are available for performing the vesicourethral anastomosis, no clear recommendations are possible due to the lack of high-certainty evidence. In practice, the chosen method should be based on surgeon experience and individual preference [702-707].
For minimally invasive (laparoscopic and robotic-assisted) RP, unidirectional barbed suture is associated with reduced anastomosis time, operative time and posterior reconstruction time versus conventional non-barbed suture during robotic-assisted radical prostatectomy (RARP) with no differences in post-operative leak rate, length of catheterisation and continence rate. However, no definitive conclusions could be drawn due to the relatively low quality of the data [703].
Effect of anterior and posterior reconstruction on continence
Prior to vesicourethral anastomosis, the effect of posterior and anterior reconstruction of surrounding support structures to return to continence has been tested in several small RCTs with conflicting results [708-712]. Four RCTs, including anterior suspension, have also shown conflicting results [713-716], where anterior suspension may result in earlier return to continence, but no long-term difference.
Variation in many aspects hampers reliable pooling of data and definitive recommendations. As there is conflicting evidence on the effect of anterior and/or posterior reconstruction on return to continence post-RP, no recommendations can be made. However, no studies showed an increase in adverse oncologic outcome or complications with reconstruction.
6.2.2.c.5. Retzius-sparing radical prostatectomy
The majority of operative approaches to both open and minimally-invasive RP involve displacement of the bladder and surrounding peritoneum away from the anterior abdominal wall and conducting of the operation in the Space of Retzius. An alternative technique is to preserve the Space of Retzius, conducting the operation from a posterior-only approach, termed “Retzius-sparing” (rsRARP). The motivations for the Retzius-sparing approach include reduced operative dissection (and possibly improved perioperative parameters) and continence outcomes. The majority of data, both from prospective series and small RCTs, indicate improved early and overall continence recovery compared to standard RARP [717,718]. Higher positive surgical margin rates have been consistently reported for rsRARP (OR 0.45, p < 0.05); however, no significant differences in BCR have been reported [718,719].
6.2.2.d. Other surgical technique considerations
Management of the dorsal venous complex
Management of the dorsal venous complex will largely depend on surgical choice, as neither early (prior to) nor standard (after bladder neck incision) ligation was shown to be beneficial for functional or oncological outcomes in a single-centre RCT [720]. Ligation of the DVC can be performed with standard suture or using a vascular stapler. A single-centre RCT comparing stapler to suture ligation and suture ligation with suspension to the pubic bone reported no difference between the stapler (88%) or suture (88%) groups for continence recovery (0 pads/day with or without security pad) at three and fifteen months (99%) [721].
Removal of seminal vesicles
For oncological clearance, the seminal vesicles (SV) have traditionally been removed intact with the prostate specimen [722]. An RCT comparing nerve-sparing RP with and without an SV-sparing approach found no difference in margin status, PSA recurrence, continence or erectile function outcomes. [723]. Whilst complete SV removal should be the default, preservation of the SV tips may be considered in cases of low risk of involvement.
Bladder neck mucosal eversion
During open RP, mucosal eversion of the bladder neck aims for a mucosa-to-mucosa vesicourethral anastomosis to reduce anastomotic stricture; however, this has not been shown to reduce anastomotic stricture rate [724]. The strongest predictor of anastomotic stricture in RP is current cigarette smoking [725], but also surgical approach (open more likely than RARP) [726].
Pneumoperitoneum pressure
Use of pneumoperitoneum during minimally invasive (laparoscopic, robotic-assisted) RP is likely to reduce bleeding at the expense of increased abdominal pressure and associated physiological changes. In a randomised triple-blinded study comparing RARP (with standard DVC ligation, n=98) low-pressure (7 mmHg) versus standard-pressure (12 mmHg) pneumoperitoneum, low pressure was associated with better postoperative pain and other parameters on postoperative day one at the expense of statistically higher blood loss of questionable clinical relevance (mean 227 mL vs. 159.9mL; p = 0.001)[727].
6.2.2.e. Postoperative considerations
Urinary catheter
Recommendations for use of prophylactic antibiotics at time of indwelling catheter (IDC) removal to reduce UTI are provided by the EAU Guidelines on Urological Infections. Clinicians should refer to their local institutional guidelines on thromboprophylaxis for deep venous thrombosis prophylaxis recommendations for RP.
Cystography prior to catheter removal
Cystography prior to catheter removal can check for a substantial anastomotic leak, which may defer catheter removal to allow further healing and sealing of the anastomosis. Men with LUTS, large prostates, previous TURP or bladder neck reconstruction, or intraoperative leak may benefit most from postoperative cystography as these factors have been associated with leakage [728,729]. Heterogeneity in quality of available data, including variable prevalence of leakage and unclear long term impact, mean that recommendations on use of routine cystography cannot be provided.
Pelvic drain
A pelvic drain has traditionally been used in RP for potential drainage of urine leaking from the vesico-urethral anastomosis, blood or lymphatic fluid when a PLND has been performed. Two RCTs have been performed in the robotic-assisted laparoscopic setting [730,731]. Patients with urine leak at intraoperative anastomosis watertight testing were excluded. Both trials showed noninferiority in complication rates when no drain was used. When the anastomosis is found to be watertight intraoperatively, it is reasonable to avoid inserting a pelvic drain. There is no evidence to guide usage of a pelvic drain in PLND.
6.2.2.f. Acute and chronic complications of radical prostatectomy
An important consideration is whether complications are reduced by using newer techniques such as RARP.
Postoperative outcomes
Table 6.1.4 lists the intra- and peri- operative complications of RRP and RARP. Robot-assisted RP generally requires Trendelenberg position with head down, often using lithotomy positioning with leg stirrups. Surgeons and patients should be aware of the risk of neuropathy, affecting up to 11% of patients mostly in the lower limbs [732,733]. An SR and meta-analysis of unplanned hospital visits and readmissions post-RP analysed 60 studies with over 400,000 patients over a 20-year period up to 2020. The SR found an emergency room visit rate of 12% and a hospital readmission rate of 4% at 30 days postoperatively [734].
Functional outcomes
Systematic reviews have documented complication rates after RARP [735-739] and can be compared with contemporaneous reports after RRP [740]. A prospective controlled non-RCT of patients undergoing RP in 14 centres using RARP or RRP showed that, twelve months after RARP, 21.3% of patients were incontinent, as were 20.2% after RRP (adjusted OR: 1.08, 95% CI: 0.87–1.34) [741]. Erectile dysfunction was observed in 70.4% after RARP and 74.7% after RRP. The adjusted OR was 0.81 (95% CI: 0.66–0.98) [741].
An RCT comparing RARP and RRP reported outcomes at 12 weeks in 326 patients and functional outcomes at two years [742]. Urinary function scores did not differ significantly between RRP versus RARP at six and 12 weeks post-surgery (74–50 vs. 71–10, p = 0.09; 83–80 vs. 82–50, p = 0.48), with comparable outcomes for sexual function scores (30–70 vs. 32–70, p = 0.45; 35–00 vs. 38–90, p = 0.18). In the RRP group, 14 (9%) patients had postoperative complications versus six (4%) in the RARP group. A similar single-centre RCT with multiple surgeons comparing RARP to RRP in 327 patients reported lower bleeding, shorter hospitalisation duration and complications (11% vs. 17%, p = 0.1) for RARP [743]. Higher continence rates were observed for RARP at three (80% vs. 65%, p = 0.002), six (90% vs. 82%, p = 0.04) and eighteen months (95% vs. 79%, p < 0.001). Similar trends were observed for potency at three and six months.
A subsequent meta-analysis of five RCTs (1,205 patients) that compared RARP with LRP showed no difference in continence at 12 months (OR 1.95, 95% CI 0.67 – 5.62) or oncological outcomes (positive margin rate, biochemical recurrence). RARP, however, resulted in better three- (OR 1.81) and six-month (OR 1.88) continence outcomes, as well as erectile recovery in preoperatively potent patients (OR 4.05, p = 0.003) [744]. At ten years follow-up, RARP and LRP were shown to have comparable continence and potency rates, however quality of continence (totally dry) and potency (erection quality) were higher for RARP with similar oncological outcomes [745].
Recommendations for management of post-RP erectile dysfunction are provided by the EAU Guidelines on Sexual and Reproductive Health.
Table 6.2.4: Intra-and perioperative complications of retropubic RP, laparoscopic RP and RARP (adapted from [735])
| Intra-and perioperative complications of retropubic RP, laparoscopic RP and RARP | |||
| Predicted probability of event | RARP (%) | Laparoscopic RP (%) | RRP (%) |
| Bladder neck contracture | 1.0 | 2.1 | 4.9 |
| Anastomotic leak | 1.0 | 4.4 | 3.3 |
| Infection | 0.8 | 1.1 | 4.8 |
| Organ injury | 0.4 | 2.9 | 0.8 |
| Ileus | 1.1 | 2.4 | 0.3 |
| Deep vein thrombosis | 0.6 | 0.2 | 1.4 |
| Predicted rates of event | RARP (%) | Laparoscopic RP (%) | RRP (%) |
| Clavien-Dindo I | 2.1 | 4.1 | 4.2 |
| Clavien-Dindo II | 3.9 | 7.2 | 17.5 |
| Clavien-Dindo IIIa | 0.5 | 2.3 | 1.8 |
| Clavien-Dindo IIIb | 0.9 | 3.6 | 2.5 |
| Clavien-Dindo IVa | 0.6 | 0.8 | 2.1 |
| Clavien-Dindo V | < 0.1 | 0.2 | 0.2 |
RARP = robot-assisted radical prostatectomy; RP = radical prostatectomy; RRP = radical retropubic prostatectomy.
6.2.2.f.1. Effect of anterior and posterior reconstruction on continence
Preservation of integrity of the external urethral sphincter is critical for continence post-RP. Less clear is the effect of reconstruction of surrounding support structures to return to continence. Several small RCTs have been conducted, however, pooling analyses is hampered by variation in the definitions of incontinence and surgical approach, such as open versus robotic and intraperitoneal versus extra-peritoneal. In addition, techniques used to perform both anterior suspension or reconstruction and posterior reconstruction are varied. Anterior suspension, for example, is performed either through periosteum of the pubis or the combination of ligated DVC and puboprostatic ligaments (PPL). Posterior reconstruction from rhabdosphincter is described to either Denonvilliers’ fascia posterior to bladder or to the posterior bladder wall itself.
Two trials assessing posterior reconstruction in RARP found no significant improvement in return to continence [708,709]. A third trial using posterior bladder wall for reconstruction showed only an earlier return to one pad per day (median 18 vs. 30 days, p = 0.024) [710]. When combining both anterior and posterior reconstruction, where for anterior reconstruction the PPL were sutured to the anterior bladder neck, another RCT found no improvement compared to a standard anastomosis with no reconstruction [711].
Four RCTs including anterior suspension have also shown conflicting results. Anterior suspension alone through the pubic periosteum, in the setting of extra-peritoneal RARP, showed no advantage [713]. However, when combined with posterior reconstruction in RRP, one RCT showed significant improvement in return to continence at one month (7.1% vs. 26.5%, p = 0.047) and three months (15.4% vs. 45.2%, p = 0.016), but not at six months (57.9% vs. 65.4%, p = 0.609) [714]. Another anterior plus posterior reconstruction RCT using the Advanced Reconstruction of Vesico-urethral Support (ARVUS) technique and the strict definition of continence of ‘no pads’, showed statistically significant improvement in continence at two weeks (43.8% vs. 11.8%), four weeks (62.5% vs. 14.7%), eight weeks (68.8% vs. 20.6%), six months (75% vs. 44.1%) and twelve months (86.7% vs. 61.3%) when compared to standard posterior Rocco reconstruction [715]. Anterior suspension alone through the DVC and PPL combined without posterior construction in the setting of RARP has shown improvement in continence at one month (20% vs. 53%, p = 0.029), three months (47% vs. 73%, p = 0.034) and six months (83% vs. 100%, p = 0.02), but not at twelve months (97% vs. 100%, p = 0.313) [716]. Together, these results suggest a possible earlier return to continence, but no long-term difference.
A novel method of urethral reconstruction with peritoneal support flaps was shown in a randomised trial compared to standard RARP (n = 96) to improve urinary continence recovery (0-1 pad) at one month (73% vs. 49%, p = 0.017) and three months (93% vs. 77%, p = 0.025); however, patient reported outcomes, complications and oncological outcomes were similar [712].
Because there is conflicting evidence regarding the effect of anterior and/or posterior reconstruction on return to continence post-RP, no recommendations can be made. However, no studies showed an increase in adverse oncologic outcome or complications with reconstruction.
6.2.2.f.2. Deep venous thrombosis prophylaxis
As with all pelvic cancer surgery lasting over one hour, there is a measurable increased risk of deep vein thrombosis, and therefore consideration should be given to chemical thrombosis prophylaxis, commonly used for three to four weeks after surgery. This should be adapted based on national recommendations, when available.
6.2.3. Radiotherapy
Intensity-modulated RT (IMRT) or volumetric-modulated arc therapy (VMAT) with image-guided RT (IGRT) is currently widely recognised as the standard treatment approach for EBRT.
6.2.3.a. External beam radiation therapy
6.2.3.a.1. Technical aspects
Intensity-modulated RT and VMAT employ dynamic multi-leaf collimators, which automatically and continuously adapt to the contours of the target volume seen by each beam. Viani et al. show significantly reduced acute and late grade ≥ 2 genitourinary (GU) and gastrointestinal (GI) toxicity in favour of IMRT, while BCR-free rates did not differ significantly when comparing IMRT with three-dimensional conformal RT (3D-CRT) in an RCT comprising 215 patients [746]. A meta-analysis by Yu et al. (23 studies, 9,556 patients) concluded that IMRT significantly decreases the occurrence of grade 2–4 acute GI toxicity, late GI toxicity and late rectal bleeding, and achieves better PSA relapse-free survival in comparison with 3D-CRT. Intensity-modulated EBRT and 3D-CRT show comparable acute rectal toxicity, late GU toxicity and OS, while IMRT slightly increases the morbidity of acute GU toxicity [747]. Therefore, IMRT plus IGRT remain the SOC for the treatment of PCa.
The advantage of VMAT over IMRT is shorter treatment times, generally two to three minutes in total. Both techniques allow for a more complex distribution of the dose to be delivered and provide concave isodose curves, which are particularly useful as a means of sparing the rectum. Radiotherapy treatment planning for IMRT and VMAT differs from that used in conventional EBRT, requiring a computer system capable of ‘inverse planning’ and the appropriate physics expertise. Treatment plans must conform to prespecified dose constraints to critical organs at risk of normal tissue damage and a formal quality assurance process should be routine.
With dose escalation using IMRT/VMAT, organ movement becomes a critical issue in terms of both tumour control and treatment toxicity. Techniques will therefore combine IMRT/VMAT with some form of IGRT (usually gold-marker or cone-beam CT), in which organ movement can be visualised and corrected for in real time, although the optimum means (number of applications per week) of achieving this is still unclear [748,749]. Tomotherapy is another technique for the delivery of IMRT, using a linear accelerator mounted on a ring gantry that rotates as the patient is delivered through the centre of the ring in a manner analogous to spiral CT scanning.
The use of MR-guided adapted RT is still investigational [750]. Planning studies confirm that MR-based adaptive RT significantly reduces doses to organs at risk (OAR), and this may translate into clinical benefit [751]. Although the rates of acute GI and GU toxicity appear low - based mostly on patients treated with stereotactic RT [752], follow-up is too short for definitive conclusions [750]. Due to the daily fraction time of up to 45 minutes [750,752], the heavy MR-workflow and the limited field size (rendering most pelvic fields too large), MR-guided adapted RT implementation is not yet routine [750]. A prospective single centre RCT, the MIRAGE trial (CT-guided Stereotactic Body Radiation Therapy and MRI-guided Stereotactic Body Radiation Therapy for Prostate Cancer) demonstrates reduced acute GU and GI toxicity with MRI-guided SBRT and margin reduction from 4mm to 2mm [753]. Secondary endpoints of cumulative incidence rates of late grade ≥ 2 toxicity at two years with MRI-guided versus CT-guided SBRT were 27% (95% CI 19–39%)] versus 51% (95% CI 41–63%) for GU toxicity (p = 0.004), and 1.4% (95% CI 0.2–9.6) versus 9.5% (95% CI 4.6–19) for GI toxicity (p = 0.025). Cumulative logistic regression revealed that MRI-guided SBRT was associated with significantly lower odds of a clinically relevant deterioration in bowel function according to the Expanded Prostate Cancer Index Composite-26 score (OD 0.444, 95% CI 0.209–0.942; p = 0.035) and in the Sexual Health Inventory in Men score (OD 0.366, 95% CI 0.148–0.906; p = 0.03) [754].
6.2.3.a.2. Dose escalation
Local control is a critical issue for the outcome of RT of PCa. Local failure due to insufficient total dose has been shown to be prognostic for death from PCa, because a second wave of metastases is seen five to ten years later [755]. Several RCTs have shown that dose escalation (range 74–80Gy) has a significant impact on 10-year biochemical relapse as well as metastases and disease-specific mortality [756-763]. These trials have generally included patients from several risk groups, and the use of neoadjuvant/adjuvant ADT has varied (see Table 6.2.5). The best evidence of an OS benefit in patients with intermediate- or high-risk PCa derives from a non-randomised, but well-conducted propensity-matched retrospective analysis of the United States National Cancer Database by Kalbasi et al., including a total of 42,481 patients [764]. If IMRT/VMAT and IGRT are used for dose escalation, rates of severe late side effects (> grade 3) are 2–4% for the rectum and 2–6% for the GU tract [758,765].
The concept of a focal boost to the dominant intraprostatic lesion (DIL) visible on MRI rather than global prostate dose escalation has been successfully validated in a RCT of 571 intermediate- and high-risk patients [765,766]. Patients were randomised between 77Gy in 35 fractions of 2.2Gy and the same dose plus a focal boost up to 18Gy. Additional ADT was given to 65% of patients in both arms, however, the duration of the ADT was not reported. With a median follow-up of 106 months, there was a significant improvement of ten-year biochemical DFS (BDFS) (primary endpoint) from 71% (95%CI 65-77%) in the control arm to 86% (95%CI 81-91%) in the focal boost arm (p < 0.001). In addition, focal boosting decreased local and regional lymph node failure but did not impact DMFS and OS. No significant difference for late GU or GI toxicity grade ≥ 2 (23% and 12% vs. 28% and 13%) was documented at up to five years [765]. For grade ≥ 3 GU-toxicity, these numbers were 3.5% and 5.6% (p > 0.05). However, longer follow-up is needed to assess late GU toxicity [767]. Of note, there was a dose response, with increased boost dose associated with reduced distant metastatic failure [766]. Systematic review of MRI-defined DIL focal boost studies using standard fractionation shows good tolerability and improved BPFS [768]. Its role when using hypofractionation and ultrahypofractionation is under investigation.
6.2.3.a.3. Hypofractionation
Fractionated RT utilises differences in the DNA repair capacity of normal and tumour tissue and slowly proliferating cells are very sensitive to an increased dose per fraction [769]. A meta-analysis of 25 studies including > 14,000 patients concluded that, since PCa has a slow proliferation rate, hypofractionated RT could be more effective than conventional fractions of 1.8–2Gy [770]. Hypofractionation (HFX) has the added advantage of being more convenient for the patient at lower cost.
Moderate HFX is defined as RT with 2.5–3.4Gy/fx. Several studies report on moderate HFX applied in various techniques, including in part ADT [771-778]. A Cochrane review on moderate HFX for clinically localised PCa [779] included eleven studies (n = 8,278) with a median follow-up of 72 months showing little or no difference in PCa-specific survival (HR: 1.00) when compared to conventionally fractionated radiotherapy. Based on four studies (n = 3,848), moderate HFX to the prostate alone likely makes little or no difference to late radiation GU toxicity (RR: 1.05) or GI toxicity (RR: 1.1). An individual patient meta-analysis of seven phase III clinical trials comparing conventional with iso-toxic or dose escalated moderate HFX, shows no differences in PFS (HR 0.92, 95% CI 0.81–1.05; p = 0.21 and 0.94, 0.82–1.09; p = 0.43, respectively) [780]. The odds of grade 2 or higher gastrointestinal toxic effects were significantly higher with dose-escalated (OR 1·48, 95% CI 1·14–1·92; p=0·0035) with bowel quality-of-life decrement (OR 1·68, 95% CI 1·07–2·61; p=0·023). Dose escalated moderate HFX is therefore not recommended.
Toxicity outcomes in two RCTs recruiting high-risk patients and adding elective pelvic nodal radiation have reported. The PCS-5 multicentre RCT recruited high risk patients (25.9% T3-4) and an initial two-year toxicity analysis demonstrated comparable G2+ GI toxicity across treatment arms with lower rates of late G2+ GU toxicity with HFX [781]. No differences were seen in survival outcomes at median follow-up of five years although, as secondary endpoints, extrapolation of survival results is limited by small sample size [782]. In the single-center randomised pHART2-RCT, an increase in five-year G3+ GI toxicity was noted when HFX was combined with elective pelvic nodal RT [783]. In the postoperative setting, moderate HFX is non-inferior in terms of two-year patient-reported toxicity to conventional fractionation with similar rates of patient reported GI and GU toxicity [784].
Ultra-HFX uses even larger doses per fraction and requires IGRT or ideally stereotactic body RT (SBRT). Table 6.2.7 provides an overview of selected studies investigating the role of Ultra-HFX in treating predominantly intermediate-risk localised disease. Biochemical control is comparable to conventional fractionation. However, there are concerns about higher-grade GU toxicity, and UHFX should be avoided in patients with severe pre-existing lower urinary tract symptoms (IPSS > 19) and/or outflow obstruction with or without median lobe [785,786]. In the HYPO-RT-PC randomised trial (n = 1,200), no difference in failure-free survival was seen for conventional or ultra-HFX, but acute grade ≥ 2 GU toxicity was 23% versus 28% (p = 0.057), favouring conventional fractionation. There were no significant differences in long-term toxicity [785]. Presentation reports indicate that after a median follow-up duration of 10.6 years, UHF was shown to be non-inferior to CF in terms of failure-free survival, with ten-year failure-free survival rates of 72% for UHF compared with 65% for CF (adjusted HR = 0.84, 95% CI: 0.69-1.03) [787].
In the Intensity-modulated fractionated RT versus SBRT for PCa (PACE-B) trial, acute grade ≥ 2 GU or GI toxicities did not differ significantly between conventional fractionation and ultra-HFX [788]. At two years, treatment was well-tolerated in both arms with no differences in RTOG ≥ grade 2 GU or GI toxicities, but clinician scoring of urinary toxicity using CTCAE and patient-reported Expanded Prostate Cancer Index Composite (EPIC)-26 urinary bother scores were both higher in the SBRT arm [789]. After 74 months median follow-up, five-year biochemical/clinical failure free-rates were 94.6% (95% CI 91.9%, 96.4%) in the control arm and 95.8% (95% CI 93.3%, 97.4%) in the SBRT arm, confirming that SBRT is non-inferior (HR 0.73 90% CI 0.48-1.12, p for non-inferiority = 0.004). The cumulative five-year rate of late RTOG grade 2+ GI toxicity was similar in both arms (10%), but higher rates of cumulative five-year RTOG grade 2+ GU toxicity occurred with SBRT at 26.9% (95% CI 22.8, 31.5%) compared to the control arm at 18.3% (95% CI 14.8, 22.5%) [786]. The GU toxicity is temporary with no statistical difference in clinician-reported toxicity between groups at five years and no clinically relevant difference in patient-reported outcomes in the five years of follow-up. Adopting planning dose constraints to the penile bulb might minimise ED, particularly in younger patients [790].
6.2.3.a.4. Neoadjuvant or adjuvant hormone therapy plus radiotherapy
The combination of RT with luteinising hormone releasing hormone (LHRH) ADT has superiority compared with RT alone followed by deferred ADT on relapse, as shown by phase III RCTs [791-802] (Table 6.2.8). The main message is that, for intermediate-risk disease, a short duration of four to six months is optimal, while a longer duration of two to three years is needed for high-risk patients. The largest RCT in intermediate-risk disease comparing dose-escalated RT with or without six months of ADT failed to demonstrate an OS advantage with a median follow-up time of 6.3 years. Six months of ADT use was associated with reduced PSA failure, fewer distant metastases and improved prostate-cancer-specific mortality [802].
The question of the added value of EBRT combined with ADT has been clarified by three RCTs. All showed a clear benefit of adding EBRT to long-term ADT (Table 6.2.9).
The combination of ADT with various forms of RT has been studied extensively, with extremely strong evidence for the use of such combined modality therapy in several settings. The MARCAP (Individual Patient Data Meta- Analysis of Randomised Trials in Cancer of the Prostate) consortium conducted a meta-analysis of trials using individual patient data (IPD) and a primary endpoint of MFS. Trials were eligible if they studied the use or prolongation of ADT in patients receiving definitive RT and included 12 trials with 10,853 patients. Median follow-up was over 11 years. The use of ADT was clearly associated with significant improvements in BCR, metastatic recurrence, MFS and OS. The benefits of ADT were independent of RT dose, age and risk groups comparing NCCN unfavourable intermediate-risk, high-risk and locally advanced disease. The extension of duration of neoadjuvant ADT showed no demonstrable benefits [803].
A meta-analysis from two RCTs (RTOG 9413 and Ottawa 0101) compared neoadjuvant/concomitant versus adjuvant ADT (without substratifying between favourable and unfavourable intermediate-risk disease) in conjunction with prostate RT and reported superior PFS with adjuvant ADT, but the data heterogeneity means that this observation is hypothesis-generating only [804].
In addition, a Canadian two-arm dose-escalated (76Gy) RCT compared neoadjuvant and concomitant with adjuvant short-term ADT in 432 patients with intermediate-risk PCa. After ten years, no significant difference in OS or RT-related grade ≥ 3 GI or GU toxicity was seen [805]. Therefore, both regimens in combination with dose escalation are reasonable standards.
6.2.3.b. Proton beam therapy
In theory, proton beams are an attractive alternative to photon-beam RT for PCa, because they deposit almost all their radiation dose at the end of the particle’s path in tissue (the Bragg peak) in contrast to photons, which deposit radiation along their path. There is also a very sharp fall-off for proton beams beyond their deposition depth, meaning that critical normal tissues beyond this depth could be effectively spared. In contrast, photon beams continue to deposit energy until they leave the body, including an exit dose. The PARTIQoL Phase III RCT compared proton beam therapy (PBT) with IMRT in 450 participants with localised prostate cancer. With a median follow-up of 60.3 months, no difference in any QoL domain or PFS was found [806]. Proton beam therapy has no advantages over less resource-intensive IMRT/VMAT. However, the publication of the full study is awaited to confirm the results.
Table 6.2.5: Randomised trials of dose escalation in localised PCa
| Trial | n | PCa condition | Radiotherapy Dose | Follow-up (median) | Outcome | Results |
| MD Anderson study 2011 [763] | 301 | T1-T3, N0, M0, PSA ≤ 10ng/mL PSA 10-20ng/mL PSA > 20ng/mL | 70 vs.78Gy | 15 yr. | DM, DSM, FFF | All patients: 18.9% FFF at 70Gy; 12% FFF at 78Gy; (p = 0.042) 3.4% DM at 70Gy; 1.1% DM at 78Gy; (p = 0.018) 6.2% DSM at 70Gy; 3.2% DSM at 78Gy; (p = 0.043) No difference in OS (p > 0.05) |
| PROG 95-09 2010 [757] | 393 | T1b-T2b, PSA ≤ 15ng/mL Low risk: T1-2a, PSA < 10mg/mL, GS ≤ 6. Interim risk: PSA 10-15ng/mL or GS 7 or T2b. High risk: GS 8-10. | 70.2 vs.79.2Gy, including proton boost 19.8 vs. 28.8Gy | 8.9 yr. | 10-yr. ASTRO BCF | All patients: Low-risk patients: |
| MRC RT01 2014 [762] | 843 | T1b-T3a, N0, M0 PSA < 50ng/mL neoadjuvant ADT | 64 vs. 74Gy | 10 yr. | BFS, OS | 43% BFS at 64Gy; 55% BFS at 74Gy; (p = 0.0003) 71% OS both groups (p = 0.96) |
| Dutch RCT 2014 [761] | 664 | T1b-T4 143 pts. with (neo) adjuvant ADT | 68 vs. 78Gy | 110 mo. | Freedom biochemical (Phoenix) and/or clinical failure at 10 yr. | 43% FFF at 68Gy; 49% FFF at 78Gy; (p = 0.045) |
| GETUG 06 2011 [760] | 306 | T1b-T3a, N0, M0 PSA < 50ng/mL | 70 vs. 80Gy | 61 mo. | BCF (ASTRO) | 39% BF at 70Gy; 28% BF at 80Gy |
| RTOG 0126 2018 [756] | 1,532 | T1b-T2b ISUP GG 1 + PSA 10-20ng/mL or ISUP GG 2/3 + PSA < 15ng/mL | 70.2 vs. 79.2Gy | 100 mo. | OS, DM, BCF (ASTRO) | 75% OS at 70.2Gy; 76% OS at 79.2Gy 6% DM at 70.2Gy; 4% DM at 79.2Gy; (p = 0.05) 47% BCF at 70.2Gy; 31% BCF at 79.2Gy; (p < 0.001; Phoenix, p < 0.001) |
| FLAME Trial [765-767] | 571 | EAU risk classification: High risk (84%) | 77Gy (35fx 2.2Gy) vs. 77Gy 35fx) + focal boost (up to 18Gy) ADT (65% both arms - duration unknown) | 106 mo. | bDFS (10 yr.) | bDFS 86% at 77Gy+boost; 71% at 77Gy (p<0.001) Focal boost in favour of local and regional lymph node DFS |
ADT = androgen-deprivation therapy; BCF = biochemical failure; BFS = biochemical progression-free survival; DM = distant metastases; DSM = 50 disease-specific mortality; FFF = freedom from biochemical or clinical failure; fx = fractions; GS = Gleason score; ISUP = International Society of Urological Pathology; MFS = metastasis-free survival; mo. = months; n = number of patients; OS = overall survival; PSA = prostate-specific antigen; bDFS = biochemical disease-free survival; yr. = years.
Table 6.2.6: Major phase III randomised trials of moderate hypofractionation for primary treatment
| Study/Author | n | Risk, ISUP GG, or NCCN | ADT | RT Regimen | BED, Gy | Follow-up (median) | Outcome |
Lee, et al. 2024 [807] | 550 542 | low risk | None | 70Gy/28fx 73.8Gy/41fx | 80 69.6 | 150 mo. | 12 yr. DFS 56.1% (95% CI, 51.5 to 60.5) control arm and 61.8% (95% CI, 57.2 to 66.0) for HFX. HR 0.85 (95% CI, 0.71 to 1.03) |
Dearnaley, et al. CHHiP 2016 [774] | 1,077/19fx 1,074/20fx 1,065/37fx | 15% low 73% intermediate 12% high | 3-6 mo. before and during EBRT | 57 Gy/19fx 60 Gy/20fx 74 Gy/37fx | 73.3 77.1 74 | 62 mo. | 5 yr. BCDF 85.9% (19fx) 90.6% (20fx) 88.3% (37fx) |
| De Vries, et al. 2020 [808] | 403 392 | 30% ISUP GG 1 45% ISUP GG 2-3, 25% ISUP GG 4-5 | None | 64.6 Gy/19fx 78 Gy/39fx | 90.4 78 | 89 mo. | 8-yr. OS 80.8% vs. 77.6% (p = 0.17) 8 yr. TF 24.4% vs. 26.3% |
Catton, et al. 2017 [776] | 608 | Intermediate risk 53% T1c 46% T2a-c | None | 60 Gy/20fx | 77.1 | 72 mo. | 5 yr. BCDF both arms 85% HR: 0.96 (n.s.) |
| 598 | 9% ISUP GG 1 63% ISUP GG 2 28% ISUP GG 3 | 78Gy/39fx | 78 | ||||
Glicksman, et al. 2024 PHART-2 [783] | 186 | All high risk N0M0 T1-2 82.8% T3-4 12.2% | 22 mo. median | 68Gy to prostate (SIB) + 48Gy to pelvis in 25fx
78Gy to prostate + 46Gy to pelvis in 39fx | 82
78 | 67 mo. | No difference in acute toxicity and PROs Higher 5-yr cumulative G3+ GI in HFX 13.5% (95% CI, 7.1%-21.9%) vs 2.4% (95% CI, 0.5%-7.6%) (P = 0.01) |
Niazi, et al. 2023 PCS-5 [781] | 329 | All high risk N0M0 T1-2 73.8% T3-4 25.9% | 28 mo. - 3 mo. before during and after EBRT | 68Gy to prostate (SIB) + 45Gy to pelvis in 25fx
76Gy to prostate + 46Gy to pelvis in 38fx | 82
76 | 24 mo. | Similar 2 yr. G2+ GI toxicity (8-10%)
Reduced 2 yr G2+ GU toxicity with HFX (4.3% vs 15.9%; p=0.035) |
ADT = androgen deprivation therapy; BCDF = biochemical or clinical disease failure; BED = biologically equivalent dose, calculated to be equivalent in 2Gy fractions using an α/ß of 1.5Gy; DFS = disease-free survival; EBRT = external beam radiotherapy; fx = fractions; GG = grade group; GI = gastrointestinal; GU = genitourinary; HFX = hypofractionation; HR = hazard ratio; ISUP = International Society of Urological Pathology; mo. = months; n = number of patients; OS = overall survival; NCCN = National Comprehensive Cancer Network; n.s. = not significant; PROs = patient1397645928reported outcomes; RT = radiotherapy; SIB = simultaneous integrated boost; TF = treatment failure; yr. = years.
Table 6.2.7: Selected trials on ultra-hypofractionation for intact localised PCa
| Study | n | med FU (mo) | Risk-Group | Regimen (TD/fx) | Outcome |
| Widmark et al. 2019 HYPO-RT-PC [785] | 1,200 | 60 | 89% intermediate 11% high | 78 Gy / 39 fx, 8 wks 42.7 Gy / 7 fx, 2.5 wks No SBRT | FFS at 5 yrs 84% in both arms |
Brand et al. 2019 Tree et al. 2022 Van As et al. 2024 [789] PACE-B [788] [786] | 874 | 74 | 9.3% NCCN low 90.7% NCCN intermediate ISUP GG 3 excluded | 78Gy/39fx, 7.5 wks or 62Gy/20fx 4wks 36.25Gy/5fx, 1-2 wks SBRT | Biochemical/clinical FFS at 5 yrs 94.6% (CRT) vs. 95.6% (SBRT) Cumulative 5-yr G 2+ GI toxicity similar (10%) Cumulative 5-yr G2+ GU SBRT 26.9% (95%CI 22.8,31.5%) CRT 18.3% (95%CI 14.8,22.5%). |
CRT = control arm radiotherapy; FFS = failure-free survival; FU = follow-up; fx = number fractions; mo. = months; n = number of patients; SBRT = stereotactic body radiotherapy; TD = total dose; wks = weeks; yr. = years.
Table 6.2.8: Selected studies of use and duration of ADT in combination with RT for PCa
| Study | TNM stage | n | Trial | ADT | RT | Effect on OS |
RTOG 85-31 2005 [793] | T3 or N1 M0 | 977 | EBRT ± ADT | Orchiectomy or LHRH agonist 15% RP | 65-70Gy | Significant benefit for combined treatment (p = 0.002) seems to be mostly caused by patients with ISUP grade group 2-5 |
RTOG 94-13 2007 [797] | T1c-4 N0-1 M0 | 1,292 | ADT timing comparison | 2 mo. neoadjuvant plus concomitant vs. 4 mo. adjuvant suppression | Whole pelvic RT vs. prostate only; 70.2Gy | No significant difference between neoadjuvant plus concomitant vs. adjuvant androgen suppression therapy groups (interaction suspected) |
RTOG 86-10 2008 [794] | T2-4 N0-1 | 456 | EBRT ± ADT | Goserelin plus flutamide 2 mo. before, plus concomitant therapy | 65-70Gy RT | No significant difference at 10 yr. |
| D’Amico AV, et al. 2008 [795] | T2 N0 M0 (localised unfavourable risk) | 206 | EBRT ± ADT | LHRH agonist plus flutamide for 6 mo. | 70Gy 3D-CRT | Significant benefit that may pertain only to men with no or minimal co-morbidity (HR: 0.55, 95% CI: 0.34-0.90, p = 0.01) |
RTOG 92-02 2008 [798] | T2c-4 N0-1 M0 | 1,554 | Short vs. prolonged ADT | LHRH agonist given for 2 yr. as adjuvant after 4 mo. as neoadjuvant | 65-70Gy | p = 0.73, p = 0.36 overall; significant benefit (p = 0.044) (p = 0.0061) in subset with ISUP grade group 4-5 |
| EORTC 22961 2009 [799] | T1c-2ab N1 M0, T2c-4 N0-1 M0 | 970 | Short vs. prolonged ADT | LHRH agonist for 6 mo. vs. 3 yr. | 70Gy 3D-CRT | Better result with 3 yr. treatment than with 6 mo. (3.8% improvement in survival at 5 yr.) |
EORTC 22863 2010 [792] | T1-2 poorly differentiated and M0, or T3-4 N0-1 M0 | 415 | EBRT ± ADT | LHRH agonist for 3 yr. (adjuvant) | 70Gy RT | Significant benefit at 10 yr. for combined treatment (HR: 0.60, 95% CI: 0.45-0.80, p = 0.0004). |
| TROG 96-01 2011 [796] | T2b-4 N0 M0 | 802 | Neoadjuvant ADT Duration | Goserelin plus flutamide 3 or 6 mo. before, plus concomitant suppression | 66Gy 3D-CRT | No significant difference in OS reported; benefit in PCa-specific survival (HR: 0.56, 95% CI: 0.32-0.98, p = 0.04) (10 yr.: HR: 0.84, 0.65-1.08, p = 0.18) |
RTOG 99-10 2015 [800] | intermediate risk 94% T1-T2; 6% T3-4 | 1,579 | Short vs. prolonged ADT | LHRH agonist 8 + 8 vs. 8 + 28 wk. | 70.2Gy 2D/3D | 67 vs. 68%, p = 0.62, confirms 8 + 8 wk. LHRH as a standard |
| PCSIII 2020 [801] | Intermediate risk | 600 | 76Gy alone vs. 76Gy + ADT vs. 70Gy + ADT | LHRH + bicalutamide 6 mo. 4 mo. prior to RT | 70 vs. 7 Gy | Significantly improved biochemical failure-free and PCa-specific survival for ADT arms, with no difference in OS. |
RTOG 0815 2023 [802] | Intermediate risk | 1,492 | Dose - escalated RT ± ADT | LHRH agonist/antagonist + bicalutamide or flutamide 6 mo. 2 mo. prior to RT | 79.2Gy (89%) 45Gy + BT boost (11%) | No difference in OS. Significantly improved biochemical failure-free, metastatic-free survival and PCa-specific survival for ADT arm. |
3D-CRT = three-dimensional conformal radiotherapy; ADT = androgen deprivation therapy; BT = brachytherapy; CI = confidence interval; EBRT = external beam radiotherapy in standard fractionation; HR = hazard ratio; ISUP = International Society of Urological Pathology; LHRH = luteinising hormone-releasing hormone; mo. = months; n = number of patients; OS = overall survival; RP = radical prostatectomy; RT = radiotherapy; wks = weeks; yr. = years.
Table 6.2.9: Selected studies of ADT in combination with, or without, RT for PCa
| Study | TNM stage | n | Trial design | ADT | RT | Effect on OS |
| SPCG-7/SFUO-3 2016 [809] | T1b-2 WHO Grade 1-3, T3 N0 M0 | 875 | ADT ± EBRT | LHRH agonist for 3 mo. Plus continuous flutamide | 70Gy 3D-CRT vs. no RT | 34% (95% CI: 29-39%) vs. 17% (95% CI: 13-22% CSM at 12 (15) yr. favouring combined treatment (p < 0.0001 for 15-yr. results) NCIC CTG PR.3/MRC |
| PRO7/NCIC 2015 [810] | T3-4 (88%), PSA > 20ng/mL (64%), ISUP GG 4-5 (36%) N0 M0 | 1,205 | ADT ± EBRT | Continuous LHRH agonist | 65-70Gy 3D-CRT vs. no RT | 10-yr. OS = 49% vs. 55% favouring combined treatment HR: 0.7, p < 0.001) |
| Sargos, et al., 2020 [811] | T3-4 N0 M0 | 273 | ADT ± EBRT | LHRH agonist for 3 yr. | 70Gy 3D-CRT vs. no RT | Significant reduction of clinical progression; 5-yr. OS 71.4% vs. 71.5% |
3D-CRT = three-dimensional conformal radiotherapy; ADT = androgen-deprivation therapy; CSM = cancer-specific mortality; EBRT = external beam radiotherapy; HR = hazard ratio; LHRH = luteinising hormone-releasing hormone; mo. = months; n = number of patients; OS = overall survival; PSA = prostate-specific antigen; RT = radiotherapy; yr. = years.
6.2.3.c. Spacer during external beam radiation therapy
Biodegradable spacer insertion involves using a liquid gel or balloon to increase the distance between the prostate and rectum and consequently reduce the amount of radiation reaching the rectum. Various materials have been used with most evidence available for CE-marked hydrogel spacers [812]. An SR and meta-analysis including three RCTs, three prospective cohorts, and eleven retrospective cohort studies demonstrated that the use of spacer is associated with lower acute grade 2+ rectal toxicity (3.07% vs. 6.05%, RR=0.53, 95% CI=0.33–0.86, p < 0.001) and late grade 2+ rectal toxicity (1.62% vs. 9.35%, RR: 0.25, 95% CI: 0.15–0.42,p < 0.001). No difference is observed in significant grade 3+ GI (acute or late) events and there is no statistical difference in bowel-related QoL (risk difference= −0.16, 95% CI: −0.38–0.06, p = 0.15) [813]. With more widespread clinical use safety reports have described uncommon, but severe and life changing complications including prostatic abscess, fistulae and sepsis. Implantation is associated with a learning curve and should only be undertaken by teams with experience of TRUS and transperineal procedures with robust audit reporting in place [814].
6.2.3.d. Brachytherapy
6.2.3.d.1. Low-dose rate brachytherapy
Low-dose rate (LDR) BT uses radioactive seeds permanently implanted into the prostate. Low-dose rate monotherapy [815] can be offered to patients with NCCN-favourable intermediate risk and good urinary function defined as an International Prostatic Symptom Score (IPSS) < 12 and maximum flow rate > 15mL/min on urinary flow tests [816]. The RTOG phase III RCT compared LDR BT +/- EBRT in participants with ISUP GG1 and PSA < 20 or ISUP GG 2 and PSA < 10 and found that the addition of EBRT resulted in increased toxicity but no improvement in freedom from progression [817].
Patients having had a previous TURP can undergo BT without an increase in risk of urinary toxicity with due attention to dose distribution. A minimal channel TURP is recommended, leaving at least 1 cm rim of prostate tissue around the post-TURP urethral defect at the posterolateral sides of the prostate and there should be at least a 3-month interval between TURP and BT to allow for adequate healing [818-821].
The only available RCT comparing RP and LDR BT as monotherapy was closed due to poor accrual [822]. Outcome data are available from several large population cohorts with mature follow-up [823-827]. A significant correlation has been shown between the implanted dose and biochemical control [828]. A D90 (dose covering 90% of the prostate volume) of > 140Gy leads to a significantly higher biochemical control rate (PSA < 1.0ng/mL) after four years (92 vs. 68%). There is no OS benefit in adding neoadjuvant or adjuvant ADT to LDR monotherapy [829].
Low-dose-rate BT can be combined with EBRT in NCCN-unfavourable intermediate-risk PCa and high-risk patients. External beam RT (total dose of 78Gy) has been compared with EBRT (total dose 46Gy) followed by LDR BT boost (prescribed dose 115Gy) in intermediate-risk and high-risk patients in the ASCENDE-RT randomised trial with twelve months of ADT in both arms [830,831]. The LDR boost resulted in 5-, 7- and 10-year PSA PFS increase (89%, 86% and 85%, respectively, compared to 84%, 75%, 70%) but with no impact on distant metastasis or OS. This improvement in biochemical control was achieved at a cost of increased late grade 3+ GU toxicity (18% compared to 8%) and two treatment-related deaths [831,832]. Urinary toxicity was mainly in the development of urethral strictures and incontinence, and great care should be taken during treatment planning.
6.2.3.d.2. High-dose rate brachytherapy
High dose rate (HDR) BT uses a radioactive source temporarily introduced into the prostate to deliver radiation. Table 6.2.10 outlines the technical differences. The use of the GEC (Groupe European de Curietherapie)/ESTRO Guidelines is strongly recommended [833]. High-dose-rate BT can be delivered in single or multiple fractions and is often combined with EBRT of at least 45Gy, conventionally fractionated [834]. A retrospective analysis on 1,641 intermediate- and high-risk patients demonstrated a better distant-metastasis free survival when a HDR BT boost was added to 50–54Gy EBRT. The difference mounted to 12% at ten years [835]. An SR of non-RCTs and data from population studies suggest outcomes with EBRT plus HDR BT are superior to EBRT alone [836,837].
A single-centre RCT of EBRT (55Gy in 20 fractions) vs. EBRT (35.75Gy in 13 fractions), followed by HDR BT (17Gy in two fractions over 24 hours) has been reported [838]. In 218 patients with T1–3 N0M0 PCa, the combination of EBRT and HDR BT showed a significant improvement in the biochemical disease-free rate (p = 0.04) at five and ten years (75% and 46% compared to 61% and 39%). However, an unexpectedly high rate of early recurrences was observed in the EBRT arm alone, even after two years, possibly due to a dose lower than the current standard used [838].
Supporting, but not definitive, evidence of the benefit of HDR boost is available from the TROG 03.04 RADAR trial. This multi-centre study had upfront radiation dose escalation (non-randomised) with dosing options of 66, 70 or 74Gy EBRT, or 46Gy EBRT plus HDR BT boost and randomised men with locally advanced PCa to 6- or 18-months ADT. After a minimum follow-up of ten years, HDR boost significantly reduced distant progression, the study primary endpoint (HR: 0.68, 95% CI: 0.57–0.80; p < 0.0001), when compared to EBRT alone and, independent of duration of ADT, HDR boost was associated with increased IPSS of three points at 18 months post-treatment resolving by three years but decreased rectal symptoms when compared to EBRT [839]. Although radiation dose escalation using BT boost provides much higher biological doses, the TROG 03.04 RADAR RCT and SRs show ADT use independently predicts better outcomes regardless of radiation dose intensification [829,839,840]. Omitting ADT may result in inferior OS and based on current evidence ADT use and duration should be in line with that used when delivering EBRT alone.
Fractionated HDR BT as monotherapy can be offered to patients with intermediate-risk PCa, who should be informed that results are only available from limited series in very experienced centres. Five-year PSA control rates of 93.5% for intermediate-risk PCa are reported, with late grade 3+ GU toxicity rates < 5% and no, or very minimal, grade 3+ GI toxicity rates [841]. Single fraction HDR monotherapy should not be used as it has inferior biochemical control rates compared to fractionated HDR monotherapy with local failure rates at eight years of 35.9% versus 11.2% (p<0.001) [842].
Table 6.2.10: Difference between LDR and HDR brachytherapy
| Differences in prostate brachytherapy techniques | |
| Low dose rate (LDR) | Permanent seeds implanted |
| Uses Iodine-125 (I-125) (most common) | |
| Palladium-103 (103Pd-) or Cesium-131 isotopes | |
| Radiation dose delivered over weeks and months | |
| Acute side effects resolve over months | |
| Radiation protection issues for patient and carers | |
| High dose rate (HDR) | Temporary implantation |
| Iridium-192 (IR-192) isotope introduced through implanted needles or catheters | |
| Radiation dose delivered in minutes | |
| Acute side effects resolve over weeks | |
| No radiation protection issues for patient or carers | |
6.2.3.e. Acute side effects of external beam radiotherapy and brachytherapy
Gastrointestinal and urinary side effects are common during and after EBRT. In the EORTC 22991 trial, approximately 50% of patients reported acute GU toxicity of grade 1, 20% grade 2, and 2% of grade 3. In the same trial, approximately 30% of patients reported acute grade 1 GI toxicity, 10% grade 2, and less than 1% grade 3. Common toxicities included dysuria, urinary frequency, urinary retention, haematuria, diarrhoea, rectal bleeding and proctitis [843]. In addition, general side effects such as fatigue are common. It should be noted that the incidence of acute side effects is greater than that of late effects, implying that most acute effects resolve.
In an RCT comparing patient reported QoL after LDR or HDR boost combined with external beam radiotherapy to the pelvis, more intense and prolonged acute urinary side effects are noted with LDR boost [844]. In an RCT of conventional-dose EBRT vs. EBRT and LDR BT, the incidence of acute proctitis was reduced in the BT arm, but other acute toxicities were equivalent [830]. In a pooled analysis of 864 patients treated using extreme HFX and stereotactic RT, declines in urinary and bowel domains were noted at three months which returned to baseline - or better - by six months [845].
6.2.4. Investigational therapies
6.2.4.a. Background
In addition to RP, EBRT and BT, other modalities have emerged as potential therapeutic options in patients with clinically localised PCa [846-848]. These new modalities have been developed as minimally invasive procedures with the aim of providing equivalent oncological safety, reduced toxicity and improved functional outcomes. In this section, both whole-gland and focal treatment [849,850] will be considered, looking particularly at high-intensity focused US (HIFU), cryotherapeutic ablation of the prostate (cryotherapy), focal photodynamic therapy (PDT), and irreversible electroporation (IRE), as sufficient data are available to form the basis of some initial judgements. Other options such as radiofrequency ablation (RFA) and focal laser ablation (FLA), among others, are considered to be in the early phases of evaluation [849].
High-intensity focused US consists of focused US waves emitted from a transducer that cause tissue damage by mechanical and thermal effects as well as by cavitation [851]. The goal of HIFU is to heat malignant tissue above 65°C, so that it is destroyed by coagulative necrosis. High-intensity focused US is performed under general or spinal anaesthesia, with the patient lying in the lateral or supine position. Since the ultrasound energy is most often delivered from the rectal cavity, HIFU faces challenges in delivering energy to the anterior part in large prostates.
Cryotherapy uses freezing techniques to induce cell death by dehydration resulting in protein denaturation, direct rupture of cellular membranes by ice crystals and vascular stasis and microthrombi, resulting in stagnation of the microcirculation with consecutive ischaemic apoptosis [846-848]. Freezing of the prostate is ensured by the placement of 17-gauge cryo-needles under TRUS guidance, placement of thermosensors at the level of the external sphincter and rectal wall and insertion of a urethral warmer. Two freeze-thaw cycles are used under TRUS guidance resulting in a temperature of -40°C in the mid-gland and at the neurovascular bundle. Currently, third and fourth generation cryotherapy devices are mainly used.
Irreversible electroporation (IRE) uses high-volage low-energy electric pulses applied among transperineally placed electrodes to induce nanopores within cell walls. These pores modify of cell membrane permeability and induce cell death by disruption of cellular homeostasis [852].
6.2.4.b. Whole-gland therapies
Whole gland treatments using cryosurgery and HIFU were investigated as a replacement for surgery or radiotherapy, with limited success. The main adverse effects of whole-gland cryosurgery are ED (18%), urinary incontinence (2–20%), urethral sloughing (0–38%), rectal pain and bleeding (3%) and rectourethral fistula formation (0–6%) [853]. There is a lack of prospective comparative data regarding oncological outcomes of whole-gland cryosurgery as a curative treatment option for men with localised PCa, with most studies being non-comparative single-arm case series with short follow-up [853].
High-intensity focused US has previously been widely used for whole-gland therapy with the following adverse effects: acute urinary retention (10%), ED (23%), urethral stricture (8%), rectal pain or bleeding (11%), rectourethral fistula (0–5%) and urinary incontinence (10%) [853]. An SR including 375 retrospective and prospective trials with more than 6500 patients showed a wide variation of oncological and functional outcomes [854]. Combining whole-gland HIFU treatment with TURP has been shown to reduce the rate of urethral strictures and to maintain the level of incontinence, but to increase the rate of ED [855].
Overall, the lack of any long-term prospective comparative studies and data to suggest poor long-term oncological outcomes for men with high-risk localised disease [856] prevents whole-gland HIFU from being considered as a reasonable alternative to the established curative treatment options [853]. In addition, the expected improvements in functional outcome failed to materialise with 12% of patient developing incontinence and 61% developing ED [857].
6.2.4.c. Focal therapy
Over the past two decades, there has been a trend towards earlier diagnosis of PCa as a result of greater public and professional awareness, leading to the adoption of both formal and informal screening strategies. The effect of this change has been that men are identified at an earlier stage with smaller tumours, with a greater propensity for unifocal disease [858-860]. There is also greater awareness of the risks of the consequences of treatment leading to attempts to ablate only a region of the prostate containing the tumour, thereby limiting toxicity by sparing the neurovascular bundles, sphincter, and urethra [861-863]. The question remains which if any of these small unifocal tumours require treatment.
An SR included data from 5,827 patients across 72 studies and covered various energy sources, including HIFU, cryotherapy, Photodynamic Therapy (PDT), laser interstitial thermotherapy, focal BT, irreversible electroporation (IRE) and radiofrequency ablation (RFA) [864]. The review favours HIFU and PDT for their higher quality data, over 95% of pad-free incontinence and 85–90% of patients without clinically significant cancer in short-term analysis. This must be critically analysed, because 45% of all patients with a focal approach included in this SR had an ISUP Grade GG 1 cancer. The overall quality of the evidence was low, due to most studies being single-centre, non-comparative and retrospective in design, heterogeneity of definitions and approaches, follow-up strategies, outcomes and duration of follow-up. Although the review finds high-quality evidence that focal therapy has favourable functional outcomes and minimises AEs, definitive evidence of oncological benefit remains unavailable.
A more stringent SR including only prospective studies and per protocol posttreatment biopsies found that, after one year, 8.8% of patients had an infield failure with ≥ ISUP GG 2 cancers and 13.0% had ≥ ISUP GG 2 cancers anywhere in the prostate [865]. This work did not include any definition of clinically relevant cancer and included 35% of patients with ISUP GG 1 at initial diagnosis. Focal ablation showed only a 9% reduction in sexual function scores at one year, compared to 43% for whole-gland ablation.
At this time, the largest analysis on oncologic outcomes following focal HIFU includes 1,379 men with a median follow-up of 32 months (65% of patients were D’Amico intermediate risk and 28% high risk) [866]. In this study, one repeated focal HIFU session was allowed and performed in 18% of all patients. Parametric MRI was performed if consecutive PSA rises were identified and biopsies were offered if the mpMRI was suspicious. Eighty percent of patients had at least one follow-up mpMRI and 44% had a follow-up biopsy. The primary outcome was failure-free survival (FFS), which was defined as evidence of cancer requiring whole-gland salvage treatment. At seven years the FFS for intermediate- and high-risk cancers was 68% and 65%, respectively [866].
An SR on irreversible electroporation (IER) including 19 studies and 1,452 treated patients showed that the infield recurrence rate after one year was comparable to other focal therapy energy sources, with 0% - 39% in repeat biopsy [867]. Additionally, the clinically significant out-field cancer rate was high (0%-31%). The retreatment rate using IRE, radical prostatectomy, radiotherapy and HIFU was 8%-36%. While this review showed over 95% pad-fee patients after treatment. The assessment of erectile function by IIEF and EPIC showed greater heterogeneity and warrants a more detailed investigation. These findings were underlined by an international multicentre study with 411 patients confirming the good functional outcome with a four point drop in EEEF-5 at three months and residual ISUP GG 2 cancers found in 24.1% after one year. However, only a quarter of patients had a biopsy after IRE in this study [868].
There is currently no well-defined pathway for focal therapy or follow-up, and the field is still developing. The optimal energy source for tumours at various locations, the need for double treatments during initial therapy and the use of MRI or PSA for follow up are still a matter of research. The high rate of out-field recurrences after treatment indicates that the pretreatment evaluation should be maximised by targeted and systematic biopsies [865]. Whether modern imaging modalities (e.g. PSMA-PET) could support the mpMRI is not sufficiently evaluated. The Guideline panel acknowledges the challenges for interventional RCTs [869-871]. The interim analysis and meeting reports demonstrate slow recruitment, patients declining consent and rejecting their treatment allocation into the RP group (approx. 25%). In an attempt to overcome this challenge, propensity-matched analysis using prospective multicentre databases have been performed for comparison of focal therapy versus radical therapy [872,873]. Such analyses are always susceptible to unmeasured selection biases in who was selected for each treatment.
Oncological follow-up data up to eight years can be used to counsel patients in treatment decisions [872,873]. Patients managed by focal therapy had a HIFU or cryotherapy, with one retreatment, if required. Of these, 17.1% of patients in the focal arm received a retreatment. The primary outcome was FFS defined as ‘need for local or systemic salvage treatment or metastasis’. Both groups included 246 patients with an average PSA of 7.9ng/mL and 60% ISUP GG 2/3 cancers. The cancer core length was 5–6mm, with 45% having bilateral cancer. The authors report similar cancer control eight years after therapy, with FFS and BCR of 83% and 23.9% for focal therapy versus 79% and 24.8% for RP, respectively. Comparable results were demonstrated in a cohort-based analysis with a follow-up of six years [873]. The use of various definitions for oncological failure in the two arms is another limitation of these studies. While any recurrence after RP was seen as failure, a second HIFU was permitted in the focal group. The current data from the HIFU Evaluation and Assessment of Treatment (HEAT) registry indicates that a repeat HIFU does not significantly decrease urinary or erectile function [874]. However, this change of failure definition will have to be re-evaluated. It is important to note, that these results were achieved in centres with a dedicated focal program in which all patients had an mpMRI with targeted and systematic biopsies or full template-mapping biopsies. Therefore, it seems necessary to perform systematic biopsies in patients who are candidates for focal therapy.
The impact of salvage therapies after focal therapy was investigated in small series in specialised centres [875,876]. If a salvage RP is required after focal therapy, the reported functional and oncological outcomes are comparable to treatment-naive patients [877,878]. In a recent SR including 482 patients from 12 studies, the authors conclude that, when compared to primary surgery, the salvage radical prostatectomy after focal therapy has a higher PSM rate of 27% and a slightly worse incontinence rate. Although the early complication rate was also higher, most of them could be managed conservatively [879]. An SR of salvage radiotherapy after HIFU or cryotherapy was analysed showed a favourable toxicity profile and a comparable overall biochemical relapse rate of approximately 20% [880].
One comparative RCT was conducted in a very-low risk population for which there is currently a strong movement away from any form of active treatment. This study was comparing padeliporfin-based vascular targeted PDT versus AS and found at a median follow-up of 24 months that fewer patients progressed in the PDT arm compared with the AS arm (adjusted HR: 0.34, 95% CI: 0.24–0.46), and required less radical therapy (6% vs. 29%, p < 0.0001). Updated results were published in 2018, showing that these benefits were maintained after four years [881]. Nevertheless, limitations of the study include an unusually high observed rate of disease progression in the AS arm (58% in two years) and more patients in the AS arm chose to undergo radical therapy without a clinical indication, which may have introduced confounding bias. Finally, the AS arm did not undergo any confirmatory biopsy or any MRI scanning, which is not representative of contemporary practice. A matched-pair analysis comparing focal cryotherapy to AS with 76% ISUP GG 1 cancers failed to demonstrate any significant advantages for MFS and OS [882].
The available evidence indicates that focal therapy is associated with fewer AEs than whole-gland or radical treatments. Many of the patients included in these trials would currently be considered to have been overtreated. Robust prospective trials reporting standardised 15-year oncological outcomes [883] are needed in patients with clinically significant cancers before unrestricted recommendations in support of focal therapy for routine clinical practice can be made [849,883,884]. Currently, focal therapy using HIFU or cryotherapy should be performed within the context of a prospective registry.
All other ablative modalities and treatment strategies should only be offered in a well-designed prospective trial setting. To allow quality analysis of the collected data, the prospective registry should adhere to the EMA recommendations (Guideline on registry-based studies EMA/426390/2021), which emphasises the need for clear follow-up timelines and timely recording, completeness of core data of consecutive patients enrolled, an analysis plan in defined intervals and a data quality management.
6.3. Management by disease stages
6.3.1. Management of low-risk disease
6.3.1.a. Watchful waiting
For patients with a life expectancy of < 10 years (based on comorbidities and age) for whom curative treatment would not be an option in the case of progression after AS, WW is standard of care.
6.3.1.b. Active surveillance
Active surveillance should be considered standard of care for all patients with a life expectancy > 10 years (based on comorbidities and age) and where curative treatment would be considered in the case of disease progression.
6.3.1.b.1. Androgen deprivation monotherapy
The Early Prostate Cancer (EPC) Trial Programme found that, in patients with localised disease, ADT monotherapy did not improve PFS or OS in any of the subgroups compared with placebo [885]. Instead, there was a statistically insignificant numerical trend towards worse OS with ADT in the WW subgroup (HR: 1.16, 95% CI: 0.99–1.37; p = 0.07). Although the trial did not directly address men with low-risk disease, it offered some evidence suggesting that otherwise asymptomatic men with localised disease should not receive ADT monotherapy.
6.3.1.c. Other therapeutic options
Other treatments, such as whole-gland therapy (e.g. RP or RT) or focal ablative therapy, remain highly likely to be overtreatment in the setting of low-risk disease and should not be used outside a trial setting.
6.3.1.d. Recommendations for the management of low-risk disease
| Recommendations | Strength rating |
| Manage patients with a life expectancy < 10 years with watchful waiting. | Strong |
| Manage patients with a life expectancy > 10 years and low-risk disease with active surveillance. | Strong |
6.3.2. Management of intermediate-risk disease
6.3.2.a. Watchful waiting
For patients with a life expectancy of < 10 years (based on comorbidities and age), where curative treatment is not a direct option or would not be an option in the case of progression after AS, WW is standard of care.
6.3.2.b. Active Surveillance
Although men with less-favourable disease characteristics have worse outcomes after any treatment, the question is whether a delay in curative treatment due to initial active surveillance leads to additionally unfavourable outcomes. Intuitively, the higher risk disease, the higher risk of adverse outcomes due to an initial delay. Inclusion is based on favourable disease characteristics as discussed in Section 6.2.1.b.2.
6.3.2.c. Radical prostatectomy
Patients with intermediate-risk PCa should be informed about the results of two RCTs (SPCG-4 and PIVOT) comparing RP versus WW in localised PCa. In the SPCG-4 study, death from any cause (RR: 0.71, 95% CI: 0.53–0.95), death from PCa (RR: 0.38, 95% CI: 0.23–0.62) and distant metastases (RR: 0.49, 95% CI: 0.32–0.74) were significantly reduced in intermediate-risk PCa at 18 years. After 30-years follow-up overall (not risk-stratified), RP reduced death from any cause (RR: 0.74, 95% CI: 0.64–0.87), and death from PCa (RR: 0.52, 95% CI: 0.40–0.67) for a mean of 2.2 life years (95%CI 1.4-2.9) gained [886]. Survival benefit was most likely if alive for more than 20 years, with a number needed to treat to avert one PCa death of six [887]. In the PIVOT trial, according to a preplanned subgroup analysis among men with intermediate-risk tumours, RP significantly reduced all-cause mortality (HR: 0.69, 95% CI: 0.49–0.98), but not death from PCa (0.50, 95% CI: 0.21–1.21) at ten years [888]. In the ProtecT trial, 24% of the population were intermediate risk (at baseline) and no significant difference in prostate cancer deaths was seen for RP versus active monitoring (with delayed active treatment, HR 0.68 (0.11–4.05). A meta-analysis based on the findings of SPCG-4, PIVOT and ProtecT demonstrated a benefit from RP over-observation with a significantly decreased risk of death of 9% and of disease progression of 43% [889]. However, no stratification by disease stages was performed. A large study found 2.9% of LN invasion in a contemporary cohort of 6,883 patients undergoing RP and LND for intermediate risk PCa [890].
6.3.2.d. Radiation therapy
6.3.2.d.1. IMRT/VMAT
Ultra-hypofractionated IMRT/IGRT or SBRT, using either 36.25Gy (40Gy to prostate) in 5fx or 42.7Gy in 7fx, can be offered to patients with NCCN-favourable intermediate and good urinary function. Additional ADT is not required in GG2 disease [786]. Patients undergoing conventional or moderate hypofractionation and suitable for ADT can be treated with short-term ADT (four to six months) [891-893]. The RTOG 0815 RCT demonstrated improved BRFS, metastasis-free and prostate CSS with the addition of six-months ADT to dose-escalated RT [802]. For patients unsuitable (e.g. due to comorbidities) or unwilling to accept ADT (e.g. to preserve their sexual health), the recommended treatment is IMRT/VMAT (76–78Gy or equivalent moderate HFX) or a combination of IMRT/VMAT and BT, as described below. A secondary analysis of the PCS III trial has suggested that patients with NCCN-favourable intermediate-risk disease can safely omit ADT if their RT dose is 76Gy, but this is based on an unplanned subgroup analysis and only 138 patients had favourable intermediate-risk disease [801]. An individual discussion between the physician and the patient of the possible benefits and harms of omitting ADT in this group is essential [778].
6.3.2.d.2. Brachytherapy
Systematic review recommends LDR BT monotherapy can be offered to patients with NCCN-favourable intermediate-risk disease and good urinary function (see Section 4.4) [894]. Fractionated HDR BT as monotherapy can be offered to selected patients with intermediate-risk PCa, although they should be informed that results are only available from small series in very experienced centres. Five-year PSA control rates over 90% are reported, with late grade 3+ GU toxicity rates < 5% and no, or very minimal, grade 3+ GI toxicity rates [841]. There are no direct data to inform on the use of ADT in this setting. Trimodality therapy with IMRT plus BT boost and short-term ADT can be considered for NCCN-unfavourable intermediate-risk PCa (see Section 6.2.3.d), but patients should be made aware that the potential improvements in biochemical control are accompanied with an increased risk of long-term urinary problems [830,832,837].
6.3.2.e. Other therapeutic options
6.3.2.e.1. Focal therapy
The available evidence indicates that focal therapy is associated with less AEs than whole gland or radical treatments. Robust prospective trials reporting standardised fifteen-year oncological outcomes [883], are needed in patients with clinically significant cancers before unrestricted recommendations in support of focal therapy for routine clinical practice can be made [849,883,884].
6.3.2.e.2. Androgen deprivation therapy monotherapy
Data regarding the use of ADT monotherapy for intermediate-risk disease have been inferred indirectly from the EORTC 30891 trial, which was an RCT comparing deferred ADT versus immediate ADT in 985 patients with T0–4 N0–2 M0 disease [895]. The trial showed a small, but statistically significant, difference in OS in favour of immediate ADT monotherapy, but there was no significant difference in CSS, predominantly because the risk of cancer-specific mortality was low in patients with PSA < 8ng/mL. Consequently, the use of ADT monotherapy for this group of patients is not considered standard, even if they are not eligible for radical treatment.
6.3.2.f. Recommendations for the management of intermediate-risk disease*
| Recommendations | Strength rating |
| Expectant management | |
| Offer watchful waiting in asymptomatic patients with life expectancy < 10 years (based on co-morbidities and age). | Strong |
| Offer active surveillance (AS) to selected patients with ISUP grade group (GG) 2 disease, e.g. < 10% pattern 4, PSA < 10 ng/mL, ≤ cT2a, low disease extent on imaging and low extent of tumour in biopsies (≤ 3 positive cores with ISUP GG 2 and ≤ 50% cancer involvement/core) or another single element of intermediate-risk disease with low disease extent on imaging and low biopsy extent, accepting the potential increased risk of metastatic progression. | Weak |
| Patients with ISUP GG 3 disease should be excluded from AS protocols. | Strong |
| Reclassify patients with low-volume ISUP GG 2 disease included in AS protocols if repeat non-magnetic resonance imaging (MRI)-based systematic biopsies performed during monitoring reveal > 3 positive cores or maximum cancer involvement > 50%/core of ISUP GG 2 disease. | Weak |
| Radical prostatectomy (RP) | |
| Offer RP to patients with a life expectancy of > ten years. | Strong |
| Offer nerve-sparing surgery to patients with a low risk of extracapsular disease on that side. | Strong |
| Radiotherapeutic treatment | |
| Offer low-dose rate (LDR) brachytherapy to patients with good urinary function and NCCN-favourable intermediate-risk disease. | Strong |
| Offer intensity-modulated radiotherapy (IMRT)/volumetric modulated arc therapy (VMAT) plus image-guided radiotherapy (IGRT), with a total dose of 76–78 Gy or moderate hypofractionation (60 Gy/20 fx in four weeks or 70 Gy/28 fx in six weeks), in combination with short-term androgen deprivation therapy (ADT) (four to six months). | Strong |
| Offer focal boosting to MRI-defined dominant intraprostatic tumour when using conventionally fractionated IMRT/IGRT (1.8-2.0 Gy per fraction), ensuring that Organ at Risk constraints are not exceeded. | Weak |
| Offer ultra-hypofractionated IMRT/IGRT or stereotactic body radiation therapy, using either 36.25 Gy (40 Gy to prostate) in 5 fx or 42.7 Gy in 7 fx delivered alternate days in patients with favourable intermediate risk considering urinary function. | Weak |
| Offer LDR brachytherapy boost combined with IMRT/VMAT plus IGRT to patients with good urinary function and NCCN unfavourable intermediate-risk disease, in combination with short-term ADT (four to six months). | Weak |
| Offer high-dose rate brachytherapy boost combined with IMRT/VMAT plus IGRT to patients with good urinary function and NCCN unfavourable intermediate-risk disease in combination with short-term ADT (four to six months). | Weak |
| Other therapeutic options | |
| Only offer whole-gland ablative therapy (such as cryotherapy, high-intensity focused ultrasound, etc.) or focal ablative therapy within clinical trials or registries. | Strong |
| Do not offer ADT monotherapy to asymptomatic men not able to receive any local treatment. | Weak |
*All recommendations are based on conventional imaging with isotope bone scan and CT/MR abdomen/pelvis.
6.3.3. Management of high-risk localised disease
Patients with high-risk PCa are at an increased risk of PSA failure, need for secondary therapy, metastatic progression and death from PCa. Nevertheless, not all high-risk PCa patients have a uniformly poor prognosis after RP [896]. When managed with noncurative intent, high-risk PCa is associated with 10-year and 15-year PCSM rates of 28.8 and 35.5%, respectively [897]. There is no consensus regarding the optimal treatment of men with high-risk PCa.
Some evidence suggests that radical treatment for high-risk PCa can be delayed up to three months after the diagnosis without any oncological consequences [898,899]. Systematic reviews suggest that there is a higher risk of biochemical recurrence and worse pathological outcomes when definitive treatment is given beyond a six- to nine-months delay. However, there is no conclusive data regarding stronger endpoints (CSS or OS).
6.3.3.a. Radical prostatectomy
Radical prostatectomy is a standard option in selected patients with a low tumour volume, provided that the tumour is not fixed to the pelvic wall or there is no invasion of the urethral sphincter. Patients should be aware preoperatively that surgery may be part of multimodal treatment, with adjuvant SRT or ADT; however, almost half of patients will be free from recurrence without multimodal treatment at five years [900,901]. Neoadjuvant therapy using ADT is not indicated [902].
6.3.3.b. External beam radiation therapy
For high-risk localised PCa, a combined modality approach should be used consisting of IMRT/VMAT plus long-term ADT. The duration of ADT must take into account PS, comorbidities and the number of poor prognostic factors. It is important to recognise that in several studies, EBRT plus short-term ADT did not improve OS in high-risk localised PCa and long-term ADT (at least two to three years) is currently recommended for these patients [794,795,803]. Moderate HFX is an option in high-risk patients with localised disease. The CHHiP study included 12% high-risk patients (n = 386), but limited entry to those with a PSA < 30ng/mL and a Roach formula risk of SV involvement < 30% [774]. Patients were ineligible if they had both T3a tumours and ISUP grade group 4 or higher. The PCS-5 RCT used moderate HFX and elective nodal irradiation and efficacy was equivalent in both groups [781,782].
6.3.3.b.1. Lymph node irradiation in cN0
There is no clear evidence for prophylactic irradiation of the pelvic LNs in intermediate- and high-risk disease. The long-term results of the NRG/RTOG 9413-trial, which randomised intermediate-risk and high-risk localised PCa patients (1,322 cN0 patients were enrolled), showed that neoadjuvant HT plus whole pelvic RT improved PFS only compared with neoadjuvant ADT plus prostate RT and whole pelvic RT plus adjuvant ADT [903]. However, at the increased risk of ≥ grade 3 GI-toxicity.
A well-conducted single-centre RCT randomised 224 patients comparing prostate-only RT (PORT) versus whole pelvic RT (WPRT) in localised high-risk- and locally advanced tumours (cN0) with a risk of > 20% of positive nodes (Roach formula). With a median follow-up of 68 months, there was a significant improvement of distant MFS (95.9% vs. 89.2%, HR: 0.35, p = 0.01) and DFS (89.5% vs.77.2%, p = 0.02). However, there was a significant higher rate of late GU ≥ 2 effects (17.7% vs. 7.5%, p = 0.02), the trial was relatively small in size with additional limitations and these findings are therefore insufficient to define a change in practice [904,905]. The benefits of pelvic nodal irradiation using IMRT/VMAT merit further investigation in large scale RCTs.
6.3.3.b.2. Brachytherapy boost
In men with NCCN-unfavourable intermediate- or high-risk PCa, BT boost with supplemental EBRT and HT may be considered. See Sections 6.2.3.d.1 and 6.2.3.d.2 for details on RCTs comparing EBRT alone and EBRT with LDR or HDR boost, respectively.
6.3.3.c. Recommendations for the management of high-risk localised disease*
| Recommendations | Strength rating |
| Expectant management | |
| Offer watchful waiting to asymptomatic patients with life expectancy < 10 years. | Strong |
| Radical prostatectomy (RP) | |
| Offer RP to selected patients. | Strong |
| Extended pelvic lymph node dissection (ePLND) | |
| In patients undergoing a lymph node dissection you should perform an ePLND. | Strong |
| Do not perform a frozen section of nodes during RP to decide whether to proceed with, or abandon, the procedure. | Strong |
| Radiotherapeutic treatment | |
| Offer intensity-modulated radiotherapy (IMRT)/volumetric modulated arc therapy (VMAT) plus image-guided radiotherapy (IGRT), with a total dose of 76–78 Gy or moderate hypofractionation (60 Gy/20 fx in four weeks or 70 Gy/28 fx in six weeks), in combination with long-term androgen deprivation therapy (ADT) (two to three years). | Strong |
| Offer focal boosting to magnetic resonance imaging (MRI)-defined dominant intraprostatic tumour when using normo-fractionated IMRT/IGRT (1.8-2.0 Gy per fraction) ensuring that Organ at Risk constraints are not exceeded. | Strong |
| Offer patients with good urinary function IMRT/VMAT plus IGRT with brachytherapy boost (either high-dose rate or low-dose rate) in combination with long-term ADT (two to three years). | Weak |
| Other therapeutic options | |
| Do not offer either whole gland or focal therapy. | Strong |
| Only offer ADT monotherapy to those patients unwilling or unable to receive any form of local treatment if they have a prostate-specific antigen (PSA)-doubling time < 12 months, and either a PSA > 50 ng/mL or a poorly differentiated tumour. | Strong |
*All recommendations are based on conventional imaging with isotope bone scan and CT/MR abdomen/pelvis.
6.3.4. Management of locally advanced PCa
In the absence of high-level evidence, an SR could not define the most optimal treatment option [906]. Randomised controlled trials are only available for EBRT. A local treatment combined with a systemic treatment provides the best outcome, provided the patient is fit enough to receive both. The initial results of the SCPG-15 trials suggested that randomisation between surgery and EBRT is feasible, but oncologic outcomes are awaited [907].
6.3.4.a. Radical prostatectomy
Surgery for locally advanced disease as part of a multimodal therapy has been reported [897,908,909]. However, the comparative oncological effectiveness of RP as part of a multimodal treatment strategy versus upfront EBRT with ADT for locally advanced PCa remains unknown. A prospective phase III RCT (SPCG-15) comparing RP (with or without adjuvant or salvage EBRT) against primary EBRT and ADT among patients with locally advanced (T3) disease is currently recruiting [910]. Data from retrospective case series demonstrated over 60% CSS at 15 years and over 75% OS at ten years [877,897,900,908,909,911,912]. For cT3b–T4 disease, PCa cohort studies showed 10-year CSS of over 87% and OS of 65% [878,913]. The indication for RP in all previously described stages assumes the absence of clinically detectable nodal involvement (cN0), based on conventional imaging. In case of suspected positive LNs during RP (initially considered cN0) the procedure should not be abandoned, since RP may have a survival benefit in these patients. Intraoperative frozen section analysis is not justified in this case [534].
6.3.4.b. Treatment of cN1 M0 PCa
Lymph-node-metastasised PCa is an entity in which options for local therapy and systemic therapies overlap. Approximately 5 to 10% of newly diagnosed PCa patients have synchronous suspected pelvic nodal metastases on conventional imaging (CT/bone scan) without bone or visceral metastases (cN1 M0 stage). Individual patient data from RCTs of RT plus long term ADT indicate that cN1 disease carries highest risk of metastasis (HR 1.86 [1.56–2.21]; five-year MFS 67%, ten-year MFS 36%) and death (HR 1.77 [1.45–2.15], ten-year OS 47%) among patients with high-risk localised/locoregional disease [914].
6.3.4.b.1. Consideration of molecular imaging
Meta-analyses have shown that molecular imaging, such as PSMA-PET/CT, prior to primary treatment in advanced PCa detected disease outside the prostate in 32% of cases despite prior negative conventional imaging using bone scan and pelvic CT/MRI [514]. A RCT assessing PSMA-PET/CT as staging tool in high-risk PCa confirmed these findings and showed a 32% increase in accuracy compared with conventional imaging for the detection of pelvic nodal metastases [553]. Notably, more sensitive imaging also caused a stage shift with more cases classified as N1 on “molecular imaging” (miN1), but with, on average, lower nodal disease burden compared to cases classified as cN1. After follow-up (median 41 months), miN1 status was prognostic for treatment failure (adjusted HR 2.0 [1.10–3.64], p = 0.007), but N1 status per conventional imaging was not (HR 0.6 [0.1–2.4], p = 0.45) [915].
The definition of miN1 is a subject of ongoing discussion, given that multiple guidelines exist because detection can be influenced by size of the lymph nodes and PSMA expression [114,916,917]. For patients with high or equivocal PSMA expression but normal size (< 10 mm), there is a lack of knowledge of the best treatment option and prospective data are encouraged [918].
6.3.4.b.2. Local treatment of cN1 M0 PCa
The management of cN1M0 PCa is historically based on long-term ADT combined with a local treatment with radiotherapy more commonly used than RP/pelvic nodal dissection. No randomised evidence is available and the potential benefit of adding local treatment to ADT has been assessed in a nonrandomised post-hoc analysis of STAMPEDE and retrospective studies summarised by Yaow et al. [919]. Pooled meta-analysis was performed for local treatment versus no local treatment (four studies, n = 4,597, local treatment n = 2,646) and showed improved estimated overall survival at all time points to 10 years (OR: 1.49-1.81). The majority of patients underwent RT as local therapy. Assessment of RT versus no local therapy (four studies, n=3,768) showed similar estimates for improvements in overall survival. Not included in this pooled analysis was STAMPEDE control arm data, which showed improvements in failure-free survival (adjusted HR: 0.48, 95% CI: 0.29-0.79) without severe toxicity [920] at median follow-up of 17 months. Comparisons between local treatment modalities were limited by inclusion of retrospective studies, which fail to describe clearly how cN1 was defined.
Local treatment of cN1M0 disease in the era of taxane chemotherapy and ARPIs is understudied. Extended follow-up of STAMPEDE, reported as exploratory sub-analyses of patients who received docetaxel or control according to receipt of RT after median follow-up of 81.2 months, maintained failure-free survival benefit (HR: 0.68) in N+ patients but no prostate cancer-specific survival (HR: 0.81) or overall survival (HR: 0.77) benefit was demonstrated [921]. Greatest benefits from RT were seen in the control (without docetaxel) group, because no significant benefits of RT receipt were seen in any category for the docetaxel group. Two RCTs from the STAMPEDE platform protocol reported a pre-planned meta-analysis of men with de novo high-risk/locally advanced M0 disease or relapse after primary curative therapy with high-risk features. Thirty-nine percent of patients (n = 774) were N1 on conventional imaging [922]. Radiotherapy in addition to long-term ADT was administered in at least 71% of N1 patients. Data on survival according to whether RT was planned in N1 patients was not presented.
6.3.4.b.3. Systemic treatment of cN1 M0 PCa
The intensification of systemic treatment from initial ADT to other agents has been assessed within data from the STAMPEDE multi-arm RCT with a preplanned meta-analysis in M0 patients. In cN1 M0 patients (39% of the cohort), improved metastasis-free (HR: 0.49, 95% CI: 0.38-0.64) and overall (HR: 0.53, 95% CI: 0.39-0.70) survival was observed with intensification (abiraterone and enzalutamide or abiraterone alone) above standard of care (ADT +/- prostate radiotherapy in 85% of the whole cohort) in cN1M0 patients [922].
Considering intensification with docetaxel, exploratory sub-analyses of STAMPEDE nonmetastatic (cN0/N1M0) patients who received docetaxel or control showed failure-free survival benefit (HR: 0.70, 95% CI: 0.56-0.88), but no metastatic progression-free (HR: 0.89) or overall survival (HR: 0.88) benefit [921]. Similar trends were observed in the N0 and N+ subgroups. Radiotherapy was delivered to 77% of the cohort (see Section 6.3.4.b). The AFU-GETUG 12 trial compared the impact of docetaxel plus estramustine in addition to ADT and 29% of included high-risk non-metastatic PCa patients had a nodal involvement (pN1) at randomisation [923]. Relapse-free survival rates were higher for cN1 patients receiving docetaxel plus estramustine but did not achieve statistical significance (HR: 0.66; 86 0.43–1.01). A meta-analysis of docetaxel trials in N0/N1-M0 patients showed an 8% four-year failure-free survival advantage for docetaxel compared with ADT alone without OS benefit (HR: 0.87, 95% CI: 0.69-1.09) [924].
Given the MFS and OS benefits observed in the overall population (see Section 6.3.4.b), additional abiraterone (for 2 years) above standard of care (combined ADT for 3 years with prostate +/- WPRT) should be a SOC in cN1 patients.
Table 6.3.1: Selected studies assessing local treatment in (any cT) cN1 M0 prostate cancer patients
| Study | n | Design | Study period/ follow-up | Treatment arms | Effect on survival |
| ADT only | |||||
| Bryant, et al. 2018 [925] | 648 | Retrospective (National Veterans Affairs) | 2000-2015 61 mo. | ADT ± EBRT | Significant benefit for combined treatment only if PSA levels less than the median (26ng/mL) All-cause mortality HR: 0.50 CSS, HR: 0.38 |
| Sarkar, et al. 2019 [926] | 741 | Retrospective (National Veterans Affairs) | 2000-2015 51 mo. | ADT ± local treatment (surgery or RT) | Significant benefit for RP All-cause mortality HR 0.36 CSS, HR: 0.32
No statistical difference for RP vs. RT (p ≥ 0.1) All-cause mortality HR: 047 CSS, HR: 0.88 |
Lin, et al. 2015 [927] | 983 before propensity score matching | Retrospective (NCDB) | 2004-2006 48 mo. | ADT ± EBRT | Significant benefit for combined treatment 5-yr. OS: 73% vs. 52% HR: 0.5 |
| Tward, et al. 2013 [928] | 1,100 | Retrospective (SEER) | 1988-2006 64 mo. | EBRT (n = 397) vs. no EBRT (n=703) No information on ADT) | Significant benefit for EBRT 5-yr. CSS: 78% vs. 71% HR: 0.66 5-yr. OS: 68% vs. 56%, HR: 0.70 |
| Rusthoven, et al. 2014 [929] | 796 | Retrospective (SEER) | 1995-2005 61 mo. | EBRT vs. no EBRT (no information on ADT) | Significant benefit for EBRT 10-yr. OS: 45% vs. 29% HR: 0.58 |
| Seisen, et al. 2018 [930] | 1,987 | Retrospective (NCDB) | 2003-2011 50 mo. | ADT ± local treatment (surgery or RT) | Significant benefit for combined treatment 5-yr. OS: 78.8% vs. 49.2% HR: 0.31 No difference between RP and RT |
| Chierigo, et al. 2022 [931] | 4,685 | Retrospective (SEER) | 2004-2016 | RP or RT (unknown ADT status) | Propensity score matching 5-yr OS: 84.6% (RP) vs. 75% (RT), HR 0.62, p < 0.001 5-yr CSS: 90.7% (RP) vs. 83% (RT), HR 0.62, p < 0.001 5-yr other cause mortality, 6.1% RP vs. 8.0% RT, HR 0.71, p = 0.04 |
| James, et al. 2016 [920] | 177 | Unplanned subgroup analysis RCT | 2005-2014
17 mo. | ADT ± EBRT (EBRT encouraged) | Significant benefit for combined treatment 5-yr. OS: 93% vs. 71% 2-yr. FFS: 81% vs. 53% FFS, HR: 0.48 |
| Elumalai, et al. 2023 [932] | 337 | Retrospective 4 centres UK | 2022-2019 | ADT +/- EBRT | Significant benefit for combined treatment 5-yr.OS: 87% vs. 56% HR: 0.27 5-yr. BPFS: 74.1% vs. 34.2% HR: 0.33 |
| Other systemic therapies | |||||
James, et al. 2022 [921] | 258 (N1 patients) | Planned subgroup analysis RCT | 2005-2018 81.2 mo | Standard of care (ADT +/- EBRT) +/- docetaxel
(EBRT planned for 55% SOC, 40% of docetaxel) | 5-year estimated Metastatic PFS (SOC + docetaxel vs SOC, HR: 0.79) OS (RT 78% vs. no RT 71%, HR: 0.77)* CSS (RT 84% vs. no RT 79%, HR: 0.81)* FFS (RT 51% vs. no RT 36%, HR: 0.68)* *No stratification for docetaxel use |
Attard, et al. 2022 [922] | 774 (N1) | Planned subgroup analysis RCT | 2011-2016
72 mo | Standard of care (ADT +/- EBRT) +/- Abiraterone with or without enzalutamide (EBRT planned for 71% of N1 patients) | MFS (SOC + Abiraterone with or without enzalutamide vs SOC alone, HR: 0.49, 95% CI: 0.38-0.64)
OS (SOC + Abiraterone with or without enzalutamide vs SOC alone, HR: 0.53, 95% CI: 0.39-0.70) |
ADT = androgen deprivation therapy; BPFS = biochemical progression-free survival; CSS = cancer-specific survival; EBRT = external beam radiotherapy; FFS = failure-free survival; HR = hazard ratio; mo. = months; n = number of patients; OS = overall survival; PSA = prostate-specific antigen; RP = radical prostatectomy; RT = radiotherapy; yr. = years.
6.3.4.c. Options other than surgery or radiotherapy for primary treatment
6.3.4.c.1. Investigational therapies
Cryotherapy, HIFU and focal therapies currently have no place in the management of locally advanced PCa.
6.3.4.c.2. Androgen deprivation therapy monotherapy
The deferred use of ADT as single treatment modality was answered by the EORTC 30891 trial [895]. Nine hundred and eighty-five patients with T0–4 N0–2 M0 PCa received ADT alone, either immediately or after symptomatic progression or occurrence of serious complications. After a median follow-up of 12.8 years, the OS favoured immediate treatment (HR: 1.21, 95% CI: 1.05–1.39). Surprisingly, no different disease-free or symptom-free survival was observed, raising the question of survival benefit. In locally advanced T3–T4 M0 HSPC unsuitable for surgery or RT, immediate ADT may only benefit patients with a PSA > 50ng/mL and a PSA-DT < twelve months or those that are symptomatic [895,933]. The median time to start deferred treatment was seven years. In the deferred treatment arm, 25.6% of patients died without needing treatment.
Patients with locally-advanced disease are more likely to require multi-modality therapy which might delivered sequentially (e.g. RP followed by ADT with or without RT) or in combination (e.g. RT plus ADT plus abiraterone). There is currently no randomised data to inform patients or clinicians which of these strategies is the most effective and an open discussion about the risks and benefits is important to allow shared-decision making.
6.3.4.d. Recommendations for management of locally advanced disease*
| Recommendations | Strength rating |
| Radical prostatectomy (RP) | |
| Offer RP to selected patients with cN0 disease as part of multi-modal therapy. | Weak |
| Extended pelvic lymph node dissection (ePLND) | |
| In patients undergoing a lymph node dissection you should perform an ePLND. | Strong |
| Radiotherapeutic treatments | |
| Offer patients with cN0 disease intensity-modulated radiation therapy (IMRT)/volumetric modulated arc therapy (VMAT) plus image-guided radiation therapy in combination with long-term androgen deprivation therapy (ADT). | Strong |
| Offer patients with cN0 disease and good urinary function, IMRT/VMAT plus IGRT with brachytherapy boost (either high-dose rate or low-dose rate), in combination with long-term ADT. | Weak |
| Offer long-term ADT for at least two years. | Strong |
| Offer IMRT/VMAT plus IGRT to the prostate in combination with long-term ADT and two years of abiraterone to cN0M0 patients with ≥ 2 high-risk factors (cT3-4, Gleason ≥ 8 or prostate-specific antigen ≥ 40 ng/mL). | Strong |
| Offer IMRT/VMAT plus IGRT to the prostate plus pelvis in combination with long-term ADT and two years of abiraterone to cN1M0 patients. | Strong |
| Other therapeutic options | |
| Do not offer whole gland treatment or focal treatment. | Strong |
*All recommendations are based on conventional imaging with isotope bone scan and CT/MR abdomen/pelvis.
6.3.5. Adjuvant treatment after radical prostatectomy
6.3.5.a. Introduction
Adjuvant treatment is, by definition, additional to the primary or initial therapy with the aim of decreasing the risk of relapse, despite the apparent full control following surgery. A postoperative detectable PSA is an indication of persistent prostate cells (see Section 6.3.6). All information listed below refers to patients with a postoperative undetectable PSA.
6.3.5.b. Risk factors for relapse
Patients with ISUP GG > 2 in combination with EPE (pT3a), and particularly those with SV invasion (pT3b) and/or positive surgical margins, are at high risk of progression, which can be as high as 50% after five years [934]. Irrespective of the pT stage, the number of removed nodes [935-942], tumour volume within the LNs and capsular perforation of the nodal metastases are predictors of early recurrence after RP for pN1 disease [943]. An LN density (defined as ‘the percentage of positive LNs in relation to the total number of analysed/removed LNs’) of over 20% was found to be associated with poor prognosis [944]. The number of involved nodes appears to be a major factor for predicting relapse [937,938,945]; the threshold considered is less than three positive nodes from an ePLND [521,937,945]. However, prospective data are needed before defining a definitive threshold value.
6.3.5.b.1. Biomarker-based risk stratification after radical prostatectomy
The Decipher® gene signature consists of a 22-gene panel representing multiple biological pathways and was developed to predict systemic progression after definitive treatment. A meta-analysis of five studies analysed the performance of the Decipher® Genomic Classifier (GC) test on men post-RP. The authors showed in multivariable analysis that Decipher® GC remained a statistically significant predictor of metastasis (HR: 1.30, 95% CI: 1.14–1.47, p < 0.001) per 0.1 unit increase in score and concluded that it can independently improve prognostication of patients post-RP within nearly all clinicopathologic, demographic and treatment subgroups [946]. An SR of the evidence for the Decipher® GC has confirmed the clinical utility of this test in post-RP decision-making [947]. Further studies are needed to establish how to best incorporate Decipher® GC in clinical decision-making.
6.3.5.c. Immediate (adjuvant) postoperative external irradiation after RP (cN0 or pN0)
Four prospective RCTs have assessed the role of immediate postoperative RT (adjuvant RT [ART]) (undetectable PSA mostly defined as PSA < 0.1ng/mL), demonstrating an advantage (endpoint, development of BCR) in high-risk patients (e.g. pT2 with positive surgical margins and ISUP grade group 3–5 or pT3/4 with or without positive surgical margins and ISUP grade group 3–5) post-RP (Table 6.3.2). In the ARO 96-02 trial, 80% of the pT3/R1/GS 8–10 patients randomised to observation developed BCR within ten years [948]. It must be emphasised that PSA was undetectable at inclusion only in the ARO 96-02 trial, which presents a major limitation interpreting these findings as patients with a detectable PSA would now be considered for salvage therapy rather than ART [948].
6.3.5.d. Comparison of adjuvant and salvage radiotherapy
Two retrospective matched studies (510 and 149 patients receiving ART) failed to show an advantage for MFS [949,950]. However, both studies were underpowered for high-risk patients (pT3b/R1/ISUP grade group 4–5 PCa). In contrast to these studies, a propensity score-matched retrospective analysis of two cohorts of 366 pT3 and/or R1 patients found that compared to SRT at a PSA between 0.1 and 0.5ng/mL, ART given at an undetectable PSA (< 0.1ng/mL) improved all three endpoints: BCR, MFS and OS [951].
Both approaches (ART and early SRT), together with the efficacy of adjuvant ADT, are compared in three prospective RCTs: the Medical Research Council (MRC) Radiotherapy and Androgen Deprivation In Combination After Local Surgery (RADICALS) trial [952]; the Trans-Tasman Oncology Group (TROG) Radiotherapy Adjuvant Versus Early Salvage (RAVES) trial [953]; and the Groupe d’Etude des Tumeurs Uro- Genitales (GETUG-AFU 17) trial [954]. In addition, a preplanned meta-analysis of all three trials has been published (Table 6.3.3) [955].
Two trials closed early after randomising 333/470 patients (RAVES) and 424/718 (GETUG-AFU-17) patients. RADICALS-RT included 1,396 patients, 93% (648/697) in the ART group. At the time of the ten-year analysis, 39% (270/699) of the Savage-RT-Policy Group started SRT with a median pre-SRT PSA-level of 0.2ng/ml. With the option of subsequent inclusion in RADICALS-HT, 154/649 (24%) of patients starting in the adjuvant RT group also received neoadjuvant or adjuvant HT: 90 patients for six months, 45 patients for two years and 19 patients outside RADICALS-HT. From the SRT group, 61/228 (27%) received neoadjuvant or adjuvant HT for six months (n = 33) and two years (n = 13). Fifteen of these patients were treated outside the trial [952]. All men in the GETUG-AFU-17 trial (n = 424) received six months of HT. Altogether, 684 out of 2,153 patients received additional ADT for at least six months across both trials [955]. Radiotherapy to the pelvic lymphatics was permitted in the GETUG-AFU and in the RADICALS-RT trials.
The primary endpoint for RAVES and GETUG-AFU 17 was biochemical PFS, while the primary endpoint for RADICALS-RT was MFS. So far, only RADICALS-RT have reported the ten-year primary endpoint data [956]. With a median follow up of 7.8 years, the 10-year FFDM was 93% (ART) versus 90% (SRT) (HR 0.68, p = 0.095), although based upon just 80 events in 1,396 patients. BPFS and OS also showed no significant difference (Table 6.3.3). With a median follow-up of between 4.9 years and 6.25 years in the ARTISTIC-Meta-analysis, no statistically significant difference was found for biochemical PFS for both treatments in all three trials (see Table 6.2.3). Additionally, there was a significant lower rate of grade ≥ 2 GU late side effects and grade 3–4 urethral strictures in favour of early SRT, which may also be caused by the low number of patients with PSA-progression and subsequent need for early SRT at the time of analysis (40% of patients) [955]. It should be noted that the side-effect profile may have been impacted with a larger proportion of ART patients receiving treatment with older 3D-treatment planning techniques as compared to SRT patients (GETUG-AFU 17: ART, 69% 3D vs. 46% SRT), and patients treated more recently were more likely to undergo IMRT techniques with a proven lower rate of late side effects [746]. Based on these three trials, however, patients with ‘low risk factors’ of biochemical progression after RP should be closely followed up with ultrasensitive assays and SRT should be discussed, if needed, as soon as PSA starts to rise, which must be confirmed by a second PSA measurement.
The proportion of patients with adverse pathology at RP (ISUP GG 4–5 and pT3 with or without positive margins) in all three trials was low (between 10–20%), and therefore even the meta-analysis may be underpowered to show an outcome in favour of SRT [955,957,958]. The subset analysis of this primary endpoint based on the pre-randomisation strata (i.e. the high-risk features ISUP GG 4 vs. ISUP GG 1-3 and pT3b-4 vs. ≤ pT3a) is still awaited to indicate whether these high-risk groups benefit from ART compared with SRT. However, a retrospective multi-centre study comparing ART and SRT in 26,118 patients, 2,424 of whom had high-risk features (pN1 or ISUP GG 4–5 and pT3/4 tumours) after RP [959], does support ART. With a median follow-up of 8.2 years and after excluding men with persistent PSA after RP ART showed a significantly lower acute mortality risk when compared with early SRT (p = 0.02, HR: 0.33). Therefore, ART continues to be a recommended treatment option in highly selected patients with adverse pathology (‘high-risk patients’), i.e. ISUP GG 4–5 and pT3 with or without positive margins [960,961].
In conclusion, the vast majority of patients with an undetectable PSA (< 0.1ng/ml) after RP do not need ART. However, in patients with high risk factors (pT3/4 and ISUP GG 4-5), ART to the prostatic bed should be given as they were under-represented in RADICALS, as well as in the meta-analysis [952-955] on the one hand, and the proven effect in RCT‘s on the other [948,962,963].
Table 6.3.2: Overview of all four randomised trials for adjuvant surgical bed radiation therapy after RP* (without ADT)
| Study | n | Inclusion criteria | Randomisation | Definition of BCR PSA (ng/mL) | Median FU (mo.) | Biochemical Progression- free survival | Overall survival |
SWOG 8794 2009 [962] | 431 | pT3 cN0 ± involved SM | 60-64Gy vs. observation | > 0.4 | 152 | 10 yr.: 53% vs. 30% (p < 0.05) | 10 yr.: 74% vs. 66% Median time: 15.2 vs. 13.3 yr., p = 0.023 |
EORTC 22911 2012 [963] | 1,005 | pT3 ± involved SM pN0 pT2 involved SM pN0 | 60Gy vs. observation | > 0.2 | 127 | 10 yr.: 60.6% vs. 41% (p < 0.001) | 81% vs. 77% n.s. |
ARO 96-02 2014 [948] | 388 | pT3 (± involved SM) pN0 PSA post-RP undetectable | 60Gy vs. observation | > 0.05 + confirmation | 112 | 10 yr.: 56% vs. 35% (p = 0.0001) | 10 yr.: 82% vs. 86% n.s. |
FinnProstate Group 2019 [964] | 250 | pT2,R1/ pT3a | 66.6Gy vs. observation (+ SRT) | > 0.4 (in 2 successive measure- ments) | 112 vs. 103 (patients alive) | 10 yr.: 82% vs. 61% p < 0.001 | 10 yr.: 92% vs. 87% n.s. |
*See Section 6.3.5 for delayed (salvage) post-radical prostatectomy external irradiation.BCR = biochemical recurrence; FU = follow-up; mo. = months; n = number of patients; n.s. = not significant; PSA = prostate-specific antigen; RP = radical prostatectomy; SM = surgical margin; SRT = salvage radiotherapy.
Table 6.3.3: Overview of all three randomised trials and one meta-analysis for patients treated with adjuvant vs. early salvage RT after RP
| Study | n | Inclusion criteria | Randomisation | Definition of BCR PSA (ng/mL) | Median FU (yr) | BPFS | OS or MFS | Side effects |
RAVES TROG 08.03/ ANZUP 2020 [953] | 333 target was 470 closed early | pT3a/pT3b any T - SM+PSA post- RP: < 0.1ng/mL | 64Gy ART PSA: < 0.1ng/mL vs. 64Gy early SRT at PSA > 0.2ng/mL med. pre-SRT: n.r. | > 0.4 post RT | 6.1 | 5 yr.: 86% vs. 87% (p > 0.05) | n.r. | LT grade ≥ GU: 70% vs. 54% (p = 0.002) |
| RADICALS-RT [952] | 1,396 | pT3a/pT3b/pT4 or PSA > 10ng/mL or pre-RP or any T, SM+ or Gleason 7-10. PSA post RP: < 0.2ng/mL | 52.5Gy (20Fx) or 66Gy (33Fx) ART early SRT identical at PSA > 0.1 med.pre-SRT: 0.2ng/mL | > 0.4 or 2 at any time | 7.8 | 10 yr.: 76% vs. 75% (p = 0.82 HR: 0.97) | OS: 87.6% vs. 87.4% (p = 0.92 HR: 0.98) FFDM: 93% vs. 90% (p = 0.095 HR:0.68) | SR urinary incontinence 1 yr.: 4.8 vs. 4 (p = 0.023) Urethral stricture grade 3/4 >2 yr.: 3% vs. 5% (p = 0.001) |
| GETUG-AFU 17 2020 [954] | 424 target was 718 closed early | pT3a/pT3b/pT4a and SM+PSA post-RP: < 0.1ng/mL | 66Gy (ART) vs. 66Gy early SRT at PSA 0.1 both groups: 6 mo. LHRH-A med. pre-SRT 0.24 | > 0.4 | 6.25 | 5 yr.: 92% vs. 90% (p = 0.42) | n.r. | LT grade ≥ 2 GU: 27% vs. 7% (p < 0.001) ED: 28% vs. 8% (p < 0.001) |
| ARTISTIC 2020 [955] | 2,153 | see above | see above | see above | 4.9- 6.25 | 5 yr.: 89% vs. 88% (p = 0.7) | n.r. | n.r. |
ART = adjuvant radiotherapy; BCR = biochemical recurrence; BPFS = biochemical progression-free survival; ED = erectile dysfunction; FFDM= Freedom from Distant Mets; FU = follow-up; fx = fraction; GU = genitourinary; LHRH = luteinising hormone-releasing hormone; LT = late toxicity; mo. = months; med = median; MFS = metastasis-free survival; n.r. = not reported; OS = overall survival; PSA = prostate-specific antigen; RP = radical prostatectomy; RT = radiotherapy; SRT = salvage radiotherapy; + = positive; yr. = years.
6.3.5.e. Adjuvant systemic therapy in N0 disease
The TAX3501 trial comparing the role of leuprolide (18 months) with and without docetaxel (six cycles) ended prematurely due to poor accrual. A phase III RCT comparing adjuvant docetaxel against surveillance after RP for locally advanced PCa showed that adjuvant docetaxel did not confer any oncological benefit [965]. Consequently, adjuvant chemotherapy after RP should only be considered in a clinical trial [966].
6.3.5.f. Adjuvant treatment in pN1 disease
6.3.5.f.1. Adjuvant androgen ablation alone
The combination of RP and early adjuvant HT in pN+ PCa has been shown to achieve a 10-year CSS rate of 80% and has been shown to significantly improve CSS and OS in prospective RCTs [967,968]. However, these trials included mostly patients with high-volume nodal disease and multiple adverse tumour characteristics, and these findings may not apply to men with less-extensive nodal metastases.
6.3.5.f.2. Adjuvant radiotherapy combined with ADT in pN1 disease
In a retrospective multicentre cohort study, maximal local control with RT to the prostatic fossa appeared to be beneficial in PCa patients with pN1 after RP, treated ‘adjuvantly‘ with continuous ADT (within six months after surgery irrespective of PSA). The beneficial impact of adjuvant RT on survival in patients with pN1 PCa was highly influenced by tumour characteristics. Men with low-volume nodal disease (< 3 LNs), ISUP grade group 2–5 and pT3–4 or R1, as well as men with three to four positive nodes were more likely to benefit from RT after surgery, while the other subgroups did not [969]. In contrast, a retrospective multicentre study including 1,614 patients and a median follow-up of 7.02 years assessed ART + ADT. Adjuvant RT compared to SRT was associated with a decreased all-cause mortality and this reduction increased with each additional positive pelvic LN, from the first one on and the highest effect was for more than three positive nodes [970]. These data correspond with a United States National Cancer Database analysis based on 5,498 patients [971]. Another United States National Cancer Database study including 8,074 pN1 patients reports improved OS after ADT plus EBRT (including pelvic LNs) versus observation and versus ADT alone in all men with single or multiple adverse pathological features. Men without any adverse pathological features did not benefit from immediate adjuvant therapy [972].
In an SR of the literature, RT with or without ADT was associated with improved survival in men with locally advanced disease and a higher number of positive nodes [973]. Radiotherapy to the pelvic lymphatics and the prostate fossa plus long-term ADT can be offered to patients with pN1 disease [969,974]. However, the optimal duration of ADT is still unknown.
6.3.5.f.3. Observation of pN1 patients after radical prostatectomy and extended lymph node dissection
Several retrospective studies and an SR addressed the management of patients with pN1 PCa at RP [945,969,973-975]. A subset of patients with limited nodal disease (1–2 positive LNs) showed favourable oncological outcomes and did not require additional treatment.
An analysis of 209 pN1 patients with one or two positive LNs at RP showed that 37% remained metastasis-free without need of salvage treatment at a median follow-up of 60.2 months [975]. Touijer et al., reported their results of 369 pN1-positive patients (40 with and 329 without adjuvant treatment) and showed that higher pathologic grade group and > 3 positive LNs were significantly associated with an increased risk of BCR on multivariable analysis [945]. Biochemical-free survival rates in pN1 patients without adjuvant treatment ranged from 43% at four years to 28% at ten years [973]. Reported CSS rates were 78% at five years and 72% at ten years. The majority of these patients were managed with initial observation after surgery, had favourable disease characteristics, and 63% had only one positive node [973]. Initial observation followed by early salvage treatment at the time of recurrence may represent a safe option in selected patients with a low disease burden [973].
6.3.5.g. Recommendations for adjuvant treatment for pN0 and pN1 disease after radical prostatectomy*
| Recommendations | Strength rating |
| Do not prescribe adjuvant androgen deprivation therapy (ADT) to pN0 patients. | Strong |
| In pN0 patients with ISUP GG 4–5 and pT3 ± positive margins, offer adjuvant intensity-modulated radiation therapy (IMRT)/volumetric modulated arc therapy (VMAT) plus image-guided radiation therapy (IGRT). | Weak |
In pN1 patients, after an extended lymph node dissection (eLND), discuss three management options, based on nodal involvement characteristics:
| Weak |
*All recommendations are based on conventional imaging with isotope bone scan and CT/MR abdomen/pelvis.
6.3.6. Persistent PSA after radical prostatectomy
6.3.6.a. Definition of PSA persistence
Detectable PSA levels after RP may result from persistent local disease, pre-existing metastases, or residual benign prostate tissue. PSA persistence, defined as a PSA ≥ 0.1ng/mL within four to eight weeks of surgery, is observed in 5–20% of patients [976,977]. However, timing of PSA measurement after RP might influence its interpretation [978,979]. Among patients tested within three weeks of RP, 77% had PSA > 0.1ng/mL, but only 21% remained detectable on repeat testing. When PSA was measured more than five weeks after RP, only 10% had PSA > 0.1ng/mL, and 20% of these became undetectable on confirmatory testing [978]. Patients with preoperative PSA ≥ 20ng/mL are particularly prone to misclassification if tested too early, as delayed measurements (≥ 3 months) have shown that some of these patients eventually reach undetectable PSA levels [979].
6.3.6.b. Predictors of PSA persistence after RP
A meta-analysis of consecutive patient cohorts reported that persistent PSA was more likely when risk factors such as high D’Amico risk, ISUP GG 4-5, pT stage ≥ 8 and presence of extraprostatic extension, seminal vesicle invasion, lymph node involvement and positive margin were present [980]. Cribriform pattern or intraductal carcinoma have also been associated with persistent PSA [981]. Moreover, an uptake in the pelvic nodes at PSMA PET represents one of the strongest predictors of persistently detectable PSA levels after surgery, with more than 50% of patients with miN1 PCa and more than two positive spots and/or extraprostatic extension experiencing PSA persistence [982,983].
6.3.6.c. Natural history of persistently elevated PSA after RP
Two SRs addressing persistent PSA confirmed a strong correlation of PSA persistence with poor oncologic outcomes [976,977]. In a meta-analysis of retrospective cohorts, patients with persistent PSA (≥ 0.1 ng/mL) had worse BCR-free, metastasis-free and cancer-specific survival [980]. A retrospective study (n = 11,605) showed that persistent PSA is prognostic of an increased risk of metastasis and death [984]. At fifteen years post RP, MFS rates, OS and CSS rates were 53 versus 93% (p < 0.001), 65 versus 81% (p < 0.001) and 75 versus 96% (p < 0.001) for persistent versus undetectable PSA, respectively. In multivariable Cox regression models, persistent PSA represented an independent predictor for metastasis (HR: 3.59, p < 0.001), death (HR: 1.86, p < 0.001) and cancer-specific mortality (HR: 3.15, p < 0.001). However, not all patients with persistent PSA after RP experience disease recurrence. One study showed a 50% five-year BCR-free survival in men who had a persistent PSA level > 0.1 but ≤ 0.2ng/mL at six to eight weeks after RP [985]. Another study assessed the clinical outcome of 160 men with a persistently detectable PSA level after RP [986]. In multivariable analysis, the PSA slope ≥ 0.05 after RP (as calculated using PSA levels three to twelve months after surgery) and pathological ISUP grade group (≥ 3 vs. ≤ 2) were significantly associated with the development of distant metastases among patients with persistent PSA. Prostate-specific antigen slope is more commonly reported as PSA doubling time (calculated by log [PSA slope]) [987]. Increasing PSA levels are associated with a higher risk of PCa-specific mortality particularly in patients with more aggressive disease [988]. Patients with persistent PSA levels ≥ 1 ng/ml had higher all-cause and PCa-specific mortality risk compared to those with PSA levels < 1 ng/mL [979].
6.3.6.d. Imaging in patients with persistently elevated PSA after RP
Compared to conventional imaging, PSMA PET/CT is characterised by superior accuracy compared to conventional imaging. However, studies of patients with PSA persistence after RP are limited compared to those inclusive of patients with BCR with/without persistent PSA.
A study of 129 patients who had either persistent PSA (52%) or BCR (48%) after RP, showed that men with a persistent PSA had significantly more pelvic nodal involvement on PSMA PET/CT than those with an initially undetectable PSA [989]. When focusing on patients with PSA persistence a multicentre retrospective study included 191 patients with persistently elevated PSA after RP and 68Ga-PSMA-PET/CT was positive in 68%, 35% of whom had disease confined to the pelvis (obturator, presacral/mesorectal most common) and 33% had distant metastases [525]. A subgroup analysis of 33 patients with pre- and post-RP imaging showed PET-persistence in 45%, new lesions in 24% and negative post-RP PET in 30%. Another retrospective study included 150 patients with persistent PSA after RARP who were restaged with both 68Ga-PSMA and 18F-DCFPyL PSMA. The authors found that, in the presence of persistent PSA, the majority of patients already had involved pelvic LNs (33%) or distant metastases (26%) that would support a role of PSMA PET/ CT imaging in guiding (salvage) treatment strategies [990].
Taken together, these findings support the role of PSMA PET/CT in identifying sites of residual or metastatic disease in patients with persistent PSA after RP, enabling the implementation of metastasis-directed therapies.
6.3.6.e. Management options for patients with persistent PSA
6.3.6.e.1. Comparison with biochemical recurrence (BCR)
Persistent PSA after RP is clearly a poor prognostic indicator, likely representative of low volume synchronous metastatic disease rather than metachronous disease as in BCR. A retrospective analysis of the RTOG 9601 trial of SRT +/- ADT (bicalutamide) for biochemical failure after RP considered patients with persistent PSA (n = 90) or BCR (n = 670) as the cause for biochemical failure and showed higher 10-year metastatic progression rate (29% vs. 10%, p < 0.0001), higher 10-year overall mortality rate (25% vs. 12%, p = 0.03), and higher local progression rate (3.2% vs. 1.4%, p = 0.0001) [991]. In the ARO 96-02, a prospective RCT, 74 patients with PSA persistence (20%) received immediate SRT only (66Gy per protocol [arm C]). The 10-year clinical relapse-free survival was 63% and showed worse 10-year metastasis-free survival (67% vs 83%) and overall survival (68% vs 84%) than BCR patients [992]. Therefore, it is likely that outcomes are worse than for men with persistent PSA than those experiencing BCR [993]. Indeed, studies investigating PSA persistence were excluded from the EAU Guidelines Biochemical Recurrence risk groups [994].
6.3.6.e.2. Postoperative RT
The benefit of postoperative RT (adjuvant or salvage) in patients with persistent PSA remains unclear due to a lack of RCTs specifically focusing on this setting. AN SR reported that SRT was associated with improved survival outcomes, although the available evidence is of low quality and did not include patients restaged with PSMA/PET [977].
Another study compared oncological outcomes of patients with persistent PSA who received SRT vs. those who did not [984]. In the subgroup of patients with persistent PSA, after 1:1 propensity score matching between those treated with SRT versus no RT, 10-year overall- and cancer-specific survival rates were 87 versus 73% and 94 versus 82% (p < 0.01), respectively. These differences remained statistically significant when patients were stratified according to margin status, pathologic stage, ISUP GG, and lymph node involvement. In multivariable models SRT was associated with lower risk of death (HR: 0.42, p = 0.02) and lower cancer-specific death (HR: 0.29, p = 0.03). SRT dose was 46Gy for the 54% of patients with available data, but SRT field and ADT use was unavailable. A retrospective analysis of 313 patients with PSA persistence (median follow-up 4.5 years) suggested a benefit of SRT, with or without ADT, compared to no SRT in improving metastases-free (HR 0.39, p = 0.001), cancer-specific (HR 0.34, p = 0.03) and overall survival (HR 0.24, p = 0.001) [995]. These benefits were associated with higher incidence of bowel symptoms (34 vs. 19%, p = 0.01) and bothersome incontinence if given within six months of surgery (p < 0.001).
Poor oncological outcomes are driven by the level of pre-RT PSA, the presence of ISUP GG ≥ 4 in the RP histology and pT3b disease [988,992,996-999]. Two studies suggested that only men with a persistent PSA after RP and ISUP GG ≤ 3 benefit significantly [995,1000], where positive margins, higher T stage, pN1 and lower ISUP GG were most likely to benefit from SRT, although this was not supported by Preisser et al. [984,995].
The current data do not allow clear treatment recommendations.
6.3.6.e.3. Multimodal therapy (ADT with postoperative RT)
The RTOG 9601 trial randomised patients with either PSA persistence defined as PSA nadir after surgery > 0.5 ng/mL (n = 45, 12%) or BCR (n = 338, 88%) to 24 months of bicalutamide versus placebo during and after SRT [1001]. Although the study demonstrated a benefit of antiandrogen therapy in terms of OS at a median follow-up of 13 years, no subgroup analyses were performed focusing only on men with PSA persistence. Similarly, the NRG Oncology/RTOG 0534 SPPORT trial included patients with PSA persistence after prostatectomy, alongside those with rising PSA, and demonstrated improved freedom from progression with the addition of four to six months of ADT and pelvic lymph node radiotherapy [1002]. However, no subgroup analysis assessing patients with PSA persistence was reported. Other randomised trials which assessed the role of concomitant ADT and its duration at the time of SRT on oncologic control such as the RADICALS-HD and the GETUG-AFU 16 both excluded patients with PSA persistence and, therefore, their results are not applicable to this setting [1003,1004].
The phase II GETUG-22 trial comparing RT (46Gy pelvis with 66Gy prostate bed boost) with RT plus short-term ADT for post-RP PSA persistence (0.2–2.0ng/mL) in 125 patients reported good tolerability of the combined treatment. The oncological endpoints are yet to be published [1005]. A multicentre, retrospective study from Japan considered 383 patients with pN1 and persistent PSA after RP and reported that the addition of SRT (median 66Gy; prostate bed with pelvis 67%, prostate bed alone 24%) to ADT showed better castration resistance-free (5-year p < 0.001, 10-year p = 0.02) and metastasis-free (5-year p < 0.001, 10-year p = 0.15), but not overall survival, than ADT alone in patients with pretreatment PSA ≥ 0.52ug/L [1006].
6.3.6.f. Conclusion
The available data suggest that patients with PSA persistence after RP have worse outcomes and serve to benefit most from early aggressive multimodality treatment, however, the lack of prospective RCTs makes firm recommendations difficult.
6.3.6.g. Recommendations for the management of persistent PSA after radical prostatectomy
| Recommendations | Strength rating |
| Offer a prostate-specific membrane antigen (PSMA) positron emission tomography/computed tomography (PET/CT) scan to males with a persistent prostate-specific antigen (PSA) and rising if the results will influence subsequent treatment decisions. | Weak |
| Treat males with persistent PSA and no evidence of distant metastatic disease with salvage radiotherapy and additional hormonal therapy. | Weak |
6.4. Management of PSA-only recurrence after treatment with curative intent
Follow-up will be addressed in Chapter 7 and is not discussed in this section.
6.4.1. Background
Between 27% and 53% of all patients undergoing RP or RT develop a rising PSA (PSA recurrence). Whilst metastatic progression is universally preceded by rising PSA levels, physicians must inform the patient that the natural history of PSA-only recurrence may be prolonged and that a measurable PSA may not necessarily lead to clinically apparent metastatic disease. Physicians treating patients with PSA-only recurrence face a difficult set of decisions in attempting to delay the onset of metastatic disease and death while avoiding overtreating patients whose disease may never affect their OS or QoL. It should be emphasised that the treatment recommendations for these patients should be given after discussion in a multidisciplinary team.
6.4.1.a. PSA velocity and doubling time
Various PSA kinetics definitions have been proposed with different methods of calculation (log transformed or not) and eligible PSAs:
- PSA velocity (PSAV): absolute annual increase in serum PSA (ng/mL/year); and
- PSA doubling time (PSA-DT): measures the exponential increase in serum PSA over time.
Prostate-specific antigen velocity is easier to calculate by subtracting the initial value from the final value, dividing by time. However, by ignoring middle values, not all PSA values are accurately taken into account.
Prostate-specific antigen DT is calculated assuming an exponential rise in serum PSA. The formula takes into account the natural logarithm of two divided by the slope obtained from fitting a linear regression of the natural log of PSA over time [1007]. However, many different PSA-DT calculations have been assessed according to the mathematical formula used and to the included PSA values (number, time period, intervals) [1008]. The ‘MSKCC’ method, for example, calculates a regression slope integrating all PSA values. Other methods transform PSA before calculating the slope and do not include all PSA values (different time frames and minimal intervals) [1009]. O’Brien and colleagues identified more than 20 different definitions of PSAV and PSA-DT and demonstrated that obtained values could vary widely between definitions [1009].
However, some rules can be considered for PSA-DT calculation [1007]:
- At least three PSA measurements are required.
- A minimum period between measurements (four weeks) is preferable due to potential statistical ‘noise’ when PSA values are obtained too close together (this statement can be reconsidered in case of highly active disease).
- All included PSA values should be obtained within the past twelve months at most, to reflect the current disease activity.
- PSA-DT is often expressed in months, or in weeks in highly active disease.
These measurements do not provide additional information compared with PSA alone [604,1009-1011]. In the post-local therapy relapse setting, PSA-DT has been correlated with distant progression and with poorer outcomes after salvage treatments [1012,1013]. Prostate-specific antigen DT has been linked with metastasis-free and OS in non-metastatic CRPC (nmCRPC) and identifies patients with high-risk nmCRPC who benefit from intensified therapy (PSA-DT threshold < ten months) [1014].
6.4.2. Controversies in the definitions of clinically relevant PSA relapse
The PSA level that defines treatment failure depends on the primary treatment. Patients with rising PSA after RP or primary RT have different risks of subsequent symptomatic metastatic disease based on various parameters, including the PSA level. Therefore, physicians should carefully interpret BCR endpoints when comparing treatments. Clinicians should interpret a PSA rise bearing in mind the EAU BCR risk groups [994].
After RP, the threshold that best predicts further metastases is a PSA > 0.4ng/mL and rising [1015]. However, with access to ultrasensitive PSA testing, a rising PSA much below this level will be a cause for concern for patients.
After primary RT - with or without short-term hormonal manipulation - the RTOG-ASTRO Phoenix Consensus Conference definition of PSA failure (with an accuracy of > 80% for clinical failure) is ‘any PSA increase > 2ng/mL higher than the PSA nadir value, regardless of the serum concentration of the nadir’ [1016].
After HIFU or cryotherapy, no endpoints have been validated against clinical progression or survival. Therefore, it is not possible to offer a firm recommendation of an acceptable PSA threshold after these alternative local treatments [994].
6.4.3. Natural history of biochemical recurrence
Once a PSA recurrence has been diagnosed, it is important to determine whether the recurrence has developed at local or distant sites. A SR and meta-analysis investigated the impact of BCR on clinical endpoints and concluded that patients experiencing BCR are at an increased risk of developing distant metastases, as well as PCa-specific and overall mortality [994]. The effect size of BCR as a risk factor for mortality, however, is highly variable. After primary RP, its impact ranges from HR 1.03 (95% CI: 1.004–1.06) to HR 2.32 (95% CI: 1.45–3.71) [1017,1018]. After primary RT, OS rates are approximately 20% lower at eight to ten years follow-up, even in men with minimal comorbidity [1019,1020]. Still, the variability in reported effect sizes of BCR remains high and suggests that only certain patient subgroups with BCR might be at an increased risk of mortality.
The risk of subsequent metastases, PCa-specific and overall mortality may be predicted by the initial clinical and pathologic factors (e.g. T category, PSA, ISUP grade group) and PSA kinetics (PSA-DT and interval to PSA failure), which was further investigated by the SR [994].
For patients with BCR after RP, the following outcomes were found to be associated with significant prognostic factors, including:
- distant metastatic recurrence: positive surgical margins, high RP specimen pathological ISUP GG, high pT category, short PSA-DT, high pre-SRT PSA;
- prostate-cancer-specific mortality: high RP specimen pathological ISUP grade group, short interval to biochemical failure as defined by investigators, short PSA-DT; and
- overall mortality: high RP specimen pathological ISUP grade group, short interval to biochemical failure, high PSA-DT.
For patients with BCR after RT, the corresponding outcomes are:
- distant metastatic recurrence: high biopsy ISUP grade group, high cT category, short interval to biochemical failure;
- prostate-cancer-specific mortality: short interval to biochemical failure; and
- overall mortality: high age, high biopsy ISUP grade group, short interval to biochemical failure, high initial (pretreatment) PSA.
Based on this meta-analysis, the proposal is to stratify patients into two risk categories since not all patients with BCR will have similar outcomes (see Table 6.4.1). The stratification into ‘EAU Low-Risk’ or ‘EAU High-Risk’ BCR after RP has been validated in a European cohort [1021].
Table 6.4.1: EAU risk categories for patients developing biochemical recurrence
| EAU Low Risk BCR | EAU High Risk BCR | |
| After RP | PSA-DT > 1 yr AND pathological ISUP grade group < 4 | PSA-DT ≤ 1 yr OR pathological ISUP grade group 4-5 |
| After RT | interval to biochemical failure > 18 mo. AND biopsy ISUP grade group < 4 | interval to biochemical failure ≤ 18 mo OR biopsy ISUP grade group 4-5 |
6.4.4. The role of imaging in PSA-only recurrence
Imaging is only of value if it leads to a treatment change that results in an improved outcome. In practice, however, very limited data are available regarding the outcome’s consequent on imaging at recurrence.
6.4.4.a. Assessment of metastases (including nodal)
6.4.4.a.1. Bone scan and abdominopelvic CT
As BCR after RP or RT precedes clinical metastases by seven to eight years on average [939,1022], the diagnostic yield of conventional imaging techniques (bone scan and abdominopelvic CT) is low in asymptomatic patients [1023]. In men with PSA-only recurrence after RP, the probability of a positive bone scan is < 5% when the PSA level is < 7ng/mL [1024,1025]. Only 11–14% of patients with BCR after RP have a positive CT [1024]. In a series of 132 men with BCR after RP, the mean PSA level and PSA velocity associated with a positive CT were 27.4ng/mL and 1.8ng/mL/month, respectively [1026].
6.4.4.a.2. Fluoride PET/CT
18F-NaF PET/CT has a higher sensitivity than bone scan in detecting bone metastases [1027]. However, 18F-NaF PET/CT is limited by a relative lack of specificity and by the fact that it does not assess soft-tissue metastases [1028].
6.4.4.a.3. Prostate-specific membrane antigen hybrid imaging
Radiolabelled PSMA hybrid imaging (i.e., PET/CT or PET-MRI) has shown good potential in patients with BCR. The diagnostic performance of radiolabelled PSMA PET/CT in patients with BCR has been investigated in several SRs and meta-analyses. The pooled sensitivity, specificity and AUC values for 18F-PSMA PET/CT in the diagnosis of prostate recurrence and/or metastasis were 0.93 (0.89–0.95), 0.94 (0.85–0.98) and 0.96 (0,94–0.98), respectively. The per-patient pooled sensitivity and specificity values were 0.92 (0.86–0.96) and 0.83 (0.41–0.97), respectively. The per-lesion pooled sensitivity and specificity values were identical: 0.91 (0.86–0.94) [1029]. The positivity rate increases with PSA, from 48% at PSA of 0.2-0.5 ng/ml to >90% at PSA >2 ml [1030].
A prospective multicentre, multi-reader, open-label, phase II/III trial (OSPREY) evaluated the diagnostic performance of 18F-DCFPyL in patients with presumptive radiologic evidence of recurrent or metastatic PCa on conventional imaging [964]. Median sensitivity and median PPV were 95.8% (95% CI: 87.8%–99.0%) and 81.9% (95% CI: 73.7%–90.2%), respectively.
Another prospective study evaluated the diagnostic performance of 18F-DCFPyL in 208 men with BCR after RP or RT. The primary endpoint, the correct localisation rate was achieved, demonstrating positive findings on 18F-DCFPyL PET/CT in the setting of negative standard conventional imaging [1031]. At present, there are no conclusive data concerning the comparison of such tracers [1032].
6.4.4.a.4. Whole-body and axial MRI
Whole-body MRI has not been widely evaluated in BCR because of its limited value in the detection of early metastatic involvement in normal-sized LNs [516,551,1033]. In a prospective series of 68 patients with BCR, the diagnostic performance of DW-MRI was significantly lower than that of 68Ga-PSMA PET/CT and 18NaF PET/CT for diagnosing bone metastases [1034].
6.4.4.b. Assessment of local recurrences
6.4.4.b.1. Local recurrence after radical prostatectomy
As the sensitivity of anastomotic biopsies is low, particularly for PSA levels < 1ng/mL [1023], SRT is usually decided based on BCR without histological proof of local recurrence.
Magnetic resonance imaging can detect local recurrences in the prostatic bed. The PSA threshold for MRI positivity appears to be between 0.3 and 0.5ng/mL, and PSA kinetics also influence the MRI positivity, even at low PSA values [1035]. Two single-centre studies found that a negative MRI was an independent predictor of failure of SRT [1036,1037]. Conversely, a small (≤ 0.4cc) relapse located at the vesico-urethral anastomosis is associated with excellent prognosis at salvage RT [1038]. The Prostate Imaging for Recurrence Reporting (PI-RR) system has been recently launched to standardise MRI interpretation in the context of BCR after RP or RT [1039]. Initial assessment suggests good reproducibility of the score [1040,1041].
The detection rates of 68Ga-PSMA PET/CT in patients with BCR after RP increase with the PSA level [1042]. PSMA PET/CT studies showed that a substantial part of recurrences after RP were located outside the prostatic fossa, even at low PSA levels [1043,1044]. Combining 68Ga-PSMA PET and MRI may improve the detection of local recurrences as compared to 68Ga-PSMA PET/CT alone [1045-1047].
6.4.4.b.2. Local recurrence after radiation therapy
In patients with BCR after RT, biopsy status is a major predictor of outcome, provided the biopsies are obtained at least 24 months after initial treatment. Given the morbidity of local salvage options, histological proof of the local recurrence must be obtained before treating the patient [1023].
Magnetic resonance imaging has yielded excellent results in identifying local recurrence and can be used for biopsy targeting and guiding local salvage treatment [1023,1048,1049], even if it slightly underestimates the volume of the local recurrence [1050]. Prostate-specific membrane antigen PET/CT can also detect local recurrences after RT [1051] and concordance between PSMA PET/CT and MRI is highly suggestive of cancer recurrence [1052].
In the FLAME trial PSMA PET/CT was used for recurrence after RT. Intra-prostatic recurrences in intermediate- and high-risk patients appeared at the location of the primary tumour in 98% of cases [1053].
6.4.4.c. Summary of evidence of imaging in case of biochemical recurrence
In patients with BCR, imaging can detect both local recurrences and distant metastases, however, the sensitivity of detection depends on the PSA level. After RP, PSMA hybrid imaging is the modality with the highest sensitivity at low PSA levels (< 0.5ng/mL) and may help distinguishing patients with recurrences confined to the prostatic fossa from those with distant metastases, which may impact the design and use of post-RP SRT. After RT, MRI has shown excellent results at detecting local recurrences and guiding prostate biopsy. Given the substantial morbidity of post-RT local salvage treatments, distant metastases must be ruled out in patients with local recurrences and who are fit for these salvage therapies.
6.4.4.d. Recommendations for imaging in patients with biochemical recurrence
| Recommendations | Strength rating |
| Prostate-specific antigen (PSA) recurrence after radical prostatectomy | |
| Perform prostate-specific membrane antigen (PSMA) positron emission tomography/computed tomography (PET/CT) if the PSA level is > 0.2 ng/mL and if the results will influence subsequent treatment decisions (i.e. EAU BCR risk groups). | Weak |
| PSA recurrence after radiotherapy | |
| Perform PSMA PET/CT in patients fit for curative salvage treatment. | Strong |
| Perform prostate magnetic resonance imaging to localise abnormal areas and guide biopsies in patients fit for local salvage therapy. | Weak |
6.4.5. Treatment of PSA-only recurrences
The timing and treatment modality for PSA-only recurrences after RP or RT remain a matter of controversy based on the limited evidence.
6.4.5.a. Treatment of PSA-only recurrences after radical prostatectomy
6.4.5.a.1. Salvage radiotherapy for PSA-only recurrence after radical prostatectomy (pTxcN0M0,without PET/CT)
Early SRT provides the possibility of cure for patients with an increasing PSA after RP. Boorjian et al. reported a 75% reduced risk of systemic progression with SRT when comparing 856 SRT patients with 1,801 non-SRT patients [1054]. The RAVES and RADICALS trials assessing SRT in post-RP patients with PSA levels exceeding 0.1–0.2ng/mL showed five-year freedom from BCR and BCR-free survival rates of 88% [1040,1055]. Tilki et al. demonstrated the results of a matched-pair analysis of 1,832 patients with BCR: 32.9% (n = 603) received SRT without ADT, while 1,229 (67.1%) had an observational strategy. The median follow-up was 95.9 months. Median total SRT dose was 70.2Gy. After 1:1 propensity score matching, at fifteen years after RP, MFS and OS rates were 84.3 versus 76.9% (p < 0.001) and 85.3 versus 74.4% (p = 0.04) for SRT and no RT, respectively [1056].
The PSA level at BCR was shown to be prognostic [1054]. More than 60% of patients who are treated before their PSA level rises to > 0.5ng/mL will achieve an undetectable PSA level [1057-1059], corresponding to an approximate 80% chance of being progression-free five years later [1060]. A retrospective analysis of 635 patients who were followed after RP and experienced BCR and/or local recurrence and either received no salvage treatment (n = 397) or SRT alone (n = 160) within two years of BCR showed that SRT was associated with a threefold increase in PCa-specific survival relative to those who received no salvage treatment (p < 0.001). Salvage RT has been shown to be effective mainly in patients with a short PSA-DT [1061].
In a retrospective multicentre study including 25,551 patients with at most one high-risk factor after RP (ISUP GG 4-5 or pT3/4), initiating SRT above a PSA level of 0.25 ng/mL was associated with increased ACM risk. After a median follow-up of six years, patients who received SRT at a PSA level > 0.25 ng/mL had a significantly higher ACM risk (AHR, 1.49; 95% CI, 1.11 to 2.00; p = 0.008) compared with men who received SRT when the PSA was ≤ 0.25 mg/mL [1062]. For an overview of SRT, see Table 6.4.3.
Although biochemical progression is now widely accepted as a surrogate marker of PCa recurrence, metastatic disease, disease-specific and OS are more meaningful endpoints to support clinical decision-making. A SR and meta-analysis on the impact of BCR after RP reports SRT to be favourable for OS and PCSM. SRT in particular should be initiated in patients with rapid PSA kinetics after RP and with a PSA cut-off of 0.4ng/mL [994]. An international multi-institutional analysis of pooled data from RCTs has suggested that MFS is the most valid surrogate endpoint with respect to impact on OS [1063,1064]. Table 6.4.4 summarises the results of recent studies on clinical endpoints after SRT.
The EAU BCR definitions have been externally validated and may be helpful for individualised treatment decisions [994,1021]. For men with limited life expectancy, salvage options are not indicated. A ‘wait and see‘ strategy is an option for the EAU BCR ‘Low-Risk’ group [994,1021].
6.4.5.a.2. Salvage radiotherapy combined with androgen deprivation therapy (pTxcN0, without PET/CT)
Data from RTOG 9601 suggest that both CSS and OS benefit when adding two years of bicalutamide (150 mg o.d.) to SRT [1001]. The OS benefit improved at 18 years (median follow up for surviving patients 18.9 years) to 53% for the combination treated patients (95% CI, 47%-58%) versus 43% (95% CI, 38%-49%) compared with 5% at 12 years. This improvement was also consistent with the initial report, with reduced incidence of metastatic PCs and PCa death for RT+HT [1065]. According to GETUG-AFU 16, six months of treatment with an LHRH-analogue can also significantly improve 10-year BCR, biochemical PFS and, modestly, MFS. However, SRT combined with either goserelin or placebo showed similar DSS and OS rates [1004].
In addition, Pollack et al., reported on the results of a randomised three-arm phase III trial (NRG Oncology/RTOG 0534 SPPORT), adding six months’ treatment with an LHRH analogue to SRT of the prostate bed (PBRT) (group 2) compared with PBRT alone (group 1) or the former combination with PBRT-RT and pelvic LN RT (PLNRT) (group 3) [1002]. The primary endpoint was freedom from progression (FFP) after five years. However, using the Phoenix definition of biochemical progression (nadir + 2ng/mL used for definitive RT) and not the criterion of nadir + 0.2 as is commonly used (but without clear evidence) will have resulted in a later diagnosis of progression in the SPPORT trial.
With a median follow-up of 8.2 years of the surviving patients, FFP increased significantly for group 3 (87.4%) compared with group 2 (81.3%) (p = 0.0027) and group 1 (70.9%) (p < 0.0001) [1002]. The difference between group 2 and group 1 was also significant (p < 0.0001). Distant metastasis incidence rates (secondary endpoint) were lowest in group 3 (including RT of the pelvic lymphatics) and were significantly lower compared only with group 1 (PBRT only, HR: 0.52), similar to the rate of PCa deaths (HR: 0.51). No significant difference was seen for OS. A significantly higher risk of both acute and late side effects was identified in group 3. Therefore, the role of additional PLNRT remains unclear and should be further proven in RCTs including PSMA PET-CT [1066].
RADICALS HD investigated the role of RT without ADT (n = 737) versus RT plus six months of ADT (n = 747) and RT plus six months of ADT (n = 761) versus RT plus 24 months’ long-term ADT (n = 762) in both the salvage and adjuvant settings [1003,1067,1068]. The design of RADICALS HD was complex and included components of the RADICALS RT trial together with the RADICALS HD component. RADICALS RT was a phase III comparison of ART versus observation and early SRT and has been published previously [956] (see Table 6.3.5.2).
RADICALS HD included men after prostatectomy (indications for ART or early SRT), median pre-SRT-PSA was 0.2ng/ml with conventional staging imaging (M0) without PET-CT. Due to the complex design, some patients were enrolled in a three-way randomisation (including patients from RADICALS RT, n = 492) and in a two-way randomisation (SRT + 6 months or 24 months of ADT, n = 1,197). The randomisation was influenced by physician preference. For this reason, more patients had high-risk factors in the short-term ADT versus long-term ADT study (ISUP >3: 29% versus 11% and for > pT3B tumours: 31% vs. 17%) compared with the no ADT and the short-term ADT study [1069].
With a median FU of nine years, the ten-year MFS (primary endpoint, inclusion of deaths from PCa only) for no ADT versus short-term ADT showed no significant difference for both arms (88.1% vs. 89.9%, p > 0.05) but for ‘Clinical progression free survival’ (68.3% vs. 79.4%, p < 0.0001 with some evidence of unproportional hazards) and ‘10-year freedom from non-protocol ADT’ (73.3% vs. 82.3%, p< 0.0001, but with clear evidence of unproportional hazards). Maximum GU-Tox grade 3 was 16% (no ADT) versus 13% (short term ADT) (p > 0.05). With a median FU of 8.9 years, the 10-year MFS (primary endpoint) in the second randomisation showed a moderate significant difference (78.1% vs. 71.9%) in favour of the long-term ADT arm compared with the short-term ADT arm (p = 0.029, HR 0.773). Comparable significant differences were seen for ‘Clinical progression-free survival’ and for ‘10-year freedom from non-protocol ADT’. Maximum GU-Toxicity grade 3 was 14% (short-term ADT) versus 20% (long-term ADT).
The authors concluded that the findings for short-term ADT versus no ADT do not support the use of ADT in this patient population. For the comparison of long-term ADT versus short-term ADT, the conclusion was that ‘individuals who can accept the additional duration of adverse effects, long-course ADT should be offered’ with SRT.
The duration of androgen suppression with postoperative radiotherapy for non-metastatic PCa was analysed in an SR and meta-analysis [1070] from the DADSPORT group. The SR included five RCTs with 4,411 patients, the median follow up across the trials was 6.2 - 13 years, the primary endpoint was OS, secondary endpoints were MFS and PCSS. Overall survival was slightly improved with an absolute effect of 2% at eight years (HR: 0.86 CI: 0.74-1.00, p = 0.057) with no clear effect of duration of HT (p = 0.6). There was no evidence of a significant difference in OS between short- and long-course HT. The study noted that any benefit appeared to be confined to man with higher pre-SRT PSA levels (p = 0.07) and CAPRA-S scores (p = 0.09). Hormonal therapy improved MFS (HR 0.78, CI 0.69-0.88, p < 0.001) and PCSS (HR: 0.61, CI: 0.47-0.79, p < 0.001) with an absolute improvement of 4% in both groups after eight years. Post-hoc comparisons were not done for combinations of risk factors such as ISUP GG 4-5 and pT3. Only 247/ 679 patients (5.8% of total patients) were classified as dying from PCa clearly showing that the majority of patients had favourable prognostic tumours. In summary the SR concluded that the improvements of short- or long course HT in these patients are unlikely to be clinically meaningful and might be limited to people with higher-risk factors and should be balanced against the potential impact of long-term HT side effects on QoL.
One of these RCTs reports improved OS (RTOG 96-01), another (GETUG-AFU 16) moderately improved MFS (7%) at ten years. The two arm comparison (SRT versus SRT + 6 months ADT) of RADICALS HD did not improve MFS at ten years, this is in contrast to the results of the GETUG-AFU 16 trial. Only the comparison of SRT + 6 months ADT versus SRT + 24 months ADT of RADICALS HD moderately improved ten year MFS (6.2%). This improvement came with the cost of increased side effects of the additional 18 months ADT including a doubling of patients with testosterone suppression after ten years compared with six months of ADT [1071].
Table 6.4.5 provides an overview of these RCTs.
Due to methodological discrepancies and also related to follow-up and risk, it is, as yet, not evident which patients should receive ADT, which type of ADT, and for how long. Men at high risk of further progression (e.g. with a PSA ≥ 0.7ng/mL and GS ≥ 8) may benefit from SRT combined with two years of ADT. For those at lower risk (e.g. PSA < 0.7ng/mL and GS = 8), SRT combined with six months of ADT may be sufficient [1001]. Men with a low risk profile (PSA < 0.5ng/mL and GS < 8) and a PSA level < 0.5ng/mL may receive SRT alone. In an unplanned subgroup analysis [1072] (RTOG 96-01) of men with a PSA of 0.61 to 1.5 (n = 253), an OS benefit was associated with antiandrogen assignment (HR: 0.61, 95% CI: 0.39–0.94) [1072]. In those receiving early SRT (PSA < 0.6ng/mL, n = 389), there was no improvement in OS (HR: 1.16, 95% CI: 0.79–1.70), with increased other-cause mortality (sub-distribution HR: 1.94, 95% CI: 1.17–3.20, p = 0.01) and increased odds of late grades 3–5 cardiac and neurologic toxic side effects (OR: 3.57, 95% CI: 1.09–15.97, p = 0.05). These results suggest that pre-SRT PSA level may be a prognostic biomarker for outcomes of antiandrogen treatment with SRT.
An SR addressing the benefit from combining HT with SRT suggested risk stratification of patients based on the pre-SRT PSA (< 0.5, 0.6–1, > 1ng/mL), margin status and ISUP GG as a framework to individualise treatment [1073]. In addition, potential risk factors that should be considered are (short) PSA-doubling time and pT3b-4 tumours [957,958,1067-1069].). If PSMA-PET was used for staging, this discussion is restricted to patients with a negative or cN0 PSMA-PET-CT. For patients with cN1 in PSMA-PET-CT see Section 6.4.5.a.2.c.
In conclusion, regarding the ‘weak’ recommendation ‘offer hormonal therapy in addition to SRT to men with BCR’, we have different results of three RCTs for additional short-term ADT (six months) to SRT. One of the RCTs showed an increase of MFS [1004], while the second and third RCTs did not [1002,1068]. Of two RCTs with long-term ADT in addition to SRT, one RCT showed a significant better OS [1001] while the second did not [1068], but this second RCT showed a moderate increase in MFS, at the cost of a higher rate of severe side effects. Additionally, in RADICALS HD, no subgroup analysis of risk factors was carried out. These conclusions were supported by the DADSPORT meta-analysis [1070]. There is a suggestion that absolute benefits of HT vary by prognostic risk and the number of prognostic risk factors in individual patients, especially in an early salvage setting (PSA ≤ 0.5 ng/mlL. The potential for a greater benefit of long- over short-course ADT seems to be small and must be weighed against increased toxicity and reduced QoL.
6.4.5.a.2.a. Target volume, dose, toxicity
Various attempts have been made to define common outlines for ‘clinical target volumes‘ for pN0 PCa [1074,1075] and for organs at risk of normal tissue complications [1074]. However, given the variations of techniques and dose constraints, a satisfactory consensus has not yet been achieved. A benefit in biochemical PFS, but not MFS, has been reported in patients receiving whole-pelvis SRT (± ADT), but the advantages must be weighed against possible side effects [1066]. This is supported by data from the SPPORT Trial (NRG Oncology/RTOG 0534 SPPORT), but it remains controversial [1002].
The optimal SRT dose has not been well defined, but should be at least 64Gy to the prostatic fossa (± the base of the SVs, depending on the pathological stage after RP) [961,1076]. In an SR, the pre-SRT PSA level and SRT dose both correlated with BCR, showing that relapse-free survival decreased by 2.4% per 0.1 ng/mL PSA and improved by 2.6% per Gy, suggesting that the treatment dose above 70Gy should be administered at the lowest possible PSA level [1077]. The combination of pT stage, margin status and ISUP grade group and the PSA at SRT seems to define the risk of biochemical progression, metastasis and overall mortality [949,1078]. In a study of 894 node-negative PCa patients, doses ranging from 64 to > 74 Gy were assigned to twelve risk groups defined by their pre-SRT PSA classes < 0.1, 0.1–0.2, 0.2–0.4, and > 0.4 ng/mL and ISUP grade group < 1 versus 2/3 versus > 4 [1079]. The updated Stephenson nomograms incorporate the SRT and ADT doses as predictive factors for biochemical failure and distant metastasis [1080].
Two RCTs have been published (as shown in Table 6.4.6). Intensity-modulated radiation therapy plus IGRT was used in 57% of the patients in the SAKK trial [1081] and in all patients of a Chinese trial [1082]. No patient had a PSMA PET/CT before randomisation. The primary endpoint in both trials was ‘freedom from biochemical progression’, which was not significantly improved with higher doses. In the Chinese trial, however, a subgroup analysis showed a significant improvement of this endpoint for patients with Gleason 8-10 tumours (66.5% vs. 30.2%, p = 0.012) and for multiple positive surgical margins (82.5% vs. 57.5%, p = 0.037) [1082]. In this trial, patients were treated with ART or SRT and the number of patients was relatively small (n = 144). At this time, it seems difficult to draw final conclusions about the optimal total RT dose and longer follow-up should be awaited, but subgroups of high-risk patients might benefit from higher total doses.
In contrast to definitive RT with moderate and extreme hypofractionated RT as additional standard procedures, normofractionation (single dose 1.8 Gy-2 Gy) remains the standard of care in the SRT-setting. In the randomised NRG-GU-003 phase III (HYPORT) trial 296 patients with a detectable PSA (≥ 0.1 ng/mL) with pT2/3 cN0 disease or an undetectable PSA (< 0.1 ng/mL) with pT3 or pT2 SM+ were randomised to receive 62.5 Gy in 25 fractions (HYPORT) or 66.6 Gy in 37 fractions (COPORT) [784]. The co-primary endpoints were the two-year change baseline scores for the bowel and urinary domains of the EPIC questionnaire. Median follow up was 2.1 years. The mean GI change scores for HYPORT and COPORT were both clinically significant and different in statistical significance at the end of RT (-15.52 and -7.06, p < 0.001) but were resolved at six and twelve months. There was no significant difference for biochemical failure. The trial concluded that HYPORT should be standard practice for patients receiving SRT. In addition, a retrospective study of 161 patients treated with HYPORT (a total dose of 65 Gy compared with 62.5 Gy in the RCT [1083]), with a median follow up of 106 months, reported 2% grade 3 GU-toxicities. However, 44 (27.3%) patients experienced 58 grade 3-5 complications, of which 55 were GU-related. Longer follow-up is required before a new standard of care for SRT can be defined in this setting.
Salvage RT is associated with toxicity. In one report on 464 SRT patients receiving a median 66.6 (maximum 72) Gy, acute grade 2 toxicity was recorded in 4.7% for both the GI and GU tract. Two men had late grade 3 reactions of the GI tract, but overall, severe GU tract toxicity was not observed. Late grade 2 complications occurred in 4.7% (GI tract) and 4.1% (GU tract), respectively, and 4.5% of the patients developed moderate urethral stricture [1084].
In an RCT on dose escalation for SRT (n = 350), acute grade 2 and 3 GU toxicity was observed in 13.0% and 0.6%, respectively, with 64Gy and in 16.6% and 1.7%, respectively, with 70Gy. Gastrointestinal tract grades 2 and 3 toxicity occurred in 16.0% and 0.6%, respectively, with 64Gy, and in 15.4% and 2.3%, respectively, with 70Gy [1085,1086]. Late grade 2 and 3 GI toxicity was significantly increased with higher doses but without significant differences in QoL. In this study, however, the rectal wall dose constraints were rather permissive and in 44% of the patients outdated 3-D techniques were used [1081].
With dose escalation over 72Gy and/or up to a median of 76Gy, the rate of severe side effects, especially GU symptoms, clearly increases, even with newer planning and treatment techniques [1087,1088]. In particular, when compared with 3D-CRT, IMRT was associated with a reduction in grade 2 GI toxicity from 10.2 to 1.9% (p = 0.02), but no effect on the relatively high level of GU toxicity was shown (5-year, 3D-CRT 15.8% vs. IMRT 16.8%) [1087]. However, in a RCT comparing 66Gy and 72Gy with all patients having IMRT plus IGRT (n = 144), no significant differences for GI and GU-toxicity were demonstrated [1089]. After a median salvage IMRT dose of 76Gy, however, the five-year risk of grade 2–3 toxicity rose to 22% for GU and 8% for GI symptoms, respectively [1088]. Doses of at least 64Gy and up to 72Gy in patients without PET/CT can be recommended [1084,1085].
Table 6.4.3: Selected studies of post-prostatectomy salvage radiotherapy, stratified by pre-salvage radiotherapy PSA level* (pTxcN0M0, without PET/CT)
| Study | n | Median FU (mo) | pre-SRT PSA (ng/mL) median | RT dose ADT | bNED/PFS (year) | 5-yr. results |
| Bartkowiak, et al. 2018 [1084] | 464 | 71 | 0.31 | 66.6Gy | 54% (5.9) | 73% vs. 56%; PSA < 0.2 vs. ≥ 0.2ng/mLp < 0.0001 |
| Stish, et al. 2016 [1057] | 1,106 | 107 | 0.6 | 68Gy 16% ADT | 50% (5) 36% (10) | 44% vs. 58%; PSA < 0.5 vs. > 0.5ng/mL |
| Tendulkar, et al. 2016 [1080] | 2,460 | 60 | 0.5 | 66Gy 16% ADT | 56% (5) | Pre-SRT PSA 71% 0.01–0.2ng/mL 63% 0.21–0.5ng/mL 54% 0.51–1.0ng/mL 43% 1.01–2.0ng/mL 37% > 2.0ng/mL p < 0.001 |
| Tilki, et al. 2023 [1062] | 25,551 SRT at: PSA < 0.25 n = 1,556 PSA > 0.25 n = 1,677 No RT: n = 21,645 | 72 | Not given | Med. 68.4Gy SRT+ADT: 1,489 ART: n = 673 ADT: 208 | Not given | ACM (six years): HR 1.49 of higher risk when SRT at start was > 0.25 (p = 0.008) |
*Androgen deprivation therapy can influence the outcome ‘biochemically no evidence of disease (bNED)’ or ‘progression-free survival.’ To facilitate comparisons, 5-year bNED/PFS read-outs from Kaplan-Meier plots are included.
ADT = androgen deprivation therapy; bNED = biochemically no evidence of disease; FU = follow up; mo. = months; n = number of patients; PFS = progression-free survival; PSA = prostate-specific antigen; RT = radiotherapy; SRT = salvage radiotherapy.
Table 6.4.4: Selected studies reporting clinical endpoints after SRT (cTxcN0M0, without PET/CT) (the majority of included patients did not receive ADT)
| Study | n | Median FU (mo) | Regimen | Outcome | |
| Bartkowiak, et al. 2018 [1084] | 464 | 71 | 66.6 (59.4-72) Gy no ADT | 5.9 yr. OS post-SRT PSA < 0.1ng/mL 98% post-SRT PSA ≥ 0.1ng/mL 92% p = 0.005 | |
| Jackson, et al. 2014 [1090] | 448 | 64 | 68.4Gy no ADT | 5 yr. DM post-SRT PSA < 0.1ng/mL 5% post-SRT PSA ≥ 0.1ng/mL 29% p < 0.0001 5 yr. DSM post-SRT PSA < 0.1ng/mL 2% post-SRT PSA ≥ 0.1ng/mL 7% p < 0.0001 | OS post-SRT PSA < 0.1ng/mL 97% post-SRT PSA ≥ 0.1ng/mL 90% p < 0.0001 |
| Stish, et al. 2016 [1057] | 1,106 | 107 | 68 (64.8-70.2) Gy 39% 2D treatment planning incl. 16% ADT | 5 and 8.9 yr. DM SRT: PSA ≤ 0.5ng/mL 7% and 12% SRT: PSA > 0.5ng/mL 14% and 23% p < 0.001 5 and 8.9 yr. DSM SRT: PSA ≤ 0.5ng/mL < 1% and 6% SRT: PSA > 0.5ng/mL 5% and 10% p = 0.02 5 | 8.9 yr. OS SRT: PSA ≤ 0.5ng/mL 94% and 86% SRT: PSA > 0.5ng/mL 91% and 78% p = 0.14 |
| Tendulkar, et al. 2016 [1080] | 2,460 | 60 | 66 (64.8-68.4) Gy incl. 16% ADT | 10-yr. DM (19% all patients) Pre-SRT PSA 9% 0.01–0.2ng/mL; 15% 0.21–0.5ng/mL; 19% 0.51–1.0ng/mL; 20% 1.01–2.0ng/mL; 37% > 2.0ng/mL p < 0.001 | |
ADT = androgen deprivation therapy; DM = distant metastasis; DSM = disease specific mortality; FU = follow up; mo. = months; n = number of patients; OS = overall survival; PSA = prostate specific antigen; SRT = salvage radiotherapy; yr. = years.
Table 6.4.5: Randomised controlled trials comparing salvage radiotherapy combined with androgen deprivation therapy vs. salvage radiotherapy alone
| Study | n | Risk groups | Median FU (mo) | Regimen | Outcome | |
| GETUG-AFU 16 2019 [1004] | 369 SRT + ADT 374 RT | ISUP GG ≤ 2/3 89% SUP GG ≥ 4 11% cN0 | 112 | 66Gy PBRT+ 6 mo. LHRH analogue 66Gy PBRT | 10 yr. PFS: RT + ADT, 64% PFS: RT, 49% p < 0.0001 | MFS: RT + ADT, 75% MFS: RT, 69% p = 0.034 |
RTOG 9601 2017 [1001] | 384 SRT + ADT 376 SRT | pT2 R1, pT3 cN0 | 156 | 64.8Gy PBRT + bicalutamide 24 mo. 64.8Gy PBRT + placebo | 12 yr. cumulative DM RT + ADT: 14% RT + placebo: 23% p = 0.005 OS RT + ADT: 76% RT + placebo: 71% p = 0.04 | DSM RT + ADT: 5.8% RT + placebo: 13.4% p < 0.001 |
| NRG Oncology/RTOG 0534 SPPORT [1002] | 564 SRT 578 SRT + ADT 574 SRT + PBRT + ADT | pT2 or pT3 ISUP GG <5 Pre SRT PSA: 0.1-2.0 | survivors: 8.2 years | 64.8–0.2Gy PBRT 64.8–70.2Gy PBRT 6 mo. LHRH analogue 64.8–70.2Gy PBRT + 45Gy PLNRT 6 mo. LHRH analogue | 5 yr. FFP (primary endpoint) 70.9% Group 1 81.3% Group 2 87.4% Group 3 | Comparisons: G 3 vs. G 1: p < 0.0001 G 2 vs. G 1: p < 0.0001 G 3 vs. G 2: p < 0.0027 |
RADICALS HD 0 vs. 6 mo. ADT [1067] | 737 SRT 747 SRT+ADT | ISUP >7 (11%) ≥ pT3b (17%) R1 (62%) PSA: < 0.3 (61%) ≥ 0.5 (19%) R1 (62%) N1 (3%) | 108 | 52.5Gy, 20fx PBRT (29%) 66Gy, 33fx PBRT (69%) LHRH analogue (83%) | 10 yr. MFS: SRT: 79.2% SRT+ADT: 80.4% p = 0.71; HR: 0.89 CPFS: SRT: 68.3% SRT+ADT: 79.4% p = 0.071, HR:0.54 | Max.GU-Tox G 3: SRT: 16% SRT+ADT: 13% p>0.05 |
RADICALS HD 6 versus 24 months ADT [1068] | 761 6 mo. ADT
762 24 mo. ADT | ISUP > 7 (29%) ≥ pT3b (31%) Med. Pre SRT PSA: 0.23 R1 (63%) N1 (8%) | 107 | 52.5Gy 20fx PBRT (19%) 66Gy, 33fx PBRT (79%) LHRH analogue (84%) | 10 yr. MFS: SRT+6 mo.: 71.9% SRT+24 mo.: 78.1% p = 0.029, HR: 0.77 | Max.GU-Tox G3: SRT+6 mo.: 14% SRT+24 mo.:20% p = 0.025 |
ADT = androgen deprivation therapy; CPFS = clinical progression free survival; DM = distant metastasis; DSM = disease specific mortality; PFS = progression free survival; FFP = Freedom From Progression; FU = follow-up; fx = fractions; LHRH = luteinising hormone-releasing hormone; MFS = metastasis-free survival; OS = overall survival; mo. = months; n = number of patients; yr. = years, PBRT = prostate bed radiotherapy; PFS = progression-free survival; PLNRT = pelvic lymph node radiotherapy; RT = radiotherapy; SRT = salvage ratiotherapy.
Table 6.4.6: Randomised trials investigating dose escalation for SRT without ADT and without PET-CT
| Trial | n | PCa condition | Radiotherapy Dose | Follow-up (median) | Outcome | Results |
| SAKK 09/10 trial, 2021 [961] | 350 | pT2a-3b R0 – R1 pN0 or cN0 PSA post op undetectable (< 0.1ng/mL) or persistent (> 0.1ng/mL < 0.4ng/mL) | 64Gy vs.70Gy
No ADT allowed
VMAT+ IGRT: 57% 3-D planning: 43% | 6.2 yr. | Primary endpoint: FFBP | 6 yr. FFBP: 62% vs. 61% OS: no difference Late side effects: GI grade 2: 7.3% vs. 20% GI grade 3: 4.2% vs. 2.3% p for ≥ grade 2/3: 0.009 |
Phase-III-Trial Qi X, et al., 2024 [1082] | 144 ART: 33% SRT: 67% | pT2-4 R0-R1 pN0 or cN0 Med. PSA pre-RT: 0.2ng/mL | 66Gy vs.72Gy All patients VMAT+ IGRT No ADT allowed High risk (pT3-4, GS: 8-10, PSA >20ng/mL): whole pelvis RT: 126 (87.5%) | 89.5 mo. | Primary endpoint: FFBP | 7 yr. FFBP: 70.3% vs. 61.2% (p > 0.05) High risk (GS: 8–10): 66.5% vs. 30.2% p < 0.012 HR: 0.73 Multiple SR+: 82.5% vs. 57.5% p = 0.037 HR: 0.36 Late side effects: GI + GU grade 2 p > 0.05 No grade 3 |
ADT = androgen deprivation therapy; ART = adjuvant radiotherapy; FFBP = freedom from biochemical failure; GI = gastrointestinal; GU = genitourinary; Gy = Gray; IGRT = image guided radiotherapy; mo. = months; n = number of patients; PSA = prostate-specific antigen; RT = radiotherapy; SRT = salvage radiotherapy; VMAT = volumetric arc radiation therapy; vs. = versus; yr. = years.
6.4.5.a.2.b. Salvage radiotherapy with or without ADT (pTx cN0/1) with PET/CT
In a prospective multicentre study of 323 patients with BCR, radiolabelled PSMA PET/CT changed the management intent in 62% of patients as compared to conventional staging. This was due to a significant reduction in the number of men in whom the site of disease recurrence was unknown (77% vs. 19%, p < 0.001) and a significant increase in the number of men with metastatic disease (11% vs. 57%) [1091]. A prospective study in a subgroup of 119 BCR patients with low PSA (< 0.5ng/mL) reported a change in the intended treatment in 30.2% of patients [1044]. However, no data exist on the impact on final outcome.
Another prospective study in 272 patients with early biochemical recurrent PCa after RP showed that 68Ga-PSMA PET/CT may tailor further therapy decisions (e.g. local vs. systemic treatment) at low PSA values (0.2–1ng/mL) [1092].
A multicentre retrospective study evaluated patients who underwent SRT for BCR after RP, without any signs of distant metastatic disease on PET/CT. After case-control matching, an analysis was made of two cohorts (n = 108 patients each) with and without PSMA PET/CT prior to SRT. In the cohort without PSMA PET/CT, 23 patients (21%) had BCR at one year after SRT versus nine patients (8%) who underwent restaging with PSMA PET/CT prior to SRT (p = 0.007). Radiolabelled PSMA-PET/CT was found to be associated with an improved oncological outcome in patients with BCR after RP receiving SRT to the prostatic fossa [1093]. It is worth mentioning that, in this study, the median biologically effective radiation dose administered in the PSMA-cohort was significantly higher than in the historical cohort (70Gy vs. 66Gy, respectively, p < 0.001). The role of SRT to the prostatic fossa for patients with a negative PSMA-PET-CT for PSA progression after RP was investigated in a retrospective multicentre analysis of 1,222 patients [1094]. Exclusion of patients with pathological LN metastasis, PSA persistence after RP, LN metastasis, nodal irradiation or ADT led to a cohort of 341 patients with local positive PET or negative PET. The total dose to the fossa was significantly higher for patients with local PET positive findings (76.8% > 70 Gy vs. 77.5% < 70 Gy, p <0.001) and median pre SRT-PSA value was comparable (>0.2 and < 0.5 ng/mL) in both groups. With a median follow-up of 28 months the three-year BPFS was 71.6% in PET negative cases and 80.0% in locally PET positive cases (p > 0.05 in multivariate analysis). These data emphasise the importance of early SRT, even in PET-CT negative cases.
A single-centre open-label, phase II/III RCT (EMPIRE-1) evaluated the role of 18F-fluciclovine-PET/CT compared with conventional imaging for SRT. Three hundred and sixty-five patients with detectable PSA after RP but negative results on conventional imaging were randomised to RT directed by conventional imaging alone or to conventional imaging plus PET/CT. Patients with M1 disease in the PET/CT group (n = 4) were excluded. Patients with cN1 were irradiated to the pelvic lymphatics but without a boost to the metastasis. Median follow-up was 3.5 years. In adjusted analyses, the study group was significantly associated with an improvement of the event-free survival (HR: 2.04, 95% CI: 1.06–3.93, p = 0.0327) [1095].
6.4.5.a.2.c. Nodal-directed therapy for rcN1 (with PET/CT)
Radiolabelled PSMA PET/CT is increasingly used as a diagnostic tool to assess metastatic disease burden in patients with BCR following prior definitive therapy. A review including 30 studies and 4,476 patients showed overall estimates of positivity in a restaging setting of 38% in pelvic LNs and 13% in extrapelvic LN metastases [1051]. The percentage positivity of PSMA PET/CT was proven to increase with higher PSA values [1051]. Results of this review demonstrated a high sensitivity and specificity of 68Ga-PSMA in advanced PCa with a per-lesion-analysed sensitivity and specificity of 75% and 99%, respectively.
A large retrospective international study included patients with LN-recurrent PCa (cN1 and M1a) and PSA progression following multimodality treatment (surgery and postoperative RT) [1096]. The aim of the study was to compare SOC with nodal metastasis-directed therapy (MDT). The nodal MDT-group showed significantly better CSS than the SOC control group (five-year survival 98.6% vs. 95.7%, p < 0.01, respectively) [1096].
Another retrospective study compared stereotactic body radiation therapy (SABR) with elective nodal irradiation (ENRT) in nodal oligo-recurrent PCa (n = 506 patients, 365 of which with N1 pelvic recurrence). With a median follow-up of 36 months, ENRT (n = 197) was associated with a significant reduction of nodal recurrences (p < 0.001), compared with SABR (n = 309) of 2% versus 18%, respectively. In a multivariable analysis, patients with one LN at recurrence had longer adjusted MFS after ENRT (HR: 0.50, 95% CI: 0.30–0.85, p = 0.009). The tendency to relapse was higher for pelvic than for extrapelvic nodes (p < 0.001) [1097]. For patients presenting with two or more pelvic or extra-pelvic LNs, adjusted MFS was not significantly different (HR: 0.92, 95% CI: 0.54–1.59, p = 0.8). In these situations, SABR should be used in highly selected patients in prospective cohorts or clinical trials only before any recommendations can be made.
A systematic review and pooled analysis concluded, although heterogeneity was high across all studies, the pooled rates of PSA- image- and systemic progressions were higher in LND compared with RT for BCR patients with PET-positive LNs [1098]. The pooled incidence from 19 publications for PSA-response were 51.1% (LND) and 74.3% (RT) and for PSA-progression 69.8% (LND) and 26.9% (RT).
The phase II randomised PEACE V-STORM trial compared salvage metastases directed therapy (MDT) and elective nodal RT for oligorecurrent nodal PCa in 196 patients [1099]. Patients in the MDT-group had salvage LND or SBRT (30 Gy in three fractions every other day), patients in the ENRT group received 45 Gy in 28 fractions with a simultaneous integrated boost to the PET-positive nodes (65 Gy), both groups had six months of ADT. Fifty-three patients (55%) in the MDT group and 58 patients (62%) in the ENRT group had one positive node, up to five positive nodes were eligible. PSA value at inclusion was 1 (0.53-2.32) in the MDT group and 0.85 (0.42-2.09) in the ENRT group, more than 80% had a PSMA-PET-CT. The primary endpoint was MFS. With a medium follow-up of 50 months the four-year MFS was 76% in the ENRT group compared with 63% in the MDT group (p = 0.063, HR: 0.62). The predominant recurrence pattern after MDT was locoregional (39%) compared with 18% in the ENRT group. However, in the STORM trial all 28 patients with pelvic nodal recurrence in the MDT arm received salvage SBRT, demonstrating that effective salvage treatments after SBRT are possible. The optimal duration of ADT is uncertain and durations > 6 months are likely to be more effective. These data have to be confirmed by a phase III trial.
6.4.5.a.3. Salvage lymph node dissection
Salvage lymph node dissection (SLND) is one form of metastases-directed therapy (MDT) which aims at surgically removing the site of nodal recurrence detected at PET imaging in patients who experience BCR following curative-intent therapies. Among the available phase II randomised trials assessing the role of MDT on oncologic control, only the STOMP study included a small number (n=5) of patients with oligo-recurrent PCa detected by choline PET/CT who were treated with SLND [1100], limiting its applicability to this setting. Therefore, current evidence is based on retrospective series.
An SR synthesised data from 27 series including patients restaged with choline or PSMA PET imaging and reported complete biochemical response after surgery in 13-79% of the cases with five-year BCR-free and OS rates between 6%-31% and 84%, respectively [1101]. Prostate specific antigen value at salvage surgery and number and location of positive nodes at preoperative imaging represented preoperative predictors of biochemical response [1101]. The number of positive nodes at final pathology, pT stage and ISUP GG were postoperative predictors of biochemical control [1101]. An SR including 995 patients, with PET-detected nodal recurrence after RP, treated with SLND reported a pooled incidence of 51% in PSA response with PSA progression rates of 70% during follow-up [1098]. A large multi-centre retrospective study evaluated the long-term outcomes of 189 patients who underwent SLND for oligo-recurrent PCa and reported BCR-free survival of 11% at ten years. Patients with a PSA response after SLND and patients receiving ADT within six months from SNLD had a lower risk of death from PCa [1102]. Most of the patients (81%) received a choline PET and median PSA at SLND was 2.5 ng/mL. The addition of RT to the lymphatic template after salvage LN dissection may improve the BCR rate [1103]. High-level evidence for the oncological value of SLND (including adjuvant RT of the LNs) is still lacking [1101]. This limited evidence should be balanced against the risk of adverse postoperative events. Despite being considered safe, SLND is associated with a non-negligible risk of complications: up to 14% of patients in retrospective series experienced Clavien-Dindo grade 3b complications [1101]. This risk should be discussed during preoperative counselling, and the procedure is best performed in high-volume centres.
Studies including patients restaged with PSMA PET hybrid imaging focused on the role of radioguided surgery (RGS) have been performed. Data suggest that, in patients with node-recurrent PCa treated with SLND, RGS may offer important surgical guidance for surgeons, thus eventually improving oncologic outcomes [1104]. An SR of thirteen series showed that this technique achieved a decline in PSA levels > 90% in 22-100% of cases and BCR-free survival rates of 50-62% at a median follow-up of seventeen months [540]. Higher preoperative PSA, higher number of PSMA-avid lesions, multiple (pelvic plus retroperitoneal), and retroperitoneal localisation of lesions at preoperative imaging were independent predictors of BCR after PSMA-RGS [1105]. Moreover, patients achieving biochemical response of < 0.1 ng/ml were at lower risk of additional therapies at two-year follow-up [1105].
6.4.5.b. Management of PSA failures after radiation therapy
Therapeutic options in these patients are ADT or salvage local procedures, as well as a ‘wait and see’ approach, based on EAU BCR risk categories at relapse. A SR and meta-analysis included studies comparing the efficacy and toxicity of salvage RP, salvage HIFU, salvage cryotherapy, SBRT, salvage LDR BT, and salvage HDR BT in the management of locally recurrent PCa after primary radical EBRT [1106]. The outcomes were BCR-free survival at two and five years. No significant differences with regards to recurrence-free survival (RFS) between these modalities was found. Five-year RFS ranged from 50% after cryotherapy to 60% after HDR BT and SBRT. The authors reported that severe GU toxicity exceeded 21% for whole-gland HIFU and RP, whereas it ranged from 4.2% to 8.1% with reirradiation. Differences in severe GI toxicity also appeared to favour re-irradiation, particularly HDR BT [1106]. Due to the methodological limitations of this review (the majority of the studies included were uncontrolled single-arm case series and there was considerable heterogeneity in the definitions of core outcomes) the available evidence for these treatment options is of low quality and strong recommendations regarding the choice of any of these techniques cannot be made. The following is an overview of the most important findings for each of these techniques.
6.4.5.b.1. Salvage radical prostatectomy
Salvage RP may be a curative-intent option for selected patients with biopsy-confirmed local recurrence after RT without evidence of nodal or distance metastases at restaging (namely, PSMA PET). This procedure is associated with a higher likelihood of AEs compared to primary surgery because of the risk of fibrosis and poor wound healing due to radiation [1107].
6.4.5.b.1.a. Oncological outcomes
An SR evaluated 3,836 patients in 55 studies with median follow-up ranging from 4.6–94 months and showed that SRP provided five-year BCR occurrence 48-59%, cancer-specific survival 13.4-98%, and overall survival 62-100% [1108]. These figures are similar to those reported by Chade et al. in 2011, with five-year and 10-year BCR-free survival estimates ranging from 47–82% and from 28–53%, respectively. The 10-year CSS and OS rates ranged from 70–83% and from 54–89%, respectively. Importantly, most of these findings were based on patient cohorts restaged using conventional imaging at the time of BCR. It may be hypothesised that the adoption of more accurate imaging modalities, such as PSMA/PET, could preoperatively identify patients with nodal or distant metastases who would not be eligible for SRP, thereby improving patient selection and potentially enhancing oncological outcomes. A study evaluating 568 patients with rising PSA levels after primary radiotherapy who underwent PSMA PET/CT found that isolated local recurrence was detected in only 32% of patients meeting the Phoenix criteria for BCR after RT, while distant metastases were present in 49% of these cases [1109]. The pre-SRP PSA value and initial prostate biopsy ISUP grade group were the strongest predictors of the presence of organ-confined disease, progression, and CSS [1110]. In a multicentre analysis including 414 patients, five-year BCR-free survival, CSS and OS were 56.7%, 97.7% and 92.1%, respectively [1111]. Pathological T stage ≥ T3b (OR: 2.348) and GS (up to OR: 7.183 for ISUP GG 4-5) were independent predictors for BCR. Appropriate risk-stratification according to EAU Guidelines Biochemical Recurrence criteria may better select for SRP, with higher metastasis-free (90% vs 76%, p < 0.01) and overall survival (89% vs. 84%, p = 0.01) for low versus high EAU risk [1112,1113].
Lymphadenectomy was performed in most cases (79%), with 20.5% of patients staged N+ at final pathology [1108]. Detailed analysis of a multi-institutional series of 853 SRP patients reported that 87% underwent lymphadenectomy, 21% were pN1 and these patients suffered worse overall and cancer-specific survival [1114]. As in primary surgery, patients with persistent PSA after SRP (42%) had worse BCR-free (6.6 vs. 59%), metastasis-free (71 vs. 88%) and overall survival (77 vs. 94%) after median follow-up of 84 months according to a retrospective, multi-institutional series of 580 patients [1115]. Persistent PSA after SRP was shown to be an independent predictor for BCR and death.
6.4.5.b.1.b. Morbidity
Most reported cases have been open (60%) and robotic-assisted (38%), resulting in an overall complication rate of 34%, with major (Clavien grade ≥ 3) complications occurring across a range of 0- 64% [1108]. Compared to primary open RP, SRP is associated with a higher risk of later anastomotic stricture (47 vs. 5.8%), urinary retention (25.3% vs. 3.5%), urinary fistula (4.1% vs. 0.06%), abscess (3.2% vs. 0.7%) and rectal injury (9.2 vs. 0.6%) [1116]. These complications appear to be less common with robotic compared to open surgery [1107,1110,1117]. An SR including 1,630 patients treated with SRP from 33 series reported intraoperative complication rates of up to 9%, while postoperative complication rates ranged from 0% to 90% [1118].
Functional outcomes are also worse compared to primary surgery, considering urinary incontinence (47.9%, range 21%- 90%) and ED in nearly all patients [1108,1110,1117].
6.4.5.b.1.c. Summary of salvage radical prostatectomy
In general, SRP should be considered only in selected patients with biopsy-proven local recurrence disease after RP with a PSMA PET negative for nodal or distant metastases, low comorbidity burden, a life expectancy of at least ten years, a pre-SRP PSA < 10 ng/mL, initial biopsy ISUP grade group ≤ 2/3, and those whose initial clinical staging was T1 or T2 [1110].
6.4.5.b.2. Salvage cryoablation of the prostate
6.4.5.b.2.a. Oncological outcomes and morbidity
Salvage cryoablation of the prostate (SCAP) has been proposed as an alternative to salvage RP, because it has a potentially lower risk of morbidity and equal efficacy.
In an SR, a total of 32 studies assessed SCAP, recruiting a total of 5,513 patients. The overwhelming majority of patients (93%) received whole-gland SCAP. The adjusted pooled analysis for two-year BCR-free survival for SCAP was 67.49% (95% CI: 61.68–72.81%), and for five-year BCR-free survival was 50.25% (95% CI: 44.10–56.40%). However, the certainty of the evidence was low. Table 6.4.7 summarises the results of a selection of the largest series on SCAP to date in relation to oncological outcomes (BCR only) [1106]. An SR and meta-analysis including 36 studies confirmed an RFS of 67.6%, 59.5% and 47.3% at two, three and five years, respectively [1119]. The authors describe a lower recurrence rate when PSA was < 5ng/mL, the time from primary radiation was > 70 months, when whole gland cryotherapy was performed, and adjuvant ADT was used. Overall, the morbidity was acceptable with 8.5% urinary incontinence, 3% sloughing/stenosis and 1% recto-vesical fistula.
Table 6.4.7: Oncological results of selected salvage cryoablation of the prostate case series, including at least 250 patients
| Study | n | Median FU (mo) | Time point of outcome measurement (yr) | BCR-free probability | Definition of failure |
| Ginsburg, et al. 2017 [1120] | 898 | 19.0 | 5 yr. | 71.3% | Phoenix criteria |
| Spiess, et al. 2010 [1121] | 450 | 40.8 | 3.4 yr. | 39.6% | PSA > 0.5 ng/mL |
| Campbell et al. 2023 [1122] | 419 | 72 | 5 yr. | 74.6% Cyo as primary 78.5% RT as primary | Phoenix criteria |
| Li, et al. 2015 [1123] | 486 | 18.2 | 5 yr. | 63.8% | Phoenix criteria |
| Kovac, et al. 2016 [1124] | 486 | 18.2 | 5 yr. | 75.5% (nadir PSA < 0.4ng/mL); 22.1% (nadir PSA ≥ 0.4ng/mL) | Phoenix criteria |
| Ahmad, et al. 2013 [1125] | 283 | 23.9 | 3 yr. | 67.0% (nadir PSA ≤ 1ng/mL); 14.0% (nadir PSA > 1ng/mL) | Phoenix criteria |
| Pisters, et al. 2008 [1126] | 279 | 21.6 | 5 yr. | 58.9% (ASTRO) 54.5% (Phoenix) | ASTRO and Phoenix criteria |
ASTRO = American Society for Therapeutic Radiology and Oncology; BCR = biochemical recurrence; FU = follow-up; mo. = months; n = number of patients; PSA = prostate-specific antigen; Cyo = cryoablation; RT = radiotherapy; yr. = years.
6.4.5.b.3. Salvage re-irradiation
6.4.5.b.3.a. Salvage brachytherapy for radiotherapy failure
Carefully selected patients with a good PS, primary localised PCa, good urinary function and histologically proven local recurrence are candidates for salvage BT using either HDR or LDR.
In an SR, a total of 16 studies (four prospective) and 32 studies (two prospective) assessed salvage HDR and LDR BT, respectively, with the majority (> 85%) receiving whole-gland BT rather than focal treatment [1106]. The adjusted pooled analysis for two-year BCR-free survival for HDR was 77% (95% CI: 70–83%) and 81% for LDR (95% CI:74–86%). The five-year BCR-free survival for HDR was 60% (95% CI: 52–67%) and for LDR was 56% (95% CI: 48–63%). As noted above, BT techniques are associated with lower rates of severe GU toxicity when compared to RP or HIFU, at 8% for HDR (95% CI: 5.1–11%) and 8.1% for LDR (95% CI: 4.3–13%). Rates of severe GI toxicity are reported to be very low at 0% for HDR (95% CI: 0–0.2%) and 1.5% for LDR (95% CI: 0.2–3.4%). High-dose-rate or LDR BT are effective treatment options with an acceptable toxicity profile. However, the published series are small and likely under-report toxicity. Consequently, this treatment should be offered in experienced centres, ideally within randomised clinical trials or prospective registry studies (see Table 6.4.8).
Table 6.4.8: Treatment-related toxicity and BCR-free probability in selected salvage brachytherapy studies including at least 100 patients.
| Study | Study design | n and BT type | Median FU (mo) | Treatment toxicity | BCR-free probability |
| Lopez, et al. 2019 [1127] | multi-centre retrospective | 75 HDR 44 LDR | 52 | 23.5% late G 3+ GU | 5 yr. 71% (95% CI: 65.9-75.9%) |
| Crook, et al. 2019 [1128] | multi-centre prospective | 100 LDR | 54 | 14% late G 3 combined GI/GU | n.r. |
| Smith, et al. 2020 [1129] | single-centre retrospective | 108 LDR | 76 | 15.7%/2.8% late G 3 GU/GI | 5 yr. 63.1% 10 yr. 52% |
| Lyczek, et al. 2009 [1130] | single-centre retrospective | 115 HDR | n.r. | 12.2%/0.9% late G 3+ GU/GI | 60% at 40 mo. |
BCR = biochemical recurrence; BT = brachytherapy; CI = confidence interval; FU = follow-up; G = grade; GI = gastrointestinal; GU = genitourinary; HDR = high dose rate; LDR = low dose rate; mo. = months; n = number of patients; n.r. = not reported; yr. = years.
6.4.5.b.3.b. Salvage stereotactic ablative body radiotherapy for radiotherapy failure
6.4.5.b.3.b.1.Oncological outcomes and morbidity
Stereotactic ablative body radiotherapy (CyberKnife® or linac-based treatment) is a potentially viable new option to treat local recurrence after RT. Carefully selected patients with good IPSS score, without obstruction, good PS and histologically proven localised local recurrence are potential candidates for SABR. In a meta-analysis and SR five mostly retrospective studies including 206 patients were treated with CyberKnife® or linac-based treatment showing two-year RFS estimates (61.6%, 95% CI: 52.6–69.9%) [1106]. In a retrospective multicentre study (n = 100) the median pre-salvage PSA was 4.3ng/mL with 34% of patients having received ADT for twelve months (median). All recurrences were biopsy proven. Patients were treated with the CyberKnife® with a single dose of 6Gy in six daily fractions (total dose 36Gy). With a median follow-up of 30 months the estimated three-year second BCR-free survival was 55% [1131].
In a smaller retrospective series including 50 men with histologically proven local recurrence with a median pre-salvage PSA of 3.9ng/mL only 15% had received additional ADT. The estimated five-year second BCR-free survival was 60% (median follow-up of 44 months) which is an outcome comparable to series treating patients with RP, HIFU or BT [1132]. Table 6.4.9 summarises the results of the two larger SABR series addressing oncological outcomes and morbidity.
Table 6.4.9: Treatment-related toxicity and BCR-free survival in selected SABR studies
| Study | Study design | n and RT type | Median FU (mo.) | Fractionation (SD/TD) | ADT | Treatment toxicity | BCR-free survival |
| Bergamin, et al. 2020 [1133] | single-centre prospective | 25 LINAC based | 25 | SD 6-6.2 TD 36-38Gy | 0/25 | 2 yr. late G1 GI 8% G2 GU 4% | 2 yr. 80% |
Fuller, et al. 2020 [1132] | single-centre retrospective | 50 Cyber Knife | 44 | SD 6.8Gy TD 34Gy | 7/50 | 5 yr: 8% late G3+ GU | 5 yr. 60% |
Pasquier, et al. 2020 [1131] | multicentre retrospective | 100 Cyber Knife | 30 | SD 6Gy TD 36Gy | 34/100 median 12 mo. | 3 yr. grade 2+ GU 20.8% GI 1% | 3 yr. 55% |
ADT = androgen deprivation therapy; BCR = biochemical recurrence; FU = follow-up; G = grade; GI = gastrointestinal; GU = genitourinary; mo. = months; n = number of patients; RT type = type of radiotherapy; SD = single dose; TD = total dose; yr. = years.
6.4.5.b.3.b.2.Morbidity
In a retrospective single-centre study with 50 consecutive patients, chronic significant toxicity was only seen for the GU domain with five-year grade 2+ and grade 3+ GU rates of 17% and 8%, respectively. No GI toxicity > grade 1 was seen. Of note, of the fifteen patients who were sexually potent presalvage SBRT, twelve subsequently lost potency [1132]. In a retrospective French (GETUG) multicentre series (n = 100) the three-year late grade 2+ GU and GI toxicity was 20.8% (95% CI: 13–29%) and 1% (95% CI: 0.1–5.1%), respectively [1131]. A SR and meta-analysis demonstrated salvage SABR resulted in comparable rates of G3+ GU toxicity when compared to salvage cryotherapy and brachytherapy, but substantially lower rates than salvage HIFU [1134].
6.4.5.b.3.b.3.Summary of salvage stereotactic ablative body radiotherapy
Despite the encouraging results so far, the number of patients treated with SABR is relatively limited. In view of the rates of higher-grade 2+ GU side effects, SABR should only be offered to selected patients, in experienced centres as part of a clinical trial or well-designed prospective study.
6.4.5.b.4. Salvage high-intensity focused ultrasound
6.4.5.b.4.a. Oncological outcomes
Salvage HIFU has emerged as an alternative thermal ablation option for radiation-recurrent PCa. Being relatively newer than SCAP the data for salvage HIFU are even more limited. A SR and meta-analysis included 20 studies (n = 1,783) assessing salvage HIFU [1106], which was also confirmed by another SR and meta-analysis [1134]. The overwhelming majority of patients (86%) received whole-gland salvage HIFU. The adjusted pooled analysis for two-year BCR-free survival for salvage HIFU was 54.14% (95% CI: 47.77–60.38%) and for five-year BCR-free survival 52.72% (95% CI: 42.66–62.56%). However, the certainty of the evidence was low. Table 6.4.10 summarises the results of a selection of the largest series on salvage HIFU to date in relation to oncological outcomes (BCR only).
Table 6.4.10: Oncological results of selected salvage cryoablation of the prostate case series,including at least 250 patients
| Study | n | Median FU (mo.) | Time point of outcome measurement | BCR-free probability | Definition of failure |
Crouzet, et al. 2017 [1135] | 418 | 39.6 | 5 yr. | 49.0% | Phoenix criteria |
Murat, et al. 2009 [1136] | 167 | Mean 18.1 | 3 yr. | 25.0% (high-risk) 53.0% (low-risk)* | Phoenix criteria or positive biopsy or initiation of post-HIFU salvage therapy |
Kanthabalan, et al. 2017 [1137] | 150 | 35.0 | 3 yr. | 48.0% | Phoenix criteria |
Jones, et al. 2018 [1138] | 100 | 12.0 | 1 yr. | 50.0% | Nadir PSA > 0.5 ng/mL or positive biopsy |
**Results stratified by pre-EBRT D’Amico risk groups.
BCR = biochemical recurrence; FU = follow-up; HIFU = high-intensity focused ultrasound; mo. = months; n = number of patients; yr. = years.
6.4.5.b.4.b. Morbidity
The main adverse effects and complications relating to salvage HIFU include urinary incontinence, urinary retention due to bladder outflow obstruction, rectourethral fistula and ED. The SR and meta-analysis showed an adjusted pooled analysis for severe GU toxicity for salvage HIFU of 22.66% (95% CI: 16.98–28.85%) [1106]. The certainty of the evidence was low. Table 6.4.11 summarises the results of a selection of the largest series on salvage HIFU to date in relation to GU outcomes.
Table 6.4.11: Peri-operative morbidity, erectile function and urinary incontinence in selected salvage HIFU case series, including at least 100 patients
| Study | n | Time point of outcome measurement (yr.) | Incontinence* (%) | Obstruction/ retention (%) | Rectourethral fistula (%) | ED (%) |
Crouzet, et al. 2017 [1135] | 418 | Median 39.6 | 42.3 | 18.0 | 2.3 | n.r. |
Murat, et al. 2009 [1136] | 167 | Median 18.1 | 49.5 | 7.8 | 3.0 | n.r. |
| Kanthabalan, et al. 2017 [1137] | 150 | 24 | 12.5 | 8.0 | 2.0 | 41.7 |
Jones, et al. 2018 [1138] | 100 | 12 | 42.0 | 49.0 | 5.0 | 74.0 |
*Incontinence was heterogeneously defined; figures represent use of at least one pad.
ED = erectile dysfunction; n.r. = not reported; n = number of patients; yr. = years.
6.4.5.b.4.c. Summary of salvage high-intensity focused ultrasound
There is a lack of high-certainty data, which prohibits any recommendations regarding the indications for salvage HIFU in routine clinical practice. There is also a risk of significant morbidity associated with its use in the salvage setting. Consequently, salvage HIFU should only be performed in selected patients in experienced centres as part of a clinical trial or well-designed prospective cohort study.
6.4.6. Hormonal therapy for relapsing patients
The aim of hormonal therapy (ADT) in relapsing patients is to improve OS, delay the onset of distant metastases, and maintain QoL. Biochemical response alone to ADT holds no clinical benefit for a patient. An SR including studies published from 2000 onwards [1139], found conflicting results on the clinical effectiveness of ADT after previous curative therapy. Some studies reported a favourable effect of ADT, including the only RCT addressing the research question of this review (86% vs. 79% advantage in OS in the early ADT group) [1140]. Other studies did not find any differences between early versus delayed, or no, ADT. One study found an unfavourable effect of ADT [1141]. Variability appears to be driven by heterogeneous tumour biology with only a minority progressing to metastases or PCa-related death. For older patients and those with comorbidities, the side effects of ADT may even decrease life expectancy. Cardiovascular risk factors in particular must be considered [1142,1143]. The benefit of early ADT seems most evident in high-risk patients, mainly defined by a high ISUP GG and a short PSA-DT (most often less than six months) and a long-life expectancy [1144].
This is supported in a three-arm randomised phase III trial (EMBARK) which evaluated response in patients with prostate cancer who had high-risk biochemical recurrence defined as a PSA-DT of ≤ 9 months and a PSA level of ≥ 2ng/mL above the nadir after radiation therapy or ≥ 1ng/mL after radical prostatectomy with or without postoperative radiation therapy [1145]. Patients were randomly assigned 1:1:1 to receive enzalutamide daily plus leuprolide every 12 weeks (combination group), placebo plus leuprolide (leuprolide-alone group), or enzalutamide monotherapy (monotherapy group). The primary end point was MFS, as assessed by blinded independent central review, in the combination group as compared with the leuprolide-alone group. A key secondary end point was MFS in the monotherapy group as compared with the leuprolide-alone group. Other secondary end points were patient-reported outcomes and safety. A total of 1,068 patients were randomised. After a median follow-up of 60.7 months, the five-year MFS was 87.3% (95% CI, 83.0-90.6) in the combination group, 71.4% (95% CI, 65.7-76.3) in the leuprolide-alone group, and 80.0% (95% CI, 75.0-84.1) in the monotherapy group. The combination of enzalutamide plus leuprolide was superior to leuprolide alone with regards to the MFS (HR 0.42; 95% CI, 0.30-0.61; p < 0.001). Enzalutamide monotherapy also showed a superior MFS compared to leuprolide alone (HR 0.63; 95% CI, 0.46-0.87; p = 0.005). These results led to the FDA approval for enzalutamide alone or in combination with ADT for patients with high-risk biochemical recurrence in November 2023 [1146], followed by EMA approval in April 2024 [1147].
The 2025 update of this study present eight-year OS. In the combination group the OS was 78.9% (95% CI 73.9-83.1) and 69.5% (95% CI 64.0-74.3) in the leuprolide-alone group; the HR for death was 0.60 (95% CI 0.44-0.80; p < 0.001). The eight-year OS with monotherapy was 73.1% (95% CI 67.6-77.9), which did not differ significantly from that with leuprolide alone (HR 0.83; 95% CI 0.63-1.10; p = 0.19). This led to the conclusion that OS was significantly longer with the combination of enzalutamide and leuprolide than with leuprolide alone among patients with PCa with high-risk BCR. Enzalutamide monotherapy was not superior to leuprolide alone in the analysis of OS. These findings support the use of enzalutamide with ADT in high-risk BCR patients with a PSA-DT ≤ 9 months and a PSA above predefined thresholds following local therapy [1148].
A Scandinavian phase-III trial (SPCG-14) [1149] evaluated the effect of docetaxel added to bicalutamide in hormone-naive nonmetastatic PCa with a rising PSA after radical treatment (prostatectomy or radiotherapy, n = 315) or not suitable for curative treatment (n = 3). Between 2009 and 2018, 348 patients were randomised, and median follow up was 4.9 years. As no MFS data are available and the primary endpoint was PFS only, no recommendation for the addition of docetaxel in this setting of PSA recurrence can be made at this time.
6.4.7. Recommendations for second-line therapy after treatment with curative intent
| Recommendations for local salvage treatment | Strength rating |
| Biochemical recurrence (BCR) after radical prostatectomy | |
| Offer early salvage intensity-modulated radiotherapy/volumetric arc radiation therapy plus image-guided radiotherapy to men with two consecutive prostate-specific antigen (PSA) rises. | Strong |
| Offer monitoring, including PSA, to EAU low-risk BCR patients. | Weak |
| Do not wait for a PSA threshold before starting treatment. Once the decision for salvage radiotherapy (SRT) has been made, SRT (at least 64 Gy) should be given as soon as possible. | Strong |
| Offer hormonal therapy in addition to SRT to men with BCR. | Weak |
| Follow-up after radical prostatectomy or radiotherapy | |
| Routinely follow-up asymptomatic patients by obtaining at least a disease-specific history and serum PSA measurement. | Strong |
| At recurrence, only perform imaging if the result will affect treatment planning. | Strong |
| BCR after radiotherapy | |
| Offer monitoring, including PSA, to EAU Low-Risk BCR patients. | Weak |
| Offer highly selected patients with biopsy-proven local recurrence salvage radical prostatectomy, brachytherapy, stereotactic body radiotherapy in experienced centres. | Strong |
| Offer highly selected patients with biopsy-proven local recurrence high-intensity focused ultrasound, or cryosurgical ablation within a clinical trial setting or well-designed prospective cohort study. | Weak |
| Systemic salvage treatment | |
| Do not offer androgen deprivation therapy (ADT) to M0 patients with a PSA-doubling time > 12 months. | Strong |
| Offer enzalutamide with ADT to EMBARK-like patients (M0 patients on conventional imaging and PSA doubling time of ≤ 9 months). | Strong |
6.5. Systemic treatments for prostate cancer
6.5.1. Hormonal therapy
Androgen deprivation can be achieved by suppressing the secretion of testicular androgens in various ways.
6.5.1.a. Castration level
The castration level of testosterone is < 50ng/dL (1.7 nmol/L), defined more than 40 years ago when testosterone testing was less sensitive. Current methods have shown that the mean value after surgical castration is 15ng/dL [1035]. Therefore, a preferred target level should be defined as < 20ng/dL (< 0.7 nmol/L). This definition is important because better results are repeatedly observed in ADT monotherapy cohorts with lower testosterone levels compared to 50ng/dL [1036-1038]. However, the castrate level considered by the regulatory authorities and in clinical trials addressing castration in PCa is still the historical < 50ng/dL (1.7 nmol/L).
6.5.1.b. Bilateral orchiectomy
Bilateral orchiectomy or subcapsular pulpectomy is still considered the primary treatment modality for ADT. It is a simple, cheap and low-complication procedure. It is easily performed under local anaesthesia and is the quickest way to achieve a castration level that is usually reached within less than twelve hours. It is irreversible and therefore does not allow for intermittent treatment [1039].
6.5.1.c. Luteinising-hormone-releasing hormone agonists
Long-acting LHRH agonists are currently the main stay of ADT. These synthetic analogues of LHRH are administered as depot injections on a monthly, three-monthly, six-monthly or yearly schedule. The first injection induces a transient rise in luteinising hormone (LH) and follicle-stimulating hormone (FSH), leading to the ‘testosterone surge’ or ‘flare-up’ phenomenon, which starts two to three days after administration and lasts for approximately one week. This transient hormonal rise has historically been associated with potentially detrimental clinical effects (the clinical flare), such as increased bone pain, acute bladder outlet obstruction, obstructive renal failure, spinal cord compression and cardiovascular death due to hypercoagulation status [1150]. Patients at risk are typically those with high-volume symptomatic bone metastases. Concomitant therapy with an anti-androgen decreases the incidence of clinical flare but does not completely remove the risk. Anti-androgen therapy is usually continued for four weeks but neither the timing nor the duration of anti-androgen therapy are based on strong evidence. Moreover, the long-term benefit of flare prevention remains unclear [1151]. Notably, more recent analyses question the clinical relevance of this testosterone flare in most patients, suggesting limited evidence for significant PSA increases, disease progression, or complications directly attributable to the flare. The need for anti-androgens at initiation may therefore be limited to selected high-risk cases [1151,1152].
Chronic exposure to LHRH agonists results in the downregulation of LHRH receptors, suppressing LH and FSH secretion and therefore testosterone production. A castration level is usually obtained within two to four weeks [1153]. Although there is no formal direct comparison between the various compounds, they are considered to be equally effective [1154]. No survival difference between LHRH agonists and orchiectomy has been reported, due to the lack of high-quality comparative trials [1155]. The various products have practical differences that must be considered in everyday practice, including the storage temperature, whether a drug is ready for immediate use or requires reconstitution, and whether a drug is given by subcutaneous or intramuscular injection.
6.5.1.d. Luteinising-hormone-releasing hormone antagonists
Luteinising-hormone-releasing hormone antagonists immediately bind to LHRH receptors, leading to a rapid decrease in LH, FSH and testosterone levels without any flare. The practical shortcoming of these compounds as injections is the lack of a long-acting depot formulation with, so far, only monthly formulations being available. Degarelix is a LHRH antagonist. The standard dosage is 240 mg in the first month, followed by monthly injections of 80 mg. Most patients achieve a castrate level at day three [1153]. A phase III RCT compared degarelix to monthly leuprorelin following up patients for twelve months, suggesting a better PSA PFS for degarelix 240/80 mg compared to monthly leuprorelin [1156]. An SR did not show a major difference between agonists and degarelix and highlighted the paucity of on-treatment data beyond twelve months, as well as the lack of survival data [1157]. Its definitive superiority over the LHRH analogues remains to be proven. Short-term follow-up data from a meta-analysis indicate that the use of LHRH antagonist is associated with significantly lower overall mortality and cardiovascular events as compared with agonists. On the other hand, other adverse effects, such as decreased libido, hot flushes, ED, weight gain and injection site reactions, are seen less often with the agonists [1158,1159].
Relugolix is an oral LHRH antagonist. It was compared to the LHRH agonist leuprolide in a randomised phase III trial [1160]. The primary endpoint was sustained testosterone suppression to castrate levels through 48 weeks. There was a significant difference of 7.9 percentage points (95% CI: 4.1–11.8) showing noninferiority and superiority of relugolix. The incidence of major adverse cardiovascular events was significantly lower with relugolix (prespecified safety analysis). Relugolix has been approved by the FDA [1161] and EMA [1162] for hormone sensitive PCa.
6.5.1.e. Anti-androgens
These oral compounds are classified according to their chemical structure as:
- steroidal, e.g. cyproterone acetate (CPA), megestrol acetate and medroxyprogesterone acetate; and
- non-steroidal or pure, e.g. nilutamide, flutamide and bicalutamide.
Both classes compete with androgens at the receptor level. This leads to an unchanged or slightly elevated testosterone level. Conversely, steroidal anti-androgens have progestational properties leading to central inhibition by crossing the blood-brain barrier.
6.5.1.e.1. Steroidal anti-androgens
These compounds are synthetic derivatives of hydroxyprogesterone. Their main pharmacological side effects are secondary to castration (gynaecomastia is quite rare) whilst the non-pharmacological side effects are cardiovascular toxicity (4–40% for CPA) and hepatotoxicity.
Cyproterone acetate was the first licensed anti-androgen but the least studied. Its most effective dose as monotherapy is still unknown. It appears to be associated with a poorer OS when compared with LHRH analogues and there is no benefit when compared with flutamide [1163,1164].
6.5.1.e.2. Non-steroidal anti-androgens
Non-steroidal anti-androgen monotherapy with e.g. nilutamide, flutamide or bicalutamide does not suppress testosterone secretion and it is claimed that libido, overall physical performance and bone mineral density (BMD) are frequently preserved [1165]. Non-androgen-related pharmacological side effects differ between agents. Bicalutamide shows a more favourable safety and tolerability profile than flutamide and nilutamide [1166]. The dosage licensed for use in combination with LHRH blockade is 50mg/day, and 150mg/day for monotherapy. The androgen pharmacological side effects are mainly gynaecomastia (70%) and breast pain (68%). However, non-steroidal anti-androgen monotherapy offers clear bone protection compared with LHRH analogues and probably LHRH antagonists [1165,1167]. All three agents share the potential for liver toxicity (occasionally fatal), requiring regular monitoring of patients’ liver enzymes.
6.5.1.e.3. New androgen receptor pathway inhibitors (ARPIs)
Once on ADT, the development of castration-resistance (CRPC) is only a matter of time. It is considered to be mediated through two main overlapping mechanisms: androgen-receptor (AR)-independent and AR-dependent mechanisms. In CRPC, the intracellular androgen level is increased compared to androgen-sensitive cells and an overexpression of the AR has been observed, suggesting an adaptive mechanism [1168]. This has led to the development of several compounds targeting the androgen axis. Table 6.5.1 summarises the status of the various ARPIs [1169-1174]. For the updated approval status, see the EMA and FDA websites [1146,1175-1178].
Table 6.5.1: Status of the different ARPIs
High-risk localised & locally advanced** | High-risk BCR | mHSPC | nmCRPC | mCRPC | |
| Abiraterone | X* | X | X | ||
| Enzalutamide | X | X | X | X | |
| Apalutamide | X | X | |||
| Darolutamide | X | X |
* Unlicenced indication;
** STAMPEDE definition
6.5.1.e.3.a. Abiraterone acetate
Abiraterone acetate is a CYP17 inhibitor (a combination of 17α-hydrolase and 17,20-lyase inhibition). By blocking CYP17, abiraterone acetate significantly decreases the intracellular testosterone level by suppressing its synthesis at the adrenal level and inside the cancer cells (intracrine mechanism). This compound must be used together with prednisone/prednisolone to prevent drug-induced hyperaldosteronism [1175,1177].
6.5.1.e.3.b. Apalutamide, darolutamide, enzalutamide and rezvilutamide
These agents are novel non-steroidal antiandrogens with a higher affinity for the AR receptor than traditional non-steroidal antiandrogens. In addition, while previous non-steroidal antiandrogens still allow transfer of ARs to the nucleus and would act as partial agonists, all four agents also block AR transfer and therefore suppress any possible agonist-like activity [1169,1170,1178,1179]. Darolutamide has structurally unique properties. In particular, in preclinical studies darolutamide was shown not to cross the blood-brain barrier [1180,1181].
6.5.2. Cytotoxic drug treatment
6.5.2.a. Taxanes
Paclitaxel derivatives promote the assembly of microtubules and inhibit the subsequent depolymerisation, impairing the tubulin dynamics that foster the mitotic spindle assembly during interphase in mitosis [1182]. Docetaxel binds ß-tubulin dimers in a 1:1 stoichiometric ratio, exhibiting a stronger dynamic instability using its inhibitory effect in tubulin depolymerisation [1183]. It also activates NF-kB causing apoptosis via a mitochondria-dependent pathway [1184]. Docetaxel shows significant activity against prostate tumours. Cabazitaxel also works by binding to the microtubules. This prevents cellular mitosis and stabilises the tumour cells. As a result, the cells do not divide. In addition, it inhibits androgen receptors by binding to the microtubules and microtubule-associated motor protein dynein. As a consequence, androgen receptor nuclear translocation is prevented [1182]. Common side effects include peripheral neuropathy, myalgias, neutropenia and arthralgia.
6.5.3. Nonhormonal noncytotoxic drug treatments
6.5.3.a. Poly (ADP-ribose) polymerase inhibitors (PARPi)
Poly (ADP-ribose) polymerase inhibitors (PARPi) block the enzyme poly ADP-ribose polymerase (PARP) and were developed aiming to selectively target cancer cells harbouring BRCA mutations and other mutations inducing homologous recombination deficiency and a high level of replication pressure with a sensitivity to PARPi treatment. Due to the oncogenic loss of some DNA repair effectors and incomplete DNA repair repertoire, some cancer cells are addicted to certain DNA repair pathways such as Poly (ADP-ribose) polymerase (PARP)-related single-strand break repair pathway. The interaction between BRCA and PARP is a form of synthetic lethal effect, which means the simultaneously functional loss of two genes leads to cell death, while a defect in any single gene only has a limited effect on cell viability [1185]. BRCA mutations predispose patients to the development of PCa whilst also making tumours more responsive to PARPi. Therefore, BRCA status should be assessed in patients with metastatic PCa for optimal treatment selection.
6.5.3.b. Immune checkpoint inhibitors
Checkpoint inhibitors target the molecules CTLA4, programmed cell death protein 1 (PD-1), and programmed death-ligand 1 (PD-L1). For advanced PCa patients who are microsatellite instability-high/deficient mismatch repair (MSI-H/dMMR), the PD-1 inhibitor pembrolizumab has been approved by the FDA but not by the EMA. The label is tumour agnostic [1186,1187].
6.5.3.c. Radiopharmaceutical therapy
Radiopharmaceutical therapy (RPT) is based on the delivery of radioactive isotopes to tumour-associated targets. The mechanism of action for RPT is radiation-induced killing of cells. Radionuclides with different emission properties are used to deliver radiation. The most commonly used radionuclides are represented by β-particles (e.g. 177Lu) or α-particles (e.g. 223Ra, 225Ac). 223Ra based on its biochemical similarity to Calcium, is integrated in bones with increased osteoblastic activity, thus targeting skeletal PCa metastases. 177Lu is increasingly used because of its optimal imaging range (100–200keV), favourable half time (6.6 days), and appropriate β-particle energy for therapy. The short path of the β-particles (0.05–0.08 mm) results in minimal toxic effects in adjacent healthy tissue. These properties enable such radionuclides to be used as theranostics (i.e. the same radionuclide may be used for both diagnostic and therapeutic purposes). However, an essential requirement prior to any RPT is to assess the targeting of the agent, mainly using PET techniques which show the tumour expression and the extent of cancer [1188]. The FDA has approved 177Lu for the treatment of adult patients with PSMA-positive mCRPC who have been treated with ARPI and taxane-based chemotherapy [1189,1190].
6.5.3.d. AKT inhibitors
AKT inhibitors are orally administered ATP-competitive molecules that target the three AKT isoforms. AKT propagates signals from the PI3K/AKT/mTOR pathway which is central for promoting cell survival, proliferation, metabolism, and growth. AKT inhibitors interrupt key growth and survival signals in cancer cells by locking AKT into an inactive state and preventing downstream phosphorylation.
6.6. Management of metastatic prostate cancer
6.6.1. Introduction
Most prospective data available rely on the definition of M1 disease based on CT scan or MRI and bone scintigraphy. Modern, more accurate imaging is increasingly being applied in clinical practice; however, its influence on treatment and patient outcomes has not yet been validated in prospective RCTs.
6.6.2. Molecular testing
Different techniques may be applied for molecular testing including immunohistochemistry (IHC), fluorescence in situ hybridization (FISH), and next generation sequencing (NGS) of RNA or DNA. Treatment should be based on predictive [1191] and not prognostic genetic aberrations. Patients potentially eligible for treatment intensification should be offered somatic DNA sequencing using panel-based assays [1191]. Assay failure rates are lower on more recently collected primary tumour tissue or on tissue from metastases. Although useful information has been extracted from tissue up to five to ten years old [1192]. Genetic testing on circulating tumour DNA (ctDNA) is an alternative option and has been used in some trials. The FoundationOne® Liquid CDx, has been FDA approved [1220]. Certified (accredited) institutions should be used for NGS multiplication procedure (minimum depth of coverage of 200 X). A critical asset is the decision support helping to rate the mutations according to their clinical relevance [1193,1194]. Defective MMR assessment can be performed by IHC for MMR proteins (MSH2, MSH6, MLH1 and PMS2) and/or by next generation sequencing (NGS) assays [1195]. For patients with metastatic disease and assay failure germline testing is recommended to identify BRCA 1/2 alterations. Germline molecular testing is discussed in Section 5.1.6 and recommendations for germline testing are provided in Section 5.2.8. Level 1 evidence for the use of PARP inhibitors has been reported [341,1196-1207]. Microsatellite instability (MSI) high (or MMR deficiency) is rare in PCa, but for those patients, pembrolizumab has been approved by the FDA and could be a valuable additional treatment option [1187,1208].
6.6.3. Prognostic and predictive factors
Median survival of patients with newly diagnosed metastases (synchronous mHSPC) is approximately 50 months with ADT alone, however, it is highly variable since the M1 population is heterogeneous [1209]. Several prognostic factors for survival have been suggested, including the number and location of bone metastases, presence of visceral metastases, ISUP GG, performance status and initial PSA and alkaline phosphatase level, but only few have been validated [1210-1213].
‘Volume’ of disease as a potential predictor was introduced by CHAARTED (Chemohormonal Therapy versus Androgen Ablation Randomised Trial for Extensive Disease in Prostate Cancer) [1213-1215] (Table 6.6.1) and subsequently, in STAMPEDE, was shown to be predictive in an adequately powered subgroup analysis for benefit of addition of prostate RT to ADT in the subgroup of patients with low volume/burden disease [1216] (Table 6.6.1).
‘Metachronous’ metastatic disease (after radical local treatment of the primary tumour) vs. synchronous (or de novo) metastatic disease has also been shown to have generally a better prognosis [1217].
Based on a large SWOG 9346 cohort, the PSA level after seven months of ADT was used to create three prognostic groups (Table 6.6.2) [1218]. A PSA ≤ 0.2ng/mL at seven months has been confirmed as a prognostic marker for men receiving ADT for metastatic disease in the CHAARTED study independent of the addition of docetaxel [1219]. Similarly, reaching PSA levels of ≤ 0.1ng/ml after six months were shown to be correlated with long-term outcomes in the LATITUDE study [1220]. Also for patients treated with ADT and apalutamide, a deep PSA decline defined by ≥ 90% from baseline or to PSA ≤ 0.2ng/mL at a landmark of three months was associated with longer OS [1221] for patients. An SR and meta-analysis confirmed that PSA response following initiation of ADT plus ARPI is strongly associated with OS across all stages of advanced PCa, including mHSPC, nmCRPC, and mCRPC. Achieving undetectable PSA levels or PSA decline of ≥ 90% was consistently associated with prolonged OS, underscoring PSA response as a robust early prognostic biomarker, although its role as a surrogate endpoint for OS remains to be validated [1222].
Table 6.6.1: Definition of high and low volume in CHAARTED and high and low risk in LATITUDE
| High | Low | |
| CHAARTED (volume) | ≥ 4 bone metastases including ≥ 1 outside vertebral column or pelvis AND/OR Visceral metastasis* | Not high |
LATITUDE (risk) | > 2 high-risk features of:
| Not high |
*Lymph nodes are not considered as visceral metastases.
Table 6.6.2: Prognostic factors based on the SWOG 9346 study
| PSA after 7 months after start of ADT | Median survival on ADT monotherapy |
| < 0.2ng/mL | 75 months |
| 0.2 ≤ 4ng/mL | 44 months |
| > 4ng/mL | 13 months |
6.6.4. First-line hormonal treatment
Primary ADT has been the standard of care for over 50 years [1223]. No high-level evidence is available in favour of a specific type of ADT for oncological outcomes - neither for orchiectomy, nor for a LHRH agonist or antagonist. The level of testosterone is reduced much faster with orchiectomy and LHRH antagonist; therefore, they are recommended in patients with impending spinal cord compression or other potential impending complications.
Cardiovascular side effects may be less frequent in patients treated with LHRH antagonists than patients treated with LHRH agonists [1160,1224-1226]. Therefore, LHRH antagonists may be preferred in patients with pre-existing cardiovascular disease or other cardiovascular risk factors.
6.6.4.a. Non-steroidal anti-androgen monotherapy
Older generation non-steroidal anti-androgen (NSAA) monotherapy is inferior to ADT (either medical or surgical) in terms of OS, clinical progression, treatment failure and treatment discontinuation due to AEs [1227] and is generally not recommended. For combination treatment of mHSPC ADT is the standard of care.
6.6.4.b. Intermittent versus continuous androgen deprivation therapy
Three independent reviews [1228-1230] and two meta-analyses [1231,1232] looked at the clinical efficacy of intermittent androgen deprivation (IAD) therapy. All these reviews included eight RCTs, of which only three were conducted in patients with exclusively M1 disease.
So far, the SWOG 9346 is the largest trial addressing IAD in M1b patients [1233]. Of 3,040 screened patients, only 1,535 patients met the inclusion criteria. This highlights the fact that only about 50% of M1b patients can be expected to be candidates for IAD, i.e. the best PSA responders. This was a noninferiority trial leading to inconclusive results: the actual upper limit was above the prespecified 90% upper limit of 1.2 (HR: 1.1, CI: 0.99–1.23), the prespecified noninferiority limit was not achieved, and the results did not show a significant inferiority for any treatment arm. However, based on this study, inferior survival with IAD cannot be completely ruled out, even in this highly selected subgroup. The use of intermittent ADT has been superseded as continuous ADT based combination therapy has become SOC.
6.6.4.c. Early versus deferred androgen deprivation therapy
Early treatment before the onset of symptoms is recommended in the majority of patients with metastatic hormone-sensitive disease. A Cochrane analysis from 2019 regarding the topic concluded that early ADT probably extends time to death of any cause and time to death from PCa [1234], but the analysis included only a very limited number of metastatic patients. Randomised phase III data is lacking in this specific setting and specifically not with the combination therapies that are standard nowadays, however, data is accumulating for the use of long-term ADT earlier in the disease pathway.
Delaying ADT, often using highly sensitive imaging techniques like PSMA-PET/CT or whole bode MRI, in order to offer RT/SABR for men with oligometastatic PCa is the focus of multiple studies (see Section 6.6.7) [1235].
6.6.5. Combination therapies
All of the following combination therapies have been studied with continuous ADT, not intermittent ADT.
6.6.5.a.‘Combined’ androgen blockade with older generation NSAA (bicalutamide, flutamide, nilutamide)
Systematic reviews have shown that combined androgen blockade using a NSAA appears to provide a small survival advantage (< 5%) versus monotherapy (surgical castration or LHRH agonists) [1236,1237]. This minimal survival advantage must be balanced against the increased side effects, especially because the newer combination therapies are more effective as shown specifically for enzalutamide, which was tested against NSAA in phase III trials [1238,1239].
6.6.5.b. Androgen deprivation combined with other agents
6.6.5.b.1. Combination with an ARPI alone (abiraterone, apalutamide, enzalutamide, rezvilutamide, darolutamide)
Two large RCTs (STAMPEDE, LATITUDE), assessed the addition of abiraterone acetate (1000 mg daily) plus prednisone (5 mg daily) to ADT in men with mHSPC [1173,1240,1241] (Table 6.6.3). The primary objective of both trials was an improvement in OS. Both trials showed a significant OS benefit. In LATITUDE with only de novo high-risk metastatic patients included, the HR reached 0.62 (0.51–0.76) [1173]. The HR in STAMPEDE was very similar, with 0.63 (0.52–0.76) in the total patient population (metastatic and nonmetastatic) and an HR of 0.61 in the subgroup of metastatic patients [1240]. While only high-risk patients were included in the LATITUDE trial, a post-hoc analysis from STAMPEDE showed the same benefit whatever the risk or the volume category was [1242].
All secondary objectives, such as PFS, time to radiographic progression, time to pain, or time to chemotherapy, were in favour of the combination. No difference in treatment-related deaths was observed with the combination of ADT plus AAP compared to ADT monotherapy (HR: 1.37 [0.82–2.29]). However, twice as many patients discontinued treatment due to toxicity in the combination arms in STAMPEDE (20%) compared to LATITUDE (12%) [1241]. Based on these data upfront AAP combined with ADT has become a standard option in men presenting de novo metastatic PCa.
In five large RCTs, the addition of AR antagonists to ADT in men with mHSPC was tested [1171,1172,1238]. In ARCHES, the primary endpoint was rPFS which was significantly improved for the combination of enzalutamide and ADT (HR 0.39; 95% CI 0.3–0.5). Approximately 36% of the patients had low-volume disease: approximately 25% had prior local therapy and 18% of the patients had received prior docetaxel. In the final prespecified analysis, the key secondary endpoint OS was significantly improved (HR 0.66; 95% CI 0.53-0.81) and a significant benefit for rPFS was maintained (HR 0.63; 95% CI 0.52–0.76) [1243].
In ENZAMET, the primary endpoint was OS. The addition of enzalutamide to ADT in the first analysis improved (HR 0.67; 95% CI 0.52–0.86) compared to ADT plus a non-steroidal antiandrogen. Approximately half of the patients had concomitant docetaxel: about 40% had prior local therapy and about half of the patients had low-volume disease [1172]. In a planned later analysis with a median follow-up of 68 months, the OS benefit of adding enzalutamide was maintained (HR 0.7 95% CI 0.58-0.84) (Table 6.6.4) [1244].
In the TITAN trial, ADT plus apalutamide was used and rPFS and OS were co-primary endpoints. In the primary analysis, rPFS was significantly improved through the addition of apalutamide (HR 0.48; 95% CI 0.39–0.6). OS at 24 months was improved for the combination (HR 0.67; 95% CI 0.51–0.89). In the final analysis, the HR for OS was 0.65 (0.53–0.79) without adjustment for crossover. In this trial, 16% of patients had prior local therapy, 37% had low-volume disease, and 11% received prior docetaxel [1171,1245] (Table 6.6.4). A secondary analysis of the Titan study found that nearly half of the patients developing subsequent radiographic progression had no concomitant PSA progression, suggesting that heavy reliance on PSA monitoring may be inadequate for assessing disease activity in this context [1246].
In the CHART trial, ADT plus rezvilutamide was evaluated versus ADT plus bicalutamide in patients with high-volume de novo metastatic disease. Ninety percent of the patients were recruited in China. Overall survival and rPFS were co-primary endpoints. At the preplanned interim analysis, rezvilutamide significantly improved rPFS compared with bicalutamide (HR 0.44; 95% CI 0.33–0.58) and OS (HR 0.58; 95% CI 0.44–0.77) (Table 6.6.5) [1239]. Patient reported outcomes as assessed by FACT-P and BPI-SF were superior in men receiving rezvilutamide compared to bicalutamide, delaying both deterioration of pain and the FACT-P functional status [1247].
In ARANOTE, darolutamide plus ADT was randomised 2:1 versus placebo plus ADT. It proved to significantly improve rPFS, which was the primary endpoint (HR 0.54; 95% CI 0.41-0.71], with consistent benefits across subgroups, including high- and low-volume disease [1174]. Adverse events were similar in the two groups. Overall survival was not statistically different in the final analysis (HR 0.81; 95% CI 0.59-1.12) (Table 6.6.5) [1174].
In summary, the addition of the new AR antagonists significantly improves clinical outcomes with no convincing evidence of differences between subgroups. The majority of patients had de novo metastatic disease. The effect in the subgroup analyses seemed to be consistent and therefore, a combination should also be offered for men progressing after radical local therapy [1244,1248,1249].
Table 6.6.3: Results from the STAMPEDE arm G and LATITUDE studies
| STAMPEDE [1240] | LATITUDE [1173] | |||
| ADT | ADT + AA + P | ADT + placebo | ADT + AA + P | |
| N | 957 | 960 | 597 | 602 |
| Newly diagnosed N+ | 20% | 19% | 0 | 0 |
| Newly diagnosed M+ | 50% | 48% | 100% | 100% |
| Key inclusion criteria | Patients scheduled for long-term ADT • newly diagnosed M1 or N+ situations • locally advanced (at least two of cT3 cT4, ISUP grade ≥ 4, PSA ≥ 40ng/mL) • relapsing locally treated disease with a PSA > 4ng/mL and a PSA-DT < 6 mo.
or PSA > 20ng/mL or nodal or metastatic relapse | Newly diagnosed M1 disease and 2 out of the 3 risk factors: ISUP GG ≥ 4, ≥ 3 bone lesions, measurable visceral metastasis | ||
| Primary objective | OS | OS; rPFS | ||
| Median follow-up | 40 mo. | 30.4 mo. | ||
| 3-yr. OS | 83% (ADT + AA + P) 76% (ADT) | 66% (ADT + AA + P) 49% (ADT + placebo) | ||
| HR (95% CI) | 0.63 (0.52-0.76) | 0.62 (0.51-0.76) | ||
| M1 only | ||||
| N | 1,002 | 1,199 | ||
| 3-yr. OS | NA | 66% (ADT + AA + P) 49% (ADT + placebo) | ||
| HR (95% CI) | 0.61 (0.49-0.75) | 0.62 (0.51-0.76) | ||
| HR | FFS (biological, radiological, clinical or death): 0.29 (0.25-0.34) | rPFS: 0.49 (0.39-0.53) | ||
AA = abiraterone acetate; ADT = androgen deprivation therapy; CI = confidence interval; FFS = failure-free survival; HR = hazard ratio; ISUP = International Society of Urological Pathology; mo. = months; N = number of patients; NA = not available; OS = overall survival; P = prednisone; PSA = prostate-specific antigen; PSA-DT = prostate-specific antigen doubling time; rPFS = radiographic progression-free survival.
Table 6.6.4: Results from the ENZAMET and TITAN studies with OS as primary endpoint
| ENZAMET [1238,1244] | TITAN [1171,1245] | |||
ADT+ older antagonist ± docetaxel (SOC) | ADT + enzalutamide ± docetaxel | ADT + placebo | ADT + apalutamide | |
| N | 562 | 563 | 527 | 525 |
| Newly diagnosed M+ | 72.1% | 72.5% | 83.7% | 78.3% |
| Low volume | 47% | 48% | 36% | 38% |
| Primary objective | OS | OS; rPFS | ||
| Median follow up | 68 mo. | 30.4 mo. | ||
| OS | 5-year survival: 67% (ADT + enzalutamide) 57% (SOC) | 2-yr survival: 84% (ADT + apalutamide) 74% (ADT + placebo) | ||
| HR (95% CI) for OS | 0.70 (0.58-0.84) | 0.67 (0.51-0.89) | ||
ADT = androgen deprivation therapy; CI = confidence interval; HR = hazard ratio; mo. = months; N = number of patients; PSA-DT = prostate-specific antigen doubling time; OS = overall survival; rPFS = radiographic progression-free survival; SOC = standard of care; yr. = years.
Table 6.6.5: Results from the ARCHES, CHART and ARANOTE studies
| ARCHES [1172,1243] | CHART [1239] | ARANOTE [1174] | ||||
| ADT ± docetaxel | ADT + enzalutamide ± docetaxel | ADT + bicalutamide | ADT + rezvilutamide | ADT+ darolutamide | ADT + placebo | |
| N | 576 | 574 | 328 | 326 | 448 | 223 |
| Newly diagnosed M+ | 63% | 70% | 100% | 100% | 71.1% | 75.3% |
| Low volume | 35% | 38% | 0% | 0% | 29.4% | 29.6% |
| Use of early docetaxel | 18% (previous) | 18% (previous) | 0% | 0% | 0 | 0 |
| Primary endpoint(s) | rPFS | OS; rPFS | rPFS | |||
| Median follow-up | 44.6 mo. | 29.3 mo. | 25.3 mo. | 25.0 mo. | ||
| Median rPFS | 38.9 mo. | 49.8 mo. | 23.5 mo. | Not reached | Not reached | 25.0 mo. |
| HR (95% CI) for rPFS | 0.63 (0.52-0.76) | HR: 0.46 (0.36-0.60) | 0.54 (0.41-0.71) | |||
| Median OS | Not reached | Not reached | Not reached | Not reached | Not reached | |
| HR (95% CI) for OS | 0.66 (0.53-0.81): Main secondary endpoint | 0.58 (0.44-0.77) | 0.78 (0.58 – 1.05) | |||
ADT = androgen deprivation therapy; HR = hazard ratio; mo. = months; N = number of patients; OS = overall survival; rPFS = radiographic progression-free survival.
6.6.5.b.2. Androgen deprivation therapy combined with chemotherapy
Three large RCTs were conducted [775,1088,1114]. All trials compared ADT alone as the SOC with ADT combined with immediate docetaxel (75 mg/sqm, every 3 weeks within three months of ADT initiation). The primary objective in all three studies was to assess OS.
In the GETUG 15 trial, all patients had M1 PCa, either de novo or after a primary treatment [1250]. They were stratified based on previous treatment and Glass risk factors [1210]. In the CHAARTED trial, the same inclusion criteria applied, and patients were stratified according to disease volume [1213].
STAMPEDE is a multi-arm multistage trial in which the reference arm (ADT monotherapy) included 1,184 patients. One of the experimental arms was docetaxel combined with ADT (n = 593), another was docetaxel combined with zoledronic acid (n = 593). Patients were included with either M1 or N1 or having two of the following three criteria: T3/4, PSA ≥ 40ng/mL or ISUP GG 4–5. Relapsed patients after local treatment were also included if they met one of the following criteria: PSA ≥ 4ng/mL with a PSA-DT < 6 months or a PSA ≥ 20ng/mL, N1 or M1. No stratification was used regarding metastatic disease volume (high/low volume) [891].
In all three trials, toxicity was mainly haematological with approximately 12–15% grade 3–4 neutropenia, and 6–12% grade 3–4 febrile neutropenia. The use of granulocyte colony-stimulating factor receptor (GCSF) was shown to be beneficial in reducing febrile neutropenia. Primary or secondary prophylaxis with GCSF should be based on available guidelines [1251,1252].
Docetaxel in all three trials was used at the standard dose of 75mg/sqm every three weeks, six cycles in CHAARTED and STAMPEDE and up to nine cycles in GETUG-AFU-15. In subgroup analyses from GETUG-AFU 15 and CHAARTED, the beneficial effect of the addition of docetaxel to ADT was most evident in men with de novo metastatic high-volume disease [1214,1215], while it was in the same range whatever the volume in the post-hoc analysis from STAMPEDE [1253]. The effect of adding docetaxel was less apparent in men who had prior local radical treatment although the numbers were small and the event rates low. An SR and meta-analysis which included these 3 trials showed that the addition of docetaxel to SOC improved survival [1252]. The HR of 0.77 (95% CI: 0.68–0.87, p < 0.0001) translates into an absolute improvement in four-year survival of 9% (95% CI: 5–14). An SR and meta-analysis of individual participant data from the three trials showed that there is no meaningful beneficial effect of addition of docetaxel to ADT for patients with metachronous low volume disease. The largest absolute improvement at five years was observed for the patients with high volume and clinical stage 4 disease [1254]. Therefore, adding docetaxel alone to ADT should only be considered if no ARPI is available or all available ARPIs are contraindicated.
Triplet therapy: ADT and chemotherapy +/- ARPI
The addition of abiraterone to ADT and docetaxel improved rPFS and OS in the PEACE-1 trial [1255, 1256]. The trial has a 2x2 factorial design and participants with de novo (synchronous) metastatic PCa were randomised to standard of care (SOC), which was ADT at the beginning of the trial, later ADT plus docetaxel for six cycles (if chemotherapy-fit), versus SOC plus RT versus SOC plus abiraterone versus SOC plus RT plus abiraterone. Co-primary endpoints were rPFS and OS, which were both significantly improved in the total population. Also in the group of patients who received ADT plus docetaxel as SOC (n = 710), both rPFS and OS were increased with a HR: 0.5 (0.34–0.71) and 0.75 (0.59–0.95), respectively. Of note in this population, about 35% had low-volume disease. Toxicity was modestly increased by the addition of abiraterone, mainly hypertension.
In the ARASENS phase III trial, all patients received ADT and docetaxel for six cycles as SOC plus darolutamide or placebo [1257]. A total of 1,306 metastatic patients were included, 14% of them with relapsed disease after radical local treatment (metachronous). The primary endpoint was OS, and this was statistically significantly improved by the addition of darolutamide (HR: 0.68; 95% CI: 0.57–0.8).
In this trial the occurrence of AEs was similar in both arms. Of the included patients 77% had high volume and 70% high-risk disease. In an unplanned subgroup analysis the beneficial effect of adding darolutamide versus placebo for OS was seen in the patients with high-volume (HR 0.69; 0.57-0.82), with high-risk (HR: 0.71; 0.58-0.86) and in low-risk disease (HR: 0.62; 0.42-0.9), for the small subgroup of patients with low-volume disease the results were less clear regarding an OS benefit (HR: 0.68; 0.41-1.13) [1258].
In both PEACE I and ARASENS, docetaxel and the ARPI were given concomitantly. In ENZAMET about 45% of patients received concurrent docetaxel, in TITAN and ARCHES, 11-18% of patients received prior docetaxel as a part of SOC [1171,1172,1238,1243-1245].
ADT and ARPI +/- PARPI (AMPLITUDE)
The AMPLITUDE phase III trial (n = 696) evaluated the combination of the PARP inhibitor niraparib with abiraterone acetate plus prednisone (AAP) in patients with mHSPC with germline or somatic alterations in HRR genes, including BRCA1 or BRCA2 or at least one of seven other HRR genes [1259]. The primary endpoint was met, with a significant improvement in rPFS observed in the BRCA subgroup (HR 0.52; 95% CI: 0.37–0.72) and in the intention-to-treat population (HR 0.63; 95% CI: 0.49–0.80).
The secondary endpoint of OS was immature (HR 0.79) and showed a trend towards favouring niraparib. More grade 3–4 AEs were reported in the niraparib arm: 75% versus 59%, especially anaemia (29% vs. 4.6%), hypertension (26.5% vs. 18.4%) and the need of transfusion. The treatment was more likely to be discontinued in the niraparib arm where also one case of MDS was reported.
ADT and ARPI +/- AKT inhibitor (CAPItello-281)
The phase III RCT CAPItello-281 trial evaluated the addition of the AKT inhibitor capivasertib to standard therapy with abiraterone plus ADT in patients with de novo PTEN deficient mHSPC [1260]. Central testing of 6,003 pre-screened patients confirmed immunohistochemical (IHC) ≥ 90% PTEN loss in 1,519 (25.3%) assessed specimens. Of 1,012 included patients 507 were randomised to capivasertib plus abiraterone and 505 to placebo plus abiraterone with both arms receiving concomitant prednisone/prednisolone and ADT.
The primary endpoint of rPFS was significantly improved by capivasertib versus placebo (median 33.2 vs. 25.7 months; HR: 0.81; 95% CI: 0.66–0.98, p < 0.034. The secondary endpoint of OS was still immature (HR: 0.90; 95% CI: 0.71–1.15, p = 0.401).
6.6.6. Treatment selection and patient selection
Selecting patients for triplets of either docetaxel, niraparib or capivasertib in addition to ADT and ARPI is challenging. The addition of docetaxel was not formally tested against ADT and ARPI, but only tested in the setting of ARPI in addition to ADT and docetaxel. The addition of niraparib or capivasertib requires upfront molecular and IHC testing and has been shown to improve rPFS but still not OS.
In men with HRR mutations, especially in those with BRAC 1 or 2 alterations, the addition of niraparib to ADT and abiraterone plus prednisolone appears to be an important new option for this poor prognostic group of patients. Regulatory approval of this combination is pending.
The OS benefit of adding docetaxel to ADT and ARPI has not been studied directly and ADT plus ARPI remains the SOC in patients with no documented deficiency of HRR or PTEN. However, PTEN transcriptomic inactivity as assessed by NGS of RNA, revealed shorter OS on ADT and abiraterone, but not with ADT and docetaxel, suggesting a higher docetaxel benefit in men with PTEN inactive PCa [1261]. Ideally, the transcriptomic PTEN activity should be known when counselling patients with mHSPC. Of note, IHC based PTEN loss and transcriptomic PTEN inactivity were not well aligned as 30% of those with PTEN positivity by IHC were classified as transcriptomically PTEN inactive.
For patients without the above mentioned molecular aberrations the choice of treatment is between ADT plus ARPI and the addition of docetaxel for six cycles. Several network meta-analyses of the published data have concluded that combination therapy is more efficient than ADT alone, but none of the doublet combination therapies have been convincingly proven to be superior over another [1261-1266]. In an SR and meta-analysis looking at association between age and efficacy of combination therapy, patients appeared to benefit from combination therapy irrespective of age [1266]. As a consequence, patients should be offered combination treatment unless there are clear contra-indications or they present with asymptomatic disease and a very short life expectancy (based on frailty assessment or non-cancer comorbidities).
Docetaxel as sole addition to ADT is no longer recommended in the majority of patients if an ARPI is available and there are no contra-indications to use one. The different ARPIs are similar regarding efficacy, but differ regarding their toxicity profiles. The choice of an ARPI should be based on the individual patient’s risk profile and comorbidities [1267]. For patients with metachronous low-volume PCa, ARPI doublet therapies were ranked as the potentially most efficacious treatment option and the expected outcomes were not significantly different from those achieved by triplet regimens; however, docetaxel adds toxicity [1261].
The question whether triplet therapy is superior to ADT and ARPI doublet therapy has been addressed by multiple SRs and meta-analyses. The choice between triplet therapy and the ADT and ARPI doublet therapy should be discussed with de novo and/or high-volume/high-risk disease patients [1268-1270].
6.6.7. Treatment of the primary tumour in newly diagnosed metastatic disease
The first reported trial evaluating prostate RT in men with mHSPC was the HORRAD trial. Four hundred and thirty-two patients were randomised to ADT alone or ADT plus IMRT with IGRT to the prostate. Overall survival was not significantly different (HR: 0.9 [0.7–1.14]) and median time to PSA progression was significantly improved in the RT arm (HR: 0.78 [0.63–0.97]) [1271]. The risk of obstructive PCa–related local symptoms such as bladder outlet obstruction or hydronephrosis was reduced in men who underwent RT as evidenced by interventions required in 18% vs. 30% (HR 0.61 [0.37–0.99]) after a median follow-up time of 75 months for patients who are still alive (66/328).
The STAMPEDE trial evaluated 2,061 men with mHSPC who were randomised to ADT alone versus ADT plus RT to the prostate. This trial confirmed that RT to the primary tumour did not improve OS in unselected patients [1216]. However, following the results from CHAARTED and prior to analysing the data, the original screening investigations were retrieved, and patients categorised as low or high volume. In the low-volume subgroup (n = 819), there was a significant OS benefit by the addition of prostate RT. This was confirmed by the latest analysis of long-term follow-up (median follow-up of 61 months [HR: 0.64 for OS benefit in the low-volume group]) [1272].
A secondary, not preplanned analysis of the STAMPEDE trial confirmed the benefit of prostate RT in patients with ≤ 3 bone metastases, but also showed a benefit in patients with M1a disease [1273]. No evidence of difference in time to symptomatic local events was found with median follow-up of over five years [1272].The dose used in these indications should be equivalent of up to 72Gy in 2Gy fractions. Therefore, RT of the prostate only in patients with low-volume metastatic disease should be considered. In an SR and meta-analysis including the above two RCTs, the authors found that, overall, there was no evidence that the addition of prostate RT to ADT improved survival in unselected patients (HR: 0.92, 95% CI: 0.81–1.04, p = 0.195) [1274]. However, there was a clear difference in the effect of metastatic burden on survival with an absolute improvement of 7% in three-year survival in men who had four or fewer bone metastases.
The randomised phase-III PEACE-1 trial with a 2x2 factorial design (SOC, SOC + abiraterone, SOC + RT and SOC + abiraterone + RT) demonstrated that adding prostate RT (total dose 74Gy in 37 fractions) significantly prolonged the co-primary endpoint of PFS from 4.4 years to 7.5 years in the low-volume metastatic burden group treated with SOC+ARPI. Additionally, a significant delay in the time to castration resistance was observed, although there was no improvement in OS in this group [1275].
PEACE-1 also reported a significant reduction in the incidence of serious genitourinary events such as obstruction, bleeding, insertion of double-J stent and TURP for patients treated with local RT to the prostate. This was an important secondary endpoint in the PEACE-1 study where the preventive effect of RT was observed both in the cohort of patients with low-volume metastatic disease (26% vs. 11%; delay in the time to first serious genitourinary event p = 0·0002;) and the overall cohort (22.3% vs 12.2%; p = 0·0001) [1275].
A network meta-analysis of ten RCTs, including PEACE-1, in a population with de novo mHSPC and no prior docetaxel use, did not reveal a significant OS benefit for the addition of RT to SOC plus ARPI versus SOC plus ARPI alone (HR 0.76; 95% CI 0.51–1.16) [1276].
6.6.8. Metastasis-directed therapy in M1 patients
In patients relapsing after a local treatment, a metastases-directed therapy (MDT) has been proposed, with the aim to delay systemic treatment. The same rationale can be applied in men with a single or only few metastases at diagnosis, i.e., oligometastatic disease. The definition of oligometastatic PCa varies and depends strongly on the applied imaging [1235].
A retrospective analysis on 211 patients treated with MDT aimed at defining prognostic factors for MFS, palliative ADT-free (pADT) survival and cause-specific survival (CSS). With a median follow-up of 42 months after MDT, patients with cN1 only had significantly superior five-year MFS, pADT and CSS when compared to patients with M1 disease (p<0.02). At five years, 23% of patients were free of BCR [1277].
Two randomised phase II trials assessed MDT using surgery ± SABR versus surveillance [1100] or SABR versus surveillance in men with oligo-recurrent PCa [1278]. Oligo recurrence was defined as < 3 lesions on choline PET/CT only [1100] or conventional imaging with MRI/CT and/or bone scan [1278]. The sample size was small with 62 and 54 patients, respectively, and a substantial proportion of them had nodal disease only [1100]. Androgen deprivation therapy-free survival was the primary endpoint in the STOMP study which was longer with MDT than with surveillance [1100]. The primary endpoint in the ORIOLE trial was progression after six months which was significantly lower with SBRT than with surveillance (19% vs. 61%, p = 0.005) [1278].
The combined results of STOMP and ORIOLE confirmed the significant improvement in PFS in favour of MDT (HR: 0.44, p < 0.001) [1279].
A phase II trial assessed the biochemical response after 18F-DCFPyL PET/MRI and subsequent MDT. Overall biochemical response rate, defined as ≥ 50% PSA decline, was 60%, including 22% of patients with complete biochemical response [1280].
The randomised phase II EXTEND trial investigated whether MDT, when added to SOC systemic treatment, improved PFS when compared to SOC systemic treatment alone in oligometastatic PCa patients, with oligometastatic being defined as maximally five lesions. In total, 87 patients were randomised and the vast majority presented with one or two metastatic lesions. In total, 51 patients received ADT alone, while 36 patients also received ARPI. The addition of MDT significantly improved both PFS (15.8 months vs. not reached; HR: 0.25; p<0.001) and eugonadal PFS (6.1 months vs. not reached; HR: 0.32; p = 0.03). This significant benefit was observed both in the patient group receiving ADT alone or ADT + ARPI [1281]. In analogy, the SATURN trial, which included 28 oligo-recurrent metastatic PCa patients, looked at the PFS of adding dual ARPI and MDT to existing ADT. The median PFS in SATURN was 19.3 months and 50% of the patients still had an undetectable PSA 6 months after testosterone recovery. While MDT-induced toxicity was very low, adding dual ARPI induced grade 3 toxicity in 20% of the patients [1282].
Five-year outcomes of SBRT for oligometastatic PCa from the prospective TRANSFORM phase II trial have been reported [1283]. In total 199 men (stage M1a/b and/or N1) received SBRT and 76% were hormone naïve at baseline. The primary endpoint was five-year treatment escalation free survival (TE-FS), defined as freedom from any new cancer therapy other than further SBRT for up to five lesions. Ninety-three percent had prior RP, 46% of the patients had stage N1 only, 76% were staged by PET-CT. The rate of five-year TE-FS was 21.7% (CI: 15.7%-28.7%) overall. These data suggest that SBRT based MDT may be an effective option for delaying systemic treatment escalation in highly selected patients.
The randomised phase II clinical trial (RADIOSA) investigated whether six months ADT added to SBRT versus SBRT alone for hormone-sensitive metachronous oligo-recurrent PCa improved clinical PFS [1284]. In total, 105 patients were randomised, 52 to SBRT alone and 53 to SBRT + ADT, three patients were lost to follow up. With a median follow up of 31 months the clinical PFS was 15.1 months for the SBRT-group (95% CI: 12.4-22.8) versus 32.2 months for the SBRT+ADT group (22.4-not reached; HR: 0.43 0.26-0.72, p = 0.0010). No grade > 2 late ADT- or SBRT-related toxicities were reported.
Currently there are no data to suggest an improvement in OS. Two comprehensive reviews highlighted MDT (SABR) as a promising therapeutic approach that must still be considered as investigational until the results of ongoing RCTs are available [1285,1286]. The toxicity of MDT is low, with almost no grade ≥ 3 toxicity [1287-1289].
6.6.9 Recommendations for the first-line treatment of hormone-sensitive metastatic disease*
| Recommendations | Strength rating |
| First-line treatment | |
| Discuss all patients with hormone-sensitive metastatic disease in a multidisciplinary team | Strong |
| Offer immediate systemic treatment with androgen deprivation therapy (ADT) to palliate symptoms and reduce the risk for potentially serious sequelae of advanced disease (spinal cord compression, pathological fractures, ureteral obstruction) to M1 symptomatic patients. | Strong |
| Offer short-term administration of an older generation androgen receptor (AR) antagonist to M1 patients starting luteinising hormone-releasing hormone (LHRH) agonist to reduce the risk of the ‘flare-up’ phenomenon. | Weak |
| At the start of ADT, offer LHRH antagonists or orchiectomy to patients with impending clinical complications, such as spinal cord compression or bladder outlet obstruction. | Strong |
| Do not offer AR antagonist monotherapy to patients with M1 disease. | Strong |
| Do not offer ADT monotherapy to patients whose first presentation is M1 disease if they have no contraindications for combination therapy and have a sufficient life expectancy to benefit from combination therapy (≥ 1 year) and are willing to accept the increased risk of side effects. | Strong |
| Offer ADT combined with abiraterone acetate plus prednisone or apalutamide or enzalutamide or rezvilutamide to patients with M1 disease who are fit for the regimen. | Strong |
| Offer ADT combined with darolutamide to patients with M1 disease who are fit for the regimen | Weak |
| Offer docetaxel only in combination with ADT plus abiraterone or darolutamide to patients with M1 disease who are fit for docetaxel. | Strong |
| Test patients for somatic or germline homologous recombination repair aberrations, since they may qualify for the addition of niraparib to ADT plus abiraterone in patients with M1 disease. | Weak |
| Offer ADT combined with prostate radiotherapy (using doses up to the equivalent of 72 Gy in 2 Gy fractions) to patients whose first presentation is M1 disease and who have low volume of disease by CHAARTED criteria. | Strong |
| Do not offer ADT combined with surgery to M1 patients outside of clinical trials. | Strong |
| Only offer metastasis-directed therapy to M1 patients within a clinical trial setting or a well-designed prospective cohort study. | Strong |
| Supportive care | |
| Assess osteoporosis risk factors and perform a dual emission X-ray absorptiometry scan when commencing long-term ADT, to mitigate osseous complications. | Strong |
| Offer bone protection to avoid fractures in patients receiving combination treatment. | Strong |
| Offer calcium and vitamin D supplementation when prescribing either denosumab or bisphosphonates and monitor serum calcium. | Strong |
| Treat painful bone metastases early on with palliative measures, such as radiotherapy and adequate use of analgesics. | Strong |
| In patients with spinal cord compression, start immediate high-dose corticosteroids and assess for spinal surgery, potentially followed by radiation. Offer radiation therapy alone if surgery is not appropriate. | Strong |
*All the following statements are based on metastatic disease defined by bone scintigraphy and CT scan/MRI.
6.7. Treatment: Castration-resistant PCa (CRPC)
6.7.1. Definition of CRPC
Castrate serum testosterone < 50 ng/dL or 1.7 nmol/L plus either:
a.Biochemical progression: Three consecutive rises in PSA at least one week apart resulting in two 50% increases over the nadir, and a PSA > 2 ng/mL.
b.Radiological progression: The appearance of new lesions: either two or more new bone lesions on bone scan, ideally confirmed [1290], or a soft tissue lesion using RECIST (Response Evaluation Criteria in Solid Tumours) [1291]. Symptomatic progression alone must be questioned and subject to further investigation. It is not sufficient to diagnose CRPC.
c.Unequivocal clinical progression.
6.7.2. Management of mCRPC - general aspects
Selection of treatment for mCRPC is multifactorial and in general dependent on:
- previous treatment for mHSPC and for non-mHSPC;
- previous treatment for nmCRPC and mCRPC;
- quality of response and pace of progression on previous treatment;
- known cross resistance between androgen receptor pathway inhibitor (ARPI);
- co-medication and known drug interactions (see approved summary of product characteristics);
- known genetic alterations and microsatellite instability–high (MSI-H)/mismatch repair-deficient (dMMR) status;
- known histological variants and DNA repair deficiency (to consider platinum or targeted therapy like PARPi);
- local approval status of drugs and reimbursement situation;
- available clinical trials;
- the patient and his comorbidities.
6.7.2.a. Molecular diagnostics
Metastatic patients should be offered somatic genomic testing for homologous repair and MMR defects early on. Testing can be performed on more recently collected primary tumour tissue or on tissue from metastases. Alternatively, but still less common, genetic testing on circulating tumour DNA (ctDNA) is an option and has been used in some trials. One test, the FoundationOne® Liquid CDx, has been FDA approved [1292]. Defective MMR assessment can be performed by IHC for MMR proteins (MSH2, MSH6, MLH1 and PMS2) and/or by next generation sequencing (NGS) assays [1195]. Germline testing for at least BRCA1/2, ATM and MMR is recommended for patients with high-risk localised PCa and particularly for patients with metastatic PCa.
Molecular diagnostics should be performed by a certified (accredited) institution using a standard NGS multiplication procedure (minimum depth of coverage of 200 X). The genes and respective exons should be listed; not only DNA for mutations but RNA needs to be examined for fusions and protein expression to obtain all clinically relevant information. A critical asset is the decision support helping to rate the mutations according to their clinical relevance [1193,1194].
Level 1 evidence for the use of PARP inhibitors has been reported [341,1196-1207]. Microsatellite instability (MSI) high (or MMR deficiency) is rare in PCa, but for those patients, pembrolizumab has been approved by the FDA and could be a valuable additional treatment option [1187,1208].
6.7.3. Treatment decisions and sequence of available options
Approved agents for the treatment of mCRPC in Europe are docetaxel, abiraterone/prednisolone (AAP), enzalutamide, cabazitaxel, olaparib, niraparib/AAP, talazoparib/enzalutamide, radium-223 and lutetium (177Lu) vipivotide tetraxetan. For nmCRPC, apalutamide, darolutamide and enzalutamide have been approved. In general, sequencing of ARPIs like abiraterone and enzalutamide is not recommended particularly if the time of response to ADT and to the first ARPI was short (≤ six to twelve months) and high-risk features of rapid progression are present [1293-1295].
The use of chemotherapy with docetaxel and subsequent cabazitaxel in the treatment sequence is recommended and should be applied early enough when the patient is still fit for chemotherapy. This is supported by high-level evidence [1293].
In case of a known BRCA alteration, the use of a PARP inhibitor should always be prioritised as these patients harbour an adverse prognosis and don’t respond well to most other treatments. PARP inhibitor use significantly improves rPFS and OS in these patients [1296-1299].
6.7.4. Non-metastatic CRPC
Frequent PSA testing in non-metastatic men treated with ADT has resulted in earlier detection of biochemical progression. Of these men approximately one-third will develop bone metastases within two years, detected by conventional imaging [1014].
In men with CRPC and no detectable clinical metastases using bone scan and CT scan, baseline PSA level, PSA velocity and PSA-DT have been associated with time to first bone metastasis, bone MFS and OS [1014,1300]. These factors may be used when deciding which patients should be evaluated for metastatic disease. A consensus statement by the PCa Radiographic Assessments for Detection of Advanced Recurrence (RADAR) group suggested a bone scan and a CT scan when the PSA reached 2ng/mL and if this was negative, it should be repeated when the PSA reached 5ng/mL, and again after every doubling of the PSA based on PSA testing every three months in asymptomatic men [1301]. Symptomatic patients should undergo relevant investigations regardless of PSA level. With more sensitive imaging techniques like PSMA hybrid imaging or whole-body MRI, more patients are diagnosed with early mCRPC [1302]. It remains unclear if the use of PSMA PET/CT in this setting improves outcome.
Three large phase III RCTs, PROSPER [1303], SPARTAN [1304] and ARAMIS [1305], evaluated MFS as the primary endpoint in patients with nmCRPC (M0 CRPC) treated with enzalutamide (PROSPER) vs. placebo or apalutamide (SPARTAN) vs. placebo or darolutamide (ARAMIS) versus placebo, respectively (Table 6.7.1). The M0 status was established by CT and bone scans. Only patients at high risk for the development of metastasis with a short PSA-DT of ≤ 10 months were included. Patient characteristics in the trials revealed that about two-thirds of participants had a PSA-DT of < 6 months. All trials showed a significant MFS benefit. All three trials showed a survival benefit after a follow-up of more than 30 months. In view of the long-term treatment with these AR targeting agents in asymptomatic patients, potential AEs need to be taken into consideration and the patient informed accordingly.
6.7.5. Metastatic CRPC
The remainder of this section focuses on the management of men with proven mCRPC on conventional imaging.
6.7.5.a. Conventional androgen deprivation in CRPC
Eventually men with PCa will show evidence of disease progression despite castration. Two trials have shown only a marginal survival benefit for patients remaining on LHRH analogues during second- and third-line therapies [1306,1307]. However, in the absence of prospective data, the modest potential benefits of continuing castration outweigh the risk of treatment. In addition, all subsequent treatments have been studied in men with ongoing androgen suppression, therefore, it should be continued in these patients.
6.7.6. First-line treatment of metastatic CRPC
6.7.6.a. Abiraterone
Abiraterone was evaluated in 1,088 chemo-naive, asymptomatic or mildly symptomatic mCRPC patients in the phase III COU-AA-302 trial. Patients were randomised to abiraterone acetate or placebo, both combined with prednisone [1308]. Patients with visceral metastases were excluded. The main stratification factors were ECOG PS 0 or 1 and asymptomatic or mildly symptomatic disease. Overall survival and rPFS were the co-primary endpoints. After a median follow-up of 22.2 months there was significant improvement of rPFS (median 16.5 vs. 8.2 months, HR: 0.52, p < 0.001) and the trial was unblinded. At the final analysis with a median follow-up of 49.2 months, the OS endpoint was significantly positive (34.7 vs. 30.3 months, HR: 0.81, 95% CI: 0.70–0.93, p = 0.0033) [1309]. Adverse events related to mineralocorticoid excess and liver function abnormalities were more frequent with abiraterone but mostly grade 1–2. Subset analysis of this trial showed the drug to be equally effective in an elderly population (> 75 years) [1310].
6.7.6.b. Enzalutamide
A randomised phase III trial (PREVAIL) included a similar patient population and compared enzalutamide and placebo [1311]. Men with visceral metastases were eligible but the numbers included were small. Corticosteroids were allowed but not mandatory. PREVAIL was conducted in a chemo-naive mCRPC population of 1,717 men and showed a significant improvement in both co-primary endpoints, rPFS (HR: 0.186, CI: 0.15–0.23, p < 0.0001), and OS (HR: 0.706, CI: 0.6–0.84, p < 0.001). A ≥ 50% decrease in PSA was seen in 78% of patients. The most common clinically relevant AEs were fatigue and hypertension. Enzalutamide was equally effective and well tolerated in men > 75 years [1312] as well as in those with or without visceral metastases [1313]. However, for men with liver metastases, there seemed to be no discernible benefit [1313,1314].
Enzalutamide has also been compared with bicalutamide 50 mg/day in a randomised double-blind phase II study (TERRAIN) showing a significant improvement in PFS (15.7 months vs. 5.8 months, HR: 0.44, p < 0.0001) in favour of enzalutamide [1314]. With extended follow-up and final analysis, the benefit in OS and rPFS were confirmed [1315].
6.7.6.c. Docetaxel
A statistically significant improvement in median survival of 2.0–2.9 months has been shown with docetaxel compared to mitoxantrone plus prednisone [1316,1317]. The standard first-line chemotherapy is docetaxel 75 mg/m2, three-weekly doses combined with prednisone 5 mg twice a day (BID), up to ten cycles. Prednisone can be omitted if there are contraindications or no major symptoms. The following independent prognostic factors: visceral metastases, pain, anaemia (Hb < 13 g/dL), bone scan progression, and prior estramustine may help stratify the response to docetaxel. Patients can be categorised into three risk groups: low risk (0 or 1 factor), intermediate (2 factors) and high risk (3 or 4 factors), and show three significantly different median OS estimates of 25.7, 18.7 and 12.8 months, respectively [1318].
Age by itself is not a contraindication to docetaxel [1319] but attention must be paid to careful monitoring and comorbidities as discussed in Section 6.1 [1320]. In men with mCRPC who are thought to be unable to tolerate the standard dose and schedule, docetaxel 50 mg/m2 every two weeks seems to be well-tolerated with less grade 3–4 AEs and a prolonged time to treatment failure [1321].
So far, no combination with docetaxel has proven to be superior to docetaxel alone in unselected mCRPC patients, including the combination with the checkpoint inhibitor pembrolizumab [1322].
6.7.6.d. Combinations with PARP inhibitors
Based on the suggestion that there is a synergistic antitumour effect when combining an ARPI with a PARP inhibitor, several such combination trials were conducted in first-line mCRPC patients without prior ARPI use, with different trial designs and different patient- and molecular panel selection.
Abiraterone/prednisone plus olaparib
A randomised double-blind, phase III trial (PROpel) of AAP plus olaparib (300 mg twice daily) or placebo in patients with mCRPC in the first-line setting was conducted [1198,1199]. Patients (n = 796) were randomly assigned 1:1 to study treatment regardless of homologous recombination repair gene mutation (HRRm) status which was retrospectively evaluated and determined by tumour tissue and circulating tumour DNA tests. The primary end point was imaging-based PFS (ibPFS) by investigator assessment. All patients were ARPI naïve, 24% were docetaxel pretreated. The result was significantly positive in favour of the combination with ibPFS of 24.8 versus 16.6 months (HR 0.66; 95% CI: 0.54 to 0.81; p = 0.001). In the prespecified final analyses the key secondary endpoint OS had only 47.9% maturity and did not meet the prespecified two-sided boundary for significance (0.95% CI: 0.81, 0.67-1.0, p = 0.054). The exploratory analysis of the subgroup of patients with positive HRRm status showed a rPFS HR of 0.50 (CI: 0.34 to 0.73). The BRCA mutated patients (11% of the ITT population) had an even larger benefit for rPFS (HR 0.24; 95% CI: 0.12 - 0.45) and the OS HR in these patients was 0.30 (95% CI: 0.15 - 0.59), suggesting that the overall benefit observed in the ITT population was primarily driven by patients with a BRCA mutation [1200].
The most common AEs in patients receiving olaparib plus AAP were anaemia (48%; ≥ G3 15%), fatigue (38%), nausea (30%), diarrhoea (19%), decreased appetite (16%), lymphopenia (14%), dizziness (14%), and abdominal pain (13%); 18% of patients required at least one blood transfusion and 12% required multiple transfusions [1200]. The combination of olaparib plus AAP was approved by the EMA for the treatment of adult patients with mCRPC in whom chemotherapy is not clinically indicated [1240]. In the US, the FDA has approved olaparib with AAP for mCRPC patients with deleterious or suspected deleterious BRCA mutations as determined by an FDA-approved companion diagnostic test [1201]. For patients without BRCA mutations, the FDA determined that the modest rPFS improvement, combined with clinically significant toxicities, did not demonstrate a favourable risk/benefit assessment [1297].
The combination of PARP inhibitor plus ARPI in patients with BRCA1/2 (or ATM) mutations in the first-line as opposed to the use of PARP inhibitor monotherapy or the sequential use of these agents is supported by a randomised phase II trial (BRCAAway) albeit with low patient numbers and thus a low level of evidence [1323].
Abiraterone/prednisone plus niraparib
In a randomised, double-blind, phase III trial (MAGNITUDE) AAP plus niraparib 200 mg once/daily or placebo, was evaluated [1202]. The study prospectively included two cohorts, an HRR-negative and an HRR-positive cohort. The HRR-negative cohort was closed early for futility after enrolling 200 patients. In the overall HRR-positive cohort, the addition of Niraparib to AAP resulted in a significant improvement in the first endpoint rPFS compared to AAP plus placebo (HR = 0.73; 95% CI 0.56-0.96; p = 0.0217) and the median rPFS was 16.5 vs. 13.7 months in favour of the combination. In particular, the 113 patients with BRCA 1/2 mutations [1203] who received AAP plus niraparib [1203] derived a major rPFS benefit (19.5 vs. 10.9 months; HR = 0.55 [95% CI 0.39-0.78]; nominal p = 0.0007). The final analysis of OS at median follow-up of 37.3 months revealed no difference between niraparib + AAP and placebo + AAP in the HRR+ population (HR 0.931; 95% CI 0.72-1.20; p = 0.585) or the subgroup with BRCA 1/2 alterations (HR 0.788, 95% CI 0.55-1.120 nominal p = 0.183) [1324]. The most common side effects with Niraparib plus AAP in the ITT population were anaemia (46.2%), fatigue (26.4%), hypertension (31.6%) and constipation (30.7%). The combination of niraparib plus AAP in a dual-action tablet has been approved by the EMA and the FDA for patients with mCRPC and BRCA 1/2 mutations in whom chemotherapy is not clinically indicated [1204].
Enzalutamide plus talazoparib
A randomised double-blind, phase III trial (TALAPRO-2) of the PARP inhibitor talazoparib (0.5mg daily) plus enzalutamide versus enzalutamide/placebo showed a significantly better median rPFS (first endpoint) in favour of the combination regardless of the HRR pathway status [1205]. The median rPFS was 33.1 months [95% CI 27.4–39.0] versus 19.5 months [16.6–24.7], (HR 0.67 [95% CI 0.55–0.81]; p < 0·0001).
For the subgroups of patients with HRR mutations the benefit of the combination was much more pronounced. The HRR gene-mutated population showed a median rPFS of 27.9 (16.6–not reached) for the talazoparib combination versus 16.4 (10.9–24.6) for the placebo group (0.46; 95% CI: 0.30–0.70; p = 0.0003 ) and 0.70 (0.54–0.89; p = 0.0039) in patients with a status of non-deficient or unknown. In an exploratory analysis, the HR for rPFS in patients with BRCA-mutated mCRPC was 0.23 (0.10–0.53; p = 0.0002) and, in patients with non-BRCAm HRR gene-mutated mCRPC, it was 0.66 (0.39–1.12; p = 0.12) in favour of the talazoparib combination [1205]. At a median follow-up of 52.5 months, OS was significantly improved with talazoparib plus enzalutamide compared with enzalutamide plus placebo (HR 0.80, 95% CI 0.66–0.96; p = 0·016); median OS was 45.8 months in the talazoparib group compared with 37.0 months in the control group. This effect was much more pronounced in HRR-deficient patients (n = 69; HR 0.55 [0.36–0.83]; p = 0·0035) and to a much lesser extent in HRR-non-deficient or unknown patients (n = 636; HR 0.88 [0.71–1.08]; p = 0·22). At the final updated analysis, rPFS showed a slightly higher HR (HR 0.67 [0.55–0.81]; p < 0·0001) [1325].
The most common treatment-emergent adverse events with the addition of talazoparib were anaemia, neutropenia, and fatigue; the most common grade 3–4 event was anaemia (46%), which improved after dose reduction, however, 39% required a blood transfusion, including 22% who required multiple transfusions, 8% discontinued treatment due to anaemia and 2 patients on the combination were diagnosed with myelodysplastic syndrome/acute myeloid leukaemia [1205]. In TALAPRO-2, also an HRR-deficient-only cohort (cohort 2; n = 230) was recruited. The primary analysis for the combined HRR-deficient population (n = 399) met the rPFS endpoint with a HR 0.45 (95% CI, 0.33 to 0.61; p < 0.0001; median not reached at the time of the analysis for the talazoparib group versus 13.8 months for the placebo group). At median follow-up of 44.2 months, talazoparib plus enzalutamide resulted in a statistically significant improvement in OS versus enzalutamide (HR 0.62 [95% CI 0.48–0.81]; two-sided p =0·0005) in this cohort of HRR deficient patients. The median OS was 45.1 months (95% CI 35.4 - not reached) in the talazoparib group versus 31.1 months in the control group. In the subgroup of patients with BRCA 1/2 alterations (n = 155 [39%]), median OS was not reached for talazoparib plus enzalutamide versus 28.5 months for enzalutamide (HR 0.50 [95% CI 0.32–0.78; p =0·0017); four-year OS rates were 53% in the talazoparib group versus 23% in the control group. Updated rPFS favoured talazoparib plus enzalutamide versus enzalutamide (HR 0.47 [95% CI 0.36–0.61]; p < 0·0001; median rPFS was 30.7 versus 12.3 months [1206,1326]. The expected clinical benefit in the subgroups needs to be weighed against the potential burden of side effects [1296].
The FDA approved talazoparib with enzalutamide only for HRR gene-mutated mCRPC [1207,1296,1327]. In May 2025, the FDA’s Oncologic Drugs Advisory Committee (ODAC) deemed the results from TALAPRO-2 insufficient to conclude a favourable benefit-risk profile for adding talazoparib to enzalutamide in patients with non-HRRm mCRPC [1328]. The concerns were the lack of a prespecified, statistically powered analysis in the non-HRRm subgroup, which represents the majority of the target population and thus the uncertainty about the reliability of the per se small survival effect in non-HRRm. Concerns regarding increased hematologic toxicity were also raised. The EMA has approved the combination of talazoparib and enzalutamide for the treatment of patients with mCRPC in whom chemotherapy is not clinically indicated [1329].
Regarding additional side effects of special interest, there seems to be a doubling of the risk of thromboembolic events with the use of PARPis. In a meta-analysis of 2,210 and 1,662 patients with PC and PARPi treatment vs. control, PARPi had a statistically significant increased risk of thrombosis in PCa patients (OR = 1.98, 95 % CI: 1.06–3.70, p = 0.030) with 96 (4.3 %) and 37 (2.2 %) in the PARPi and control groups, respectively [1330].
Based on 18 placebo-controlled RCTs (n = 7,307 patients, tumour agnostic), PARPis significantly increased the risk of myelodysplastic syndrome and acute myeloid leukaemia compared with placebo treatment (Peto OR 2·63 [95% CI 1·13–6·14], p = 0·026) with no between-study heterogeneity (I2=0%, χ2 p = 0·91). Median treatment duration was 9.8 months (IQR 3·6–17·4; n = 96) and median latency period since first exposure to a PARPi was 17.8 months (8·4–29·2; n = 58). Of 104 cases that reported outcomes, 47 (45%) resulted in death [1331].
Radium-223 in combination with enzalutamide
Men with mCRPC and bone metastases were randomised 1:1 to enzalutamide with or without radium-223 (ENZ vs. ENZ-RAD) in the EORTC 1333/PEACE-3 trial [1332]. After an amendment, co-administration of zoledronic acid or denosumab (bone protecting agents) was obligatory. The primary endpoint was rPFS. Of the 446 enrolled men, 87.9% in the ENZ-RAD arm completed the scheduled six cycles of RAD. Radiographic PFS was 16.4 (95% CI 13.8-19.2) months in the ENZ arm and 19.4 (95% CI 17.1-25.3) months in the ENZ-RAD arm. The HR or rPFS was 0.69 (95% CI 0.54-0.87; p = 0.0009). The HR for OS was 0.69 (95% CI 0.52-0.90; p = 0.0031), with median OS in the preplanned interim analysis of 35.0 (95% CI 28.8-38.9) months in the ENZ arm and 42.3 (95% CI 36.8-49.1) months in the ENZ-RAD arm. Treatment-emergent adverse events (TEAE) ≥ grade 3 were reported in 55.8% and 65.6% of the patients in the ENZ and ENZ-RAD arms, respectively. The most frequent grade ≥ 3 TEAE in the ENZ-RAD arm were hypertension (34%), fatigue (6%), anaemia (5%), and neutropenia (5%). No TEAE ≥ grade 3 was increased by more than 5% in the ENZ-RAD arm versus the ENZ arm.
The interim safety data analysis from the randomised phase III PEACE-3 trial comparing radium-223 combined with enzalutamide for first-line mCPRC to enzalutamide alone showed a high fracture rate in both arms. For patients who received bone protecting agent (BPA) therapy, after it became mandatory on the trial, fracture rates significantly decreased in both study arms one-year fractures decreased from 15.6% to 2.6% with a BPA in patients receiving enzalutamide monotherapy. This underscores the high risk of fracture in patients with mCRPC and the necessity of complying with guidelines regarding BPA administration in the ARPI era [1333].
6.7.7. Second-line treatment for mCRPC
All patients who receive treatment for mCRPC will eventually progress. All treatment options in this setting are presented in Table 6.7.3. There is a paucity of high-level data with regards to the sequence of treatments, particularly in case of pretreatment for mHSPC with ARPI and/or docetaxel or other agents. Treatment sequences will depend on which agents were used previously.
6.7.7.a. Cabazitaxel
Cabazitaxel is a taxane with activity in docetaxel-resistant cancers. It was studied in a large prospective, randomised, phase III trial (TROPIC) comparing cabazitaxel plus prednisone vs. mitoxantrone plus prednisone in 755 patients with mCRPC, who had progressed after or during docetaxel-based chemotherapy [1334]. Patients received a maximum of ten cycles of cabazitaxel (25 mg/m2) or mitoxantrone (12 mg/m2) plus prednisone (10 mg/day). Overall survival was the primary endpoint which was significantly longer with cabazitaxel (median: 15.1 vs. 12.7 months, p < 0.0001). There was also a significant improvement in PFS (median: 2.8 vs. 1.4 months, p < 0.0001), objective RECIST response (14.4% vs. 4.4%, p < 0.005), and PSA response rate (39.2% vs. 17.8%, p < 0.0002). Treatment-associated WHO grade 3–4 AEs developed significantly more often in the cabazitaxel arm, particularly haematological (68.2% vs. 47.3%, p < 0.0002) but also nonhaematological (57.4 vs. 39.8%, p < 0.0002) toxicity. In two post-marketing randomised phase III trials, cabazitaxel was shown not to be superior to docetaxel in the first-line setting; in the second-line setting in terms of OS, 20 mg/m2 cabazitaxel was not inferior to 25 mg/m2, but less toxic. Therefore, the lower dose should be preferred [1335,1336]. Cabazitaxel should preferably be given with prophylactic granulocyte colony-stimulating factor (G-CSF) and should be administered by physicians with expertise in handling neutropenia and sepsis [1337].
6.7.7.b. Abiraterone acetate after docetaxel for mCRPC
Positive results of the large phase III trial (COU-AA-301) were reported after a median follow-up of 12.8 months [1338] and confirmed by the final analysis [1339]. A total of 1,195 patients with mCRPC were randomised 2:1 to AAP or placebo plus prednisone. All patients had progressive disease based on the Prostate Cancer Clinical Trials Working Group 2 (PCWG2) criteria after docetaxel therapy (with a maximum of two previous chemotherapeutic regimens). The primary endpoint was OS, with a planned HR of 0.8 in favour of AAP. After a median follow-up of 20.2 months, the median survival in the AAP group was 15.8 months compared to 11.2 months in the placebo arm (HR: 0.74, p < 0.0001). The benefit was observed in all subgroups, and all the secondary objectives were in favour of AAP (PSA, radiologic tissue response, time to PSA or objective progression). The incidence of the most common grade 3–4 AEs did not differ significantly between arms, but mineralocorticoid-related side effects were more frequent in the AAP group, mainly grade 1–2 (fluid retention, oedema and hypokalaemia).
6.7.7.c. Enzalutamide after docetaxel for mCRPC
The planned interim analysis of the AFFIRM study was published in 2012 [1340]. This trial randomised 1,199 patients with mCRPC in a 2:1 fashion to enzalutamide or placebo. The patients had progressed after docetaxel treatment, according to the PCWG2 criteria. Corticosteroids were not mandatory, but could be prescribed, and were received by about 30% of the patients. The primary endpoint was OS, with an expected HR benefit of 0.76 in favour of enzalutamide. After a median follow-up of 14.4 months, the median survival in the enzalutamide group was 18.4 months compared to 13.6 months in the placebo arm (HR: 0.63, p < 0.001). This led to the recommendation to halt and unblind the study. The benefit was observed irrespective of age, baseline pain intensity, and type of progression. In the final analysis with longer follow-up the OS results were confirmed despite crossover and extensive post-progression therapies [1194]. Enzalutamide was active also in patients with visceral metastases.
All the secondary objectives were in favour of enzalutamide (PSA, soft tissue response, QoL, time to PSA, or objective progression). No difference in terms of side effects was observed in the two groups, with a lower incidence of grade 3–4 AEs in the enzalutamide arm. There was a 0.6% incidence of seizures in the enzalutamide group compared to none in the placebo arm.
6.7.7.d. Radium-223 after docetaxel for mCRPC
The only bone-specific drug that is associated with a survival benefit is the α-emitter radium-223. In a large phase III trial (ALSYMPCA) 921 patients with symptomatic mCRPC, who failed or were unfit for docetaxel, were randomised to six injections of 50 kBq/kg radium-223 or placebo plus SOC. The primary endpoint was OS. Radium-223 significantly improved median OS by 3.6 months (HR: 0.70, p < 0.001) and was also associated with prolonged time to first skeletal event, improvement in pain scores and improvement in QoL [1341]. The associated toxicity was mild and, apart from slightly more haematologic toxicity and diarrhoea with radium-223, did not differ significantly from that in the placebo arm [1341]. Radium-223 was effective and safe whether or not patients were docetaxel pretreated [1342]. Due to safety concerns following ARPI combination treatment use of radium-223 was restricted by EMA to after docetaxel and at least one AR targeted agent [1343]. In particular, the use of radium-223 in combination with AAP showed significant safety risks related to fractures and more deaths. This was most striking in patients without the concurrent use of bone health agents [1344] so that radium-223 should always be used together with bone health agents (see Section 6.7.11.b).
6.7.7.e. Rucaparib after ARPI
The phase III TRITON-3 trial randomised 405 mCRPC patients [1299]. Patients were selected for a BRCA1, BRCA2, or ATM alteration and disease progression after treatment with an ARPI for mCRPC. Treatment was as follows: rucaparib 600 mg twice daily or a physician’s choice control, either second line docetaxel or the ARPI which had not been given previously. The first endpoint rPFS in the intention-to-treat group was significantly better with rucaparib (median 10.2 months and 6.4 months, respectively; HR 0.61; 95% CI, 0.47 to 0.80; p < 0.001). The small ATM subgroup did not derive a benefit. An interim analysis revealed OS to be immature. The study design allowed for cross-over and 60% of patients received a PARP inhibitor at progression (47% rucaparib). With regards to the control arms, the median rPFS was longer with rucaparib than with docetaxel (11.2 months vs. 8.3 months; hazard ratio, 0.53; 95% CI, 0.37 - 0.77) and it was also longer than with an ARPI (11.2 months vs. 4.5 months; hazard ratio, 0.38; 95% CI, 0.25 to 0.58). The most frequent adverse events with rucaparib were fatigue, nausea and anaemia, including 24% Grade ≥ 3 anaemia and 29% of patients on rucaparib required at least one blood transfusion [1345]. Rucaparib has been approved by the FDA.
6.7.7.f. Olaparib after ARPI
See Section 6.7.8.c, ‘PARP inhibitors for mCRPC’.
6.7.7.g.177Lu-PSMA-617 after ARPI
Primary and updated analyses of rPFS for the phase III, multicentre RCT, PSMAfore, investigating taxane-naive patients with PSMA-positive mCRPC who had progressed on ARPI, have been published. Patients were 1:1 randomised between open-label, intravenous 177Lu-PSMA-617 (7.4GBq intravenously, every six weeks, for up to six cycles) and a change of ARPI. A total of 468 patients met all eligibility criteria and were randomly assigned to receive 177Lu-PSMA-617 (234 [50%] patients) or ARPI change (234 [50%]). Crossover was allowed. In the updated analysis at time of the third data cut-off (median time from randomisation to third data cut-off 24.11 months [IQR 20.24–27.40]), median rPFS was 11.60 months (95% CI: 9.30–14.19) in the 177Lu-PSMA-617 group vs. 5.59 months (4.21–5.95) in the ARPI change group (HR 0.49 [95% CI: 0.39–0.61]) [1346].
In the final analysis the key secondary endpoint of OS did not show a statistically significant difference between the 177Lu-PSMA-617 and the ARPI arms. In total 141/234 participants (60.3%) randomised to ARPI change crossed over to receive 177Lu-PSMA-617. The median OS was 24.48 months with 177Lu-PSMA-617 versus 23.13 with ARPI change (HR 0.91; 95% CI 0.72–1.14; p = 0.20); For 177Lu-PSMA-617 versus ARPI change, exposure-adjusted incidences of grade ≥ 3 and serious treatment-emergent adverse events were 60.8 versus 85.1 and 32.5 versus 49.9 per 100 patient-treatment years, respectively [1347]. Dry mouth occurred in 135/227 participants (59.5%; 2/227 grade ≥ 3) and anaemia in 62/227 (27.3%; 14/227 grade ≥ 3) in the 177Lu-PSMA-617 arm.
6.7.8. Treatment after docetaxel and one line of ARPI for mCRPC
6.7.8.a. General considerations
For men progressing quickly on AR targeted therapy (< twelve months) it is now clear that cabazitaxel is the treatment supported by the best data. The CARD trial, an open label randomised phase III trial, evaluated cabazitaxel after docetaxel and one line of ARPI (either AAP or enzalutamide) [1293]. It included patients progressing in less than twelve months on previous abiraterone or enzalutamide for mCRPC. Cabazitaxel more than doubled rPFS vs. another ARPI and reduced the risk of death by 36% vs. ARPI. The rPFS with cabazitaxel remained superior regardless of the ARPI sequence and if docetaxel was given before, or after, the first ARPI.
The choice of further treatment after docetaxel and one line of HT for mCRPC is open for patients who have a > 12 months response to first-line abiraterone or enzalutamide for mCRPC [1315]. Either second-line chemotherapy (cabazitaxel), radium-223 (if bone-only metastases), 177Lu–PSMA-617 radioligand therapy [1348,1349] and PARP inhibitors (if BRCA mutation) are valuable options.
Men previously treated with at least one ARPI or both an ARPI and docetaxel and whose tumours demonstrated homozygous deletions or deleterious mutations in DNA-repair genes showed an 88% response rate to olaparib [1350] and in another confirmatory trial a composite response of 54.3% (95% CI: 39.0–69.1) in the 400 mg cohort and in 18 of 46 (39.1%; 25.1–54.6) evaluable patients in the 300 mg cohort [1351]. In general, subsequent treatments in unselected patients are expected to have less benefit than with earlier use [1352,1353] and there is evidence of cross-resistance between enzalutamide and abiraterone [1354,1355]. This cross resistance with abiraterone should also be extrapolated to apalutamide and darolutamide. Also, the sequential use of the lutamides (apalutamide, darolutamide, enzalutamide) is not recommended as the mode of action is very similar.
6.7.8.b. Radiopharmaceuticals
6.7.8.b.1. Introduction
Historically, several radiopharmaceuticals including Phosphorous-32, Strontium-89, Yttrium-90, Samarium-153, and Rhenium-186 were developed for the treatment of bone pain secondary to metastases from PCa [1356]. They proved effective in a palliation setting, by relieving pain and improving QoL, especially in the setting of diffuse bone metastases. However, they never gained widespread adoption. The first radioisotope to demonstrate a survival benefit was Radium-223 (see Section 6.7.7.d).
6.7.8.b.2. PSMA-based therapy
The increasing use of radiolabelled PSMA hybrid imaging as a diagnostic tracer and the realisation that this allowed identification of a greater number of metastatic deposits led to attempts to treat cancer by replacing the imaging isotope with a therapeutic isotope which accumulates where the tumour is demonstrated (theranostics) [1357]. Therefore, after identification of the target, usually with diagnostic PSMA PET tracers, therapeutic radiopharmaceuticals labelled with β (Lutetium-177 or yttrium-90) or α (Actinium-225)-emitting isotopes could be used to treat metastatic PCa.
The PSMA therapeutic radiopharmaceutical supported by the most robust data is 177Lu-PSMA-617. The first patient was treated in 2014 and early clinical studies evaluating the safety and efficacy of 177Lu-PSMA therapy have demonstrated promising results, despite the fact that a significant proportion of men had already progressed on multiple therapies [1358]. The early data were based on single-centre experience [1359]. Data from uncontrolled prospective phase II trials reported high response rates with low toxic effects [1360,1361]. Positive results are also coming from a randomised phase II trial (TheraP) [1362].
In TheraP patients for whom cabazitaxel was considered the next appropriate standard treatment after docetaxel and who were highly selected by 68Ga-PSMA-11 and 18FDG PET-CT scans, were randomised to receive 177Lu-PSMA-617 (6.0–8.5 GBq intravenously, every 6 weeks, for up to 6 cycles) or cabazitaxel (20 mg/m2 for up to ten cycles). The primary endpoint was a reduction of at least 50% in PSA. The first endpoint was met (66% vs. 37% for 177Lu–PSMA-617 vs. cabazitaxel, respectively, by intention to treat; difference 29% (95% CI: 16–42; p < 0.0001; and 66% vs. 44% by treatment received; difference 23% [9–37]; p = 0.0016) [1362]. Secondary outcomes of the TheraP trial, including survival after a median follow-up of 35.7 months (IQR 31.1 to 39.2) showed that 77 (78%) participants had died in the 177Lu-PSMA-617 group and 70 (69%) participants in the cabazitaxel group. Overall survival was similar between randomly assigned patients in the two groups (19.1 vs. 19.6 months; difference -0.5, 95% CI: -3.7 to 2.7]; p = 0.77) [1363,1364].
An open-label phase III trial (VISION) compared 177Lutetium Vipivotid tetraxetan (177Lu-PSMA-617 radioligand therapy) with protocol-permitted SOC (i.e., excluded chemotherapy, immunotherapy, radium-223 and investigational drugs) in mCRPC patients, with PSMA expressing metastases on PET/CT, previously treated with at least one ARPI and one (around 53%) or two taxanes. Imaging-based PFS and OS were the alternate primary endpoints. More than 800 patients were randomised. 177Lu-PSMA-617 plus SOC significantly prolonged both imaging-based PFS and OS, as compared with SOC alone (see Table 6.6.3). Grade 3 or above AEs were higher with 177Lu-PSMA-617 than without (52.7% vs. 38.0%), but QoL was not adversely affected. 177Lu–PSMA-617 has shown to be an additional treatment option in this mCRPC population [1365]. In a post hoc analysis of the phase III VISION trial the magnitude of PSA decline was associated with improvement in clinical and patient-reported outcomes in patients with mCRPC receiving 177Lu-PSMA-617 plus standard of care in VISION [1366].
A systematic review and updated meta-analysis, investigated the proportion of patients with any or more than 50% PSA decrease, and OS. The review, including 69 articles and a total of 4,157 patients, showed that patients treated with 177Lu–PSMA 617 had a significantly higher response to therapy compared to controls, based on ≥ 50% PSA decrease (OR = 5.33, 95% CI: 1.24–22.90, p < 0.05). Meta-analysis revealed an OS of 0.26 according to pooled HRs for any PSA decline, which was significant after 177Lu–PSMA-617 therapy (95% CI: 0.18–0.37, p < 0.00001) and an OS of 0.52 for ≥ 50% PSA decrease, also significant after radioligand (RLT) (95% CI: 0.40–0.67, p < 0.00001) [1367].
A multicenter retrospective study on 124 mCRPC patients treated with PSMA radionuclide therapy showed that PSMA PET/CT by Response Evaluation Criteria in Prostate-Specific Membrane Antigen Imaging (RECIP) 1.0 after two cycles of 177Lu–PSMA-617 is prognostic for PSA-PFS, indicating that PSMA PET/CT by RECIP 1.0 may be used in earlier stages of PCa to evaluate drug efficacy and to predict PFS [1368].
177Lu-PSMA-617 can measure tumour sites after each therapy by taking whole body 3D images at different timepoints post-injection, using a small radiation wave from the treatment itself [1369]. This is called a Single Photon Emission Computed Tomography (SPECT) scan. SPECT detects gamma emissions from 177Lu, allowing visualisation of radioligand distribution within the body and confirming tumour targeting. There is growing evidence that SPECT can be used as an imaging response biomarker [1370].
The earlier use of ¹⁷⁷Lu-PSMA-617 was studied in patients progressing on the first ARPI for mCRPC (PSMAfore)[1322], see Section 6.7.7.g.
The reintroduction of 177Lu-PSMA therapy has been proposed in relapsed mCRPC patients who initially responded to PSMA-radionuclide therapy experiencing partial remission, but relapsed into progression after a certain period of remission. Several authors have investigated the feasibility of this approach in terms of safety and efficacy. In a retrospective analysis forty seven patients with mCRPC who had biochemical response to initial [177Lu]Lu-PSMA-617 RLT followed by disease progression received at least one (up to three) series of [177Lu]Lu-PSMA-617 RLT rechallenge. After one series of RLT rechallenge, a PSA decline of at least 50% was achieved in 57%. The median PFS of all patients was 8.7 months and the median OS was 22.7 months [1371].
There is an increasing interest in PSMA-targeted alpha therapy (225Ac-PSMA) due to the ability to deliver potent higher local radiation more selectively to cancer cells than PSMA-targeted beta therapy, while minimising unwanted damage to the surrounding normal tissues. Additionally, the intensive radiation to cancer cells results in more effective DNA strand breakage and reduces the development of treatment resistance. A meta-analysis, including nine studies with 263 patients, investigated the therapeutic effects of 225Ac-PSMA RLT in patients with metastatic CRPC, pretreated with chemotherapy, 177Lu-PSMA and/or radium-223. The pooled proportions of patients with more than 50% PSA decline and any PSA decline were 60.99% (95% CI: 54.92%– 66.83%) and 83.57% (95% CI: 78.62%–87.77%), respectively. The estimated mean PFS and mean OS were 9.15 months (95% CI: 6.69–11.03 months) and 11.77 months (95% CI: 9.51–13.49 months), respectively. These findings suggest that 225Ac-PSMA RLT may be an effective treatment option for patients with mCRPC [1372]. Despite the encouraging therapeutic response and survival of patients who received 225Ac-PSMA RLT, major AEs such as xerostomia and severe haematotoxicity must be considered as possible reasons for dose reduction or discontinuation of the therapy.
A retrospective, multicentre international study, WARMTH Act, pooled data of 488 men with mCRPC, who received one or more cycles of 8 MBq ²²⁵Ac-PSMA RLT, across 7 international centres [1373]. Patients were heavily pretreated (docetaxel 66%, cabazitaxel 21%, abiraterone 39%, enzalutamide 39%, ¹⁷⁷Lu-PSMA RLT 32% and 223Ra dichloride 4%). The median follow-up was 9.0 months. The median OS was 15.5 months (95% CI: 13.4–18.3) and median PFS was 7.9 months (CI: 6.8–8.9). In 347 (71%) out of 488 patients, information regarding treatment-induced xerostomia was available, with 236 (68%) of the 347 patients reporting xerostomia after the first cycle of ²²⁵Ac-PSMA RLT. Grade 3 or higher anaemia occurred in 64 (13%) of 488 patients, leukopenia in 19 (4%), thrombocytopenia in 32 (7%), and renal toxicity in 22 (5%). No serious AEs or treatment-related deaths were recorded. This study supports previous data showing that ²²⁵Ac-PSMA RLT has a substantial antitumour effect, being a viable therapy option in heavily pretreated mCRPC patients, including patients after ¹⁷⁷Lu-PSMA RLT. Comparable results, with a median OS of 15 months (95% CI: 10-19; median follow-up 22 months), were reported in a series of patients with mCRPC treated with ²²⁵Ac-PSMA (100-150 kBq/kg at least two cycles, at eight weeks), after becoming resistant to all previous anticancer agents [1374]. The side effect profile remains to be elucidated. So far, ²²⁵Ac-PSMA RLT for mCRPC has not been approved.
Combined therapies, including 177Lu-PSMA radionuclide therapy, in mCRPC have moved into the focus of clinical research. In an open-label, multicentre, randomised, phase II trial, EnzaP, participants not previously treated with docetaxel or an ARPI for mCRPC were randomly assigned (1:1) to enzalutamide 160 mg daily alone or with adaptive-dosed (two or four doses) 7.5 GBq ¹⁷⁷Lu-PSMA-617 intravenous, every 6–8 weeks, based on a 12-week interim PSMA PET/CT [1375]. The primary endpoint was PSA PFS. Overall, 83 men were assigned to the enzalutamide plus 177Lu-PSMA-RLT group, and 79 were assigned to enzalutamide alone. Median PSA PFS was 13.0 months (95% CI: 11.0–17.0) in the enzalutamide plus RLT group and 7.8 months (95% CI: 4·3–11·0) in the enzalutamide group (HR 0.43, 95% CI: 0.29–0.63, p < 0·0001). The most common AEs were fatigue (75%), nausea (47%), and dry mouth (40%) in the enzalutamide plus RLT and fatigue (70%), nausea (27%], and constipation (23%) in the enzalutamide alone group [1375]. The actual benefit of the combined use, in particular, in patients pretreated by one or two ARPIs is still to be proven in larger prospective controlled trials, and a firm recommendation would be premature.
6.7.8.c. PARP inhibitors for mCRPC
So far, two PARP inhibitors as monotherapy, olaparib and rucaparib, are licenced by the FDA (EMA only approved olaparib) and several other PARP inhibitors are under investigation or were approved only in combination with an ARPI (see Section 6.7.6.e).
A randomised phase III trial (PROfound) compared the PARP inhibitor olaparib to an alternative ARPI in mCRPC with alterations in ≥ 1 of any qualifying gene with a role in HRR and progression on an ARPI. Most patients were heavily pretreated with 1–2 chemotherapies and up to 2 ARPIs [341,1197]. Radiographic PFS by blinded independent central review in the BRCA1/2 or ATM mutated population (Cohort A) was the first endpoint and significantly favoured olaparib (HR: 0.49, 95% CI: 0.38–0.63). The final results for OS demonstrated a significant improvement among men with BRCA1/2 or ATM mutations (Cohort A) (p = 0.0175; HR: 0.69, 95% CI: 0.50– 0.97). This was not significant in men with any (other) HRR alteration (Cohort B) (HR: 0.96, 95% CI: 0.63–1.49). Of note, 66% (86 of 131) of patients in the physician’s choice of enzalutamide/abiraterone-arm who progressed crossed over to olaparib.
The most common AEs were anaemia (46.1% vs. 15.4%), nausea (41.4% vs. 19.2%), decreased appetite (30.1% vs. 17.7%) and fatigue (26.2% vs. 20.8%) for olaparib vs. enzalutamide/abiraterone. Among patients receiving olaparib 16.4% discontinued treatment secondary to an AEs, compared to 8.5% of patients receiving enzalutamide/abiraterone. Of patients receiving olaparib 4.3% of patients had a pulmonary embolism, compared to 0.8% among those receiving enzalutamide/abiraterone, none of which were fatal. There were no reports of myelodysplastic syndrome or acute myeloid leukaemia. This was the first trial to show a benefit for genetic testing and precision medicine in mCRPC.
The olaparib approval by the FDA is for patients with deleterious or suspected deleterious germline or somatic HRR gene-mutated mCRPC, who have progressed following prior treatment with enzalutamide or abiraterone. The EMA approved olaparib for patients with BRCA1 and BRCA2 alterations [1376]. The recommended olaparib dose is 600 mg daily (300 mg taken orally twice daily), with or without food.
The FDA approved olaparib for patients with deleterious BRCA mutations (germline and/or somatic) who have been treated with ARPI and a taxane-based chemotherapy [1377]. Approval was based on the results of the single-arm TRITON2 trial (NCT02952534). The confirmed ORR per independent radiology review in 62 patients with deleterious BRCA mutations was 43.5% (95% CI: 31–57) [1378]. Rucaparib second line after ARPI was studied in the TRITON 3 trial and is discussed in Section 6.7.7.e.
The combination of ARPI plus a PARP inhibitor in first-line mCRPC was studied in several RCT including AAP plus Olaparib [1198], AAP plus Niraparib [1202] and Enzalutamide plus Talazoparib [1205]. See Table 6.7.2.
6.7.8.d. Sequencing treatment
6.7.8.d.1. ARPI -> ARPI (chemotherapy-naive mCRPC patients)
The use of sequential ARPIs in mCRPC showed limited benefit in retrospective series as well as in one prospective trial [1379-1386]. In patients who had a short response to the first ARPI for mCRPC (< twelve months), this sequence should be avoided because of known cross resistance and the availability of chemotherapy and PARP inhibitors (if a relevant mutation is present). In the control arm of the contemporary PSMAfore trial, the ARPI-switch showed an rPFS of 5.59 months (4.21–5.95) [1346]. Based on an rPFS benefit Lutetium Lu 177 vipivotide tetraxetan may be approved in Europe, in this setting, soon and would be preferred to an ARPI switch. An FDA approval is already in place and Lutetium Lu 177 vipivotide tetraxetan’s indication now includes PSMA-positive mCRPC post-AR inhibitor therapy where delaying taxane-based chemotherapy is appropriate [1387].
In highly selected patients treated for more than 24 weeks with AAP, the sequence with enzalutamide showed some activity with a median rPFS of 8.1 months (95% CI: 6.1–8.3) and an unconfirmed PSA response rate of 27% [1295]. In case the patient is unfit for chemotherapy and a PARP inhibitor, best supportive care should be considered in case no other appropriate treatment option is available (clinical trial or immunotherapy if MSI-high). An ARPI-ARPI sequence should never be the preferred option but might be considered in such patients if the PS still allows for active treatment and the potential side effects seem manageable.
First prospective cross-over data on an ARPI-ARPI sequence [1379] and a SR and meta-analysis suggest that for the endpoints PFS and PSA PFS, but not for OS, abiraterone followed by enzalutamide is the preferred choice [1388].
6.7.8.d.2. ARPI -> PARP inhibitor
This sequence in patients with deleterious or suspected deleterious germline or somatic HRR gene-mutated mCRPC is supported by data from the randomised phase III PROfound trial studying olaparib [1197] and TRITON 3 studying rucaparib [1299]. A subgroup of patients in PROfound was pretreated with one or two ARPIs and no chemotherapy (35%).
The ARPI-PARP inhibitor sequence versus ARPI-ARPI or ARPI-docetaxel in patients with BRCA 1/2 (and ATM) altered tumours was studied in TRITON-3 and showed a significant rPFS benefit in favour of the PARP inhibitor following the first ARPI. These data underscore the importance of early genomic testing in mCRPC patients and prioritisation of PARP inhibitor treatment very early on in patients with BRCA or HRR mutations. This message is also supported by the AMPLITUDE study, showing a rPFS benefit with the addition of niraparib to abiraterone in patients with mHSPC and HRR mutations (see also chapter 6.7.7.e) [1259].
6.7.8.d.3. Docetaxel for mHSPC -> docetaxel rechallenge
There is limited evidence for second- or third-line use of docetaxel after treatment with docetaxel for mHSPC. Docetaxel seems to be less active than ARPI at progression to mCRPC following docetaxel for mHSPC [1389].
6.7.8.d.4. ARPI -> docetaxel or docetaxel -> ARPI followed by PARP inhibitor
Both olaparib and rucaparib are active in biomarker-selected mCRPC patients after ARPI and docetaxel in either sequence [1197,1377].
6.7.8.d.5. ARPI before or after docetaxel
There is level 1 evidence for both sequences (Table 6.7.3).
6.7.8.d.6. ARPI -> docetaxel -> cabazitaxel or docetaxel -> ARPI -> cabazitaxel
Both third-line treatment sequences are supported by level 1 evidence. Of note, there is high-level evidence favouring cabazitaxel vs. a second ARPI after docetaxel and one ARPI in particular in patients progressing ≤ 12 months on a prior ARPI. CARD is the first prospective randomised phase III trial addressing this question (Table 6.7.3) [1293].
6.7.8.e. Platinum chemotherapy
Cisplatin or carboplatin as monotherapy or combinations have shown limited activity in unselected patients in the pre-docetaxel era [1390]. The combination of cabazitaxel and carboplatin was evaluated in pretreated mCRPC patients in a randomised phase I/II trial. The combination improved the median PFS from 4.5 months (95% CI: 3.5–5.7) to 7.3 months (95% CI: 5.5–8.2; HR: 0.69, 95% CI: 0.50–0.95, p = 0.018) and the combination was well tolerated [1391]. On a histopathological and molecular level, there is preliminary evidence that platinum adds efficacy in patients with aggressive variant PCa molecular signatures including TP53, RB1, and PTEN [1392].
Patients with mCRPC and alterations in DDR genes are more sensitive to platinum chemotherapy than unselected patients [1393], also after progression on PARP inhibitors. Interestingly, in contemporary retrospective series, unselected patients as well as patients without DDR gene alterations also showed a 50% PSA decline when treated with platinum in up to 36% of patients [1360].
In a MA of 23 studies with 901 BRCA-positive mCRPC patients the PSA 50 response rates for PARPi and platinum were 69% (CI: 53–82%), and 74% (CI: 49–90%), respectively. Analyses of OS data showed no difference between PARPi and platinum treatments (HR: 0.86; CI: 0.49-1.52; p = 0.6) [1394]. This analysis supports the use of platinum in patients with BRCA alterations in particular after progression on PARPi or if PARPi are unavailable or suspended due to AEs.
In view of the excellent tolerability of e.g. carboplatin monotherapy, platinum could be offered to patients with far advanced mCRPC harbouring DDR gene aberrations after having progressed on standard treatment options. Prospective controlled trials are ongoing.
6.7.9. Treatment emergent neuroendocrine PCa and neuroendocrine subtype
Neuroendocrine prostate carcinoma (NEPC) is an aggressive variant of PCa, exhibiting characteristics comparable to small cell lung cancer (SCLC). Clinically, NEPC is differentiated based on whether the variant is already dominant at the time of diagnosis or develops only after prior ADT and/or androgen receptor pathway inhibitors (ARPI), distinguishing between de novo or primary NEPC and the treatment-related or so-called treatment-emergent NEPC [1395].
Treatment-related neuroendocrine prostate carcinoma (t-NEPC) is the only neuroendocrine histological subtype, primarily associated with prostate tumours, that is included in the 2022 WHO classification. The incidence of t-NEPC is rising and is estimated to account for between 15 and 20% of all CRPC cases [1396]. In contrast to primary neuroendocrine neoplasms (NENs), t-NEPC arises from adenocarcinomas or from the castration-resistant variant of PCa following potent anti-hormonal therapy through a process called transdifferentiation.
Clinically, it is important to acknowledge that in addition to AR-dependent castration-resistant adenocarcinomas, there is a subgroup of patients with AR-independent tumour biology. The variants of androgen-indifferent PCa (AIPC) include aggressive variant PCa (AVPC), NEPC, and double-negative PCa (DNPC) [1397]. Aggressive variant PCa has been defined as CRPC with at least one of the following characteristics [1392]:
- Histological evidence of small-cell neuroendocrine PCa.
- Exclusively visceral metastases.
- Predominantly lytic bone metastases.
- Extensive lymphadenopathy or large tumour mass in the prostate/pelvis.
- Low PSA level at initial diagnosis plus high tumour volume in the bones.
- Neuroendocrine markers in histology or serum at initial diagnosis or during disease progression plus one of the following: elevated serum lactate dehydrogenase, malignant hypercalcemia, and/or elevated carcinoembryonic antigen (CEA) in the absence of other features.
- Short interval (≤ 6 months) between initiation of hormone therapy and androgen-independent disease progression, with or without neuroendocrine markers,
A small subgroup of AVPC tumours express neither AR nor NE markers and are therefore referred to as Double-negative PCa [1398].
If the transformation into t-NEPC or a pure NEPC is suspected, a tissue biopsy is required to confirm the diagnosis (e.g. new liver metastases and low PSA or signs of AVPC characteristics).
There is no clearly defined, evidence-based standard of care established through comparative data for AVPC. Treatment recommendations are largely guided by therapeutic protocols used for SCLC. It has been suggested to treat locally advanced or metastatic NEPC with a platinum-etoposide chemotherapy regimen with or without an immune checkpoint inhibitor, whilst AR positive PCa with only minor amounts of neuroendocrine de-differentiation should be managed with either docetaxel monotherapy or rather taxane in combination with a platinum agent [1399]. However, no recommendations based on randomised phase III trials can be given.
Table 6.7.1: Phase III randomised controlled trials – nmCRPC
| Study | Intervention | Comparison | Selection criteria | Main outcomes |
| ARAMIS 2019, 2020 [1305,1400] | ADT + darolutamide | ADT + placebo | nmCRPC; baseline PSA ≥ 2ng/mL PSA-DT ≤ 10 mo. | 59% reduction of distant progression or death Median MFS: darolutamide 40.4 vs. placebo 18.4 mo.; 31% reduction in risk of death HR = 0.69 (95% CI: 0.53–0.88) p = 0.003 |
| PROSPER 2018, 2020 [1303,1401] | ADT + enzalutamide | ADT + placebo | nmCRPC; baseline PSA ≥ 2ng/mL PSA-DT ≤ 10 mo. | 71% reduction of distant progression or death Median MFS: enzalutamide 36.6 vs. placebo 14.7 months; 27% reduction in risk of death HR = 0.73 (95% CI: 0.61–0.89) p = 0.001 |
| SPARTAN 2018, 2021 [1304,1402] | ADT + apalutamide | ADT + placebo | nmCRPC; baseline PSA ≥ 2ng/mL PSA-DT ≤ 10 mo. | 72% reduction of distant progression or death Median MFS: apalutamide 40.5 vs. placebo 16.2 months; 22% reduction in risk of death HR = 0.78 (95% CI: 0.64–0.96) p = 0.0161 |
ADT = androgen-deprivation therapy; CI = confidence interval; HR = hazard ratio; MFS = metastasis-free survival; mo. = months; nmCRPC = nonmetastatic castrate-resistant prostate cancer; PSA = prostate-specific antigen;PSA-DT = prostate-specific antigen doubling time.
Table 6.7.2: Phase III randomised controlled trials - first-line treatment of mCRPC
| Study | Intervention | Comparison | Selection criteria | Main outcomes |
| DOCETAXEL | ||||
| SWOG 99-16 2004 [1403] | docetaxel/EMP, every 3 weeks, 60 mg/m2, EMP 3 x 280 mg/day | mitoxantrone, every 3 weeks, 12 mg/m2 prednisone 5 mg BID | OS: 17.52 vs. 15.6 mo. (p = 0.02, HR: 0.80; 95% CI: 0.67–0.97) PFS: 6.3 vs. 3.2 mo. (p < 0.001) | |
| TAX 327 2004, 2008 [1316,1317] | docetaxel, every 3 weeks, 75 mg/m2 prednisone 5 mg BID or docetaxel, weekly, 30 mg/m2 prednisone 5 mg BID | mitoxantrone, every 3 weeks, 12 mg/m2, prednisone 5 mg BID | OS: 19.2 for 3-weekly vs. 17.8 mo. 4-weekly and 16.3 in the control group. (p = 0.004, HR: 0.79, 95% CI: 0.67–0.93) | |
| ABIRATERONE | ||||
| COU-AA-302 2013, 2014, 2015 [1308,1309,1404] | abiraterone + prednisone | placebo + prednisone | No previous docetaxel ECOG 0–1 PSA or radiographic progression No or mild symptoms No visceral metastases | OS: 34.7 vs. 30.3 mo. (HR: 0.81, p = 0.0033) FU: 49.2 mo. rPFS: 16.5 vs. 8.3 mo. (p < 0.0001) |
| ENZALUTAMIDE | ||||
| PREVAIL 2014 [1311] | enzalutamide | placebo | No previous docetaxel ECOG 0–1 PSA or radiographic progression No or mild symptoms 10% had visceral mets | OS: 32.4 vs. 30.2 mo. (p < 0.001). FU: 22 mo. (p < 0.001 HR: 0.71, 95% CI: 0.60–0.84) rPFS: 20.0 mo. vs. 5.4 mo. HR: 0.186 (95% CI: 0.15–0.23) p < 0.0001) |
| SIPULEUCEL-T | ||||
| IMPACT 2010 [1405] | sipuleucel-T | placebo | Some with previous docetaxel ECOG 0–1 Asymptomatic or minimally symptomatic ECOG 0–1 No visceral met. No corticosteroids | OS: 25.8 vs. 21.7 mo. (p = 0.03 HR: 0.78, 95% CI: 0.61–0.98). FU: 34.1 mo. PFS: 3.7 vs. 3.6 mo. (no difference) |
| 2006 [1406] | sipuleucel-T | placebo | OS: 25.9 vs. 21.4 mo. (p = 0.1) FU: 36 mo. PFS: 11.7 vs. 10.0 wk. | |
| COMBINATIONS | ||||
| PROpel [1198,1199] | olaparib (300mg BID) + abiraterone (1000 mg/d) + prednisone (5 mg BID) | placebo + abiraterone + prednisone | ECOG 0-1 regardless of HRRm (retrospective testing) prior taxane for mHSPC allowed | ibPFS in ITT population: 24.8 vs. 16.6 mo.; HR: 0.66; 95% CI: 0.54–0.81; (p = 0.001) ibPFS in BRCA+: HR 0.24; 95% CI: 0.12-0.45 OS in ITT population: 42.1 vs. 38.9 mo.; HR 0.81; 0.95% CI: 0.81, 0.67-1.0; (p= 0,054) OS in BRCA+: HR 0.30; 95% CI: 0.15-0.59 |
| MAGNITUDE [1203,1407] | niraparib 200 mg/d + abiraterone (1,000 mg/d plus prednisone 5 mg BID) | placebo + abiraterone (1,000 mg/d plus prednisone 5 mg BID) | ECOG 0-1 AAP ≤ 4mo allowed for mCRPC HRR-biomarker-positive cohort prior docetaxel for mHSPC allowed prior ARPI for mHSPC allowed prior ARPI for mCRPC allowed | rPFS (central review) in HRR+: 16.5 vs. 13.7 mo. HR = 0.73; 95% CI: 0.56-0.96; (p = 0.022) rPFS (central review) in BRCA 1/ 2+: rPFS 19.5 versus 10.9 months; HR= 0.55; 95% CI 0.39-0.78; (nominal p= 0.0007) |
| TALAPRO-2 [1205,1296,1325-1327] | talazoparib (0.5mg/d) + enzalutamide 160mg/d | enzalutamide + placebo | ECOG 0-1 All comers: HHR deficient and HRR non-deficient or unknown prior AAP or docetaxel allowed for mHSPC | rPFS in ITT: NR (27.5-NR) vs. 21.9 mo.: HR 0.63; 95% CI: 0.51-0.78 (p<0.0001); rPFS 33.1 vs. 19.5 mo.: HR 0·67; 95% CI 0·55–0·81; (p<0·0001); rPFS in BRCA+: HR 0.23; 95% CI: 0.10-0.53 p=0.0002 Cohort 1: OS in ITT (HRR- and HRR+): 45.8 vs. 37.0 mo. - HR 0.80; 95% CI: 0.66–0.96 (p=0.016); OS in HRR+: HR 0.55 95% CI 0.36–0.83; (p=0·0035) HRR- or unknown: HR 0.88; 0.71–1.08 (p=0.22) Cohort 2: ITT= HRR+ OS 45.1 vs. 31.1 mo.: HR 0.62; 95% CI 0.48–0.81; (two-sided p=0·0005); BRCA1/2+: OS NR vs. 28.5 mo.: HR 0.50; 95% CI 0.32–0.78; (p=0·0017); 4-yr., OS 53% vs 23% |
| PEACE-3 [1332] | enzalutamide 160 mg/d+ radium 223 (6 cycles) | enzalutamide | asymptomatic or mildly symptomatic ≥ 2 bone metastases, +/- additional lymph node metastases no visceral metastases prior docetaxel or abiraterone for mHSPC allowed bone protection mandatory (after amendment) | rPFS: 19.4 vs. 16.4 mo.: HR 0.69, 95% CI 0.54-0.87, (p=0.0009), OS: 42.3 vs. 35.0 mo.: HR 0.69, 95% CI 0.52-0.90, (p=0.0031) |
AAP = abiraterone/prednisolone; ARPI = androgen receptor pathway inhibitors; BICR = blinded independent central review; BID = twice a day; BCRA+ = breast cancer gene mutation; CI = confidence interval; ECOG = Eastern Cooperative Oncology Group; EMP = estramustine; FU = follow-up; HR = hazard ratio; HRRm = homologous recombination repair genes mutation; ib = imaging based; IHC = immunohistochemistry; LN = lymph node; mets. = metastases; mo. = months; ib (imaging based); (r)PFS = (radiographic) progression-free survival; PSA = prostate-specific antigen; OS = overall survival; IHC = immunohistochemistry.
Table 6.7.3: Phase II/III randomised controlled trials in second-line/third-line mCRPC
| Study | Intervention | Comparison | Selection criteria | Main outcomes |
| ABIRATERONE | ||||
COU-AA-301 2012 [1339] | abiraterone + prednisone HR | placebo + prednisone | Previous docetaxel ECOG 0–2 PSA or radiographic progression | OS: 15.8 vs. 11.2 mo. (p < 0.0001, HR: 0.74; 95% CI: 0.64–0.86; p < 0.0001). FU: 20.2 mo. rPFS: 5.6 vs. 3.6 mo. |
COU-AA-301 2011 [1338] | OS: 14.8 vs. 10.9 mo. (p < 0.001 HR: 0.65; 95% CI: 0.54–0.77). FU: 12.8 mo. rPFS: 5.6 vs. 3.6 mo. | |||
| Radium-223 | ||||
ALSYMPCA 2013 [1341] | radium-223 | placebo | Previous or no previous docetaxel ECOG 0–2 Two or more symptomatic bone metastases No visceral metastases | OS: 14.9 vs. 11.3 mo. (p = 0.002, HR: 0.61; 95% CI: 0.46–0.81). All secondary endpoints show a benefit over best SOC. |
| CABAZITAXEL | ||||
TROPIC 2013 [1408] | cabazitaxel + prednisone | mitoxantrone + prednisone | Previous docetaxel ECOG 0–2 | OS: 318/378 vs. 346/377 events (OR: 2.11; 95% CI: 1.33–3.33). FU: 25.5 mo. OS ≥ 2 yr. 27% vs. 16% PFS |
TROPIC 2010 [1334] | OS: 15.1 vs. 12.7 mo. (p < 0.0001, HR: 0.70; 95% CI: 0.59–0.83). FU: 12.8 mo. PFS: 2.8 vs. 1.4 mo. (p < 0.0001, HR: 0.74, 95% CI: 0.64–0.86) | |||
CARD 2019 [1293] | cabazitaxel (25 mg/m2 Q3W) + prednisone + G-CSF | ARPI: abiraterone + prednisone OR Enzalutamide | Previous docetaxel Progression ≤ 12 mo. on prior alternative ARPI (either before or after docetaxel) | Med OS 13.6 vs. 11.0 mo. (p = 0.008, HR: 0.64, 95% CI: 0.46–0.89). rPFS 8.0 vs. 3.7 mo. (p < 0.001, HR: 0.54, 95% CI: 0.40–0.73). FU: 9.2 mo. |
| ENZALUTAMIDE | ||||
AFFIRM 2012 [1340] | enzalutamide | Placebo | Previous docetaxel ECOG 0–2 | OS: 18.4 vs. 13.6 mo. (p < 0.001, HR: 0.63; 95% CI: 0.53–0.75). FU: 14.4 mo. rPFS: 8.3 vs. 2.9 mo. (HR: 0.40; 95% CI: 0.35–0.47, p < 0.0001). |
| PARP inhibitor | ||||
PROfound 2020 [341,1197,1351] | olaparib | abiraterone + prednisolone or enzalutamide; cross-over allowed at progression | - Previous ARPI, alterations in HRR genes | rPFS: 7.39 vs. 3.55 mo. (p < 0.0001, HR: 0.34; 95% CI: 0.25–0.47), conf. ORR 33.3% vs. 2.3% (OR 20.86, 95% CI: 4.18–379.18). OS: 19.1 mo vs. 14.7 mo (in patients with BRCA1/2, ATM alterations) (p = 0.0175; HR 0.69; 95% CI: 0.5–0.97). |
| TRITON-3 [1299] | rucaparib (600 mg BID) | docetaxel or abiraterone acetate or enzalutamide | EOCG 0-1 Previous one ARPI BRCA 1/2 or ATM alteration | rPFS: ITT 10.2 mo vs. 6.4 mo, (HR 0.61; 95% CI, 0.47 to 0.80; p < 0.001 for both comparisons) |
| Radioligand therapy | ||||
| VISION 2021 [1365] | 177Lu-PSMA-617 SOC | SOC alone | Previous at least 1 ARPI and one or two taxane regimens; Mandatory: PSMA-positive gallium-68 (68Ga)–labelled PSMA-PET scan | Imaging-based PFS: 8.7 vs. 3.4 mo. (p < 0.001; HR 0.40; 99.2% CI: 0.29–0.57) OS: 15.3 vs. 11.3 mo. (p < 0.001; HR 0.62; 95% CI: 0.5–0.74) |
| TheraP 2021 [1362,1363] | 177Lu-PSMA-617 (8.5 GBq i.v.q. 6-weekly, decreasing 0.5 GBq/cycle; up to 6 cycles) | 177Lu-PSMA-617 1:1 randomisation cabazitaxel (20 mg/m2 i.v.q. 3-weekly, up to 10 cycles) | Post docetaxel Suitable for cabazitaxel | First endpoint PSA reduction of > 50%: 66 vs. 37 PSA responses; 66% vs. 37% by ITT; difference 29% (95% CI: 16–42; p < 0.0001; and 66% vs. 44% by treatment received; difference 23% [9–37]; p = 0.0016). Secondary endpoint OS: 19.1 vs. 19.6 mo (177Lu-PSMA vs. cabazitaxel). HR: 0.97, 95% CI: 0.7-1.4 (p = 0.99) |
| PSMAfore 2023 [1346,1347] | 177Lu-PSMA-617 at a dosage of 7·4 GBq (200 mCi) ± 10%; 6 cycles | 177Lu-PSMA-617 1:1 randomisation to ARPI- change (abiraterone or enzalutamide) | One previous ARPI for mCRPC
No previous taxane in CRPC or mHSPC | First endpoint: rPFS 3rd data cut-off : 11·60 mo (95% CI 9·30–14·19) vs 5·59 mo (4·21–5·95) (HR 0·49 [95% CI 0·39–0·61]) OS: 24.48 vs. 23.13 HR 0.91; 95% CI 0.72–1.14 (p = 0.20) |
*Only studies reporting survival outcomes as primary endpoints have been included.ARPI = androgen receptor pathway inhibitor; CI = confidence interval; CRCP = castrate-resistant prostate cancer; ECOG = Eastern Cooperative Oncology Group; FU = follow-up; GBq = gigabecquerel; G-CFS = granulocyte colony stimulating factor; HR = hazard ratio; HRR = homologous recombination repair; i.v.q. = intravenous quantity; Lu = lutetium; mHSPC = metastatic hormone-sensitive prostate cancer; mo. = months; OS = overall survival; OR = odds ratio; ORR = objective response rate; PET = positron emission tomography; PSA = prostate-specific antigen; PSMA = prostate-specific membrane antigen; (r)PFS = (radiographic) progression-free survival; SOC = standard of care; yr. = years.
6.7.10. Monitoring of treatment
Baseline examinations should include a medical history, clinical examination, as well as baseline blood tests (PSA, total testosterone level, full blood count, renal function, baseline liver function tests, alkaline phosphatase), bone scan and CT of chest, abdomen and pelvis [1409,1410]. The use of choline or PSMA PET/CT scans for progressing CRPC is unclear and most likely not as beneficial as for patients with BCR or hormone-naive disease. Flares, PSMA upregulation and discordant results compared with PSA response or progression on ARPI have been described [1411]. Prostate-specific antigen alone is not reliable enough [1412] for monitoring disease activity in advanced CRPC, since visceral metastases may develop in men without rising PSA [1413]. Instead, the PCWG2 recommends a combination of bone scintigraphy and CT scans, PSA measurements and clinical benefit in assessing men with CRPC [1290]. A majority of experts at the 2015 Advanced Prostate Cancer Consensus Conference (APCCC) suggested regular review and repeating blood profile every two to three months with bone scintigraphy and CT scans at least every six months, even in the absence of a clinical indication [1409]. This reflects that the agents with a proven OS benefit all have potential toxicity and considerable cost and patients with no objective benefit should have their treatment modified. The APCCC participants stressed that such treatments should not be stopped for PSA progression alone. Instead, at least two of the three criteria (PSA progression, radiographic progression and clinical deterioration) should be fulfilled to stop treatment. For trial purposes, the updated PCWG3 put more weight on the importance of documenting progression in existing lesions and introduced the concept of no longer ‘clinically benefiting‘ to underscore the distinction between first evidence of progression and the clinical need to terminate or change treatment [1290]. These recommendations also seem valid for clinical practice outside trials.
6.7.11. When to change treatment
The timing of treatment change for men with metastatic prostate cancer remains a matter of debate, although it is clearly advisable to start or change treatment immediately in men with symptomatic progressing metastatic disease. Any treatment change should preferably precede development of de novo symptoms or worsening of existing symptoms. Although, the number of effective treatments is increasing, head-to-head comparisons are still rare, as are prospective data assessing the sequencing of available agents. Therefore, it is not clear how to select the most appropriate ‘second-line’ treatment, particularly in patients without HRR alterations or other biomarkers. A positive example, however, is the CARD trial, which clearly established cabazitaxel as the better third-line treatment in docetaxel pretreated patients after one ARPI compared to the use of a second ARPI [1293].
The ECOG PS has been used to stratify patients. Generally, men with a PS of 0–1 are likely to tolerate treatments and those with a PS of > 2 are less likely to benefit. However, it is important that treatment decisions be individualised, particularly when symptoms related to disease progression are impacting on PS. In such cases, a trial of active life-prolonging agents to establish if a given treatment will improve the PS may be appropriate. Sequencing of treatment is discussed in the summary papers published following the 2019 and 2022 APCCC Conferences [1414,1415].
6.7.12. Symptomatic management in metastatic castration-resistant prostate cancer
Castration-resistant PCa is usually a debilitating disease often affecting the elderly male. A multidisciplinary approach is required with input from urologists, medical oncologists, radiation oncologists, nurses, psychologists and social workers [1414,1416]. Critical issues of palliation must be addressed when considering additional systemic treatment, including management of pain, constipation, anorexia, nausea, fatigue and depression.
6.7.12.a. Common complications due to bone metastases
Most patients with CRPC have painful bone metastases. External beam RT is highly effective, even as a single fraction [1417,1418]. A single infusion of a third-generation bisphosphonate could be considered when RT is not available [1419]. Common complications due to bone metastases include vertebral collapse or deformity, pathological fractures and spinal cord compression. Cementation can be an effective treatment for painful spinal fracture whatever its origin, clearly improving both pain and QoL [1420]. It is important to offer standard palliative surgery, which can be effective for managing osteoblastic metastases [1421,1422]. Impending spinal cord compression is an emergency. It must be recognised early, and patients should be educated to recognise the warning signs. Once suspected, high-dose corticosteroids must be given and MRI performed as soon as possible. A systematic neurosurgery or orthopaedic surgeon consultation should be planned to discuss a possible decompression, followed by EBRT [1423]. Otherwise, EBRT with, or without, systemic therapy, is the treatment of choice.
6.7.12.b. Preventing skeletal-related events
6.7.12.b.1. Bisphosphonates
Zoledronic acid has been evaluated in mCRPC to reduce skeletal-related events (SRE). This study was conducted when no active anticancer treatments, with the exception of docetaxel, were available. Six hundred and forty-three patients who had CRPC with bone metastases were randomised to receive zoledronic acid 4 or 8mg every three weeks for fifteen consecutive months, or placebo [1424]. The 8mg dose was poorly tolerated and reduced to 4mg but did not show a significant benefit. However, at fifteen and 24 months of follow-up, patients treated with 4mg zoledronic acid had fewer SREs compared to the placebo group (44 vs. 33%, p = 0.021), and in particular fewer pathological fractures (13.1 vs. 22.1%, p = 0.015). Moreover, the time to first SRE was longer in the zoledronic acid group. No survival benefit has been seen in any prospective trial with bisphosphonates.
6.7.12.b.2. RANK ligand inhibitors
Denosumab is a fully human monoclonal antibody directed against RANKL (receptor activator of nuclear factor κ-B ligand), a key mediator of osteoclast formation, function and survival. In M0 CRPC, denosumab has been associated with increased bone-MFS compared to placebo (median benefit: 4.2 months, HR: 0.85, p = 0.028) [1417]. This benefit did not translate into a survival difference (43.9 compared to 44.8 months, respectively) and neither the FDA, nor the EMA have approved denosumab for this indication [1425].
The efficacy and safety of denosumab (n = 950) compared with zoledronic acid (n = 951) in patients with mCRPC was assessed in a phase III trial. Denosumab was superior to zoledronic acid in delaying or preventing SREs as shown by time to first on-study SRE (pathological fracture, radiation or surgery to bone, or spinal cord compression) of 20.7 versus 17.1 months, respectively (HR: 0.82, p = 0.008). Both urinary N-telopeptide and bone-specific alkaline phosphatase were significantly suppressed in the denosumab arm compared with the zoledronic acid arm (p < 0.0001 for both). However, these findings were not associated with any survival benefit and, in a post-hoc re-evaluation of endpoints, denosumab showed identical results when comparing SREs and symptomatic skeletal events [1426].
The potential toxicity (e.g. osteonecrosis of the jaw, hypocalcaemia) of these drugs must always be kept in mind (5–8.2% in M0 CRPC and mCRPC, respectively) [1427,1428]. Patients should have a dental examination before starting therapy, as the risk of jaw necrosis is increased by several risk factors including a history of trauma, dental surgery or dental infection [1429]. In addition, the risk for osteonecrosis of the jaw increased numerically with the duration of use in a pivotal trial [1430] (one year versus two years with denosumab), but this was not statistically significant when compared to zoledronic acid [1425]. According to the EMA, hypocalcaemia is a concern in patients treated with denosumab and zoledronic acid. Hypocalcaemia must be avoided by adequate intake of calcium and vitamin D before initiating therapy [1431]. Hypocalcaemia should be identified and prevented during treatment with bone protective agents (risk of severe hypocalcaemia is 8% and 5% for denosumab and zoledronic acid, respectively) [1428]. Serum calcium should be measured in patients starting therapy and monitored during treatment, especially during the first weeks and in patients with risk factors for hypocalcaemia or on other medication affecting serum calcium. Daily calcium (> 500 mg) and vitamin D (> 400 IU equivalent) are recommended in all patients, except in case of hypercalcaemia [1428,1432,1433].
6.7.13. Summary of evidence and recommendations for life-prolonging treatments of castrate-resistant disease
| Summary of evidence | LE |
| Treatment for mCRPC will be influenced by which treatments patients have already been exposed to. | 4 |
| Recommendations | Strength rating |
| Ensure that testosterone levels are confirmed to be < 50ng/dL before diagnosing castrate-resistant PCa (CRPC). | Strong |
| Counsel, manage and treat patients with metastatic CRPC (mCRPC) in a multidisciplinary team. | Strong |
| Treat patients with mCRPC with life-prolonging agents. | Strong |
| Offer mCRPC patients somatic and/or germline molecular testing, as well as testing for mismatch repair deficiencies or microsatellite instability, if not done previously. | Strong |
6.7.14. Recommendations for systemic treatments of castrate-resistant disease
Summary statement for mCRPC first line combination therapy:
The combination of ARPI plus PARP inhibitors showed a significant rPFS benefit in RCTs for unselected patients. The OS benefit seen with enzalutamide plus talazoparib in the ITT population seems to be driven by the HRR-mutated group. The side effects of PARP inhibitors add substantial toxicity to ARPI monotherapy. Therefore, only a weak recommendation is given for the enzalutamide/talazoparib combination in patients without HRR or BRCA 1/2 mutations.
| Recommendations | Strength rating |
| Base the choice of treatment on the performance status (PS), symptoms, comorbidities, location and extent of disease, genomic profile, patient preference, and on previous treatment for hormone-sensitive metastatic PCa (mHSPC) (alphabetical order: abiraterone, cabazitaxel, docetaxel, enzalutamide, 177lutetium-PSMA-617-radioligand therapy, radium-223, sipuleucel-T and, for patients with DNA homologous recombination repair [HRR] alterations, olaparib, olaparib/abiraterone, niraparib/abiraterone, rucaparib, and talazoparib/enzalutamide). | Strong |
| Avoid sequencing of androgen receptor-targeted agents. | Weak |
| Offer chemotherapy to patients previously treated with an androgen receptor pathway inhibitor (ARPI). | Strong |
| Offer patients with metastatic castrate-resistant PCa (mCRPC) who are candidates for cytotoxic therapy and are chemotherapy naïve docetaxel with 75 mg/m2 every three weeks. | Strong |
| Offer patients previously untreated for mCRPC and harbouring an HRR or breast cancer gene (BRCA) mutation abiraterone in combination with olaparib if the patient is fit for both agents and did not previously receive an ARPI. | Strong |
| Offer patients previously untreated for mCRPC and harbouring a BRCA mutation abiraterone in combination with niraparib if the patient is fit for both agents and did not previously receive an ARPI. | Strong |
| Offer patients previously untreated for mCRPC and harbouring an HRR mutation enzalutamide in combination with talazoparib, if the patient is fit for both agents and did not previously receive an ARPI. | Strong |
| Offer genetically tested patients without known HRR mutations and previously untreated for mCRPC enzalutamide in combination with talazoparib, if the patient is fit for both agents, willing to bear additional side effects, and did not previously receive an ARPI. | Weak |
| Offer poly(ADP-ribose) polymerase (PARP) inhibitor monotherapy to pre-treated mCRPC patients with relevant DNA repair gene mutations. | Strong |
| Offer patients with mCRPC and progression following docetaxel chemotherapy further life-prolonging treatment options, which include abiraterone, cabazitaxel, enzalutamide, radium-223 and olaparib in case of DNA HRR alterations. | Strong |
| Base further treatment decisions regarding mCRPC on PS, previous treatments, symptoms, comorbidities, genomic profile, extent of disease, and patient preference. | Strong |
| Offer cabazitaxel to patients previously treated with docetaxel. | Strong |
| Offer cabazitaxel to patients previously treated with docetaxel who have progressed within 12 months of treatment with abiraterone or enzalutamide for mCRPC. | Strong |
| Offer enzalutamide plus radium-223 to asymptomatic or mildly symptomatic mCRPC patients with bone metastases without visceral metastases. | Strong |
| Offer 177Lu-PSMA-617 to ARPI and docetaxel pre-treated mCRPC patients with one or more metastatic lesions, highly expressing prostate-specific membrane antigen (PSMA) on diagnostic radiolabelled PSMA positron emission tomography/computed tomography (PET/CT) scan and lacking any relevant non-PSMA avid metastases. | Strong |
| Offer 177Lu-PSMA-617 to ARPI pre-treated mCRPC patients with one or more metastatic lesions, highly expressing PSMA (exceeding the uptake in the liver) on diagnostic radiolabelled PSMA PET/CT scan, if not fit for docetaxel. | Weak |
6.7.15. Recommendation for non-metastatic castrate-resistant disease
| Recommendation | Strength rating |
| Offer apalutamide, darolutamide or enzalutamide to patients with M0 castrate-resistant PCa and a high risk of developing metastasis (prostate-specific antigen doubling time < 10 months) to prolong time to metastases and overall survival. | Strong |
Figure 6.4: Treatment of non-metastasized (M0) – asymptomatic * Life expectancy ten years;
* Life expectancy ten years;
** Recommendation based on clinical staging using digital rectal examination, not imaging ;
*** Recommendation based on staging using combination of bone scan and CT;
**** See text, dependent on GG and (biopsy) volume.
1 EBRT: IMRT/VMAT + IGRT of the prostate.
Green = weak recommendation
ADT = androgen deprivation therapy; CT = computed tomography; DRE = digital rectal examination; EBRT = external beam radiotherapy; ECE = extracapsular extension; ePLND = extended pelvic lymph node dissection; GG = grade group; HDR = high-dose rate; IDC = intraducal carcinoma; IGRT = image-guided radiotherapy; IMRT = intensity-modulated radiotherapy; LDR = low-dose rate; PSA = prostate-specific antigen; VMAT = volumetric modulated arc therapy.
Figure 6.5: Treatment of metastasized (M1*) – disease, M+HSPC * Based on staging using combination of bone scan and CT; **Not for low volume, metachronous disease.
* Based on staging using combination of bone scan and CT; **Not for low volume, metachronous disease.
1 EBRT: IMRT/VMAT + IGRT of the prostate (equivalent of up to 72 Gy in 2 Gy fractions).
2 Triple therapy was better than ADT plus docetaxel but randomised data comparing it to ADT plus ARTA is missing.
3 Darolutamide is shown in grey due to it being a weak recommendation.
ARPI = androgen receptor pathway inhibitors; ADT = androgen deprivation therapy; BRCA = breast cancer gene; CT = computed tomography; EBRT = external beam radiotherapy; HSPC = hormone-sensitive metastatic prostate cancer; HRR = homologous recombination repair; IGRT = image-guided radiotherapy; IMRT = intensity-modulated radiotherapy; VMAT = volumetric modulated arc therapy.Note: Please be aware that the various options in the following flowcharts present a generalised approach only and cannot take the management of individual patients into account, nor the availability of resources.