5. DISEASE MANAGEMENT
5.1. Conservative treatment
5.1.1. Watchful waiting
Many men with LUTS are not troubled enough by their symptoms to require drug treatment or surgical intervention. All men with LUTS should be formally assessed prior to any allocation of treatment to establish symptom severity and to differentiate between men with uncomplicated (the majority) and complicated LUTS. Watchful waiting is a viable option for many men with non-bothersome LUTS, as few will progress to AUR and complications (e.g. renal insufficiency or stones) [147, 148], whilst others can remain stable for years [149]. In one study, approximately 85% of men with mild LUTS were stable on WW at one year [150].
A study comparing WW and transurethral resection of the prostate (TURP) in men with moderate LUTS showed the surgical group had improved bladder function (flow rates and PVR volumes), particularly in those with high levels of bother: 36% of WW patients crossed over to surgery within five years, leaving 64% doing well in the WW group [151, 152]. Increasing symptom bother and PVR volumes are the strongest predictors of WW failure. Men with mild-to-moderate uncomplicated LUTS who are not too troubled by their symptoms are suitable for WW.
5.1.2. Behavioural and dietary modifications
This type of management to includes the following components:
- education (regarding the patient’s condition)
- reassurance (that cancer is not a cause of the urinary symptoms)
- periodic monitoring
- lifestyle advice [149, 150, 153, 154] concerning topics such as:
- reduction of fluid intake at specific times aimed at reducing urinary frequency when most inconvenient (e.g. at night or when going out in public)
- avoidance/moderation of intake of caffeine or alcohol, which may have a diuretic and bladder irritant effect, thereby increase fluid output and enhancing frequency, urgency and nocturia
- use of relaxed and double-voiding techniques
- urethral milking to prevent post-micturition dribble
- distraction techniques, such as penile squeeze, breathing exercises, perineal pressure and mental tricks to take the mind off the bladder and toilet, to help control OAB symptoms
- bladder retraining that encourages men to hold on when they have urgency to increase their bladder capacity and the time between voids
- reviewing the medication and optimising the time of administration or substituting drugs for others that have fewer urinary effects (these recommendations apply in particular to diuretics)
- providing necessary assistance when there is impairment of dexterity, mobility or mental state
- treatment of constipation
Evidence exists showing that self-management as part of WW reduces both symptoms and progression [153, 154]. Men randomised to three self-care management sessions in addition to standard care had better symptom improvement and QoL than men treated with standard care only, for up to a year [153]. Two SRs and meta-analyses reported reasonable certainty in estimates that self-management interventions significantly reduced symptom severity in terms of IPSS at six months and improve QoL as compared to usual care [155, 156]. The reduction in IPSS score with self-management was similar to that achieved with drug therapy at six to twelve weeks. Self-management had a smaller, additional benefit at six weeks when added to drug therapy [155]. An RCT evaluating the addition of application-based therapeutic interventions (physical, psychological and behavioural) to standard medical care, reported significant improvements in IPSS, HRQoL, OAB-SF and overall QoL compared to medical care alone [157]. Providing patients with a standardised information booklet that offers guidance on conservative and lifestyle interventions has the potential to improve LUTS outcomes compared with usual care in the primary care setting, as supported by emerging evidence [158].
5.1.3. Practical considerations
The components of self-care management have not been individually studied. The above components of lifestyle advice have been derived from formal consensus methodology [159]. Further research in this area is required.
| Summary of evidence | LE |
| Watchful waiting is usually a safe alternative for men who are less bothered by urinary difficulty or who wish to delay treatment. The treatment failure rate over a period of five years was 21% and 79% of patients were clinically stable. | 1b |
| An additional study reported that 81% of patients were clinically stable on WW after a mean follow-up of 17 months. | 2 |
| Men randomised to three self-management sessions in addition to standard care had better symptom improvement and QoL than men treated with standard care alone at up to a year. Self-care management as part of WW reduces both symptoms and progression. | 1b |
| Self-management achieved a clinically meaningful reduction in symptom severity at six months compared to usual care. There was also a small but significant additional benefit of adding self-management to drug therapy. | 1b |
| Recommendations | Strength rating |
| Offer watchful waiting to men with mild/moderate LUTS who are minimally bothered by their symptoms. | Strong |
| Offer men with LUTS lifestyle advice and self-care information prior to, or concurrent with, treatment. | Strong |
5.2. Pharmacological treatment
5.2.1.α1-Adrenoceptor antagonists (α1-blockers)
Mechanism of action:
α1-blockers aim to inhibit the effect of endogenously released noradrenaline on smooth muscle cells in the prostate and thereby reduce prostate tone and BOO [160]. However, α1-blockers have a small effect on urodynamically determined bladder outlet resistance [161], and treatment-associated improvement of LUTS correlates poorly with obstruction [162]. Therefore, other mechanisms of action may also be relevant.
α1-adrenoceptors located outside the prostate (e.g. urinary bladder and/or spinal cord) and α1-adrenoceptor subtypes (α1B- or α1D-adrenoceptors) may play a role as mediators of effects. α 1-adrenoceptors in blood vessels, other non-prostatic smooth muscle cells and the central nervous system may mediate adverse events.
Currently available α1-blockers are: alfuzosin hydrochloride (alfuzosin); doxazosin mesylate (doxazosin); silodosin; tamsulosin hydrochloride (tamsulosin); terazosin hydrochloride (terazosin); and naftopidil. α1-blockers exist in various formulations. Although different formulations result in different pharmacokinetic and tolerability profiles, the overall difference in clinical efficacy between the difference formulations appears to be negligible.
Efficacy
All α1-blockers have a similar efficacy in appropriate doses [163] and the effects take a few weeks to develop fully, but significant efficacy over placebo can occur within hours to days [162].
Controlled studies show that α1-blockers typically reduce IPSS by approximately 30-40% and increase Qmax by approximately 20-25%. However, substantial improvements also occurred in the corresponding placebo arms [64, 164]. In open-label studies, an IPSS improvement of up to 50% and Qmax increase of up to 40% were documented [64, 164]. A SR and meta-analysis suggested that Qmax variation underestimates the real effect of α1-blockers on BPO, as small improvements in Qmax correspond to relevant improvements in BOO index in PFS [165].
An α1-blocker can reduce both storage and voiding LUTS. Prostate size does not affect α1-blocker efficacy in studies with follow-up periods of less than one year, but α1-blockers do appear to be more efficacious in patients with smaller prostates (< 40mL) in longer-term studies [66,166-169]. The efficacy of α1-blockers is similar across age groups [164]. A pooled analysis of phase III and IV trials of silodosin 8mg demonstrated that improvements in total, storage, voiding and QoL IPSS scores were similar for the severe and not severe LUTS cohorts [170]. Reduction in IPPS and Qmax improvement during α1-blocker treatment appears to be maintained over at least four years [66]. In addition, α1-blockers neither reduce prostate size nor prevent AUR in long-term studies [167-169]. However, evidence suggests that the use of α1-blockers (alfuzosin, tamsulosin and silodosin) improves resolution of AUR [171, 172].
Tolerability and safety
Tissue distribution, subtype selectivity and pharmacokinetic profiles of certain formulations may contribute to the tolerability profile of specific drugs. The most frequent adverse events of α1-blockers are asthenia, dizziness and (orthostatic) hypotension. Vasodilating effects are most pronounced with doxazosin and terazosin and are less common with alfuzosin and tamsulosin [173]. Patients with cardiovascular comorbidity and/or vasoactive comedication may be susceptible to α1-blocker-induced vasodilatation [174]. In contrast, the frequency of hypotension with the α1A-selective blocker silodosin is comparable with placebo [175]. In a large retrospective cohort analysis of men aged > 66 years treated with α1-blockers, the risks of falling (odds ratio [OR] 1.14) and of sustaining a fracture (OR: 1.16) was increased, most likely as a result of induced hypotension [176]. In terms of cardiovascular risk, a large population-based study reported an increased risk of cardiac failure with long-term α-blocker use (hazard radio [HR]: 1.22), which was higher for non-selective α-blockers [177]. Whilst there has been concern about a possible risk of dementia with long-term use of α1-blockers, a large nationwide Finnish case-control study of 24,602 cases and 98,397 controls did not find evidence of a significant association [178].
An adverse ocular event termed intraoperative floppy iris syndrome (IFIS) was reported in 2005, affecting cataract surgery [179]. A meta-analysis on IFIS after alfuzosin, doxazosin, tamsulosin or terazosin exposure showed an increased risk for all α1-blockers [180]. However, the OR for IFIS was much higher for tamsulosin. It appears prudent not to initiate α1-blocker treatment prior to scheduled cataract surgery, and the ophthalmologist should be informed about α1-blocker use.
An SR concluded that α1-blockers do not adversely affect libido or erectile function but can cause abnormal ejaculation (OR: 7.53) [181]. Originally, abnormal ejaculation was thought to be retrograde, but more recent data demonstrate that abnormal ejaculation is due to a decrease or absence of seminal fluid during ejaculation, with young age being an apparent risk factor. In a meta-analysis, ejaculatory dysfunction (EjD) was significantly more common with α1-blockers than with placebo (OR: 5.88). In particular, EjD was significantly more commonly related with tamsulosin or silodosin (OR: 8.57 and 32.5) than placebo, while both doxazosin and terazosin (OR: 0.80 and 1.78) were associated with a low risk of EjD [182]. In the meta-regression, the occurrence of EjD was independently associated with the improvement of urinary symptoms and flow rate, suggesting that the more effective the α1-blocker is the greater the incidence of EjD.
Practical considerations
α1-blockers are usually considered the first-line drug treatment for MLUTS due to their rapid onset of action, good efficacy and low rate and severity of adverse events. However, α1-blockers do not prevent occurrence of urinary retention or need for surgery. Ophthalmologists should be informed about α1-blocker use prior to cataract surgery. Elderly patients treated with non-selective α1-blockers should be informed about the risk of orthostatic hypotension. Sexually active patients treated with selective α1-blockers should be counselled about the risk of EjD.
| Summary of evidence | LE |
| α1-blockers are effective in reducing urinary symptoms (IPSS) and increasing the peak urinary flow rate (Qmax) compared with placebo. | 1a |
| Alfuzosin, terazosin and doxazosin showed a statistically significant increased risk of developing vascular-related events compared with placebo. | 1a |
| Alfuzosin, doxazosin, tamsulosin and terazosin exposure has been associated with an increased risk of IFIS. | 1a |
| Ejaculatory dysfunction is significantly more common with α1-blockers than with placebo, particularly with more-selective α1-blockers such as tamsulosin and silodosin. | 1a |
| Recommendation | Strength rating |
| Offer α1-blockers to men with moderate-to-severe LUTS. | Strong |
5.2.2.5α-reductase inhibitors
Mechanism of action
Androgen effects on the prostate are mediated by dihydrotestosterone (DHT), which is converted from testosterone by the enzyme 5α-reductase [183], and has two isoforms:
- 5α-reductase type 1: predominant expression and activity in the skin and liver
- 5α-reductase type 2: predominant expression and activity in the prostate
Two 5-ARIs are available for clinical use: dutasteride and finasteride. Finasteride inhibits only 5α-reductase type 2, whereas dutasteride inhibits both 5α-reductase types (dual 5-ARI). The 5-ARIs induce apoptosis of prostate epithelial cells [184] leading to prostate size reduction of approximately 18-28% and a decrease in circulating PSA levels of approximately 50% after six to twelve months of treatment [185]. Mean prostate volume and PSA reduction may be even more pronounced after long-term treatment. Continuous treatment reduces the serum DHT concentration by approximately 70% with finasteride and 95% with dutasteride. However, prostate DHT concentration is reduced to a similar level (85-90%) by both 5-ARIs.
Efficacy
Clinical effects relative to placebo are seen after treatment of at least six months. After two to four years of treatment, 5-ARIs improve IPSS by approximately 15-30%, decrease prostate volume by 18-28% and increase Qmax by 1.5-2.0mL/s in patients with LUTS due to BPE [66, 168,169, 186-192]. An indirect comparison and one direct comparative trial (12 months duration) indicated that dutasteride and finasteride are equally effective in the treatment of LUTS [185, 193]. Symptom reduction depends on initial prostate size.
Finasteride may not be more efficacious than placebo in patients with prostates < 40mL [194]. However, dutasteride seems to reduce IPSS, prostate volume and the risk of AUR, and to increase Qmax even in patients with prostate volumes of between 30 and 40mL [195, 196]. A long-term trial with dutasteride in symptomatic men with prostate volumes > 30mL and increased risk for disease progression showed that dutasteride reduced LUTS at least as much as the α1-blocker tamsulosin [168, 192, 197]. The greater the baseline prostate volume (or serum PSA level), the faster and more pronounced the symptomatic benefit of dutasteride as compared to tamsulosin.
5α-reductase inhibitors, but not α1-blockers, reduce the long-term (> one year) risk of AUR or need for surgery [66, 190, 198]. In the PLESS study, finasteride reduced the relative risk of AUR by 57% and need for surgery by 55% (absolute risk reduction 4% and 7%, respectively) at four years compared with placebo [190]. In the MTOPS study, finasteride reduced the relative risk of AUR by 68% and need for surgery by 64% (absolute risk reduction 2% and 3%, respectively), also at four years [66]. A pooled analysis of three RCTs with two-year follow-up data reported that treatment with finasteride decreased the relative risk of AUR by 57% and surgical intervention by 34% (absolute risk reduction 2% for both) in patients with moderately symptomatic LUTS [199]. Dutasteride has also demonstrated efficacy in reducing the risks for AUR and BPO-related surgery. Open-label trials have demonstrated relevant changes in urodynamic parameters [200,201]. A large Danish registry study reported that treatment with 5‐ARI versus α‐blocker monotherapy was associated with a reduced risk of BPO‐related surgery and AUR for up to 15 years of follow‐up, and the absolute risk reduction was 4% [202].
Additionally, finasteride might reduce blood loss during transurethral prostate surgery, probably due to its effects on prostatic vascularisation, although evidence for a clinically significant effect is mixed [203-205].
Tolerability and safety
The most common adverse events are reduced libido, ED and, less frequently, EjD such as retrograde ejaculation, ejaculation failure or decreased semen volume [66,169,185,206]. Gynaecomastia (with breast or nipple tenderness) develops in 1-2% of patients. Two studies have suggested that treatment with 5-ARIs is associated with a higher incidence of high-grade cancers, although no causal relationship has been proven [207-209].
A long-standing debate has been ongoing regarding potential cardiovascular side effects of 5-ARIs, in particular dutasteride [210]. Population-based studies in Taiwan and Ontario did not find an association between the use of 5-ARIs and increased cardiovascular side effects [210, 211]. In a British-Taiwanese population-based cohort study, the risk of type II diabetes was higher in men with 5-ARIs than in men receiving tamsulosin, but did not differ between dutasteride and finasteride [212]. A large Swedish cohort study showed an increased risk of depression with both finasteride (HR 1.61) and dutasteride (HR 1.68), but no long-term association with dementia or suicide risk [213] . An SR found no association between 5-ARI use and depression or suicide risk [214]. However, the European Medicines Agency (EMA), based on an EU-wide review of available data, have issued a warning advising patients taking finasteride to stop treatment and seek medical advice if they experience depressed mood, depression or suicidal ideation [215].
| Summary of evidence | LE |
| After two to four years of treatment, 5-ARIs improve IPSS by approximately 15-30%, decrease prostate volume by 18-28% and increase Qmax by 1.5-2.0mL/s in patients with LUTS due to prostate enlargement. | 1b |
| 5α-reductase inhibitors can reduce risk of LUTS progression with regard to AUR and the need for surgery. Due to their slow onset of action, 5-ARIs are suitable only for long-term treatment (years). | 1a |
| The most relevant urological adverse events of 5-ARIs are related to sexual function and include reduced libido, ED, and less frequently, EjD. | 1b |
| Recommendations | Strength rating |
| Use 5α-reductase inhibitors (5-ARIs) in men who have moderate-to-severe LUTS and an increased risk of LUTS progression (e.g. prostate volume > 40mL). | Strong |
| Counsel patients about the slow onset of action and side effects of 5-ARIs. | Strong |
5.2.3. Muscarinic receptor antagonists
Mechanism of action
The detrusor is innervated by parasympathetic nerves whose main neurotransmitter is acetylcholine, which stimulates muscarinic receptors (M-cholinoreceptors) on the smooth muscle cells. Muscarinic receptors are also present on other cell types, such as bladder urothelial cells and epithelial cells of the salivary glands. Five muscarinic receptor subtypes (M1-M5) have been described, of which M2 and M3 are predominant in the detrusor. The M2 subtype is more numerous, but the M3 subtype is functionally more important in bladder contractions [216, 217]. Antimuscarinic effects might also be induced or modulated through other cell types, such as the bladder urothelium or by the central nervous system [218, 219].
The following muscarinic receptor antagonists are licensed for treating OAB/storage symptoms: darifenacin hydrobromide (darifenacin); fesoterodine fumarate (fesoterodine); oxybutynin hydrochloride (oxybutynin); propiverine hydrochloride (propiverine); solifenacin succinate (solifenacin); tolterodine tartrate (tolterodine); and trospium chloride. Transdermal preparations of oxybutynin have been formulated and evaluated in clinical trials [220, 221].
Efficacy
A subanalysis of an open-label trial of OAB patients showed that age, but not gender, had an impact on urgency, frequency or urgency incontinence [222]. In a pooled analysis, which included a subanalysis of male patients, fesoterodine 8mg was superior to tolterodine extended release (ER) 4mg for the improvement of severe urgency episodes/24 hours and the OAB-q Symptom Bother score at week 12. The urinary retention rate was approximately 2% [223].
The efficacy of antimuscarinics as single agents in men with OAB in the absence of BOO have been tested [224-229]. Most trials lasted only 12 weeks. Four post hoc analyses of large RCTs on the treatment of OAB in women and men without presumed BOO were performed focusing only on the men [225, 230, 231]. Tolterodine can significantly reduce urgency incontinence, daytime or 24-hour frequency and urgency-related voiding whilst improving patient perception of treatment benefit [232]. Solifenacin significantly improved mean patient perception of bladder condition scores, mean OAB questionnaire scores and overall perception of bladder problems. Fesoterodine improved micturition frequency, urgency episodes and UUI episodes. In open-label trials with tolterodine, daytime frequency, nocturia, UUI and IPSS were significantly reduced compared with baseline values after 12 to 25 weeks [226,229]. The TIMES RCT reported that tolterodine ER monotherapy significantly improved UUI episodes per 24 hours at week 12 compared to placebo. Tolterodine ER did not significantly improve urgency, IPSS total or QoL compared with placebo [228].
A further analysis showed that men with PSA levels of < 1.3ng/mL (smaller prostates) might benefit more from antimuscarinics [233]. Two other studies found a positive effect of antimuscarinics in patients with OAB and concomitant BPO [229, 234]. In a small RCT, propiverine improved frequency and urgency episodes [234].
Tolerability and safety
Antimuscarinic drug trials generally show approximately 3-10% withdrawals, which is similar to placebo. Drug-related adverse events include dry mouth (up to 16%), constipation (up to 4%), micturition difficulties (up to 2%), nasopharyngitis (up to 3%) and dizziness (up to 5%).
Increased PVR in men without BOO is minimal and similar to placebo. Nevertheless, fesoterodine 8mg showed higher PVRs (+20.2mL) than placebo (-0.6mL) or fesoterodine 4mg (+9.6mL) [226]. Incidence of urinary retention in men without BOO was similar to placebo for tolterodine (0-1.3% vs. 0-1.4%). With fesoterodine 8mg, 5.3% had symptoms, which was higher than placebo or fesoterodine 4mg (both 0.8%). These symptoms appeared during the first two weeks of treatment and mainly affected men aged 66 years or older.
Theoretically, antimuscarinics might decrease bladder strength, and hence might be associated with PVR or urinary retention. A 12 week safety study on men with mild-to-moderate BOO showed that tolterodine increased the PVR (49mL vs. 16mL) but not AUR (3% in both arms) [235]. The urodynamic effects included larger BVs at first detrusor contraction, higher maximum cystometric capacity and decreased BCI, Qmax was unchanged. This trial indicated that short-term treatment with antimuscarinics in men with BOO is safe [230].
Practical considerations
Not all antimuscarinics have been tested in elderly men, and long-term studies on the efficacy of muscarinic receptor antagonists in men of any age with LUTS are not yet available. In addition, only patients with low PVR volumes at baseline were included in the studies. These drugs should therefore be prescribed with caution, and regular re-evaluation of symptom scores and PVR is advised. Men should be advised to discontinue medication if worsening voiding LUTS or urinary stream is noted after initiation of therapy.
| Summary of evidence | LE |
| Antimuscarinic monotherapy can significantly improve urgency, UUI and increased daytime frequency. | 2 |
| Antimuscarinic monotherapy can be associated with increased PVR after therapy, but acute retention is a rare event in men with a PVR volume of < 150mL at baseline. | 2 |
| Recommendations | Strength rating |
| Use muscarinic receptor antagonists in men with moderate-to-severe LUTS who mainly have bladder storage symptoms. | Strong |
| Do not use antimuscarinic overactive bladder medications in men with a post-void residual volume > 150mL. | Weak |
5.2.4. β3 agonist
Mechanism of action
β3 adrenoceptors are the predominant beta receptors expressed in the smooth muscle cells of the detrusor and their stimulation is thought to induce detrusor relaxation. The mode of action of β3 agonists is not fully elucidated [236].
Efficacy
Mirabegron 50mg is the first clinically available β3 agonist with approval for use in adults with OAB. Mirabegron has undergone extensive evaluation in RCTs conducted in Europe, Australia, North America and Japan [237-241]. Mirabegron demonstrated significant efficacy in treating the symptoms of OAB, including micturition frequency, urgency and UUI, and also patient perception of treatment benefit. These studies had a predominantly female study population. A meta-analysis of eight RCTs including 10,248 patients (27% male) found that mirabegron treatment resulted in reduced frequency, urgency and UUI rates, as well as an improved voided volume with a statistically significant improvement of nocturia compared with both placebo and tolterodine [242]. A meta-analysis of 28 RCTs of 27,481 patients also confirmed that mirabegron treatment resulted in reduced frequency, urgency and UUI rates when compared with placebo. Mirabegron 50mg significantly reduces micturition episodes per 24 hour compared with tolterodine [243].
Mirabegron has been evaluated in male patients with OAB in the context of LUTS either associated or not associated with BPO confirmed by UDS [244]. Mirabegron 25mg daily led to increased satisfaction and improved QoL, but symptoms assessed by validated questionnaires (IPSS and OAB-SS), only improved in non-obstructed patients. Mirabegron as an add-on therapy has been studied in OAB patients with incontinence despite antimuscarinic therapy [245], again in a predominantly female study population. An Asian study with a higher proportion of male subjects (approximately one third) reported superiority over placebo in reducing frequency of micturition but did not report the results separately for the genders [246].
In a study of more than 1,000 patients, of whom approximately 30% were male, combination therapy of mirabegron 25/50mg and solifenacin 5/10mg was associated with statistically significant improvements in patient outcomes and health-related QoL versus solifenacin 5mg and placebo, however, they did not separate out the effects in men and women [247]. In another study, in which 28% patients were male, mirabegron significantly improved patient-reported perception of their condition and QoL, whether patients were incontinent or not [248]. A phase IV study with a small proportion of male subjects reported addition of mirabegron in people with persisting urgency despite solifenacin in a Japanese population [249].
An RCT has suggested Mirabegron might lead improvement of urinary symptoms and sexual function in patients suffering from BPO and concurrent erectile dysfunction (ED) [250]. The study showed a significant improvement in the IIEF-15 total score and IPSS score in patients treated with Mirabegron + Doxazosin [250]. Nevertheless, further multicentre RCTs with large numbers of patients are required to consolidate these data.
Tolerability and safety
The most common treatment-related adverse events in the mirabegron groups were hypertension, UTI, headache and nasopharyngitis [237-240]. Mirabegron is contraindicated in patients with severe uncontrolled hypertension (systolic blood pressure ≥ 180mmHg or diastolic blood pressure ≥ 110mmHg or both). Blood pressure should be measured before starting treatment and should be monitored regularly during treatment. A combination of 13 clinical studies including 13,396 patients, 25% of whom were male, showed that OAB treatments (anticholinergics or mirabegron) were not associated with an increased risk of hypertension or cardiovascular events compared to placebo [251]. The proportion of patients with dry mouth and constipation in the mirabegron groups was notably lower than reported in RCTs of other OAB agents or of the active control tolterodine [237]. Evaluation of urodynamic parameters in men with combined BOO and OAB concluded that mirabegron did not adversely affect voiding urodynamic parameters compared to placebo in terms of Qmax, detrusor pressure at maximum flow and BCI [252]. The overall change in PVR with mirabegron is small [252].
A small prospective study (mainly focused on males) has shown that mirabegron 25mg is safe in patients aged 80 years and above with multiple comorbidities [253]. A pooled analysis of three trials, each of 12 weeks, and a one-year trial, showed a more favourable tolerability profile for mirabegron than antimuscarinics in patients aged > 65 years [254]. The PILLAR phase IV study also showed that, in a large population of 888 patients ≥ 65 years (approximately 30% of males), mirabegron 50mg was safe and effective [255]. In an 18-week study of 3,527 patients (23% male), the incidence of adverse events was higher in the combination (solifenacin 5mg plus mirabegron 25mg) group (40%) than the group prescribed mirabegron 25mg alone (32%). Events recorded as urinary retention were low (< 1%) but were reported slightly more frequently in the combined group when compared with the monotherapy and placebo groups. The PVR volume was slightly increased in the combined group compared with solifenacin 5mg and the mirabegron monotherapy and placebo groups. Combined therapy with solifenacin 5mg plus mirabegron 25mg and solifenacin 5mg plus mirabegron 50mg provided improvements in efficacy generally consistent with an additive effect [256].
In a retrospective analysis of persistence and adherence in 21,996 patients, 30% of whom were male, the median time to discontinuation was significantly longer for mirabegron (169 days) compared to tolterodine (56 days) and other antimuscarinics (30-78 days). There was no statistical difference between men and women [257].
The phase III EMPOWUR trial comparing vibegron to placebo and tolterodine showed once daily 75mg vibegron provided statistically significant reductions in micturitions, urgency episodes and UUI [258]. Treatment was well tolerated, with a favourable safety profile. However, the majority of the study population (85%) were female.
Practical considerations
Long-term studies on the efficacy and safety of mirabegron in men with LUTS are not yet available. Available studies on mirabegron in combination with antimuscarinics in OAB patients had a predominantly female study population, while further trials are still pending. Further evidence in male patients using vibegron is awaited.
| Summary of evidence | LE |
| β3 agonists improve storage LUTS, including urinary frequency, urgency and UUI. | 2 |
| Patients prescribed mirabegron remained on treatment longer than those prescribed antimuscarinics. | 3 |
| Recommendation | Strength rating |
| Use β3 agonists in men with moderate-to-severe storage LUTS. | Strong |
*Further evidence is awaited for vibegron.
5.2.5. Phosphodiesterase type 5 inhibitors
Mechanism of action
Phosphodiesterase type 5 inhibitors (PDE5Is) increase intracellular cyclic guanosine monophosphate, thus reducing smooth muscle tone of the detrusor, prostate and urethra. Nitric oxide and PDE5Is might also alter reflex pathways in the spinal cord and neurotransmission in the urethra, prostate or bladder [259]. Moreover, chronic treatment with PDE5Is appears to increase blood perfusion and oxygenation in the LUT [260]. Phosphodiesterase type 5 inhibitors could also reduce chronic inflammation in the prostate and bladder [261]. The exact mechanism of PDE5Is on LUTS remains unclear.
Although clinical trials of several selective oral PDE5Is have been conducted in men with LUTS, only tadalafil (5mg once daily) has been licensed for the treatment of MLUTS.
Efficacy
Randomised controlled trials have demonstrated that PDE5Is reduce IPSS, storage and voiding LUTS and improve QoL. However, Qmax did not significantly differ from placebo in most trials [262]. A Cochrane review included a total of 16 RCTs that examined the effects of PDE5Is compared to placebo and other standard of care drugs (α1-blockers and 5-ARIs) in men with LUTS [263]. In the updated meta-analysis, PDE5Is led to a small reduction (mean difference [MD] 1.89 lower; 95% confidence interval [CI]: 2.27 to 1.50 lower; n = 4293) in IPSS compared to placebo [263]. There was no difference between PDE5Is and α1-blockers in IPSS [264]. Most evidence was limited to short-term treatment up to 12 weeks. In other meta-analyses, PDE5Is were also found to improve IPSS and IIEF score, but not always Qmax [265, 266]. A meta-regression suggested that younger men with low body mass index and more severe LUTS benefit the most from treatment with PDE5Is [265].
In a post hoc analysis of data pooled from four blinded trials of tadalafil 5mg versus placebo once daily, a minimum improvement of 25% in IPSS score was found in 60% in the tadalafil and in 44% in the placebo group [267]. The maximum trial duration was 52 weeks [268]. A subgroup analysis of pooled data from four RCTs demonstrated a significant reduction in LUTS, regardless of baseline severity, age, previous use of α-blockers or PDE5Is, total testosterone level or predicted prostate volume [269]. In a post hoc analysis of pooled data from four RCTs, tadalafil was shown to also be effective in men with cardiovascular risk factors/comorbidities, except for patients receiving more than one antihypertensive medication. Among sexually active men > 45 years, tadalafil improved both LUTS/BPH and ED [269].
An integrated data analyses from four placebo controlled clinical studies showed that total IPSS improvement was largely attributed to direct (92.5%) versus indirect (7.5%) treatment effects by means of IIEF-erectile function improvement [270]. Another analysis showed a small but significant increase in Qmax without any effect on PVR [271]. An integrated analysis of RCTs showed that tadalafil was not superior to placebo for IPSS improvement at 12 weeks in men ≥ 75 years (with varied effect size between studies), but was for men < 75 years [272]. An open label urodynamic study of 71 patients showed significant improvements in both voiding and storage symptoms, confirmed by improvements in BOO index (61.3 to 47.1), and resolution of DO in 15 (38%) of 38 patients. Flow rate improved from 7.1 to 9.1mL/s and mean IPSS from 18.2 to 13.4 [273].
A multicentre, double blind, placebo controlled RCT compared once daily tadalafil 20mg versus placebo during 12 weeks in men with LUTS with or without BOO. Urodynamic measures including detrusor pressure at maximum urinary flow rate, Qmax, maximum detrusor pressure, BOO or bladder capacity remained largely unchanged during the study with no statistically significant or clinically adverse event differences between tadalafil and placebo [274].
A study has shown that, in patients with OAB and LUT obstruction unresponsive to previous treatment, the simultaneous administration of solifenacin, tadalafil and dutasteride could be an effective and safe choice [275].
Tolerability and safety
Reported adverse effects in RCTs comparing the effect of all PDE5Is versus placebo in men with LUTS, include flushing, gastroesophageal reflux, headache, dyspepsia, back pain and nasal congestion [265].
Tadalafil is contraindicated in patients using nitrates or guanylate cyclase stimulators, such as Riociguat, and in men with cardiac disease for whom sexual activity is inadvisable [276]. Tadalafil is also contraindicated in patients with myocardial infarction within the last 90 days; patients with unstable angina or angina occurring during sexual intercourse; patients with New York Heart Association Class 2 or greater heart failure in the last six months; patients with uncontrolled arrhythmias, hypotension (< 90/50mm Hg), or uncontrolled hypertension; patients with a stroke within the last six months; or if anterior ischaemic optic neuropathy with sudden loss of vision is known or was reported after previous use of PDE5Is [276]. Detailed information regarding tolerability/safety of all available PDE5Is for the treatment of ED in men treated with α-blockers for LUTS are provided in the EAU Guidelines on Sexual and Reproductive Health [277].
Practical considerations
To date, only tadalafil 5mg once daily has been officially licensed for the treatment of MLUTS with or without ED. Long-term experience with tadalafil in men with LUTS is limited to one trial with a one-year follow-up [268], limiting conclusions about efficacy or tolerability greater than one year. There is limited information available on reduction of prostate size and no data on disease progression.
| Summary of evidence | LE |
| Phosphodiesterase type 5 inhibitors significantly improve IPSS and IIEF score but not Qmax. | 1a |
| Recommendation | Strength rating |
| Use phosphodiesterase type 5 inhibitors in men with moderate-to-severe LUTS with or without erectile dysfunction. | Strong |
5.2.6. Plant extracts - phytotherapy
Potential mechanism of action
Herbal drug preparations are made of roots, seeds, pollen, bark or fruits. Single plant preparations (monopreparations) and preparations are available that combine two or more plants in one pill (combination preparations) [278].
Possible relevant compounds include phytosterols, ß-sitosterol, fatty acids and lectins [278]. In vitro, plant extracts can have anti-inflammatory, antiandrogenic and oestrogenic effects; decrease sexual hormone-binding globulin; inhibit aromatase, lipoxygenase, growth factor-stimulated proliferation of prostatic cells, α-adrenoceptors, 5 α-reductase, muscarinic acetylcholine receptors, dihydropyridine receptors and vanilloid receptors; and neutralise free radicals [273, 278, 279]. The in vivo effects of these compounds are uncertain, and the precise mechanisms of plant extracts remain unclear.
Efficacy
The extracts of the same plant produced by different companies do not necessarily have the same biological or clinical effects. Therefore, the effects of one brand cannot be extrapolated to others [280]. In addition, batches from the same producer may contain different concentrations of active ingredients [281]. A review of recent extraction techniques and their impact on the composition/biological activity of available Serenoa repens-based products showed that results from different clinical trials must be compared strictly according to the same validated extraction technique and/or content of active compounds [282], because the pharmacokinetic properties of the various preparations can vary significantly.
Heterogeneity and a limited regulatory framework characterise the current status of phytotherapeutic agents. The EMA has developed the Committee on Herbal Medicinal Products (HMPC). European Union (EU) herbal monographs contain the HMPC’s scientific opinion on safety and efficacy data about herbal substances and their preparations intended for medicinal use. The HMPC evaluates all available information, including non-clinical and clinical data, whilst also documenting long-standing use and experience in the EU. European Union monographs are divided into two sections: a) well-established use (marketing authorisation) - when an active ingredient of a medicine has been used for more than ten years and its efficacy and safety have been well established (including a review of the relevant literature); and b) Traditional use (simplified registration) - for herbal medicinal products that do not fulfil the requirements for a marketing authorisation, but for which sufficient safety data and plausible efficacy is available based on long-standing use and experience.
The HPMC periodically invites all interested parties to submit any scientific data that the Committee should consider during their periodic review of the monographs. Table 1 lists the available EU monographs for herbal medicinal products and the current calls for update.
Table 1: European Union monographs for herbal medicinal products [283]
| Herbal substance | HMPC evaluation | Therapeutic Indication by HMPC | Date of monograph |
| Serenoa repens, fructus (saw palmetto, fruit) Extraction solvent: hexane [284] | Well-established use | Symptomatic treatment of BPH | 14/01/2016 Addendum 1/9/21** |
| Serenoa repens, fructus (saw palmetto, fruit) Extraction solvent: ethanol [284] | Traditional use | LUTS related to BPH* | 14/01/2016 Addendum 1/9/21** |
| Cucurbita pepo L, semen (pumpkin seed) Preparation as defined in the monograph [285] | Traditional use | LUTS related to BPH or related to an OAB* | 25/03/2013 Call ended 30/4/21 |
| Prunus africana (Hook f.) Kalkm., cortex (pygeum africanum bark) Preparation as defined in the monograph [286] | Traditional use | LUTS related to BPH* | 01/09/2017 No call for update |
| Urtica dioica L., Urtica urens L., their hybrids or their mixtures, radix Preparation as defined in the monograph [287] | Traditional use | LUTS related to BPH* | 05/11/2012 Call ended 30/6/21 |
| Epilobium angustifolium L. and/or Epilobium parviflorum Schreb., herba (Willow herb) Preparation as defined in the monograph [288] | Traditional use | LUTS related to BPH* | 13/01/2016 No call for update |
*After serious conditions have been excluded by a medical doctor.
**Addendum concluded that no revision was required.
Panel interpretation
Only hexane-extracted Serenoa repens (HESr) has been recommended for well-established use by the HMPC. Based on this, a detailed scoping search covering the timeframe between the search cut-off date of the EU monograph and May 2021 was conducted for HESr.
A large meta-analysis of 30 RCTs with 5,222 men and follow-up ranging from four to 60 weeks demonstrated no benefit of treatment with S. repens in comparison to placebo for the relief of LUTS [289]. It was concluded that S. repens was not superior to placebo, finasteride or tamsulosin with regard to IPSS improvement, Qmax or prostate size reduction. However, the similar improvement in IPSS or Qmax compared with finasteride or tamsulosin could be interpreted as treatment equivalence. Importantly, in the meta-analysis, various brands of S. repens were included regardless of the presence of HESr as the main ingredient in the extract.
Another SR focused on data from 12 RCTs on the efficacy and safety of HESr [290]. This review concluded that HESr was superior to placebo in terms of improvement of nocturia and Qmax in patients with enlarged prostates. Improvement in LUTS was similar to tamsulosin and short-term use of finasteride. An updated SR analysed 15 RCTs and also included 12 observational studies. The review confirmed the results of the previous SR on the efficacy of HESr [291]. Compared with placebo, HESr was associated with 0.64 (95% CI: 0.98-0.31) fewer voids/night and an additional mean increase in Qmax of 2.75mL/s (95% CI: 0.57-4.93); both were significant. When compared with α-blockers, HESr showed similar improvements in IPSS (WMD 0.57; 95% CI: 0.27-1.42) and a comparable increase in Qmax when compared to tamsulosin (WMD 0.02; 95% CI: 0.71-0.66). Efficacy assessed using IPSS was similar after six months of treatment between HESr and 5-ARIs. Analysis of all available published data for HESr showed a mean significant improvement in IPSS from baseline of 5.73 points (95% CI: 6.91-4.54) [291].
A network meta-analysis tried to compare the clinical efficacy of S. repens (HESr and non-HESr) against placebo and α1-blockers in men with LUTS. Interestingly, only two RCTs on HESr were included in the analysis. The results of the analysis showed that S. repens achieved no clinically meaningful improvement against placebo or α1-blockers in short-term follow-up. However, S. repens showed a clinical benefit after a prolonged period of treatment, and HESr demonstrated a greater improvement than non-HESr in terms of IPSS [292].
With respect to safety and tolerability, data from the SRs showed that HESr had a favourable safety profile, with gastrointestinal disorders being the most frequent adverse effects (mean incidence 3.8%), while HESr had very limited impact on sexual function.
A cross-sectional study compared the combination of HESr with silodosin-to-silodosin monotherapy in patients treated for at least 12 months (mean duration 13.5 months) [293]. It was reported that 69.9% of the combination therapy patients achieved the predefined clinically meaningful improvement (improvement of more than three points in baseline IPSS) compared to 30.1% of patients treated with silodosin only. In addition, a greater than 25% improvement in IPSS was found in 68.8% and 31.2% of the patients in the combination and the monotherapy groups, respectively. These data suggest that a combination of an α1-blocker with HESr may result in greater clinically meaningful improvements in LUTS compared to α1-blocker monotherapy [293].
Practical considerations
Available RCTs do not use the same endpoints (e.g. IPSS). More studies on the use of HESr in combination with other pharmacotherapeutic agents for MLUTS are pending. There is a need to define the subpopulation of patients who will benefit most from therapy with HESr.
| Summary of evidence | LE |
| Hexane-extracted Serenoa repens improves Qmax and results in fewer voids/night (0.64 [95% CI: 0.98-0.31]) compared to placebo. | 2 |
| Hexane-extracted Serenoa repens has a very limited negative impact on sexual function. | 2 |
| Recommendations | Strength rating |
| Offer hexane-extracted Serenoa repens to men with LUTS who want to avoid any potential adverse events related in particular to sexual function. | Weak |
| Inform the patient that the magnitude of efficacy may be modest with hexane extracted Serenoa repens. | Strong |
5.2.7. Combination therapies
5.2.7.a. α1-blockers + 5α-reductase inhibitors
Mechanism of action
Combination therapy consists of an α1-blocker (Section 5.2.1) together with a 5-ARI (Section 5.2.2). The α1-blocker exhibits clinical effects within hours or days, whereas the 5-ARI needs several months to develop full clinical efficacy. Finasteride has been tested in clinical trials with alfuzosin, terazosin, doxazosin or terazosin, and dutasteride with tamsulosin.
Efficacy
Several studies have investigated the efficacy of combination therapy against an α1-blocker, 5-ARI or placebo alone. Initial studies with follow-up periods of six to twelve months demonstrated that the α1-blocker was superior to finasteride in symptom reduction, whereas combination therapy of both agents was not superior to α1-blocker monotherapy [187, 188, 294]. In studies with a placebo arm, the α1-blocker was consistently more effective than placebo, but finasteride was not. Data at one year in the MTOPS study showed similar results [66].
Long-term data (four years) from the MTOPS and CombAT studies showed that combination treatment is superior to monotherapy for symptoms and Qmax, and superior to α1-blocker alone in reducing the risk of AUR or need for surgery [66, 168, 169].
The CombAT study demonstrated that combination treatment is superior to either monotherapy regarding symptoms and flow rate starting from month nine, and superior to α1-blocker for AUR and the need for surgery after eight months [169]. The differences in MTOPS may therefore reflect different inclusion and exclusion criteria and baseline patient characteristics.
Discontinuation of the α1-blocker after six to nine months of combination therapy was investigated in an RCT and an open-label multicentre trial [295, 296]. The first trial evaluated the combination of tamsulosin with dutasteride and the impact of tamsulosin discontinuation after six months [295], with nearly three quarters of patients reporting no worsening of symptoms. However, patients with severe symptoms (IPSS > 20) at baseline may benefit from longer combination therapy.
A trial evaluated the symptomatic outcome of finasteride monotherapy at three and nine months after discontinuation of nine-month combination therapy [296]. Lower urinary tract symptom improvement after combination therapy was sustained at three months (IPSS difference 1.24) and nine months (IPSS difference 0.4). The limitations of studies include short duration and short follow-up period after discontinuation.
In both the MTOPS and CombAT studies, combination therapy was superior to monotherapy in preventing clinical progression as defined by an IPSS increase of at least four points, AUR, UTI, incontinence or an increase in creatinine > 50%. The MTOPS study found that the risk of long-term clinical progression (primarily due to increasing IPSS) was reduced by 66% with combined therapy versus placebo and to a greater extent than with either finasteride or doxazosin monotherapy (34% and 39%, respectively) [66]. In addition, finasteride (alone or in combination), but not doxazosin alone, significantly reduced both the risks of AUR and the need for BPO-related surgery over the four-year study. In the CombAT study, combination therapy reduced the relative risks of AUR by 68%, BPO-related surgery by 71% and symptom deterioration by 41% compared with tamsulosin, after four years [297]. To prevent one case of urinary retention and/or surgical treatment, 13 patients need to be treated for four years with dutasteride and tamsulosin combination therapy compared to tamsulosin monotherapy, while the absolute risk reduction (risk difference) was 7.7%.
The CONDUCT study compared efficacy and safety of a fixed-dose combination of dutasteride and tamsulosin to a WW approach with the potential initiation of tamsulosin (step-up approach) in a two-year RCT with a total of 742 patients. In both arms, detailed lifestyle advice was given. This fixed-dose combination resulted in a rapid and sustained improvement in men with moderate LUTS at risk of disease progression - the difference in IPSS at 24 months was 5.4 in the active arm and 3.6 in the placebo arm [298]. Moreover, tamsulosin plus dutasteride significantly reduced the relative risk of clinical progression (mainly characterised as a worsening in symptoms) by 43.1% when compared with WW, with an absolute risk reduction of 11.3% (number needed to treat [NNT] = 9).
The influence of baseline variables on changes in IPSS after combination therapy with dutasteride plus tamsulosin or either monotherapy was tested based on the four-year results of the CombAT study. Combination therapy provided consistent improvement of LUTS over tamsulosin across all analysed baseline variables at 48 months [299].
A combination of the 5-ARI finasteride and tadalafil 5mg was tested in a large-scale RCT against finasteride monotherapy. This study supports the concept of this novel combination therapy and is described in more detail in Section 5.2.5 [300].
Tolerability and safety
Adverse events for both drug classes have been reported with combination treatment [66,168,169]. The adverse events observed during combination treatment were typical of α1-blockers and 5-ARIs. The frequency of adverse events was significantly higher for combination therapy. The MTOPS study demonstrated that the incidence of treatment-related adverse events is higher during the first year of combined treatment between doxazosin and finasteride [301]. A meta-analysis measuring the impact of medical treatments for LUTS/BPH on ejaculatory function reported that combination therapy with α1-blockers and 5-ARIs resulted in a threefold increased risk of EjD compared with each monotherapy [182].
Practical considerations:
Compared with α1-blockers or 5-ARI monotherapy, combination therapy results in a greater improvement in LUTS and increase in Qmax and is superior in prevention of disease progression. However, combination therapy is also associated with a higher rate of adverse events. Combination therapy should therefore be prescribed primarily in men who have moderate-to-severe LUTS who are at risk of disease progression (higher prostate volume, higher PSA concentration, advanced age, higher PVR, lower Qmax, etc.). Combination therapy should only be used when long-term treatment (more than 12 months) is intended, and patients should be informed of this.
| Summary of evidence | LE |
| Long-term data (four years) from the MTOPS and CombAT studies showed that combination treatment is superior to monotherapy for symptoms and Qmax, and superior to α1-blocker alone in reducing the risk of AUR or need for surgery. | 1b |
| The MTOPS study found that the risk of long-term clinical progression (primarily due to increasing IPSS) was reduced by 66% with combined therapy versus placebo and to a greater extent than with either finasteride or doxazosin monotherapy. | 1b |
| The CombAT study found that combination therapy reduced the relative risks of AUR by 68%, BPH-related surgery by 71% and symptom deterioration by 41% compared with tamsulosin after four years. | 1b |
| Adverse events of both drug classes are seen with combined treatment using α1-blockers and 5-ARIs. | 1b |
| Recommendation | Strength rating |
| Offer combination treatment with an α1-blocker and a 5α-reductase inhibitor to men with moderate-to-severe LUTS and an increased risk of disease progression (e.g. prostate volume > 40mL). | Strong |
5.2.7.b. α1-blockers + muscarinic receptor antagonists
Mechanism of action
Combination treatment consists of an α1-blocker together with an antimuscarinic aiming to antagonise both α1-adrenoceptors and muscarinic receptors. The possible combinations have not all been tested in clinical trials to date.
Efficacy
Several RCTs and prospective studies investigated combination therapy lasting four to twelve weeks, either as an initial treatment in men with OAB and presumed BPO or as a sequential treatment for storage symptoms persisting while on an α1-blocker [221, 232, 297, 302-306]. Combination treatment has marginal efficacy in reducing urgency, UUI, voiding frequency, nocturia or IPSS compared with α1-blockers or placebo alone and improves QoL [232, 306]. An SR and meta-analysis of RCTs (six studies of treatment-naïve patients and five studies of men with persistent storage LUTS, despite prior treatment with α-blockers) concluded that the addition of antimuscarinics to α-blockers marginally reduced the number of micturition episodes per day (standard MD -0.19) but did not have a significant impact on the number of urgency episodes, and had a higher side-effect profile [307]. Similar findings were also reported in a Cochrane review of RCTs of men with LUTS secondary to BPO, with a small improvement in IPSS (MD 2.04) and QoL (MD 0.71) with combination therapy compared to α-blocker monotherapy, although overall certainty of evidence (COE) was deemed moderate to very low [308].
Symptom improvement is higher regardless of PSA concentration with combination therapy, whereas tolterodine alone improved symptoms mainly in men with a serum PSA of < 1.3ng/mL [233].
In a meta-analysis of 16 studies with 3,548 patients with BPH/OAB, initial combination treatment of an α1-blocker with anticholinergic medication showed no difference in total IPSS and Qmax between the two group [309].
Effectiveness of therapy is evident primarily in those men with moderate-to-severe storage LUTS [310]. Long-term use of combination therapy has been reported in patients receiving treatment for up to one year, showing symptomatic response is maintained, with a low incidence of AUR [311]. In men with moderate-to-severe storage symptoms, voiding symptoms and PVR < 150mL, the reduction in symptoms using combination therapy is associated with patient-relevant improvements in health-related QoL compared with placebo and α1-blocker monotherapy [312].
Combined behavioural and drug therapy yielded greater improvements in OAB symptoms than drug therapy alone, but not behavioural therapy alone in an RCT evaluating the effectiveness of combined behavioural strategies and drug therapy for OAB symptoms in men [313].
Tolerability and safety
Adverse events of both drug classes are seen with combined treatment using α1-blockers and antimuscarinics. The most common side effect is dry mouth. Some side effects (e.g. dry mouth or ejaculation failure) may show increased incidence which cannot simply be explained by summing the incidence with the drugs used separately. Increased PVR may be seen, but is usually not clinically significant, and risk of AUR is low up to one year of treatment [228, 314, 315]. Antimuscarinics do not cause evident deterioration in Qmax used in conjunction with an α1-blocker in men with OAB symptoms [306, 316].
An RCT investigated safety in terms of maximum detrusor pressure and Qmax for solifenacin (6mg or 9mg) with tamsulosin in men with LUTS and BOO compared with placebo [317]. The combination therapy was noninferior to placebo for the primary urodynamic variables and Qmax was increased versus placebo [317].
Practical considerations
Class effects are likely to underlie efficacy and QoL using an α1-blocker and antimuscarinic. Trials mainly used storage symptom endpoints, were of short duration, and included only men with low PVR volumes at baseline. Measuring PVR is therefore recommended during combination treatment.
| Summary of evidence | LE |
| Combination treatment with α1-blockers and antimuscarinics is effective for improving LUTS-related QoL impairment. | 2 |
| Combination treatment with α1-blockers and antimuscarinics is more effective for reducing urgency, UUI, voiding frequency, nocturia or IPSS compared with α1-blockers or placebo alone. | 2 |
| Adverse events of both drug classes are seen with combined treatment using α1-blockers and antimuscarinics. | 1 |
| There is a low risk of AUR using α1-blockers and antimuscarinics in men known to have a PVR volume of < 150mL. | 2 |
| Recommendations | Strength rating |
| Use combination treatment of a α1-blocker with a muscarinic receptor antagonist in patients with moderate-to-severe LUTS if relief of storage symptoms has been insufficient with monotherapy with either drug. | Weak |
| Do not prescribe combination treatment in men with a post-void residual volume > 150mL. | Weak |
5.2.7.c. α1-blockers + Beta-3 agonist
Mechanism of action
Combination therapy consists of an α1-blocker (Section 5.2.1) together with a β₃-agonist (Section 5.2.4) as an add-on therapy in males receiving α1-blockers with persisting OAB symptoms.
Efficacy
The MATCH study explored the effect of the addition of mirabegron 50mg to tamsulosin 0.2mg compared to tamsulosin plus placebo in 544 patients [318]. A statistically significant difference of 0.52 voids per day was seen in favour of mirabegron. Total IPSS score also improved but was not significant between the groups. Another RCT evaluated add-on therapy with mirabegron for OAB symptoms persisting after treatment with tamsulosin 0.2mg daily in men with BPO [319]. Combination therapy was associated with greater improvements in OAB symptom score, in urinary urgency and daytime frequency, as well as the storage subscore of IPSS and QoL index compared to monotherapy with tamsulosin [320].
The PLUS phase IV trial [319] compared mirabegron and placebo in a population of males treated with a standard dose of tamsulosin 0.4mg. After a four-week run-in period of treatment with tamsulosin 0.4mg alone, 715 patients were randomised between placebo and mirabegron 25mg, upgraded to 50mg after one month. While mean number of micturition’s was significantly reduced in the experimental arm, the effect size was deemed as low (mean adjusted difference of 0.39 voids per day). Similar results were seen for mean voided volume and urgency episodes, but total IPSS, IPSS subscores and OAB-q symptom score were not significantly different between the groups.
An RCT comparing the efficacy of mirabegron 50mg or fesoterodine 4mg add-on therapy to silodosin in LUTS patients with persisting OAB symptoms reported that, at three months, fesoterodine add-on therapy showed a significantly greater improvement than mirabegron add-on therapy in OAB symptom score and urgency score and IPSS-QoL [247]. Fesoterodine was also superior in alleviating DO.
An RCT (the COURAGE trial) comparing the efficacy of vibegron 75mg versus placebo as add-on therapy to an α1-blocker (initiated > three months before screening) with or without a 5 α-reductase inhibitor (initiated > six months before screening) in LUTS patients with persisting OAB symptoms, reported that vibegron add-on therapy showed a significantly greater improvement at 12 weeks than placebo in daily micturition’s and urgency episodes. Vibegron was also associated with significant improvements versus placebo at week 12 in nocturia and UUI episodes, in IPSS storage score changes and volume voided [321].
Tolerability and safety
In the MATCH study, main adverse events were in line with previous trials, and cardiovascular events were uncommon in the studied populations [318]. The PLUS phase IV trial also reported adverse events similar to those seen in previous trials (hypertension, headache and nasopharyngitis being the most frequent) [319]. Six episodes of retention were recorded (1.7%), and overall, no clinically significant specific change was seen in Qmax and PVR. An open-label, randomised, two-arm, two-sequence study reported that the addition of mirabegron or tamsulosin to patients under tamsulosin or mirabegron monotherapy did not cause clinically relevant changes in cardiovascular safety or safety profiles [322].
Solifenacin and mirabegron were also compared in another RCT that has shown comparable efficacy, but a better safety profile for mirabegron [323].
In the COURAGE trial, adverse events, including hypertension and UTI, occurred at similar rates in vibegron and placebo arms. Urinary retention occurred in < 1% of patients in both arms [321].
Practical consideration
Add-on therapy with β₃ agonists in patients with remaining symptoms under α1-blocker therapy has been evaluated only in short-term clinical trials. The short-term benefit remains uncertain, with a low effect size in urinary frequency compared to placebo, and more studies with longer follow-up are required.
| Summary of evidence | LE |
| Combination treatment with α1-blockers and β₃ agonists results in a slight decrease in number of voids and urgency episodes per day compared with α1-blockers alone. | 1b |
| Adverse events of both drug classes are seen with combined treatment using α1-blockers and mirabegron. | 1b |
| Recommendation | Strength rating |
| Use combination treatment of a α1-blocker with β₃ agonists in patients with persistent storage LUTS after treatment with α1-blocker monotherapy. | Weak |
5.2.7.d. α1-blockers + Phosphodiesterase type 5 inhibitors
Mechanism of action
Combination treatment consists of an α1-blocker together with a PDE5I (Section 5.2.5) with the intent to achieve better improvements in LUTS.
Efficacy
A meta-analysis of five RCTs (two studies with tadalafil 20mg daily, two with sildenafil 25mg, and one with vardenafil 20mg) showed that combination therapy significantly improved IPSS score (-1.8), IIEF score (+3.6) and Qmax (+1.5mL/s) compared with α-blockers alone [265]. Both an SR and Cochrane review found similar findings, and a network meta-analysis of 55 RCTs (excluding 5-ARIs) found that the combination of PDE5Is and α-blockers had greater IPSS improvement than monotherapy and any other combination therapy [263, 324, 325]. These results have been confirmed by recent prospective studies, which have shown an improvement in the IPSS QoL, IIEF-5 score and Qmax in patients taking PDE5Is and α-blockers [262, 326].
Tolerability and safety
No serious adverse events have been reported in the association of PDE5Is and α-blockers. In RCTs comparing α-blockers alone with combined therapy, adverse events occur with similar incidence across the two treatment arms, suggesting that the addition of PDE5Is to α-blockers is well tolerated [324].
Practical consideration
The combination of α-blockers and PDE5Is versus α-blockers monotherapy leads to greater improvements in LUTS, QoL, erectile function and Qmax without increase in adverse events.
Data from meta-analyses suggest that younger men with low body mass index and more severe LUTS may be the population that benefits most from this association [265]. However, further studies with large populations and longer follow-up are needed to confirm these findings.
| Summary of evidence | LE |
| Combination of PDE5Is and α-blockers improves IPSS, but magnitude of effect is of low clinical significance. | 1a |
| Recommendations | Strength rating |
| Use combination treatment of a α1-blockers + phosphodiesterase type 5 inhibitors in patients with bothersome LUTS, particularly in patients willing to improve their erectile function. | Weak |
| Inform the patients that the magnitude of the effect is modest. | Weak |
Note: All patients should be counselled regarding pharmacological treatment-related adverse events to select the most appropriate treatment for each individual patient.
5.3. Surgical treatment of benign prostatic obstruction
Surgical treatment is one of the cornerstones of LUTS/BPO management. Based on its ubiquitous availability, as well as its efficacy, monopolar TURP (M-TURP) has long been considered the reference technique for the surgical management of LUTS/BPO. In recent years, however, various techniques have been developed with the aim of providing a safe and effective alternative to M-TURP. Previously, the surgical section of the Guidelines was based on technology rather than on surgical approach. As the clinical reality is primarily reflected by surgical approach and not necessarily by a specific technology, the chapter on surgical management is divided into the following five sections:
- Resection
- Enucleation
- Vaporisation
- Alternative ablative techniques
- Nonablative techniques
In addition, most of the studies are restricted by prostate size, which is also reflected in the present Guidelines. Notably, only a small fraction of RCTs is performed in patients with a prostate > 80mL. High-level evidence for larger prostates is therefore limited.
Based on Panel consensus, timeframes defining short-, mid- and long-term follow-up of patients submitted to surgical treatments are 12, 36 and over 36 months, respectively. The durability of a technique is reflected by the reoperation rate during follow-up, the failure to wean patients off medication, as well as the initiation of novel LUTS medication after surgery. However, for the majority of techniques, only the reoperation rate is reported, and clinicians should inform patients that long-term surgical RCTs are often lacking. Some patients value sexual function and perceive higher safety over maximum efficacy, and it is therefore not surprising that some patients consciously choose an alternative ablative or non-ablative technique despite the knowledge that it might not be their definitive treatment. In contrast, many urologists are critical about these procedures due to their inferior relief of BOO.
Recommendations on new devices or interventions will only be included in the Guidelines once supported by a minimum level of evidence. To clarify this, the Panel have published their position on CoE [327]. In summary, a device or technology is only included once supported by RCTs looking at both efficacy and safety, with adequate follow-up, and secondary studies to confirm the reproducibility and generalisability of the first pivotal studies [327]. Otherwise, there is a danger that a single pivotal study can be overexploited by device manufacturers. Studies that are needed include proof of concept, RCTs on efficacy and safety, as well as cohort studies with a broad range of inclusion and exclusion criteria to confirm both reproducibility and generalisability of the benefits and harms [327]. The Panel assesses the quality of all RCTs and, if they do not meet the standard required, the intervention will continue to have no recommendation, i.e. an RCT does not guarantee inclusion in the Guidelines.
In addition, the Guidelines continue to include techniques under investigation. These are devices or technologies that have shown promising results in initial studies, however, they do not yet meet the aforementioned criteria to provide a CoE that enables the Panel to regard these devices or technologies as recommended alternatives. To account for evolving evidence, recommendations for some techniques under investigation have been made, however, these techniques remain under investigation until further studies provide the recommended CoE.
Ultimately, shared decision-making and patient counselling should accompany all therapeutic choices, ensuring that patients have realistic expectations and are aware of potential risks, possible complications, need for reintervention, and expected outcomes.
5.3.1. Resection of the prostate
5.3.1.a. Monopolar and bipolar transurethral resection of the prostate
Mechanism of action
Transurethral resection of the prostate is performed in either an M-TURP or bipolar TURP (B-TURP) fashion. Transurethral resection of the prostate removes tissue from the transition zone of the gland in various degrees, resulting in a volume and PSA reduction of 25-58%.
Contrary to M-TURP, in B-TURP systems, the energy does not travel through the body to reach a skin pad. Bipolar circuitry is completed locally. Rather, energy is confined between an active (resection loop) and a passive pole situated on the resectoscope tip (‘true’ bipolar systems) or the sheath (‘quasi’ bipolar systems) using normal saline for irrigation thereby eliminating TUR-syndrome [328, 329].
Efficacy
In a meta-analysis of 20 RCTs with a maximum follow-up of five years, M-TURP resulted in a substantial mean Qmax improvement (+162%), a significant reduction in IPSS (-70%), QoL (-69%) and PVR (-77%) [330]. Monopolar-TURP delivers durable outcomes, as shown by studies with a follow-up of eight to 22 years [331]. One study with a mean follow-up of 13 years reported a significant and sustained decrease in most symptoms and improvement in urodynamic parameters. Failures were associated with DUA rather than regrowth of BPH [110]. A secondary prostatic operation, usually re-TURP, has been reported at a constant annual rate of approximately 1-2%. An SR analysing 29 RCTs found a retreatment rate of 2.6% after a mean follow-up of 16 months [332]. Data from an Austrian nationwide study of two cohorts totalling 41,059 men submitted to M-TURP showed that the overall retreatment rates (re-TURP, urethrotomy and bladder neck incision) remained unchanged during the last decade (0.9%, 3.7%, 9.5% and 12.7%, at three months, one year, five years and eight years, respectively), and that the respective incidence of re-TURP was 0.8%, 2.4%, 6.1% and 8.3%, respectively [333, 334].
Bipolar TURP is the most widely investigated alternative to M-TURP. Pooled results from 59 RCTs have been reported to date [335]. Early pooled results at 12 months concluded that no clinically relevant differences exist in short-term efficacy (IPSS, QoL and Qmax) [335, 336]. Subsequent meta-analyses supported these conclusions, though trial quality was generally poor [330,337-340]. The largest meta-analysis published to date confirmed that B-TURP compared to M-TURP results in little to no difference in urological symptoms and bother (IPSS and QoL) at 12 months [335]. Data from RCTs with mid- to long-term follow-up (up to 60 months) showed no differences in efficacy parameters [341-349]. A meta-analysis of RCTs comparing B-TURP versus M-TURP reported similar efficacy at 36 months in terms of IPSS and Qmax [350].
Tolerability and safety
Perioperative mortality and morbidity of M-TURP have decreased over time, but morbidity remains considerable (0.1% and 11.1%, respectively) [351]. Data from an Austrian nationwide study of two cohorts totalling 41,059 men submitted to M-TURP showed a 20% reduction in mortality rate over time to 0.1% at 30 days and 0.5% at 90 days [333, 334].
The risk of TUR-syndrome decreased to < 1.1% in M-TURP [332, 352]. Data from 10,654 M-TURPs reported bleeding requiring transfusion in 2.9% of cases [351]. Short- to mid-term complications reported in an analysis of RCTs using M-TURP as a comparator were: bleeding requiring transfusion 2% (0-9%), TUR-syndrome 0.8% (0-5%), AUR 4.5% (0-13.3%), clot retention 4.9% (0-39%) and UTI 4.1% (0-22%) [330]. Long-term complications of M-TURP include UI, urinary retention and UTIs, bladder neck contracture (BNC), urethral stricture, retrograde ejaculation and ED [332].
Early pooled results concluded that no differences exist in short-term urethral stricture/BNC rates, but B-TURP is preferable to M-TURP due to a more favourable perioperative safety profile (elimination of TUR-syndrome; lower clot retention/blood transfusion rates; and shorter irrigation, catheterisation and possibly hospitalisation times) [336]. Subsequent meta-analyses supported these conclusions [330, 337-340, 350], however, trial quality was relatively poor and limited follow-up might cause underreporting of late complications, such as urethral stricture/BNC [336]. The largest meta-analysis published to date concluded that B-TURP compared to M-TURP reduced TUR syndrome and blood transfusion events by 20, and 28 fewer events per 1,000 participants, respectively [335].
An RCT-based meta-analysis has shown that TUR in saline (TURis) reduces the risk of TUR syndrome and the need for blood transfusion compared to M-TURP [340]. The meta-analysis concluded that TURis is associated with improved perioperative safety, eliminating the risk of TUR syndrome, reducing the risk of blood transfusion/clot retention and hospital stay. No significant difference was detected in urethral stricture rates.
Data from the vast majority of individual RCTs with mid- to long-term follow-up (up to 60 months) showed no differences between M-TURP and B-TURP in urethral stricture/BNC rates [341-349], in accordance with all published meta-analyses. However, two individual RCTs have shown opposing results [348, 353]. A significantly higher stricture (urethral stricture + BNC) rate was detected in the B-TURP arm performed using a ‘quasi’ bipolar system (TURis, Olympus Medical) in patients with a prostate volume > 70mL at 36-months follow-up [348]. In addition, a significantly higher BNC - but not urethral stricture- rate was detected in the B-TURP arm performed with a ‘ true’ bipolar system (Gyrus PK SuperPulse, Olympus Medical) in 137 patients at 12-months follow-up [353].
Randomised controlled trials using the erectile function domain of the IIEF (IIEF-ED) and the ejaculatory domain of the male sexual-health questionnaire (Ej-MSHQ) showed that M-TURP and B-TURP have a similar effect on erectile and ejaculatory function [354, 355]. Comparative evaluations of the effects on overall sexual function, quantified with IIEF-15, showed no differences between B-TURP and M-TURP at 12-month follow-up (erection, orgasmic function, sexual desire, intercourse satisfaction, overall satisfaction) [355, 356]. Moreover, the largest meta-analysis published to date showed that erectile function measured by IIEF-5 appears to be similar at 12-month follow-up after B-TURP and M-TURP [335].
A comparative study [357] evaluated the safety of B-TURP in patients taking therapeutic oral anticoagulation (phenprocoumon) or antiplatelet drug therapy (acetylsalicylic acid or clopidogrel), without stopping or bridging the medication. Outcomes under acetylsalicylic acid were comparable to the unmedicated control group. Under oral anticoagulation therapy catheterisation (median 41 hours vs. 24 hours) and hospitalisation time was longer (median four days vs. three days), AUR rate was higher (18% vs. 6%), but blood transfusion rates did not differ from the control group. Under antiplatelet therapy blood transfusion (19% vs. 1%) and rehospitalisation rates (19% vs. 3%) were higher. In a retrospective study, elderly patients > 80 years undergoing TURP showed a significantly higher rate of complications when compared to those undergoing holmium laser enucleation of the prostate (HoLEP), including total postoperative complications (44.7% vs. 22.0%), postoperative haematuria (9.2% vs. 1.2%) and transient urinary retention (21% vs. 9.8%) [358].
Practical considerations
Monopolar TURP is an effective treatment for moderate-to-severe LUTS secondary to BPO. The choice should be based primarily on prostate volume (30-80mL suitable for M-TURP). No studies on the optimal cut-off value exist, but the complication rates increase with prostate size [351]. The upper limit for M-TURP is suggested as 80mL (based on Panel consensus, under the assumption that this limit depends on the surgeon’s experience, choice of resectoscope size and resection speed). As surgical duration increases, there is a significant increase in the rate of complications, and the procedure is safest when performed in under 90 minutes [359].
Bipolar TURP in patients with moderate-to-severe LUTS secondary to BPO has similar efficacy with M-TURP but lower perioperative morbidity. The duration of improvements with B-TURP were documented in several RCTs with midterm follow-up. Long-term results (up to five years) for B-TURP showed that safety and efficacy are comparable to M-TURP [341-349]. The choice of B-TURP should be based on equipment availability, surgeon’s experience and patient’s preference.
| Summary of evidence | LE |
| Bipolar or M-TURP is the current standard surgical procedure for men with prostate sizes of 30-80mL and bothersome moderate-to-severe LUTS secondary of BPO. | 1a |
| Bipolar TURP achieves short-, mid- and long-term results comparable with M-TURP, but B-TURP has a more favourable perioperative safety profile. | 1a |
| Recommendation | Strength rating |
| Offer bipolar- or monopolar-transurethral resection of the prostate to surgically treat moderate-to-severe LUTS in men with prostate size of 30-80mL. | Strong |
5.3.1.b. Holmium laser resection of the prostate
With the advent of HoLEP (Section 5.3.2.c) and the fact that no relevant publications on holmium laser resection of the prostate (HoLRP) have been published since 2004, HoLRP of the prostate does not play a role in contemporary treatment algorithms.
5.3.1.c. Thulium:yttrium-aluminium-garnet laser vaporesection of the prostate
Mechanism of action
The Thulium:yttrium-aluminium-garnet laser (Tm:YAG) emits a wavelength of 2010 or 2,013nm in a pulsed or continuous wave (CW) mode. The laser is primarily used with bare-ended laser fibres [360]. Various applications, such as vaporesection (ThuVARP), have been published [361].
Efficacy
Several meta-analyses with pooled data from both RCTs and non-RCTs have evaluated ThuVARP versus M-TURP [362-364] and B-TURP [365-367]. The largest meta-analyses included nine RCTs and seven non-RCTs and reported no clinically relevant differences in efficacy (IPSS, QoL and Qmax) between ThuVARP and M-TURP or B-TURP at 12 months [366]. A multicentre RCT with 410 men reported that ThuVARP and TURP are equivalent in terms of IPSS but not Qmax, with TURP deemed superior at 12-month follow-up [368]. The beneficial effect of TURP in terms of Qmax was strengthened in men aged < 70 years and in those diagnosed with LUTS rather than urinary retention. No differences in individual patient-reported urinary symptoms were seen between arms, with the exception of some evidence to indicate potential reduction in nocturia in the TURP arm. Data from one RCT with long-term follow-up showed no difference in efficacy and reoperation rates between ThuVARP and M-TURP (2.1% vs. 4.1%, respectively) [369]. A prospective multicentre study on ThuVARP including 2,216 patients showed durable postoperative improvement in IPSS, QoL, Qmax and PVR for the entire eight years of follow-up [370].
Tolerability and safety
Two meta-analyses showed longer operation times, shorter catheterisation/hospitalisation times and less blood loss without significant differences in transfusion rates or in any other short-term complication rates for ThuVARP compared to TURP [362-367]. A significantly higher transfusion rate was reported after M-TURP in two meta-analyses [364, 366]. However, overall RCT quality was relatively low with limited follow-up, potentially accounting for under-reporting of late complications such as urethral stricture/BNC [366]. A multicentre RCT with 410 men followed up for 12 months reported that ThuVARP and TURP show similar operation, catheterisation and hospitalisation times between arms with no difference in the frequency or severity of surgical complications or in blood transfusions rate or haemoglobin change [368,371]. Patients with urinary retention had similarly positive outcomes to those with LUTS [368, 371]. Data from three RCTs with mid- to long-term follow-up (18 to 48 months) showed no differences in late complication rates between ThuVARP and TURP [369, 372, 373].
Haemoglobin drop was significantly higher in the bridging group in a retrospectively analysed case series of 103 patients who underwent ThuVARP and received either low molecular weight heparin bridging or continued antiplatelet/anticoagulant therapy [374].
Practical considerations
As a limited number of RCTs with mid- to long-term follow-up support the efficacy of ThuVARP, there is a need for ongoing investigation of the technique.
| Summary of evidence | LE |
| Laser vaporesection of the prostate using Tm:YAG laser (ThuVARP) has similar operation, catheterisation and hospitalisation times compared to TURP. ThuVARP and TURP are equivalent in terms of IPSS but not Qmax, with TURP deemed superior at 12-month follow-up. ThuVARP and TURP show similar short-term safety. Mid- to long-term results on efficacy and safety compared to TURP are very limited. | 1b |
| Recommendation | Strength rating |
| Offer laser vaporesection of the prostate using Tm:YAG laser (ThuVARP) as an alternative to transurethral resection of the prostate. | Weak |
ThuVARP = Thulium:yttrium-aluminium-garnet laser vaporesection of the prostate
5.3.1.d. Transurethral incision of the prostate
Mechanism of action
Transurethral incision of the prostate involves incising the bladder outlet without relevant tissue removal. Transurethral incision of the prostate is conventionally performed with a Collins knife using electrocautery. However, alternative energy sources such as holmium laser may be used [375]. The mainstay of this technique is in prostate sizes < 30mL without a middle lobe.
Efficacy
An RCT comparing conventional TUIP versus TUIP using holmium laser in prostates ≤ 30mL with a follow-up of 12 months found both procedures to be equally effective in relieving BOO with similarly low reoperation rates [375]. A meta-analysis of ten RCTs found similar LUTS improvements and lower but significant improvements in Qmax for TUIP [376]. In this meta-analysis, an upper limit of prostate size was reported as an entry criterion for eight studies with five < 30mL and three < 60mL. A meta-analysis of six trials showed that reoperation was more common after TUIP (18.4%) than after M-TURP (7.2%) [376].
Tolerability and safety
An RCT comparing conventional TUIP versus TUIP using holmium laser reported both procedures to be safe with low complication rates. However, the operation time and retrograde ejaculation rate was significantly lower in the conventional TUIP arm [375]. No cases of TUR-syndrome have been recorded after TUIP. The risk of bleeding after TUIP is small [376].
Practical considerations
Transurethral incision of the prostate is an effective treatment for moderate-to-severe LUTS secondary to BPO. The choice between M-TURP and TUIP should be based primarily on prostate volume (< 30mL TUIP) [376].
| Summary of evidence | LE |
| Transurethral incision of the prostate shows similar efficacy and safety to M-TURP for treating moderate-to-severe LUTS secondary to BPO in men with prostates < 30mL. | 1a |
| No case of TUR-syndrome has been recorded, the risk of bleeding requiring transfusion is negligible and retrograde ejaculation rate is significantly lower after TUIP, but the reoperation rate is higher compared to M-TURP. | 1a |
| The choice between TUIP and TURP should be based primarily on prostate volume (< 30mL and 30-80mL suitable for TUIP and TURP, respectively). | 4 |
| Recommendation | Strength rating |
| Offer transurethral incision of the prostate to surgically treat moderate-to-severe LUTS in men with prostate size < 30mL, without a middle lobe. | Strong |
5.3.2. Enucleation of the prostate
5.3.2.a. Open simple prostatectomy
Open prostatectomy is the oldest surgical treatment for moderate-to-severe LUTS secondary to BPO. Obstructive adenomas are enucleated using the index finger, approaching from within the bladder (Freyer procedure) or through the anterior prostatic capsule (Millin procedure). Open prostatectomy is used for substantially enlarged glands (> 80-100mL).
Efficacy
Open prostatectomy reduces LUTS by 63-86% (12.5-23.3 IPSS points), improves QoL by 60-87%, increases mean Qmax by 375% (+ 16.5-20.2mL/s) and reduces PVR by 86-98%. Efficacy is maintained for up to six years [377-382]. Data from an Austrian nationwide study of 2,452 men submitted to OP showed that the endourological reintervention rates after primary OP were 0.9%, 3.0%, 6.0% and 8.8% at three months, one year, five years and eight years, respectively [333].
Two meta-analyses [383, 384] evaluated the overall efficacy of OP performed via a transvesical approach versus two transurethral enucleation techniques for treating patients with large glands, namely bipolar transurethral enucleation of the prostate (B-TUEP) and HoLEP. Five RCTs compared OP with B-TUEP [382,385-388] and four RCTs compared OP with HoLEP [377, 378, 389, 390]. There were no significant differences in Qmax at three-, six-, 12- and 24-months follow-up [384]. Post-void residual, PSA, IPSS and QoL showed no significant differences during 12-month follow-up [384]. Open prostatectomy and HoLEP had similar improvements regarding Qmax, IPSS score and reoperation rates after five years in one RCT [377].
Tolerability and safety
Two meta-analyses evaluated the overall safety of OP performed via a transvesical approach versus B-TUEP and HoLEP [383,384]. Operation time did not differ significantly between OP and B-TUEP but was significantly shorter for OP compared to HoLEP. Catheterisation and hospitalisation time were significantly longer for OP, which was also associated with more blood transfusions. There were no significant differences regarding other complications. There was no significant difference in IIEF-5 at three-, six-, 12- and 24-months follow-up.
Open prostatectomy mortality has decreased significantly during the past two decades (< 0.25%) [381, 391]. Data from a study of 1,286 men submitted to OP showed mortality rates of 0.2% at 30 days and 0.4% at 90 days [334].
The estimated transfusion rate was about 7-14% [377, 380, 381, 383]. Long-term complications include persistent incontinence, BNC and urethral stricture (about 6%) [377-379,383,392].
Practical considerations
Open prostatectomy is the most invasive surgical method, but it is an effective and durable procedure for the treatment of LUTS/BPO. In the absence of an endourological armamentarium including a holmium, thulium laser or a bipolar system and with appropriate patient consent, OP is a reasonable surgical treatment of choice for men with prostates > 80mL.
| Summary of evidence | LE |
| Open prostatectomy is an effective and durable procedure for the treatment of LUTS/BPO, but it is the most invasive surgical method. | 1b |
| Open prostatectomy shows similar short- and mid-term efficacy to B-TUEP and HoLEP for treating moderate-to-severe LUTS secondary to BPO in patients with large prostates. | 1a |
| Open prostatectomy has a less favourable perioperative safety profile compared to B-TUEP and HoLEP. | 1a |
| The long-term functional results of OP are comparable to HoLEP. | 1b |
| Recommendation | Strength rating |
| Offer open prostatectomy in the absence of endoscopic enucleation of the prostate to treat moderate-to-severe LUTS/benign prostatic obstruction in men with prostate size > 80mL. | Strong |
5.3.2.b. Bipolar transurethral enucleation of the prostate
Mechanism of action
Following the principles of bipolar technology (Section 5.3.1.a), the obstructive adenoma is enucleated endoscopically by the transurethral approach. Currently, two technologies exist: plasmakinetic (PK) enucleation of the prostate (PKEP) and bipolar plasma enucleation of the prostate (BPEP) [388, 393, 394]. Bipolar transurethral enucleation of the prostate is followed by either morcellation [388, 395] or resection [393, 396-400] of the enucleated adenoma.
Efficacy
Two meta-analyses reported similar efficacy at 12 months in terms of IPSS, QoL and Qmax for B-TUEP (PKEP or BPEP) versus B-TURP [401, 402]. Another meta-analysis evaluating B-TUEP versus B-TURP reported similar efficacy at 36 months in terms of IPSS and Qmax [350]. One RCT evaluating PKEP versus M-TURP reported a significant improvement in IPSS, QoL and Qmax, with urodynamically proven de-obstruction favouring PKEP at 36-months follow-up [397]. One RCT evaluating PKEP versus B-TURP in patients with prostate volume > 80mL reported no clinically relevant differences in IPSS, QoL and Qmax at six-months follow up [403]. Another RCT evaluating BPEP versus B-TURP in patients with prostate volume > 80mL reported no clinically relevant differences in IPSS, QoL, Qmax and PVR at 24-months follow-up [404]. Two RCTs evaluated the mid-term efficacy of PKEP versus B-TURP at 36 months [398, 399] and one RCT evaluated long-term efficacy at 60 months [400]. Efficacy was significantly better for PKEP in patients with large prostates at 36, 48 and 60 months [398,400]. Comparative data on efficacy for B-TUEP versus OP and the various forms of laser enucleation are presented in Section 5.3.2.a to 5.3.2.e, respectively.
Tolerability and safety
Two meta-analyses evaluating B-TUEP versus B-TURP reported similar operation, catheterisation and hospitalisation times; lower acute urine retention rates; significantly reduced haemoglobin drop and blood transfusion rates; no difference in ED; and no difference in all other reported complication rates, including urethral stricture/BNC rates for B-TUEP at 24-months follow-up [400, 401, 403].
A meta-analyses evaluating PKEP versus TURP reported that mid-term IIEF-5 scores were comparable [405]. Another meta-analysis reported less bleeding with B-TUEP compared to M-TURP but similar UI rates and AUR after catheter removal [350]. An RCT evaluating PKEP versus M-TURP in patients with prostate volume < 80mL and 36-month follow-up reported that PKEP is superior to M-TURP in terms of catheterisation and hospitalisation time [397]. No significant differences between the arms were reported in operation time, blood transfusion rates, sexual function or any other reported complications (TUR syndrome, clot retention, incontinence, retrograde ejaculation or urethral structures/BNC) [397]. One RCT evaluating PKEP versus B-TURP in patients’ prostate volume > 80mL and six-months follow-up reported that PKEP is superior to B-TURP in terms of operation, catheterisation and hospitalisation time [403]. Significant differences were reported in blood transfusion, BNC and retrograde ejaculation rates favouring PKEP, but no differences in urethral stricture and ED rates were reported [403]. Another RCT evaluating BPEP versus B-TURP in patients with prostate volume > 80mL reported that BPEP had longer operative time but shorter catheterisation, hospitalisation time with no differences in blood transfusion, urethral stricture and UI rates at 24-months follow-up [404]. No difference in urethral stricture/BNC rates was reported at 60-months follow-up [400]. Comparative data on efficacy for B-TUEP versus OP and the various forms of laser enucleation are presented in Sections 5.3.2.a to 5.3.2.e, respectively.
| Summary of evidence | LE |
| Bipolar transurethral PKEP shows favourable mid- to long-term efficacy compared to TURP. | 1b |
| Bipolar transurethral PKEP has a favourable perioperative safety profile and demonstrates similar mid- to long-term safety compared to TURP. | 1b |
| Recommendation | Strength rating |
| Offer bipolar transurethral (plasmakinetic) enucleation of the prostate to men with moderate-to-severe LUTS/benign prostatic obstruction as an alternative to transurethral resection of the prostate. | Weak |
5.3.2.c. Holmium laser enucleation of the prostate
Mechanism of action
The holmium:yttrium-aluminium garnet (Ho:YAG) laser (wavelength 2,140nm) is a pulsed solid-state laser that is absorbed by water and water-containing tissues. Tissue coagulation and necrosis are limited to 3-4mm, which is enough to obtain adequate haemostasis [406].
Efficacy
An initial meta-analysis reported no significant differences in short-term efficacy (Qmax) and reintervention rates (4.3% vs. 8.8%) between HoLEP and M-TURP [407]. However, subsequent meta-analyses reported favourable short-term efficacy (Qmax and IPSS) for HoLEP [330, 363, 401, 408]. Meta-analyses reported similar efficacy at 24-months in terms of IPSS, and Qmax [350]. Three meta-analyses evaluating HoLEP versus B-TURP showed no significant differences in short-term efficacy (IPSS, QoL and Qmax) [350, 401, 409]. One RCT comparing HoLEP with B-TURP in patients with prostate volume < 80mL reported no significant difference in IPSS, QoL and Qmax at 24 months [410]. One RCT comparing HoLEP with M-TURP in a small number of patients with mean prostate volume < 80mL and a seven-year follow-up found that the functional long-term results were comparable [406]. Another RCT comparing HoLEP with B-TURP in patients with prostate volume > 80mL reported no significant difference in IPSS, QoL and Qmax at 36 months. However, the overall retreatment rate was significantly lower following HoLEP with fewer patients restarting α-blockers and fewer reoperations [411]. Section 5.3.2.a presents comparative efficacy data for HoLEP versus OP. One RCT evaluating HoLEP versus PKEP in patients with mean prostate volume < 80mL reported similar improvements in IPSS and Qmax at 12-month follow-up [395]. Another RCT comparing HoLEP with PkEP improvements of IPSS found that QoL, PVR and Qmax were stable at three-year follow-up without differences between the groups [412,413].
An RCT comparing HoLEP versus B-TUEP reported no significant difference in IPSS, QoL, PVR and Qmax at one-, three- and 12-month follow-up [414].
An SR and meta-analysis based on retrospective series revealed a superior enucleation efficiency associated with en-bloc and the two-lobe techniques compared to the three-lobe technique, but no superiority on functional outcomes were reported [415].
Another SR and meta-analysis of 15 retrospective studies demonstrated the safety and efficacy of HoLEP in patients with recurrent or residual adenoma of the prostate [416].
Tolerability and safety
Several meta-analyses found that HoLEP has longer operation times, shorter catheterisation and hospitalisation times, reduced blood loss and fewer blood transfusions but no significant differences in urethral strictures (2.6% vs. 4.4%) and stress urinary incontinence (SUI) (1.5% vs. 1.5%) rates compared to M-TURP [363, 401, 407, 408, 417]. Another meta-analysis reported that HoLEP has shorter catheterisation times, fewer blood transfusions, urethral strictures and UTIs but no significant differences in clot retention rates and AUR after catheter removal compared to M-TURP [350]. Three meta-analyses evaluated HoLEP versus B-TURP [401, 409, 418]. One reported longer operation times for HoLEP, but no significant differences in hospitalisation time or complication rates [401], whilst another reported no significant differences in operation and catheterisation times or short-term complication rates [409]. Data from a large national database on perioperative outcomes of 2,869 laser enucleations of the prostate and 37,577 TURP procedures supports that laser enucleation of the prostate is associated with longer operation times, shorter hospitalisation times and similar complication rates (including transfusions and reoperations), but lower rates of infectious complications [419]. An SR reported that HoLEP has lower AUR rates after catheter removal but similar haemoglobin drop, UTI, urethral stricture and UI rates [350]. An RCT comparing HoLEP with B-TURP in patients with prostate volume < 80mL reported longer operation time, shorter catheterisation and hospitalisation times and a lower risk for haemorrhage for HoLEP with no significant differences in blood transfusion rates or other complication rates at 24 months [410]. Another RCT comparing HoLEP with B-TURP in patients with prostate volume > 80mL reported shorter operation, catheterisation and hospitalisation times and lower blood transfusion rates for HoLEP but no differences in complication rates, including UI and IIEF-5 score at 36 months [411]. Section 5.3.2.a presents comparative data on the safety of HoLEP versus OP. One RCT evaluating HoLEP versus PKEP in patients with mean prostate volume < 80mL reported significantly shorter operation times for HoLEP, but similar catheterisation and hospitalisation times and complication rates at 12-month follow-up [395]. Another RCT comparing HoLEP with PKEP found a higher haemoglobin drop and a prolonged haematuria in the PKEP group, but no clinically significant blood loss in either arm occurred [412, 413]. An RCT comparing HoLEP versus bipolar B-TUEP demonstrated shorter operation and hospitalisation times and earlier catheter removal for HoLEP [414].
Another RCT comparing high- (2J, 50Hz) with medium-power (2J, 30Hz) HoLEP demonstrated comparable perioperative safety and long-term durability [420]. These results were confirmed in another RCT comparing low (1.5 J, 25Hz) with high power (2.5J, 40Hz) HoLEP in prostates larger than 80mL [421].
An SR and meta-analysis of seven studies comparing pulse modulation use (Virtual basket) with standard HoLEP, shorter enucleation, better haemostasis and shorter total surgical time was found with pulse modulating HoLEP [422].
Laser enucleation of the prostate has been safely performed in patients using anticoagulant and/or antiplatelet medications. However, current limitations include:
- a lack of RCTs;
- studies with prostate volumes < 110mL;
- limited data on short- and mid-term complications and bridging therapy; and
- data presentation does not allow for separate interpretation of either antiplatelet or anticoagulant therapy [423].
A meta-analysis of seven RCTs evaluating HoLEP versus TURP reported that short- and mid-term IIEF-5 scores were comparable, whilst long-term scores were significantly better for HoLEP [424]. Two other meta-analyses detected no difference in mid-term retrograde ejaculation rates [425].
The impact on erectile function is comparable [426, 427]. Attempts to maintain ejaculatory function with modified template HoLEP have been reported to be successful in up to 77.8% of patients [428, 429]. Median Lobe-only enucleation achieved maintained antegrade ejaculation in 88%. However, reintervention rate was 14.5% at one year follow-up [430].
Practical considerations
The experience of the surgeon is the most important factor affecting the overall occurrence of complications in HoLEP [431, 432]. Mentorship programmes are advised improving surgical performance from both an institutional and personal learning curve perspective [433-435]. Pulse modulation use in HoLEP (Virtual basket) demonstrated statistically significantly improved outcomes related to haemostasis and operation times, however, clinical and health economic benefits remain to be determined [422].
| Summary of evidence | LE |
| Laser enucleation of the prostate using Ho:YAG laser (HoLEP) demonstrates similar mid- to long-term efficacy when compared to TURP. | 1b |
| Laser enucleation of the prostate using Ho:YAG laser (HoLEP) demonstrates similar short-term safety when compared to TURP. | 1a |
| Laser enucleation of the prostate using Ho:YAG laser (HoLEP) demonstrates longer operation times, but a more favourable perioperative profile when compared to TURP. | 1a |
| Recommendation | Strength rating |
| Offer laser enucleation of the prostate using Ho:YAG laser (HoLEP) to men with moderate-to-severe LUTS/benign prostatic obstruction as an alternative to transurethral resection of the prostate or open prostatectomy. | Strong |
5.3.2.d. Thulium:yttrium-aluminium-garnet laser enucleation of the prostate
Mechanism of action
The Tm:YAG laser is described in Section 5.3.1.c. Enucleation using the Tm:YAG laser includes thulium vapoenucleation of the prostate (ThuVEP) and thulium laser enucleation of the prostate (ThuLEP) (blunt enucleation). Pulsed or CW thulium:yttrium-aluminium-garnet or thulium fibre lasers are used for laser enucleation of the prostate and are well absorbed by water and water-containing tissues.
Efficacy
Two meta-analyses evaluating ThuLEP versus M-TURP and B-TURP reported no clinically relevant differences in short-term efficacy (Qmax, IPSS and QoL) [350,401]. An RCT with five-years follow-up comparing ThuLEP with B-TURP found no difference between the two procedures for Qmax, IPSS, PVR and QoL [436]. A meta-analysis [437] evaluating ThuLEP versus HoLEP showed no clinically relevant differences in IPSS, QoL and Qmax at 12 months in accordance with one RCT showing similar results at 18 months [438]. Moreover, ThuLEP and PKEP were compared in one RCT with 12-month follow-up with no difference with regard to efficacy [439]. Mainly prospective case studies have been carried out on ThuVEP showing a significant improvement in IPSS, Qmax and PVR after treatment [440-443]. In a retrospective comparative series, no differences were found between (super)pulsed and CW ThuLEP with regard to intraoperative and perioperative data and clinical efficacy (Qmax, IPSS, QoL) [444].
Tolerability and safety
Two meta-analyses evaluating ThuLEP versus M-TURP and B-TURP reported a longer operation time and shorter catheterisation time for ThuLEP compared to M-TURP and a shorter hospitalisation time for ThuLEP compared to B-TURP [350,401]. Lower blood transfusion rates compared to M-TURP, lower clot retention rates compared to B-TURP, and no difference in the other complication rates were also reported for ThuLEP [350,401]. One meta-analysis [437] evaluating ThuLEP versus HoLEP showed a significant difference in enucleation time favouring ThuLEP, but no significant differences in operation, catheterisation and hospitalisation times, and short-term complication rates.
Evaluating ThuLEP versus HoLEP showed a significant difference in enucleation time favouring ThuLEP, but no significant differences in operation, catheterisation and hospitalisation times, and short-term complication rates. One RCT showed no urethral and bladder neck strictures at 18 months after ThuLEP and HoLEP, respectively [438]. ThuLEP and PKEP were compared in one RCT with 12-month follow-up [439]. No significant difference in complication rates was detected, but haemoglobin level decrease and catheterisation time was significantly lower for ThuLEP. An RCT comparing ThuLEP with B-TURP reported a significant difference in IIEF-5 score favouring ThuLEP at 12 months [445].
In comparative studies, ThuVEP shows high intraoperative safety [446], including in case series of patients with large prostates [440] and anticoagulation or bleeding disorders [441,442]. A study focusing on postoperative complications after ThuVEP reported adverse events in 31% of cases, with 6.6% complications greater than Clavien grade 2 [447]. One case control study on ThuVEP with 48-month follow-up reported long-term durability of voiding improvements and overall reoperation rates of 2.4% [442].
Practical considerations
ThuLEP appears to offer similar efficacy and safety when compared to TURP, bipolar enucleation and HoLEP, whereas, in an RCT comparing HoLEP with ThuVEP, no differences were found with regard to the occurrence of perioperative complications, micturition improvement, hospitalisation and catheterisation time, as well as operation time [448, 449].
| Summary of evidence | LE |
| Enucleation of the prostate using the Tm:YAG laser demonstrates similar efficacy when compared to M-TURP/bipolar transurethral (PK) enucleation, HoLEP and B-TURP in the short-, mid- and long-term, respectively. | 1b |
| Enucleation of the prostate using the Tm:YAG laser (ThuLEP) demonstrates similar safety compared to TURP/bipolar transurethral (PK) enucleation and HoLEP in the short- and mid-term, respectively. | 1b |
| Vapoenucleation of the prostate using a Tm:YAG laser (ThuVEP) appears to be safe in patients with large prostates and those receiving anticoagulant or antiplatelet therapy. | 2b |
| Recommendations | Strength rating |
| Offer enucleation of the prostate using the Tm:YAG laser (ThuLEP, ThuVEP) to men with moderate-to-severe LUTS/benign prostatic obstruction as an alternative to transurethral resection of the prostate, holmium laser enucleation or bipolar transurethral (plasmakinetic) enucleation. | Weak |
| Offer Tm:YAG laser enucleation of the prostate to patients receiving anticoagulant or antiplatelet therapy. | Weak |
ThuLEP = Thulium Laser Enucleation of the Prostate, ThuVEP = Thulium Vaporization Enucleation of the Prostate, Tm:YAG = Thulium: Yttrium-Aluminum-Garnet
5.3.2.e. Diode laser enucleation of the prostate
Mechanism of action
For prostate surgery, diode lasers with a wavelength of 940, 980, 1,318 and 1,470nm (depending on the semiconductor used) are marketed for vaporisation and enucleation. Only a few have been evaluated in clinical trials [450]. Diode laser enucleation of the prostate does not play a role in contemporary treatment algorithms of LUTS/BPO.
Practical considerations
Diode laser enucleation of the prostate does not play a role in current clinical practice, and the Panel consensus was not to provide a recommendation.
| Summary of evidence | LE |
| Laser enucleation of the prostate using the 1,318nm or 1,470 laser showed comparable short-term efficacy and safety to B-TURP. Perioperative parameters such as blood loss, catheterisation time and hospital stay are in favour of diode enucleation. | 1b |
| Laser enucleation of the prostate using the 980nm laser showed comparable short-term efficacy and safety to bipolar transurethral (PK) enucleation. | 1b |
5.3.2.f. Alternative enucleation techniques
5.3.2.f.1. Laparoscopic/robotic simple prostatectomy (L/RASP)
Mechanism of action
The acronym L/RASP includes laparoscopic simple prostatectomy (LASP) and [451] multiport or single-port robot-assisted simple prostatectomy (RASP) [452, 453]. Both LASP and RASP are performed using various personalised techniques, based on the transcapsular (Millin) or transvesical (Freyer) approach (transperitoneally or extraperitoneally).
Efficacy
An SR and meta-analyses showed that, in 27 observational studies including 764 patients, mean increase in Qmax was 14.3mL/s, and the mean improvement in IPSS was 17.2 [454]. No differences were observed in improvements in Qmax and IPSS [454]. A meta-analysis comparing L/RASP versus OP reported no significant differences regarding functional and symptom parameters between the two techniques [455]. A multicentre RCT with median follow-up of 26 months did not demonstrate any significantly different functional or perioperative results between LASP, RASP and HoLEP [456]. An SR and meta-analysis of five non-randomised comparative trials comparing RASP with LASP demonstrated a shorter length of hospital stay after RASP as well as a higher postoperative Qmax [456, 457]. An RCT comparing HoLEP versus L/RASP for large volume (≥ 120mL) prostate glands resulted in longer catheterisation time in the LASP group than RASP and HoLEP groups (P = 0.002). Moreover, L/RASP resulted in longer hospitalisation and lower rate of patients with new onset of storage LUTS [456]. The efficacy of RASP was also demonstrated in 87 patients after prior endoscopic treatment of the prostate: no differences with regards to efficacy and perioperative complications were found compared to those patients with primary RASP (433 patients) [458].
Tolerability and safety
A meta-analysis comparing L/RASP versus OP demonstrated shorter hospital stay, as well as blood loss and transfusion rates for L/RASP [455, 459]. In comparative studies to OP, length of hospital stay, length of catheter use and estimated blood loss were significantly lower in the L/RASP group, while the duration of surgery was longer. There were no differences in perioperative complications between the two procedures [454]. In a multicentre RCT comparing LASP, RASP and HoLEP, LASP demonstrated significantly longer catheterisation times than RASP and HoLEP, whilst RASP and LASP showed longer hospitalisation times and lower rates of de novo bladder storage symptoms [456]. This was confirmed in an meta-analysis of six studies comparing RASP with laser enucleation of the prostate: operative time, catheterisation time, length of stay, haemoglobin loss, transfusion rate and the occurrence of > Clavien Dindo Grade 3 complications favoured laser enucleation of the prostate [460]. In a comparative analysis of robotic versus OP for large-volume prostates, a propensity score-matched analysis was performed with five covariates. Robotic compared with OP demonstrated a significantly shorter average length of hospital stay but longer mean operative time. The robotic approach was associated with a lower estimated blood loss. Improvements in Qmax, IPSS, QoL, PVR and postoperative PSA levels were similar for both groups. No difference in complications was observed between the groups [461].
In a small retrospective case series of patients with prostates > 200mL, RASP (n = 53) was compared to HoLEP (n = 22). Although median PV was higher in RASP (225mL) as in HoLEP (204.5mL), the amount of resected tissue was higher in the HoLEP group (180g vs. 134.5g). However, there were no differences in perioperative complications and micturition improvement. The PSA was significantly lower one month postoperatively after HoLEP compared to RASP (0.4 vs. 1.8ng/mL) [462].
A retrospective cohort study found no differences with regards to perioperative complications and short-term complications comparing open simple prostatectomy (OSP)-RASP with ThuLEP (mean specimen weight 74 vs. 42g) in medium-sized prostates. ThuLEP was, however, associated with shorter catheterisation time compared with SP-RASP, while postoperative SUI was higher after ThuLEP than after SP-RASP (0 vs. 18.8%) [463].
Practical considerations
Currently, most studies on L/RASP are retrospective in nature. Adequately powered RCT against reference options are needed to compare efficacy, safety, and hospitalisation times, learning curve and costs of L/RASP, and both OP and endoscopic methods. Despite the lack of level 1 evidence, minimally invasive simple prostatectomy is increasingly being used in clinical practice.
| Summary of evidence | LE |
| Laparoscopic/robotic simple prostatectom is feasible in men with LUTS/BPO and prostate sizes > 80mL needing surgical treatment, however, RCTs are needed. | 2a |
5.3.2.f.2. 532nm (‘Greenlight’) laser enucleation of the prostate (GreenLEP)
Two approaches for potassium titanyl phosphate (KTP) and the lithium triborate (LBO) laser-based enucleation technique are available [464]. GreenLEP is an anatomical enucleation technique following the principle of blunt dissection of the adenoma with the sheath and laser energy for incision as described for ThuLEP [465]. A variation is the in-situ vaporisation of apically enucleated tissue, also referred to as anatomic vaporisation-incision technique [465, 466]. To date, no high quality adequate RCTs evaluating enucleation using the KTP/LBO laser have been carried out [467, 468].
5.3.2.g. Anatomical endoscopic enucleation of the prostate
As all transurethral enucleation procedures follow the same surgical principles, the acronym AEEP has been introduced. Anatomical endoscopic enucleation of the prostate - AEEP - describes the removal of the transition zone of the prostate regardless of the energy source used.
5.3.3. Vaporisation of the prostate
5.3.3.a. Bipolar transurethral vaporisation of the prostate
Mechanism of action
Bipolar transurethral vaporisation of the prostate (B-TUVP) utilises a bipolar electrode and a high-frequency generator to create a plasma field (thin layer of highly ionised particles) to vaporise prostatic tissue [469]. Bipolar transurethral vaporisation of the prostate displays thinner (< 2mm) coagulation zones [470], compared to monopolar TUVP (up to 10mm) [471], potentially resulting in fewer irritative side effects and SUI [470, 472, 473].
Efficacy
Bipolar-TUVP has been compared to TURP in 13 RCTs, including a total of 1,244 men with a prostate size of < 80mL [344, 474-485]. Early RCTs evaluated the PK B-TUVP system [474-478], however, during the last decade, only the ‘plasma’ B-TUVP system with the ‘mushroom- or button-like’ electrode (Olympus, Medical) has been evaluated [344, 479-485]. Results have been pooled in three meta-analyses [330,486,487], and a narrative synthesis has been produced in two SRs [330, 488].
Follow-up in most RCTs is 12 months [474-477, 479-481, 483, 485] with the longest being 36 months in a small RCT (n = 40) and 18 months in a subsequent RCT (n = 340) evaluating PK [478] and plasma B-TUVP [344], respectively. Pooled results from meta-analyses concluded that no significant differences exist in short-term efficacy (IPSS, QoL , Qmax and PVR) between PK B-TUVP and TURP [330, 350, 487], and this was confirmed in a separate SR of seven RCTs [488]. However, the promising initial efficacy profile of PK B-TUVP may be compromised by inferior clinical outcomes (IPSS and Qmax) at mid-term. Higher quality RCTs with longer follow-up are necessary to draw definite conclusions on mid and long-term outcomes [330, 478].
Tolerability and safety
Early pooled results concluded that no statistically significant differences exist for intraoperative and short-term complications between PK B-TUVP and TURP, but perioperative complications are significantly fewer after B-TUVP [330]. However, the results of a statistical analysis comparing pooled specific complication rates were not directly reported in this meta-analysis [330]. Mid-term complications (urethral stricture, ED and retrograde ejaculation) are similar [478], but larger RCTs with longer follow-up are necessary to draw definite conclusions [330, 478]. An SR of seven RCTs comparing PK and plasma B-TUVP with TURP concluded that most RCTs demonstrated shorter catheterisation (42.5 vs. 77.5 hours) and hospitalisation times (3.1 vs. 4.4 days) with B-TUVP [488], but another SR concluded that heterogeneity of RCTs and methodological limitations do not permit firm conclusions [330]. A meta-analysis reported that B-TUVP has shorter and similar catheterisation time compared to M-TURP and B-TURP, respectively; significantly fewer clot retentions/blood transfusions compared to M-TURP but not B-TURP; and no difference in other complication rates compared to either TURP technique [350]. A meta-analysis of six RCTs specifically evaluating plasma B-TUVP versus TURP concluded that no significant differences exist between the techniques in overall complication and transfusion rates [487]. However, a statistically significant difference was detected in major complication rates (Clavien 3, 4), including urethral stricture, severe bleeding necessitating reoperation and UI, and in the duration of catheterisation, favouring plasma B-TUVP.
Practical considerations
Bipolar-TUVP and PK TUVP have similar short-term efficacy to TURP, but with a favourable short-term safety profile. However, heterogeneity of RCTs, non-standardised techniques and methodological limitations do not permit firm conclusions, and multicentre, long-term RCTs are needed. A shared decision-making approach should be undertaken and cover expected recovery, possible retreatment rates and the importance of centre-specific outcomes when counselling patients.
| Summary of evidence | LE |
| Bipolar-TUVP and TURP have similar short-term efficacy. | 1a |
| Plasmakinetic B-TUVP has a favourable perioperative profile, similar mid-term safety but inferior mid-term efficacy compared to TURP. | 1a |
| Plasma B-TUVP has a lower short-term major morbidity rate compared to TURP. | 1a |
| Recommendation | Strength rating |
| Offer bipolar transurethral vaporisation of the prostate as an alternative to transurethral resection of the prostate to surgically treat moderate-to-severe LUTS/benign prostatic obstruction in men with a prostate volume of 30-80mL. | Weak |
5.3.3.b. 532 nm (‘Greenlight’) laser vaporisation of the prostate
Mechanism of action
Potassium titanyl phosphate and LBO lasers are described in Section 5.3.2.f.2.
Efficacy
Meta-analyses of RCTs comparing photoselective vaporisation of the prostate (PVP) using the 80W and 120W lasers with TURP have reported no difference in Qmax and IPSS between 80W or 120W PVP and TURP [489,490]. Another meta-analysis of four RCTs including 559 patients on the 120W laser demonstrated no significant difference in functional and symptomatic parameters at 24-month follow-up when compared to TURP [491]. A meta-analysis of two RCTs reported similar efficacy of 120W PVP compared to M-TURP at 36-months follow-up [350].
The only available RCT for the 180W laser reported non-inferiority to TURP in terms of IPSS, Qmax, PVR, prostate volume reduction, PSA decrease and QoL questionnaires. Efficacy outcomes were similar to TURP, with stable results at 24-months follow-up [492].
One RCT comparing HoLEP to PVP in patients with prostates > 60mL showed comparable symptom improvement but significantly higher flow rates and lower PVR volume after HoLEP at short-term follow-up. In addition, PVP showed a 22% conversion rate to TURP [493].
One RCT compared B-TUVP with PVP with the 180W XPS laser. Comparable improvement in IPSS and Qmax were reported at 24-months follow-up [494].
Tolerability and safety
A meta-analysis of RCTs comparing the 80W and 120W lasers with TURP showed shorter catheterisation time (MD 32 hours) and length of hospital stay (MD 1.85 days) after PVP [330]. Fewer blood transfusions and less was observed with PVP. No difference was noted in postoperative urinary retention, UTI, meatal stenosis, urethral stricture or bladder neck stenosis [330]. Similarly, a meta-analysis including trials with the 120W laser reported lower transfusion rates and shorter catheterisation time and duration of hospital stay compared to TURP. Reoperation rates and operation time favoured TURP. No significant differences were demonstrated for treatment for urethral stricture, BNC, incidence of incontinence and UTI [491]. A meta-analysis confirmed that PVP was superior to both M-TURP/B-TURP with regard to catheterisation and to M-TURP but not to B-TURP with regard to transfusion rate and clot retention [350]. In an RCT comparing the 120W HPS laser with TURP with a follow-up of 36-months, the reoperation rate was significantly higher after PVP (11% vs. 1.8%) [495].
180W Greenlight laser prostatectomy is noninferior to TURP in terms of perioperative complications. Reoperation-free survival during a 24-month follow-up was comparable between the TURP arm and the 180W XPS laser arm [492].
A retrospective feasibility study on 537 patients with a median follow-up of 31 months, 517 of whom were treated on an outpatient basis, showed that outpatient PVP with the 180W XPS laser can be performed safely with a low readmission and complication rate [496].
One retrospective long-term follow-up study on 21,869 patients treated in 20 centres in Finland compared short-term and long-term morbidity in patients undergoing M-TURP, B-TURP or any of the three generations of PVP [488]. Reoperations for bleeding were less frequent after PVP. Cumulative incidence for reoperation was higher after PVP (23.5%) than after TURP in long-term follow-up (17.8%) [497].
Based mostly on case series, the 80, 120 and 180W Greenlight laser appears to be safe in high-risk patients undergoing anticoagulation treatment [498-501]. However, patients under anticoagulation therapy were either excluded from, or represented a very small sample in, currently available RCTs. In one study, anticoagulated patients had significantly higher rates of bladder irrigation (17.2%) compared with those not taking anticoagulants (5.4%) [501]. In contrast, another retrospective study focusing on the 180W LBO laser did not find any significant differences between patients receiving or not receiving anticoagulants [502]. A retrospective study of a mixed cohort of patients treated with 80W KTP PVP and 120W LBO HPS revealed that delayed gross haematuria was common in patients (33.8%) during an average follow-up of 33 months [503]. A retrospective review of a database of patients undergoing 180W PVP without interruption of anticoagulation therapy had a 30.5% rate of perioperative adverse events with a significant occurrence of high-grade Clavien Dindo events [504].
Real-world data indicate roughly 8% of surgical reintervention at four years for both PVP (7.8%) and TURP (7.5%), thus highlighting the importance of centre-specific outcome auditing to inform patient counselling [505].
Safety in patients with urinary retention, impaired detrusor contractility, elderly patients or prostates > 80mL was shown in various prospective short-term non-randomised trials.
A retrospective study on 1,077 patients [506] with a median 18-months follow-up compared the functional results and the safety profile of PVP in patients younger and older than 75 years of age. The authors did not find any differences in terms of complications in older patients, with only 0.6% of Clavien III and an overall complication rate of 29.6%. Data on functional outcomes in the older group of patients, showed amelioration of all parameters from the baseline, with 111.7% of improvement of peak flow and 69.5% of IPSS reduction without statistical differences with the counterparts at 12 months. However, this similarity of results disappears when the study period is extended up to a median of 18 months, underlying the fact that younger patients maintain their improvement more than older patients, who nevertheless maintain a relevant improvement compared to their baseline [506].
No RCT including prostates > 100mL has been reported, therefore, comparison of retreatment rates between prostate volumes of different sizes is not possible [507-509].
A meta-analysis of five RCTs collectively comparing all three ‘Greenlight’ lasers with TURP detected no difference in retrograde ejaculation rates [425]. Additional studies have also reported no difference between OP/TURP and Greenlight PVP for erectile function [510, 511]. However, IIEF-5 scores were significantly decreased at six, 12 and 24 months in patients with preoperative IIEF-5 greater than 19 [512].
No significant difference with respect to perioperative and postoperative complications was reported in an RCT comparing B-TUVP and PVP with the 180W XPS Laser. Redo TURP for recurrent adenoma was required in 9.8% (B-TUVP) and 1.7% (PVP) of the patients during 24-months follow-up, respectively [494].
Practical considerations
The 180W XPS represents the current standard of generators for PVP, however, the number and quality of supporting publications are low, especially for large glands (> 100mL), with no long-term follow-up.
| Summary of evidence | LE |
| Laser vaporisation of the prostate using the 80W KTP and the 120W LBO laser (PVP) demonstrated higher intraoperative safety with regard to haemostatic properties when compared to TURP. Perioperative parameters such as catheterisation time and hospital stay are in favour of PVP, whereas operation time and risk of reoperation are in favour of TURP. Short-term results for the 80W KTP laser and mid-term results for the 120W LBO laser were comparable to TURP. | 1a |
| Laser vaporisation of the prostate using the 180W LBO laser (PVP) demonstrated higher intraoperative safety with regard to haemostatic properties when compared to TURP. Perioperative parameters such as catheterisation time and hospital stay favoured PVP, whereas operation time favoured TURP. Short- to mid-term results are comparable to TURP. | 1b |
| Laser vaporisation of the prostate using the 80W KTP and 120W LBO lasers appears to be safe for the treatment of patients receiving antiplatelet or anticoagulant therapy. | 2 |
| Laser vaporisation of the prostate using the 180W LBO laser appears to be safe for the treatment of patients receiving antiplatelet or anticoagulant therapy, however, the level of evidence available is low. | 3 |
| Recommendations | Strength rating |
| Offer 80W 532nm potassium titanyl phosphate (KTP) laser vaporisation of the prostate to men with moderate-to-severe LUTS/benign prostatic obstruction (BPO) with a prostate volume of 30-80mL as an alternative to transurethral resection of the prostate (TURP). | Strong |
| Offer 120W 532nm lithium borate (LBO) laser vaporisation of the prostate to men with moderate-to-severe LUTS/BPO with a prostate volume of 30-80mL as an alternative to TURP. | Strong |
| Offer 180W 532nm LBO laser vaporisation of the prostate to men with moderate-to-severe LUTS/BPO with a prostate volume of 30-80mL as an alternative to TURP. | Strong |
| Offer laser vaporisation of the prostate using 80W KTP, 120 or 180W LBO lasers for the treatment of patients receiving antiplatelet or anticoagulant therapy with a prostate volume < 80mL. | Weak |
5.3.3.c. Vaporisation techniques under investigation
Alternative vaporisation techniques have been investigated, but do not play a role in current clinical practice (e.g. ThuVAP, Ho:YAG, Blue-wavelength diode) and other emerging techniques, such as transperineal laser ablation (TPLA), as discussed in Section 5.3.4.c.2.
Refer to previous editions of the Guidelines for full rationale.
5.3.4. Alternative ablative techniques
5.3.4.a. Aquablation - image-guided robotic waterjet ablation: AquaBeam
Mechanism of action
Aquablation uses the principle of hydrodissection to ablate prostatic parenchyma while sparing collagenous structures such as blood vessels and the surgical capsule. A targeted high-velocity saline stream ablates prostatic tissue without the generation of thermal energy under real-time transrectal US guidance. After completion of ablation, haemostasis is performed with a Foley balloon catheter on light traction or diathermy or low-powered laser if necessary [513]. Early catheter removal (< day two) is commonly achievable with this approach [514].
Efficacy
In a double-blind, multicentre, pivotal RCT, 181 patients with a prostate size of 30-80mL were randomised to TURP or Aquablation [515, 516]. Mean total operative time was similar for Aquablation and TURP (33 vs. 36 minutes), but resection time was significantly lower for Aquablation (4 vs. 27 minutes). At six months, patients treated with Aquablation and TURP experienced large IPSS improvements (-16.9 and -15.1, respectively), satisfying the noninferiority hypothesis. At one-year follow-up, mean IPSS reduction was 15.1 with a mean reduction in IPSS score of 67% for both groups. No significant difference in improvement of IPSS, QoL, Qmax and reduction of PVR was reported between the groups [517].
Improvements in IPSS and Qmax were maintained after five years in both groups [518]. Mean IPSS decrease was 16.9 and 15.1 points in the Aquablation and TURP groups, respectively. Similarly, five-year improvements in Qmax were 125% and 89% compared to baseline for Aquablation and TURP, respectively. Over five years, surgical retreatments were 5.1% and 1.5%, respectively [518].
Results of Aquablation in patients with large prostates (80-150mL) were evaluated in a cohort study of 101 men (WATER II) [515]. After 12 months, significant improvements were seen in IPSS (mean decrease of 17 points), Qmax (increase of 12.5cc/sec.) and PVR (a drop of 171cc in those with PVR > 100 at baseline). At five-years follow-up, 3% of patients required surgical retreatment [519].
The five-year follow-up of Aquablation therapy confirms sustained outcomes, minimal irreversible complications and low retreatment rates across prostate volumes ranging from 30-150mL. In pooled analysis of the WATER and WATER II cohorts, mean IPSS reduction was 15.1-15.9 points, Qmax increase was 8.7-9.2mL/s and QoL score reduction was 3.2-3.3 points, demonstrating durable efficacy across small and large glands [514].
In an SR and network meta-analysis including 23 studies comparing Aquablation and HoLEP, HoLEP provided greater objective improvements with a Qmax increase of +3.24mL/s (95% CI: 0.52-5.96) and PVR reduction of -23.03mL (95% CI: -39.39 to -6.66), whereas IPSS (-1.11 points) and QoL (-0.06 points) were similar between the two techniques [520].
Urodynamic studies of 66 patients enrolled in the WATER trial at six-months follow-up showed significant changes in pdetQmax (reductions of 35 and 34cm H20, respectively) and large improvements in BOO index in both groups [521].
Tolerability and safety
Results for the WATER trial have shown comparable hospital stay and catheterisation duration (1.4 and 1 day, respectively) [515]. One case of blood transfusion was reported after Aquablation and none after TURP. In an SR of seven patient groups involving 446 patients treated by Aquablation, although there was a significant haemoglobin drop (2.06g/dL), this did not translate into increased transfusion rates. In WATER, fewer men in the Aquablation group had a persistent Clavien-Dindo grade 1 or 2 or higher adverse events compared to TURP (26% vs. 42%) at three months. Among sexually active men, the rate of anejaculation was lower in those treated with Aquablation compared to TURP (10% vs. 36%, respectively). There were no procedure-related adverse events after six months [517].
In patients with a prostate volume of 80-150mL (WATER II trial), bleeding-related events were observed in 14 patients (13.9%), eight of which (7.9%) occurred prior to discharge and six (5.9%) occurred within one month of discharge. Blood transfusions were required in eight patients, return to the theatre for fulguration in three patients and both transfusion and fulguration in two patients [522]. Maintenance of antegrade ejaculation was slightly lower in WATER II at 81% compared to 90% in the smaller prostates of WATER I [523]. In WATER II, there was a 2% de novo incontinence rate at 12 months [524].
In an analysis of procedural differences including data from WATER I and II, compared to a single pass of ablation, the use of two or more passes during Aquablation resulted in lower IPSS scores by four points, and lower IPSS QoL by 0.7 points at 24 and 36 months. Similarly, 36-month Qmax values were higher by 5mL/sec. in those with two or more passes than in those with one pass [525].
In a network meta-analysis comparing Aquablation and HoLEP, HoLEP was associated with a significantly higher risk of incontinence (RR: 4.48; 95% CI: 1.02 to 19.65) but a lower risk of blood transfusion (RR = 0.14; 95% CI: 0.00-4.21) [520].
According to long-term data from the WATER and WATER II trials [514], a five-year surgical retreatment-free survival of over 94% was reported following Aquablation with low rates of de novo incontinence (2%) and high preservation of antegrade ejaculation (81-90%). These findings support the favourable safety profile and functional preservation associated with Aquablation, including in large-volume prostates.
Practical considerations
During long-term follow-up, Aquablation provides noninferior functional outcomes compared to TURP in patients with LUTS and a prostate volume of 30-80mL. Management of bleeding and bladder neck cautery is often performed at the end of the procedure.
This is particularly suitable when preservation of ejaculatory function and early recovery are priorities. In gland volumes of 80-150mL (WATER II), symptom and flow improvements are sustained for up to five years [514, 526]. Short-term comparative studies against enucleation have shown similar efficacy and safety at six months and favourable results regarding ejaculatory function and SUI. Durability against enucleation remains to be established.
| Summary of evidence | LE |
| Aquablation appears to be as effective as TURP, both subjectively and objectively. | 1b |
| Aquablation appears to have a low incidence of EjD. | 1b |
| Aquablation is feasible in prostates > 80mL, however, additional RCTs are needed. | 2a |
| Recommendation | Strength rating |
| Offer Aquablation to patients with moderate-to-severe LUTS/benign prostatic obstruction and a prostate volume of 30-80mL as an alternative to transurethral resection of the prostate, particularly in patients interested in preserving ejaculatory function. | Strong |
5.3.4.b. Prostatic artery embolisation
Mechanism of action
Prostatic artery embolisation (PAE) can be performed as a day procedure under local anaesthesia with access through the femoral or radial arteries. Digital subtraction angiography displays arterial anatomy, and the appropriate prostatic arterial supply is selectively embolised to effect stasis in treated prostatic vessels. Various technical variations (including bead size) have been described for PAE, which requires specific training [527, 528].
Efficacy
Superior efficacy of PAE compared with a sham procedure was found in a six-month randomised, single-blind, sham-controlled trial in 80 patients with LUTS, refractory to medical treatment. The decrease in IPSS at six months was 5.03 +/- 8.13 in the sham group and 17.1 +/- 7.25 in the PAE group [529].
An SR and meta-analysis including RCTs and two non-RCTs comparative studies (n = 708 patients) showed that TURP achieved a significantly higher mean postoperative difference for IPSS and IPSS-QoL - 3.80 and 0.73 points, respectively - compared to PAE [530]. All of the functional outcomes assessed were significantly superior after TURP: 3.62mL/s for Qmax, 11.51mL for prostate volume, 11.86mL for PVR and 1.02ng/mL for PSA [530].
According to a Cochrane network meta-analysis, PAE may result in little to no difference in urologic symptoms scores as well as QoL compared to TURP at short-term follow-up of three to 12 months [531]. A network meta-analysis included outcome data at three- to six-months follow up and concluded that improvement of IPSS was similarly high after TURP and PUL.
A meta-analysis of 11 RCTs comparing TURP and PAE found no significant difference between TURP and PAE for patient-reported outcomes, including IPSS and QoL at 12 months. Prostatic artery embolisation was less effective regarding improvements in most functional outcomes such as maximum flow rate, PSA [532].
In a randomised, single-centre, non-inferiority trial with five-year follow-up, PAE was shown to result in inferior improvements in IPSS (−7.8 vs. −11.6), Qmax (+3.6 vs. +9.3mL/s) and PVR (-28 vs. -220mL) compared to TURP [533].
A SR confirmed that reintervention after PAE is higher than after TURP or enucleation techniques [534].
A Cochrane review reported that, compared to TURP, the impact on urological symptoms and QoL improvement as perceived by patients appears to be similar following PAE [535]. This review did reveal major uncertainty as to how major adverse events compare.
Tolerability and safety
Available RCTs as well as SR and meta-analyses show conflicting results about the comparative rate of adverse events after PAE or TURP depending on studies included, definition of adverse events and follow-up. In an SR of comparative studies, PAE resulted in significantly more adverse events than TURP/OP (41.6% vs. 30.4%). The frequency of AUR after the procedures was significantly higher in the PAE group (9.4% vs. 2.0%) [536]. In another compilation of studies, PAE was associated with significantly fewer overall adverse events but similar rates of severe side effects, as well as shorter hospitalisation times (MD = -1.94 days), but longer procedural times (MD = 51.43 min.) [537].
Another SR and meta-analysis of four studies (506 patients) comparing PAE and TURP found no significant difference in the postoperative complication rate between TURP and PAE [538].
According to a Cochrane network meta-analysis, PAE may also result in a larger reduction in major adverse events than TURP, but the confidence interval includes substantial benefits and harms [531]. In addition, uncertainty exists about the effect of PAE on retreatment compared to TURP at follow-up from 12 to 60 months. A meta-analysis did reveal major uncertainty as to how major adverse events compare between TURP and PAE [535].
According to a non-inferiority trial, 42% of patients who initially had PAE underwent TURP within five years [533].
Concerning sexual adverse events, the mean differences in IIEF-5 score were not significantly different between TURP and PAE in a meta-analysis [537]. Another meta-analysis of two RCTs detected no difference in retrograde ejaculation rates [425]. Postoperative erectile function measured by IIEF-5 was in favour of PAE with MD in change of 2.56 points. In another updated meta-analysis, PAE was consistently associated with lower sexual dysfunction than TURP (OR 0.24) [539].
According to a Cochrane network meta-analysis, uncertainty exists on the effect of PAE on erectile function and ejaculatory function as compared to TURP [531].
Concerns persist regarding non-target embolisation reported in earlier studies [540], however, more recent studies report fewer incidents [530, 541]. In the PROSTATE registry, periprocedural adverse events occurred in ±11.5% of patients, with most being self-limiting. No major non-target embolisations were reported [542]. An SR of 22 studies reporting radiation exposure during PAE with a 20-fold range of exposures estimated that the median risk for a 66-year-old patient of a cancer-related death was 0.117%, equivalent to a reduced life expectancy of 5.4 days. Radiation exposure therefore should be part of the counselling for patients considered for PAE. These data suggest there is potential for significant radiation reduction in some centres using appropriate protocols [543].
Practical considerations
A multidisciplinary team approach of urologists and radiologists is mandatory, and patient selection should be done by urologists and interventional radiologists. The investigation of patients with LUTS to indicate suitability for invasive techniques should be performed by urologists only. This technically demanding procedure should only be carried out by an interventional radiologist with specific mentored training and expertise in PAE [544]. There are data suggesting that larger prostates have a higher chance of a superior outcome with PAE in post hoc analysis of RCTs, but larger trials are required to clarify the most suitable patients for PAE [522, 545].
Recent long-term RCT and large registry data underline the importance of counselling patients on the higher probability of retreatment compared to TURP and on the radiation exposure inherent to PAE; centre-level expertise and optimised protocols can reduce these risks [533,542].
Further data with medium- and long-term follow-up are still required, and comparison with other minimally invasive techniques would be valuable. However, current evidence of safety and efficacy of PAE appears adequate to support the use of this procedure for men with moderate-to-severe LUTS, provided proper arrangements for consent and audit are in place. Therefore, a recommendation has been given, but PAE remains under investigation.
| Summary of evidence | LE |
| Prostatic artery embolisation is less effective than TURP at improving symptoms and urodynamic parameters such as flow rate. | 1a |
| Procedural time is longer for PAE compared to TURP, but blood loss, catheterisation and hospitalisation time are in favour of PAE. | 1b |
| Prostatic artery embolisation results in higher retreatment rates compared to TURP. | 1a |
| Recommendations | Strength rating |
| Offer prostatic artery embolisation (PAE)* to men with moderate-to-severe LUTS/benign prostatic obstruction who wish to consider minimally invasive treatment options and accept less-optimal outcomes compared with transurethral resection of the prostate. | Weak |
| Perform PAE only in units where the work up and follow-up is performed by urologists working collaboratively with trained interventional radiologists for the identification of PAE suitable patients. | Strong |
* PAE remains under investigation
5.3.4.c. Alternative ablative techniques under investigation
5.3.4.c.1. Convective water vapour energy (WAVE) ablation: The water vapour thermal therapy system
Mechanism of action
The water vapour thermal therapy (WVTT) system uses radiofrequency power to create thermal energy in the form of water vapour, which in turn deposits the stored thermal energy when the steam phase shifts to the liquid phase upon cell contact. The steam disperses through the tissue interstices and releases stored thermal energy onto prostatic tissue effecting cell necrosis. The procedure can be performed in an office-based setting. Usually, one to three injections are needed for each lateral lobe, and one to two injections may be delivered into the median lobe (depending on the prostate size).
Efficacy
In a multicentre RCT, 197 men were enrolled and randomised in a 2:1 ratio to treatment with water vapour energy ablation or sham treatment [546]. At three months, relief of symptoms (measured by a change in IPSS and Qmax) was significantly improved and maintained after WAVE therapy compared to the sham arm, although only the active treatment arm was followed up to 12 months. No relevant impact was observed on PVR. Quality of life outcome was significantly improved with a meaningful treatment response of 52% at 12 months. Further validated objective outcome measures, such as BPH impact index (BPHII), Overactive Bladder Questionnaire Short Form for OAB bother, and impact on QoL and ICS Male Item Short Form Survey for male incontinence, demonstrated improvements of symptoms at three months follow-up with sustained efficacy throughout the study period of 12 months. The reported two-year results in the WVTT cohort arm of the same study and the recently reported five-year results confirmed durability of the clinical outcome after convective water vapour energy ablation [547]. Surgical retreatment rate was 4.4% over five years.
Limited comparative data for WVTT versus reference treatments exists, however, a single-centre RCT with 24-month follow-up comparing WVTT with B-TURP reported greater improvements in IPSS, Qmax, PVR and prostate volume after B-TURP. However the WVTT arm achieved -55.3% improvement in IPSS, +62.5% in Qmax, -50% in PVR, and preserved antegrade ejaculation in 98% of patients at 12-months, with shorter operative time and hospital stay [548].
A Cochrane review found no studies comparing convective radiofrequency water vapour thermal therapy to any other active treatment form, such as TURP [549]. Another recent network meta-analysis included outcome data at three- to six-months follow-up and concluded that improvement of IPSS was similarly high after TURP and WAVE [550]. Objective outcomes (PVR and Qmax) were improved to the greatest extent after TURP, with moderate improvement after WAVE. Water vapour thermal therapy demonstrates a retreatment rate of 15.8% at four years, which includes both surgical reintervention and reinitiation of pharmacotherapy, underscoring the importance of mid-term follow-up and patient counselling [551].
Tolerability and safety
Safety profile was favourable with adverse events documented to be mild-to-moderate and resolving rapidly. Preservation of erectile and ejaculatory function after convective WVTT was demonstrated utilising validated outcome instruments such as the IIEF and the MSHQ-EjD [546]. In the 24-month RCT versus B-TURP, Rezūm (WVTT) showed a lower perioperative complication rate [548].
Practical considerations
There are two SRs of the WFTT cohort studies. One concludes that WFTT provides improvement in BPH symptoms that exceeds established minimal clinically important difference thresholds, preserves sexual function and is associated with low surgical retreatment rates over four years, therefore suggesting that it may be a valuable addition to the urological armamentarium to treat LUTS in men with BPH [552]. The other, a Cochrane review, reported that the CoE ranged from moderate to very low, with study limitations and imprecision being the most common reasons for downgrading of the evidence [549]. A Cochrane network meta-analysis reported uncertainty about the effects of WVTT on retreatment compared to sham treatment at three-months follow-up and the CoE is very low [531]. Evidence at up to 24 months supports the durability of symptom relief and functional improvement, particularly in men prioritising preservation of sexual function and outpatient recovery, although objective improvements are generally greater after TURP [548]. Randomised controlled trials against a reference technique are needed to confirm the first promising clinical results and to evaluate mid- and long-term efficacy and safety of water vapour energy treatment.
In an SR and meta-analysis evaluating WVTT in prostates volumes > 80mL (up to 150mL), favourable outcomes have resulted in an extension in indications in some countries [553].
5.3.4.c.2. Transperineal interstitial laser ablation of the prostate
Mechanism of action
Transperineal interstitial laser ablation (TPLA) uses a CW diode laser, commonly at 1,064nm, that is delivered through transperineal needles guided by US. The procedure results in localised thermal coagulative necrosis within the prostatic tissue, which decreases prostate volume and alleviates obstruction [554].
Efficacy
Transperineal interstitial laser ablation of the prostate has been compared to TURP in two RCTs. One study assigned 50 patients to either TPLA or TURP. After one year, the improvement in Qmax was better in the TURP group, while IIEF-5 score was similar between groups [555]. In another RCT, 51 patients were randomised to TPLA (26 patients) or TURP (25 patients). The primary endpoint was a change in ejaculatory function assessed by the Ej-MSHQ at one month after surgery. The distribution of ejaculatory function assessed remained unmodified after TPLA (P = 0.2), while a median 30% decrease in EJ-MSHQ score was observed after TURP (P = 0.01). The Qmax was more pronounced after TURP (mean 23.9mL/s improvement) versus 6.0mL/s after TPLA and IPSS showed a mean decrease of 11.6 after TURP versus 5.8 after TPLA with respect to baseline [556].
Tolerability and safety
TPLA is characterised by a low incidence of complications, predominantly minor in nature (Clavien-Dindo grades I-II). Adverse events include transient urinary retention, UTI and perineal pain, while major complications occur infrequently (< 5%). The procedure is generally well tolerated with local anaesthesia, and most patients are discharged within 24 hours [554, 557].
Across studies, > 85% of sexually active patients maintain antegrade ejaculation, and erectile function is generally preserved or improved. This contrasts favourably with TURP and other more invasive procedures, which are associated with higher rates of sexual dysfunction [557-559].
Practical consideration
Despite the encouraging data, adequately powered RCTs with longer follow-up comparing TPLA to a reference technique are required.
5.3.5. Nonablative techniques
5.3.5.a. Prostatic urethral lift
Mechanism of action
Prostatic urethral lift (PUL) is a minimally invasive approach under local or general anaesthesia. Encroaching lateral lobes are compressed by small permanent suture-based implants delivered under cystoscopic guidance, resulting in an opening of the prostatic urethra leaving a continuous anterior channel through the prostatic fossa.
Efficacy
Several reports have shown that PUL achieves a significant improvement in IPSS (-39% to -52%), Qmax (+32% to +59%) and QoL (-48% to -53%) [560-565]. In a meta-analysis of retrospective and prospective trials, pooled estimates showed an overall improvement following PUL, including IPSS, Qmax and QoL [565]. A Cochrane review of the sham RCT and the RCT against TURP concluded that PUL appears less effective than TURP in improving urological symptoms (IPSS, Qmax) in both the short- and long-term, while QoL outcomes may be similar [566]. Prostatic urethral lift was evaluated versus sham in a multicentre study with one- [562], three- [567] and five-years [568] follow-up of the treated cohort. Improvements in IPSS, QoL and Qmax were durable with improvement rates of 36%, 50% and 44% at 60-month follow-up, respectively [568]. A network meta-analysis included outcome data at 3- to 6-months follow up and concluded that improvement of IPSS was similarly high after TURP and PUL. Objective outcomes (PVR and Qmax) were improved to the greatest extent after TURP, with less improvement after PUL [550].
A retrospective observational study of 1,413 consecutive patients from North America and Australia split patients into those still voiding (Group A) and those in retention (Group B). The results from Group A were comparable to the results from the clinical trials and of the 165 patients in Group B 69% were catheter-free after five days, 83% after one month and 89% by study’s end [569].
A real-world analysis of over 50,000 patients undergoing LUTS/BPO procedures [505] found that UroLift was associated with higher long-term surgical reintervention rates, reaching 16.1% at four years, compared to 7.5% for TURP and 7.8% for PVP. The average annual increase in reintervention after year one was 3.6%, suggesting reduced procedural durability of UroLift relative to resective and ablative techniques [505].
Tolerability and safety
The most common complications reported postoperatively included haematuria (16-63%), dysuria (25-58%), pelvic pain (5-17.9%), urgency (7.1-10%), transient incontinence (3.6-16%) and UTI (2.9-11%) [562, 565, 567, 568]. Most symptoms were mild-to-moderate in severity and resolved within two to four weeks after the procedure. In an RCT comparing PUL to TURP, surgical recovery was measured using a validated instrument. They found that recovery from PUL is more rapid and more extensive in the first three to six months [570]. An SR and meta-analysis found that sexual function with regard to erectile and ejaculatory function remained stable or improved slightly during the 24-month follow-up [565, 566, 571].
In an RCT comparing PUL to TURP, PUL resulted in superior quality of recovery and ejaculatory function preservation. Ejaculatory function and bother scores did not change significantly in either treatment arm [570]. When counselling, clinicians should also discuss the comparatively higher mid-term reintervention likelihood after PUL as supported by real-world analyses and network meta-analytic evidence [505].
An SR of surgical reinterventions of 11 studies (2,016 patients), among which TURP/laser (51%), repeat PUL (32.7%) and device explant (19.6%) were most common, revealed an annual rate of surgical reintervention of 6% per year (95% CI: 3.0-8.9) [572]. The retreatment rate was 13.6% over five years in a multicentre study comparing PUL versus sham [568].
Practical considerations
Only limited data is available on treating patients with an obstructed/protruding middle lobe [573]. The effectiveness in large prostate glands has not yet been shown. Long-term studies are needed to evaluate the duration of the effect in comparison to other techniques. Current real-world and network-level evidence reinforces shared decision-making about durability, balancing the advantages of quicker recovery and ejaculatory preservation against a higher probability of reintervention versus TURP/PVP at ± four years [505].
| Summary of evidence | LE |
| Prostatic urethral lift improves IPSS, Qmax and QoL. These improvements are inferior to TURP at 24 months. | 1b |
| Prostatic urethral lift has a low incidence of sexual side effects. | 1b |
| Patients should be informed that risk of retreatment has been shown to be higher than after TURP. | 3 |
| Recommendation | Strength rating |
| Offer Prostatic urethral lift (Urolift®) to men with LUTS/benign prostatic obstruction interested in preserving ejaculatory function, with prostates < 70mL and no middle lobe. | Strong |
| Advise patients that retreatment rates are higher after PUL compared to transurethral resection of the prostate. | Weak |
5.3.5.b. Intra-prostatic injections
Botulinum toxin-A (BoNT-A), fexapotide triflutate (NX-1207) and PRX302 techniques have been investigated and have been proven to have no benefit in MLUTS/BPO management.
| Recommendation | Strength rating |
| Do not offer intraprostatic injection treatment to patients with LUTS//benign prostatic obstruction.* | Strong |
* Refer to previous editions of the Guidelines for full rationale.
5.3.5.c. Non-ablative techniques under investigation
5.3.5.c.1.(i)TIND
Practical considerations
The iTIND is a nitinol device composed of three elongated struts and an anchoring leaflet. Under direct visualisation, iTIND is deployed inside the prostate in expanded configuration. The intended mode of action is to compress obstructive tissue by the expanded device, thereby exerting radial force leading to ischaemic necrosis resulting in a Turner Warwick like incision. The iTIND device is left in position for five days and removed in an outpatient setting by standard urethroscopy.
Efficacy
A single-arm, prospective study of 32 patients with a three-year follow-up was conducted to evaluate feasibility and safety of the procedure [574]. The change from baseline in IPSS, QoL and Qmax was significant at every follow-up [575]. In a multicentre RCT, 175 men were randomised 2:1 between iTIND and sham procedures. Patients were assessed at baseline, 45 days, three and 12 months postoperatively. A total of 78.6% of patients in the iTIND arm showed a reduction of ≥ 3 points in IPSS, versus 60% of patients in the control arm at three months. At 12-month follow-up, the iTIND group reported a mean decrease of 9.25 in IPSS and 1.9 points in QoL, and a 3.52mL/s increase in Qmax compared to baseline [576]. In a prospective multicentre study, 81 patients were enrolled and treated with a second generation iTIND device. At twelve-month follow-up, mean IPSS decreased from 22.5 to 8.8 and Qmax at one month and increased from 7.3 to 14.7mL/s at 12-month [577].
Another recent network meta-analysis included outcome data at three- to six-months follow up and concluded that IPSS improvement was lower after iTIND compared to TURP. Improvements in objective outcomes (PVR and Qmax) were also lower after iTIND [550].
iTIND is associated with a retreatment rate of 5% at three years [551].
Tolerability and safety
The device was reported as well-tolerated by all patients. Four early complications (12.5%) were recorded, including one case of urinary retention (3.1%), one case of transient incontinence due to device displacement (3.1%) and two cases of infection (6.2%). No further complications were recorded during the 36-month follow-up period [575]. In the RCT against sham study, adverse events were typically mild and transient. Most were Clavien-Dindo grade 1 or 2, with 38.1% in the iTIND arm and 17.5% in the control arm [576]. No new EjD or ED has been reported [576, 577].
In a prospective multicentre study, 81 patients were enrolled and treated with a second generation iTIND device. During the 12-month period, two patients required medical therapy, two patients required TURP and ten patients were lost to follow-up [577].
Practical considerations
Randomised controlled trials comparing iTIND to a reference technique are ongoing.
5.3.5.c.2. Paclitaxel-coated ballon dilatation of the prostate (Optilume)
Mechanism of action
Optilume represents a minimally invasive therapeutic option for BPO and urethral strictures, integrating mechanical dilation with targeted drug delivery. This system employs a drug-eluting balloon, which serves both to mechanically dilate the affected segment and deliver medication. The balloon is inflated within the prostatic urethra to perform an anterior commissurotomy. Its surface is coated with paclitaxel, facilitating localised drug administration that suppresses smooth muscle cell proliferation and minimises scar tissue formation at the treatment site [578].
Efficacy
Optilume demonstrates rapid and sustained improvements in LUTS, as measured by IPSS. Studies report a ≥ 40% improvement in IPSS in 75-81% of patients at three months, with benefits persisting through one and even four years. The Qmax doubled post-treatment, with improvements maintained long-term (e.g. from 10.9 to 18.4mL/s at one year; +5.6mL/s at four years) [578-580].
Tolerability and safety
Most complications observed are mild and transient in nature, such as haematuria, UTI, dysuria and urinary retention. Serious adverse events related to the device or procedure are uncommon. Notably, larger balloon diameters have been associated with increased incidences of bleeding and incontinence [578-580].
Practical consideration
Randomised controlled trials comparing Optilume to a reference technique are required.
5.4. Patient selection for LUTS/BPO treatment
The choice of treatment depends on the assessed findings of patient evaluation, ability of the treatment to change the findings, treatment preferences of the individual patient, and the expectations to be met in terms of speed of onset, efficacy, side effects, QoL and disease progression.
Behavioural modifications, with or without medical treatments, are usually the first choice of therapy. Figure 3 provides a flow chart illustrating treatment choice according to evidence-based medicine and patient profiles. Surgical treatment is usually required when patients have experienced recurrent or refractory urinary retention, overflow incontinence, recurrent UTIs, bladder stones or diverticula, treatment-resistant macroscopic haematuria due to BPH/BPE, or dilatation of the upper urinary tract due to BPO, with or without renal insufficiency (absolute operation indications, need for surgery).
Additionally, surgery is usually needed when patients have not obtained adequate relief from LUTS or PVR using conservative or medical treatments (relative operation indications). The choice of surgical technique depends on prostate size, comorbidities of the patient, ability to have anaesthesia, patients’ preferences, willingness to accept surgery-associated specific side effects, availability of the surgical armamentarium, and experience of the surgeon with these surgical techniques. An algorithm for surgical approaches according to evidence-based medicine and the patient’s profile is provided in Figure 4.
Figure 3: Treatment algorithm of male LUTS using medical and/or conservative treatment options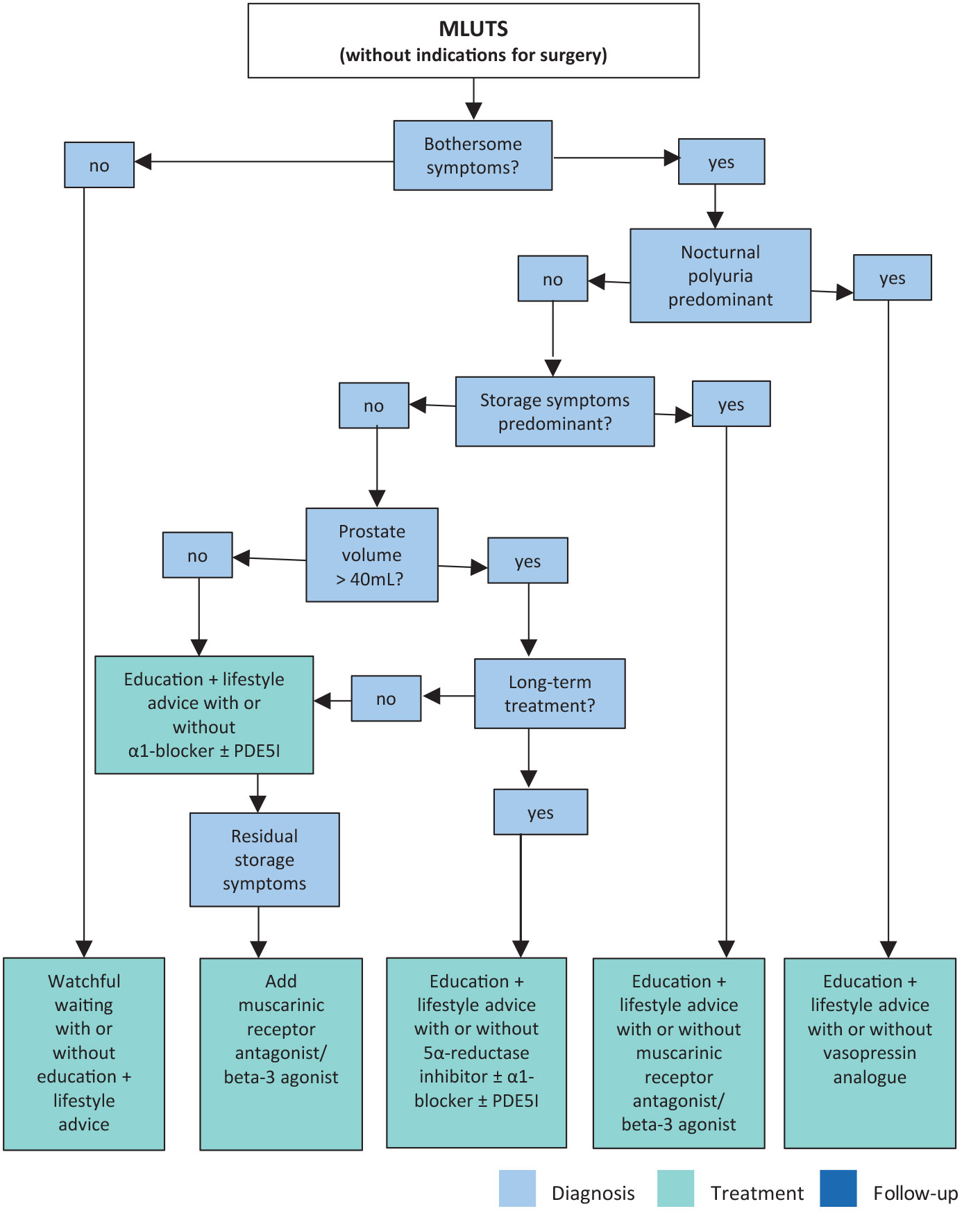 Treatment decisions depend on results assessed during initial evaluation.
Treatment decisions depend on results assessed during initial evaluation.
Note that patients’ preferences may result in different treatment decisions.
α1-blocker = alpha-1 adrenergic receptor blocker; 5α-reductase inhibitor = 5-alpha-reductase inhibitor; LUTS = lower urinary tract symptoms; MLUTS = male lower urinary tract symptoms; PDE5I = phosphodiesterase type 5 inhibitor.
Figure 4: Treatment algorithm of bothersome LUTS refractory to conservative/medical treatment or in cases of absolute operation indications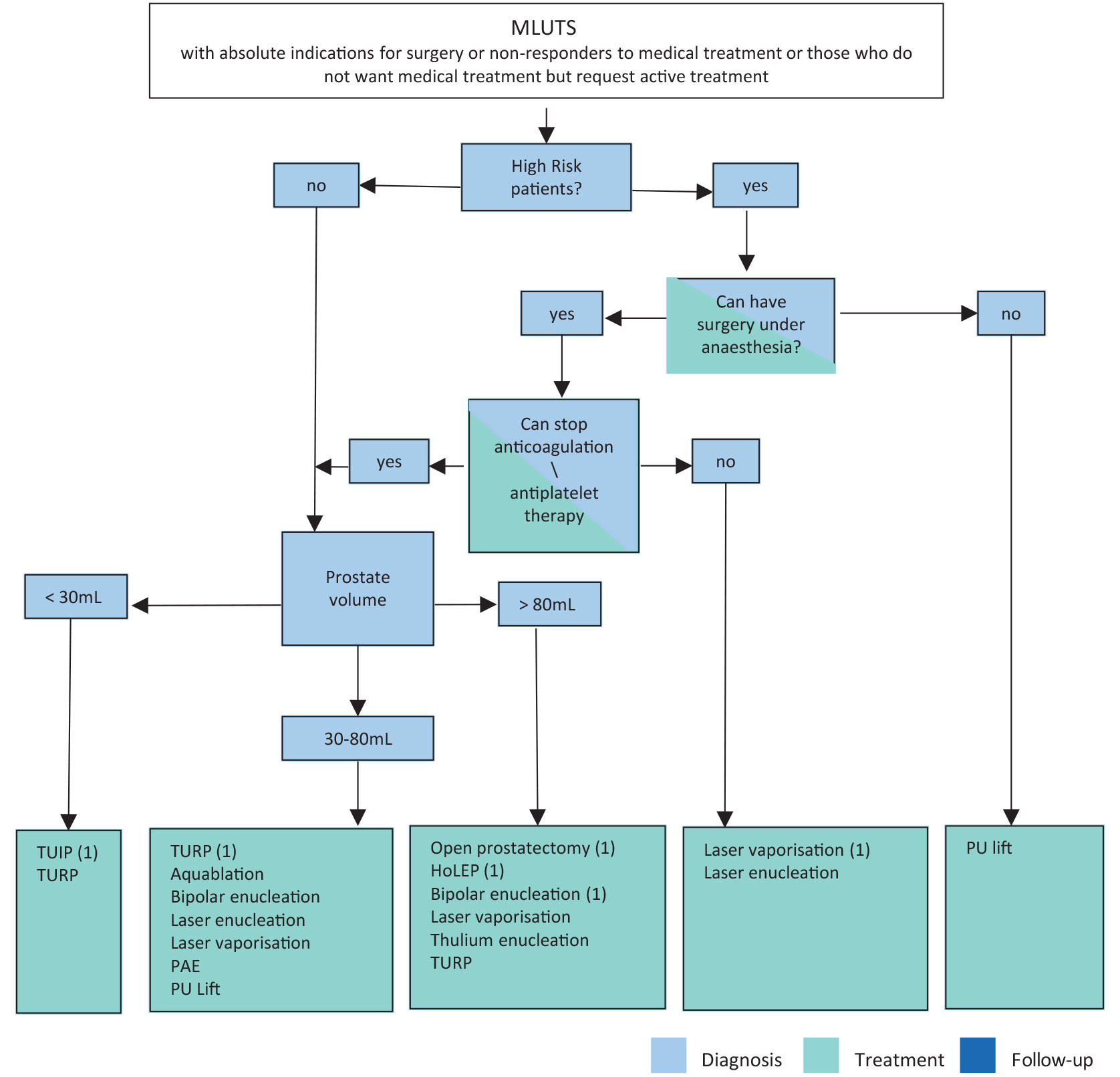 (1) Current standard /first choice. The alternative treatments are presented in alphabetical order.
(1) Current standard /first choice. The alternative treatments are presented in alphabetical order.
Laser vaporisation includes GreenLight, thulium and diode laser vaporisation.
Laser enucleation includes holmium and thulium laser enucleation.
HoLEP = holmium laser enucleation; TUIP = transurethral incision of the prostate; TURP = transurethral resection of the prostate; PU = prostatic urethral.
The flowchart is stratified by the patient’s ability to have anaesthesia, cardiovascular risk and prostate size [416, 421]
5.5. Management of nocturia in men with lower urinary tract symptoms
The following section reports an SR of therapy for the management of nocturia in men with LUTS. It also emphasises the need to consider the wide range of possible causes of nocturia [581].
Nocturia has been defined as the complaint of waking at night to void [3]. The ICS Standardisation Steering Committee has introduced the concept of main sleep period, defined as ‘the period from the time of falling asleep to the time of intending to rise for the next day’ [582].
Nocturia reflects the relationship between the amount of urine produced while asleep, and the ability of the bladder to store the urine received. Nocturia can occur as part of lower urinary tract dysfunction (LUTD), such as OAB and chronic pelvic pain syndrome. Nocturia can also occur in association with other forms of LUTD, such as BOO, but here it is debated whether the link is one of causation or simply the co-existence of two common conditions. Crucially, nocturia may have behavioural, sleep disturbance (primary or secondary) or systemic causes unrelated to LUTD (Table 2). Differing causes often coexist and each must be considered in all cases. Only where LUTD is contributory should nocturia be termed a LUTS.
Table 2: Categories of nocturia
| CATEGORY | Disproportionate urine production (at all times or during sleep) | Low volume of each void (at all times or overnight) |
| Behavioural | Inappropriate fluid intake | ‘Bladder awareness’ due to secondary sleep disturbance |
| Systemic | Water, salt and metabolite output | |
| Sleep disorder | Variable water and salt output | ‘Bladder awareness’ due to primary sleep disturbance |
| LUTD | Impaired storage function and increased filling sensation |
5.5.1. Diagnostic assessment
Evaluation is outlined in Figure 5.
- Evaluate for LUTD in accordance with the relevant Guidelines. The severity and bother of individual LUTS should be identified with a symptom score, supplemented by directed questioning if needed. A validated three-day bladder diary is mandatory.
- Review whether behavioural factors affecting fluid balance and sleep are contributing.
- Review medical history and medications, including directed evaluation for key conditions, such as renal failure, diabetes mellitus, cardiac failure and obstructive sleep apnoea. If systemic factors or sleep disorders are potentially important, consider involving appropriate medical expertise (see Figure 6). This is appropriate where a known condition is suboptimally managed, or symptoms and signs suggest an undiagnosed condition.
Figure 5: Evaluation of Nocturia in non-neurogenic MLUTS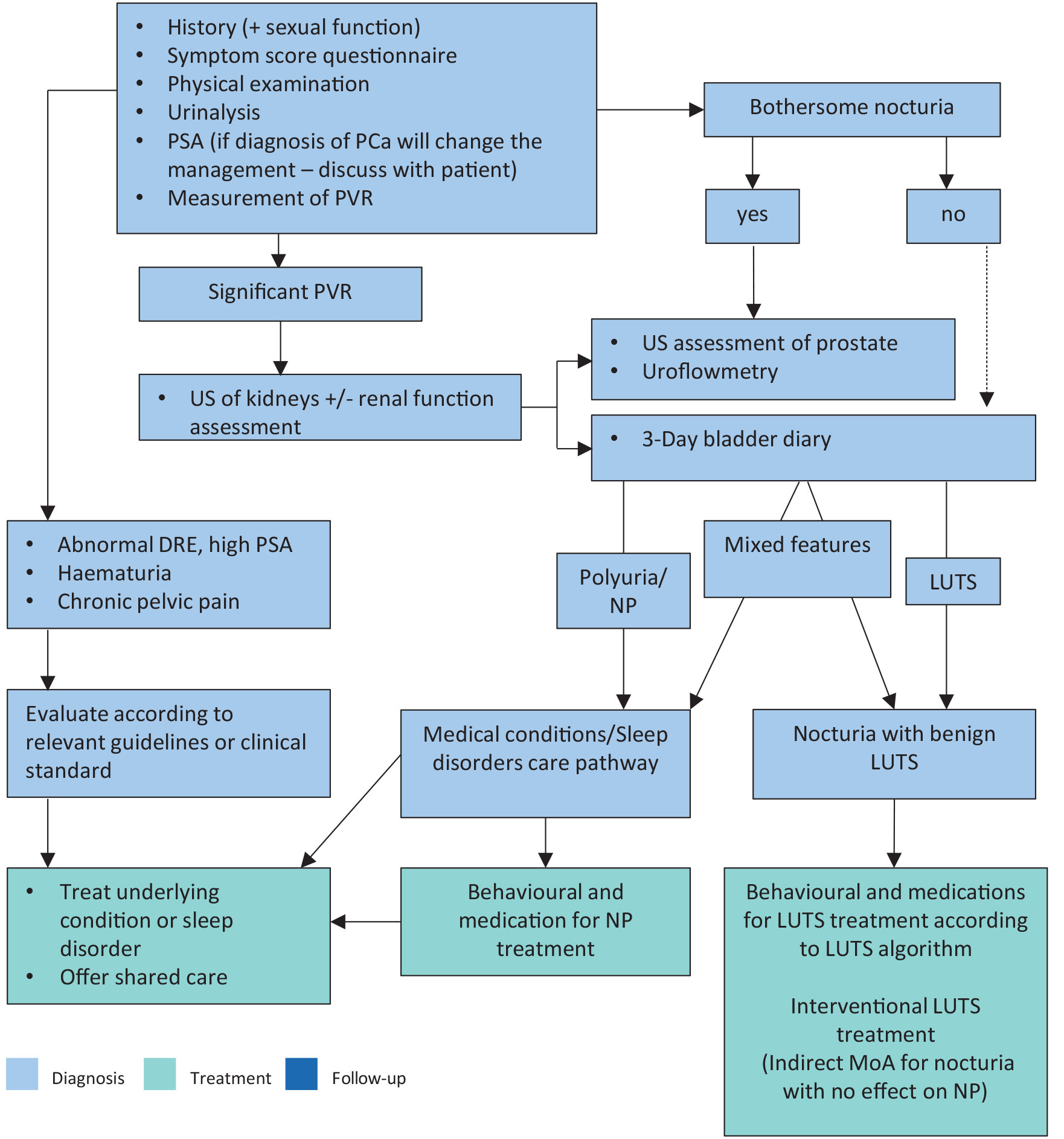 Assessment must establish whether the patient has 24-hour (global) polyuria, LUTS, a sleep disorder or a combination of these. Therapy may be driven by the bother it causes, but non bothersome nocturia may warrant assessment with a FVC (indicated by the dotted line) depending on history and clinical examination since potential presence of a serious underlying medical condition must be considered.
Assessment must establish whether the patient has 24-hour (global) polyuria, LUTS, a sleep disorder or a combination of these. Therapy may be driven by the bother it causes, but non bothersome nocturia may warrant assessment with a FVC (indicated by the dotted line) depending on history and clinical examination since potential presence of a serious underlying medical condition must be considered.
DRE = digital rectal examination; FVC = frequency volume chart; LUTS = lower urinary tract symptoms; MLUTS = male lower urinary tract symptoms; MoA = mechanism of action; NP = nocturnal polyuria; PCa = prostate cancer; PVR = post-void residual; PSA = prostate-specific antigen; US = ultrasound.
5.5.2. Medical conditions and sleep disorders shared care pathway
Causative categories for nocturia comprise [583]:
- Bladder storage problems
- 24-hour polyuria (> 40mL/kg urine output over a 24-hour period)
- Nocturnal polyuria (defined as excessive production of urine during the individual’s main sleep period, i.e. nocturnal output exceeding 20% of 24-hour urine output in young people, or 33% of urine output in people > 65 years [3], or > 90mL/min. in men [584])
- Sleep disorders
- Mixed aetiology
Potentially relevant systemic conditions are those that impair physiological fluid balance. These include factors that alter free water, salt and other solute levels, or plasma oncotic pressure; endocrine regulators such as antidiuretic hormone and natriuretic peptides; cardiovascular and autonomic control; renal function; and neurological mechanisms, including circadian regulation of the pineal gland and renal innervation. As nocturia is commonly referred to the specialty without full insight into cause, the urologist must review the likely mechanisms underlying a presentation with nocturia and instigate review by relevant specialties accordingly. Therefore, the managing urologist must evaluate nocturia patients in a context where additional medical expertise is available (Table 3). They should not proceed along any LUTD management pathway unless a causative link with LUTD is justifiably suspected, and systemic or sleep abnormalities have been considered.
In patients with non-bothersome nocturia, the medical evaluation (history and physical examination) should consider the possibility of early stages of systemic disease, and whether there is possibility of earlier diagnosis or therapy adjustment.
Some important, potentially treatable non-urological causes of nocturia include obstructive sleep apnoea, congestive cardiac failure, poorly controlled diabetes mellitus and medications (e.g. diuretics or lithium).
Table 3: Shared care pathway for nocturia, highlighting the need to manage potentially complex patients using relevant expertise for the causative factors
| Urological contribution | Shared care | Medical contribution |
Diagnosis of LUTD
| Diagnosis of conditions causing NP
| |
Conservative management
Interventional therapy
| Conservative management
| Management
* Potential causes of polyuria NEPHROLOGICAL DISEASE
CARDIOVASCULAR DISEASE
ENDOCRINE DISEASE
NEUROLOGICAL DISEASE
RESPIRATORY DISEASE
BIOCHEMICAL
|
IC = intermittent catheterisation; LUTD = Lower urinary tract dysfunction; LUTS = lower urinary tract symptoms; NP = nocturnal polyuria.
5.5.3. Treatment for nocturia
5.5.3.a. Antidiuretic therapy
The antidiuretic hormone arginine vasopressin (AVP) plays a key role in body water homeostasis and control of urine production by binding to V2 receptors in the renal collecting ducts. Arginine vasopressin increases water reabsorption and urinary osmolality, thus decreasing water excretion and total urine volume. Arginine vasopressin also has V1 receptor mediated vasoconstrictive/hypertensive effects and a very short serum half-life, which makes the hormone unsuitable for treating nocturia/NP.
Desmopressin is a synthetic analogue of AVP with high V2 receptor affinity and no relevant V1 receptor affinity. Desmopressin has been investigated for treating nocturia [585], with specific doses, titrated dosing, differing formulations and options for route of administration. Most studies have short follow-up. Global interpretation of existing studies is difficult due to the limitations, imprecision, heterogeneity and inconsistencies of the studies.
An SR of randomised or quasi-randomised trials in men with nocturia found that desmopressin may decrease the number of nocturnal voids by -0.46 compared to placebo over short-term follow-up (up to three months). Over intermediate-term follow-up (three to 12 months), there was a change of -0.85 in nocturnal voids in a substantial number of participants without increase in major adverse events [586].
Another SR of comparative trials of men with nocturia as the primary presentation and LUTS including nocturia or NP found that antidiuretic therapy using dose titration was more effective than placebo in relation to nocturnal voiding frequency and duration of undisturbed sleep [581]. Adverse events include headache, hyponatremia, insomnia, dry mouth, hypertension, abdominal pain, peripheral oedema and nausea. Three studies evaluating titrated-dose desmopressin in which men were included reported seven serious adverse events in 530 patients (1.3%) with one death. There were 17 cases of hyponatraemia (3.2%) and seven of hypertension (1.3%). Headache was reported in 53 patients (10%) and nausea in 15 (2.8%) [581].
Hyponatremia is the most important concern, especially in patients > 65 years of age, with potentially life-threatening consequences. Baseline values of sodium over 130mmol/L have been used as inclusion criteria in some research protocols. Assessment of sodium levels must be undertaken at baseline after initiation of treatment or dose titration and during treatment. Desmopressin is not recommended in high-risk groups [581].
Desmopressin oral disintegrating tablets (ODT) have been studied separately in the sex-specific pivotal trials CS41 and CS40 in patients with nocturia [587, 588]. Nearly 87% of included patients had NP and approximately 48% of the patients were > 65 years. The coprimary endpoints in both trials were change in number of nocturia episodes per night from baseline and at least a 33% decrease in the mean number of nocturnal voids from baseline during three months of treatment. The mean change in nocturia episodes from baseline was greater with desmopressin ODT compared to placebo (difference: women = -0.3 [95% CI: -0.5 to -0.1]; men = -0.4 [95% CI: -0.6 to -0.2]). The 33% responder rate was also greater with desmopressin ODT compared to placebo (women: 78% vs. 62%; men: 67% vs. 50%).
Analysis of three published placebo-controlled trials of desmopressin ODT for nocturia showed that clinically significant hyponatraemia was more frequent in patients aged ≥ 65 years than in those aged < 65 years in all dosage groups, including those receiving the minimum effective dose for desmopressin (11% of men aged ≥ 65 years vs. 0% of men aged < 65 years receiving 50mcg; 4% of women ≥ 65 aged years vs. 2% of women aged < 65 years receiving 25mcg). Severe hyponatraemia, defined as ≤ 125mmol/L serum sodium, was rare, affecting 22 of 1,431 (2%) patients overall [589]. In a recent cases series including men aged of more than 60 treated with 50mcg ODT, only 12/80 men had hyponatremia (no severe) after one week. Hyponatraemia persistence was noted in 4/12 cases after reduction to 25mcg [590].
Low-dose desmopressin ODT has been approved in Europe, Canada and Australia for the treatment of nocturia with ≥ two episodes in gender-specific low doses 50mcg for men and 25mcg for women. However, it initially failed to receive United States Food and Drug Administration (FDA) approval, with the FDA citing uncertain benefit relative to risks as the reason. Following resubmission to the FDA in June 2018 desmopressin acetate sublingual tablet, 50mcg for men and 25mcg for women, was approved for the treatment of nocturia due to NP in adults who awaken at least two times per night to void with a boxed warning for hyponatraemia.
Desmopressin acetate nasal spray is a new low-dose formulation of desmopressin and differs from other types of desmopressin formulation due to its bioavailability and route of administration. Desmopressin acetate nasal spray has been investigated in two RCTs including men and women with nocturia (over two episodes per night) and a mean age of 66 years. The average benefit of treatment relative to placebo was statistically significant but low: -0.3 and -0.2 for the 1.5mcg and 0.75mcg doses of desmopressin acetate, respectively. The number of patients with a reduction of more than 50% of nocturia episodes was 48.5% and 37.9%, respectively, compared with 30% in the placebo group [591]. The reported adverse event rate of the studies was rather low, and the risk of hyponatremia was 1.2% and 0.9% for desmopressin acetate 1.5mcg and 0.75mcg, respectively. Desmopressin acetate nasal spray was approved by the FDA in 2017 for the treatment of nocturia due to NP, but it is not available in Europe.
Practical considerations
A complete medical assessment should be made, to exclude potentially non-urological underlying causes, for example, sleep apnoea, before prescribing desmopressin in men with nocturia due to NP. The optimal dose differs between patients, in men < 65 years desmopressin treatment should be initiated at a low dose (0.1mg/day) and may be gradually increased up to a dosage of 0.4mg/day every week until maximum efficacy is reached. Desmopressin is taken once daily before sleeping. Patients should avoid drinking fluids at least one hour before and for eight hours after dosing. Low dose desmopressin may be prescribed in patients > 65 years. In men ≥ 65 years or older, low dose desmopressin should not be used if the serum sodium concentration is below normal: all patients should be monitored for hyponatremia. Urologists should be cautious when prescribing low-dose desmopressin in patients under-represented in trials (e.g. patients > 75 years) who may have an increased risk of hyponatraemia.
5.5.3.b. Medications to treat LUTD
Where LUTD is diagnosed and considered causative of nocturia, relevant medications for storage (and voiding), LUTS may be considered. Applicable medications include; selective α1-adrenergic antagonists [592], antimuscarinics [593-595], 5-ARIs [596] and PDE5Is [597]. However, effect size of these medications is generally small, or not significantly different from placebo when used to treat nocturia [581]. Data on OAB medications (antimuscarinics, beta-3 agonist) generally had a female-predominant population. No studies specifically addressing the impact of OAB medications on nocturia in men were identified [581]. Benefits with combination therapies were not consistently observed.
5.5.3.c. Other medications
Agents to promote sleep [598], diuretics [599], non-steroidal anti-inflammatory agents (NSAIDs) [600] and phytotherapy [601] were reported as being associated with response or QoL improvement [581]. Effect size of these medications in nocturia is generally small, or not significantly different from placebo. Larger responses have been reported for some medications, but larger scale confirmatory RCTs are lacking. One recent RCT has compared melatonin + tamsulosin compared to placebo + tamsulosin and found no difference on IPSS change [602]. Agents to promote sleep do not appear to reduce nocturnal voiding frequency but may help patients return to sleep.
| Summary of evidence | LE |
| No clinical trial of pathophysiology-directed primary therapy has been undertaken. | 4 |
| No robust clinical trial of behavioural therapy as primary intervention has been undertaken. | 4 |
| Antidiuretic therapy reduces nocturnal voiding frequency in men with baseline severity of two or more voids per night. | 1b |
| There is an increased risk of hyponatremia in patients 65 years of age or older under antidiuretic therapy. | 1b |
| Antidiuretic therapy increases duration of undisturbed sleep. | 1b |
| Alpha 1-blocker use is associated with improvements in undisturbed sleep duration and nocturnal voiding frequency, which are generally of only marginal clinical significance. | 2 |
| Antimuscarinic medications can reduce night-time urinary urgency severity, but the reduction in overall nocturia frequency is small or non-significant. | 2 |
| Antimuscarinic medications are associated with higher incidence of dry mouth compared with placebo. | 2 |
| 5α-reductase inhibitors reduce nocturia severity in men with baseline nocturia severity of two or more voids per night. | 2 |
| A trial of timed diuretic therapy may be offered to men with nocturia due to NP. Screening for hyponatraemia should be undertaken at baseline and during treatment. | 1b |
| Recommendations | Strength rating |
| Treat underlying causes of nocturia, including behavioural, systemic condition(s), sleep disorders (e.g. obstructive sleep apnoea), lower urinary tract dysfunction, or a combination of factors. | Strong |
| Discuss behavioural changes with the patient to reduce nocturnal urine volume and episodes of nocturia and improve sleep quality. | Strong |
| Offer desmopressin to decrease nocturia due to nocturnal polyuria in men < 65 years of age. | Weak |
| Screen for hyponatraemia at baseline, day three and day seven, one month after initiating therapy and periodically during treatment. Measure serum sodium more frequently in patients > 65 years of age and in patients at increased risk of hyponatraemia. | Strong |
| Discuss with the patient the potential clinical benefit relative to the associated risks from the use of desmopressin, especially in men > 65 years of age. | Strong |
| Offer α1-adrenergic antagonists for treating nocturia in men who have nocturia associated with voiding LUTS. | Weak |
| Offer antimuscarinic drugs for treating nocturia in men who have nocturia associated with overactive bladder. | Weak |
| Offer 5α-reductase inhibitors for treating nocturia in men who have nocturia associated with LUTS and an enlarged prostate (> 40mL). | Weak |
| Do not offer phosphodiesterase type 5 inhibitors for the treatment of nocturia. | Weak |
5.6. Management of male urinary incontinence
The aim of the following Section is to provide evidence-based recommendations for the management of male UI.
5.6.1. Epidemiology and pathophysiology
Urinary incontinence is defined as an unintentional loss of urine and is reported to have a prevalence of 11% in men aged 60-64 years old to 31% in men ≥ 85 years and to affect up to 32% of men with LUTS [603-605]. Urinary incontinence can be further classified into three types: SUI, UUI and mixed urinary incontinence (MUI). Overflow urinary incontinence, post-micturition dribble, nocturnal enuresis and total incontinence are specific forms of UI that are outside the current scope of these Guidelines. Table 4 provides an overview of the epidemiology and pathophysiology of male UI.
Table 4: Epidemiology and pathophysiology overview of male UI
| Type | Definition | Causes and associated factors | Pathophysiology | Clinical presentation |
| Stress UI: prevalence < 10% | Urine loss during movement, on effort or in general during increased abdominal pressure. |
| Sphincter deficiency | Symptoms: UI during physical activity, exercises, e.g. during coughing, sneezing, no leakage during sleep Voiding diary/pad test: with activity Voiding diary/Pad test: with activity Cough stress test: leakage can coincide with coughing |
| Urgency UI: prevalence 40-80% | Urine loss concomitant or immediately following an urgency episode. |
|
| Symptoms: urgency, usually associated with, increased frequency and nocturia
Voiding diary: urgency, frequency and nocturia, incontinence |
| Mixed UI: prevalence 10-30% | Any combination of SUI and UUI. | Causes of both SIU and UUI | Combination of SUI and UUI | Symptoms: UI on effort and with a sense of urgency Voiding diary: variable Cough stress test: may show leakage with coughing |
BPO = benign prostatic obstruction; GI = gastrointestinal; SUI = stress urinary incontinence; UI = urinaryincontinence; UTI = urinary tract infection; UUI = urgency urinary incontinence.
5.6.2. Diagnostic evaluation
Medical history and physical examination of males with UI is the same as for MLUTS (Figure 2) and should allow UI to be categorised into SUI, UUI or MUI and to identify other types of UI (overflow UI, nocturnal enuresis), or those who need rapid referral to an appropriate specialist (e.g. pelvic diseases, neurological conditions).
Specific validated questionnaires, such as the ICIQ-UI-SF, can help to quantify UI severity. However, a detailed description of the various urinary symptoms questionnaires and Patient-Reported Outcome Measures is beyond the scope of these Guidelines. For more information on available questionnaires, see the 7th ICI review on patient reported outcomes assessment [611].
Voiding diaries are a standardised method of objectively assessing symptom severity, including frequency and extent of UI episodes, voided volume and 24-hour or nocturnal total urine volume [44].
Pad tests can be used to quantify severity of UI and to monitor patient’s response to treatment although the usefulness of these tests in differentiating between the different types of incontinence or predicting outcome of treatment is uncertain. Despite this, early postoperative testing with pad tests may predict future continence in men after radical prostatectomy [612, 613].
5.6.2.a. Urodynamic testing
Urodynamic testing
Urodynamic studies allow for an objective characterisation of the type of UI and the identification of additional storage and/or voiding dysfunctions [614].
UDS in patients with UI following BPO surgery
Sphincter injury is the most common finding in incontinent men after BPO surgery in some published urodynamic series [615]. However, data coming mostly from case series and reviews, show that besides sphincter incompetence, other LUTD, assessed via UDS, play an incidental role in post BPO surgery incontinence.
Urodynamics has been performed in a retrospective study on 125 patients with incontinence after surgery for BPO. Sphincter injury was the most common finding (66.7% and 79.6% of patients who underwent TURP and OP, respectively [615]). However, decreased detrusor compliance and DO have also emerged as a cause of incontinence on urodynamic examination. Interestingly, both impaired detrusor contraction and DO alone were responsible for UI in 4 to 14% of patients [615].
UDS in patients with UI following radical prostatectomy
Also in this case, UI is mainly due to sphincter deficiency. However, an SR on UDS findings in UI patients following radical prostatectomy showed that while intrinsic urethral sphincter deficiency was reported as the sole cause of UI following radical prostatectomy in 8-71% of cases, it was variably associated with detrusor dysfunction in 0-88% of cases. In details, sphincter weakness was associated with DO in 0-100% of patients, with reduced bladder compliance in 18-58% and with both DO and reduced bladder compliance in 4-64%. Interestingly, incontinent patients who did not complain of sphincter impairment showed detrusor dysfunction as follows: DO in 0%-4%, reduced bladder compliance in 1-12%, and DO plus impairment of bladder compliance in 1-7% [616].
UDS in patients with storage symptoms
In a retrospective evaluation of 668 UDS studies involving men with symptoms of urgency with or without urgency UI, DO was documented in 258 patients (38.6%) and 293 patients (43.9%) had evidence of BOO during PFS. The symptom UI correlated with the presence of DO [617].
The aforementioned evidence emphasises how urodynamic examination serves to clarify the origin of incontinence and the presence of any other associated LUTS. The level of the evidence for the impact of UDS in the evaluation/management/outcome prediction of male patients with UI remains low due to lack of studies [618] and does not allow the Panel to produce any recommendations.
Therefore, UDS should be considered in selected patients with UI or persistent/de novo LUTS/UI after surgery, mainly when invasive treatments are considered.
Post-void residual volume measurement can be applied with caution to men with non-neurogenic UI, because the prevalence, severity and clinical application of PVR in men with UI is uncertain. However, a high PVR measurement may indicate further testing is required, like invasive UDS or renal US [619].
Urethrocystoscopy can be performed in selected patients to exclude urethral or bladder neck strictures. Some tests have been proposed to evaluate any sphincter function, for example, repositioning tests, but with poor reliability. The examination of the bladder helps to exclude tumours, stones and diverticula that may exacerbate UI [620].
Imaging (US, MRI, CT scan) can improve the understanding of the anatomical and functional abnormalities that may cause UI and thus help its management [621].
Urinalysis: Reagent strip (‘dipstick’) urinalysis may indicate UTI, proteinuria, haematuria, or glycosuria, requiring further tests as recommended according to other EAU Guidelines, for example, Guidelines on urinary tract cancers and urological infections [49-52].
| Summary of evidence | LE |
| Validated specific symptom score questionnaires and voiding diaries assist in the screening for and categorisation of UI. | 3 |
| Pad weight test can be used to quantify the presence and severity of UI, as well as a patient’s response to treatment. | 3 |
| There is limited evidence that UDS and PVR affect treatment choice in men with uncomplicated UI. | 3 |
| Recommendations | Strength rating |
| Take a complete medical history including symptoms and comorbidities, medications and a focused physical examination in the evaluation of men with urinary incontinence (UI). | Strong |
| Use a validated symptom score questionnaire, bladder diary and pad weight test to assess UI. | Strong |
| Measure post-void residual in the assessment of UI. | Strong |
| Perform invasive urodynamics for UI when considering invasive treatment. | Weak |
5.6.3. Conservative treatment
5.6.3.a. Simple clinical interventions
5.6.3.a.1. Lifestyle interventions
Examples of lifestyle factors that may be associated with UI include obesity, smoking, level of physical activity, and diet. Modification of these factors may improve UI, but most of the evidence for these interventions come from studies with predominately female study populations. As many of these interventions are now generalised public health measures, their recommendation is in line with general medical practice [622-624]. Current evidence does not allow adequate personalisation of lifestyle interventions according to the type of UI.
Modification of fluid intake, particularly restriction, is a strategy commonly used by people with UI to relieve symptoms. Advice on fluid intake given by healthcare professionals should be based on 24-hour fluid intake and urine output measurements. From a general health point of view, it should be advised that fluid intake should be sufficient to avoid thirst and that low or high 24-hour urine output should be investigated [622, 625, 626]. A cross-sectional population survey found no statistical association between caffeine intake and UI [627]. Conversely, an RCT showed that reduction of caffeine intake, associated with behavioural therapy, resulted in reduced urgency but not UI compared to behavioural therapy alone [628].
5.6.3.a.2. Treatment of comorbidities
Urinary incontinence, especially in the elderly, has been associated with multiple comorbid conditions [629]. Improvement of associated disease may reduce the severity of urinary symptoms, however, this is often difficult to assess as patients frequently suffer from more than one condition. Interventions may be combined and individualised, making it impossible to decide which alteration in an underlying disease has affected a patient’s UI.
In patients with existing UI, particularly the elderly, it may be difficult or impossible to distinguish between the effects of medication, comorbidities or ageing on UI. Although changing drug regimens for underlying diseases may be considered as a possible early intervention, there is limited evidence of benefit [630]. There is also a risk that stopping or altering medication may result in greater harm than benefit.
5.6.3.a.3. Constipation
One RCT with a majority female population found that a multimodal intervention in elderly patients involving assisted toileting, fluid intake and so on reduced the occurrence of UI and constipation, while behavioural therapy appeared to improve both [631]. However, there is no evidence to show whether treating constipation improves UI, although both constipation and UI appear to be improved by certain behavioural interventions.
5.6.3.a.4. Containment
Containment methods include the use of absorbent pads, urinary catheters, external collection devices and penile clamps. An SR of six RCTs comparing various types of pads found that pads filled with super absorbent material were better than standard pads [632]. For men with light UI, a randomised crossover trial found that a leaf-shaped type of pad was preferred to rectangular pads [633].
A Cochrane review compared various types of long-term indwelling and intermittent [634] catheters and found no evidence that one catheter material or type of catheter was superior to another [635]. An SR of non-randomised studies found no differences in UTI outcome or upper urinary tract changes between the use of suprapubic or urethral catheter drainage. However, patients with suprapubic catheters were less likely to have urethral complications [636].
An RCT in 56 men with post-prostatectomy incontinence (PPI) compared sheath drainage system, body-worn urinal, penile clamp and absorbent pads. The results showed that the three devices and absorbent pads have different strengths and limitations that make them more (or less) suitable for particular activities. Most men prefer to use a combination of devices and pads to meet their lifestyle needs. A hinge-type penile clamp was effective for brief, vigorous activities, because it was the most secure, least likely to leak and most discreet, although almost all men described it as uncomfortable or painful [637].
| Summary of evidence | LE |
| There is limited evidence that lifestyle interventions, for example, weight reduction, smoking cessation or diet modification, improves UI in males. | 3 |
| There is limited evidence that improving comorbidities or changing drug regimens for underlying disease improves UI in males. | 3 |
| Pads and/or penile sheaths are palliative options for management of both SUI and UUI. | 1b |
| Recommendations | Strength rating |
| Inform the patient that lifestyle modifications may improve urinary incontinence (UI); however, patients should be informed that the evidence for these interventions is lacking. | Strong |
| Review medical conditions and medication associated with the development or worsening of UI. | Strong |
| Discuss containment options (pads, sheaths, clamps) as a palliative option for the management of UI in a shared decision-making process. | Strong |
5.6.3.b. Behavioural and physical therapies
Behavioural and physical therapies encompass all treatments which require a form of self-motivated personal retraining by the patient and include techniques that are used to augment this effect. Usually in clinical practice, these will be introduced as part of a package of care including lifestyle changes, patient education and possibly cognitive therapy, as well. Further details for behavioural treatments are outlined in Section 5.1.2 of these Guidelines.
5.6.3.b.1. Prompted or timed voiding
With prompted voiding, carers rather than the patient initiate the decision to void. Two SRs confirmed a positive effect on continence outcomes for prompted voiding in comparison to standard care [638,639]. Timed voiding is defined as fixed, predetermined, time intervals between toileting, applicable for those with or without cognitive impairment. A Cochrane review of timed voiding, included two RCTs, found inconsistent improvement in continence compared with standard care in cognitively impaired adults [640].
5.6.3.b.2. Bladder training
Bladder training goals are to correct faulty habit patterns of frequent urination, improve control over bladder urgency, prolong voiding intervals, increase bladder capacity, reduce incontinent episodes, and restore patient confidence in controlling bladder function. The ideal form or intensity of a BT program for UI is unclear. It is also unclear whether BT can prevent the development of UI. The addition of BT to anticholinergic therapy did not improve UI compared to antimuscarinics alone, but it did improve frequency and nocturia [641]. In seven RCTs, BT was compared to drug therapy alone and showed only a benefit for oxybutynin in cure and improvement of UI [641].
5.6.3.b.3. Pelvic floor muscle training
A Cochrane review concluded that there was no overall benefit at 12 months post-surgery for men who received postoperative pelvic floor muscle training (PFMT) for the treatment of PPI and that the benefits of conservative treatment of PPI remain uncertain [642]. However, a subsequent SR and meta-analysis showed that PFMT either alone or in combination with biofeedback and/or electrical stimulation was effective for treating PPI, significantly reducing the time to continence recovery [643]. A further meta-analysis demonstrated that the addition of guided programs using biofeedback and/or pelvic floor muscle electric stimulation significantly improved continence recovery rates at one- and three-month intervals post-surgery compared to PFMT alone [644].
Two subsequent SRs in patients who underwent robotic-assisted radical prostatectomy (RARP) demonstrated that the addition of PFMT to the postoperative management plan shortened the time to continence recovery [645, 646].
Other RCTs demonstrated that, like PFMT, increased pelvic floor muscle strength and quicker return to continence may be achieved with the pilates method [647], the oscillating rod [648], a combination of biofeedback with electrostimulation [649] and whole-body vibration training [650]. Moreover, quicker return to continence and improved QoL may be achieved, even with extended and continuing nursing care [651].
The role of supervised PFMT remains controversial.
Pelvic floor muscle training is commonly started as soon as possible after surgery (i.e. between seven and ten days after the withdrawal of the urethral catheter) [652]. However, one RCT found that PFMT was helpful in men who had been incontinent for at least one year after prostatectomy and who had had no previous therapy [653].
Two RCTs have shown that written instructions alone offer similar levels of improvement to supervised PFMT [654, 655]. An RCT comparing the outcomes of an eight-week home-based PFMT with action and cue observation (n = 46) and without (control, n = 46) in patients with UI after RP revealed significantly larger improvements in 24-hour pad test in the active treatment group (MD: 106.1g; 95% CI: 13.4g, 199.1g, p = 0.037) [656].
An RCT compared supervised PFMT with a physiotherapist (started two months before RARP and continued for 12 months after) versus verbal instructions plus a brochure about PFMT plus lifestyle advice. Significantly higher UI improvements were observed following supervised PFMT in terms of 24-hour pad weight at three months (5.0g vs. 21.0g; effect size: 0.34). A significant improvement was also seen in the intervention group in terms of continence rates (no pad use) at 12 months after surgery (65.2% vs. 31.6%) [657].
An RCT showed that adding comprehensive lifestyle recommendations + Knack manoeuvre to conventional PFMT provides greatest improvement in terms of subjective severity and impact of UI, objective severity of UI, health-related QoL and patient global impression of severity and improvement at eight weeks [658].
One RCT compared PFMT to no treatment in men undergoing TURP. No demonstrable difference was observed in the incidence of postoperative incontinence up to 12 months [659]. On the other hand, an RCT in men who underwent HoLEP demonstrated that PFMT, when started preoperatively, promoted early recovery of continence [660].
The role of preoperative PFMT role remains to be established.
5.6.3.b.4. Electrical stimulation
Electromagnetic stimulation has been promoted as a treatment for UI, but only weak evidence of the short- and long-term effects has been reported in SRs [661, 662].
An SR found insufficient evidence to suggest that electrical stimulation is beneficial for male patients with PPI [663].
An RCT of 70 PPI men receiving surface or intra-anal electrostimulation reported a significant reduction in UI in terms of grams of urine loss and a significant improvement in QoL from baseline, but no statistically significant difference between treatment arms [664].
A Cochrane review of six RCTs on electrical stimulation in men with UI concluded that there was some evidence that electrical stimulation enhanced the effect of PFMT in the short-term but not after six months. Electrical stimulation was also more effective than sham stimulation at six, but not 12 months; however, there were more adverse effects including pain and discomfort with electrical stimulation [665].
5.6.3.b.5. Posterior tibial nerve stimulation
Posterior tibial nerve stimulation (PTNS) has been studied as a treatment of UI, especially UUI. Electrical stimulation of the posterior tibial nerve delivers electrical stimuli to the sacral micturition centre via the S2-S4 sacral nerve plexus. Stimulation is done either percutaneously using a fine, 34-G needle inserted just above the medial aspect of the ankle (P-PTNS) or transcutaneously using surface electrodes (T-PTNS). Percutaneous-PTNS treatment cycles typically consist of 12 weekly treatments of 30 minutes, and T-PTNS treatment cycles typically consists of self-administered 20-minute daily sessions after adequate education.
A female-predominant, sham-controlled RCT assessed the effectiveness of P-PTNS in OAB population. The treatment and sham arms included 22.8% and 20% males, respectively [666]. Overactive bladder symptoms improved significantly in 54.5% of patients in the P-PTNS arm compared to 20.9% in the sham arm. A noninferiority RCT comparing T-PTNS to P-PTNS reported significant improvements in daytime frequency, urgency and UUI episodes without significant difference between treatment arms after 12 weeks of therapy [667]. An SR on T-PTNS in idiopathic and neurogenic female-predominant (males < 10%) population, reported significant improvement in OAB symptoms in 48-93% of patients and cure of UUI episodes in 25-45% [668].
For T-PTNS, mild pain and discomfort at the puncture site is the most common adverse event [669]. Small hematomas, swelling, leg tingling and vasovagal reaction to needle placement have also been reported [666]. Treatment adherence is generally high at 89.7% [667]. Contraindications include a cardiac pacemaker and skin disease at the site of stimulation.
There is some evidence that (T- or P-) PTNS may benefit male patients with OAB, but due to insufficient data, no recommendation on PTNS in males can be made at this time. However, considering the safety of this therapy, PTNS can be offered to male patients as an alternative option prior to more invasive treatments.
| Summary of evidence | LE |
| Prompted voiding, either alone or as part of a behavioural modification programme, improves continence in elderly, care-dependent people. | 1b |
| The combination of BT with antimuscarinic drugs does not result in greater improvement of UI but may improve frequency and nocturia. | 1b |
| There is conflicting evidence on whether the addition of BT, electrostimulation or biofeedback increases the effectiveness of PFMT alone. | 1b |
| Preoperative PFMT does not confer additional benefit to men undergoing radical prostatectomy. | 1b |
| Electrical stimulation may add benefit to PFMT up to six months. | 2 |
| There is limited evidence for the effectiveness of PTNS in male population. | 2 |
| There is no evidence that PTNS cures UUI in male population. | 2 |
| Recommendations | Strength rating |
| Implement prompted voiding for patients with urinary incontinence (UI) where appropriate. | Strong |
| Offer bladder training as a complementary treatment for UI. | Strong |
| Offer pelvic floor muscle training alone or in combination with biofeedback and/or electrostimulation to men undergoing radical prostatectomy to speed recovery from UI. | Weak |
5.6.4. Pharmacological management
5.6.4.a. Drugs for urgency urinary incontinence (UUI)
Muscarinic receptor antagonists [670-673] and beta-3 agonists [318-320, 674-677] are currently the first-line pharmacological treatments for UUI. The mechanism of action, efficacy and safety and tolerability profiles of both classes of drugs are discussed in detail in Sections 5.2.3 and 5.2.4, respectively.
5.6.4.b. Drugs for stress urinary incontinence
An SR of eight studies evaluating the efficacy of duloxetine in postprostatectomy SUI reported that duloxetine resulted in a mean dry rate of 58% (25-89%), mean improvement in pad number of 61% (12-100%), and mean improvement in one-hour pad weight of 68% (53-90%) at short-term follow-up (mean one to nine months) [678]. However, mean adverse event rates were high, and treatment was discontinued in 38% of cases. The most common adverse events included fatigue, somnolence and nausea, and were reported in 18% of patients [678]. The overall certainty of evidence was low due to study heterogeneity and methodological limitations. Further RCTs with long-term follow-up are required.
Duloxetine is currently prescribed in an off-label setting in men with SUI.
| Summary of evidence | LE |
| Antimuscarinic monotherapy can significantly improve urgency, UUI and increased daytime frequency. | 1b |
| Beta-3 agonist is superior to placebo and as efficacious as antimuscarinics for improvement of UUI. | 1b |
| Duloxetine led to a short-term improvement in postprostatectomy SUI symptoms and QoL improvement, however, a considerable proportion of men discontinued treatment. | 1b |
| Recommendations | Strength rating |
| Offer antimuscarinic drugs or beta-3 agonists* to adults with urgency urinary incontinence who failed conservative treatment. | Strong |
| Offer duloxetine to men with stress urinary incontinence. | Weak |
| Inform patients about the possible adverse events of duloxetine and that its use is off-label for this indication in Europe. | Strong |
*Further evidence is awaited for Vibegron.
5.6.5. Surgical treatment for stress urinary incontinence
5.6.5.a. Bulking agents in men
Mechanism of action
Urethral bulking agents work by adding bulk and improving the coaptation of a damaged sphincter zone. They represent a treatment option for men with either small volume leak or for those unfit for more invasive treatment options [679].
Efficacy
A Cochrane review on surgical treatment of PPI identified only one RCT that fulfilled the inclusion criteria. This trial randomised 45 men to Macroplastique injection or artificial urinary sphincter (AUS) implantation and compared their outcomes at 48 months [679]. Significant improvement was reported in both groups for men with minimal incontinence, but in men with total incontinence there was a significant difference in continence rates favouring AUS implantation (72% vs. 23%) [680]. An SR of eight studies (n = 142) in men using Macroplastique, Opsys, Durasphere and Urolastic showed short-term improvement and reported dry rates between 0 and 83% [679]. A propensity score-matched analysis of 104 men with post-prostatectomy stress urinary incontinence (PP-SUI), compared submucosal injection of Macroplastique to transobturator male sling (TiLOOP male) [681]. At 12-month follow-up, the reported failure free rates were 15.4% and 76.9%, the daily use of zero to one pads was 21.2%, and 67.3% and the satisfaction rate was 3.8% and 71.2%, respectively. Several small cohort studies of several different bulking agents have not shown any benefit.
A narrative review including data from 25 articles reported a success rate with all bulking procedures of 54.3%, with a 37.5% symptoms improvement and nearly 30% of dryness [682].
In an SR and meta-analysis, three studies addressed bulking agents. Two of them, involving a total of 384 participants, showed a pooled short-term cure rate of 26.1%, and a single study on 68 subjects reported a 10.3% long term cure rate. Short- and long-term reoperation rates were not described [683].
Tolerability and safety
Bulking agent-associated dysuria and haematuria are frequently reported to be transient and self-resolving [679]. The risk of urinary retention requiring intermittent catheterisation (IC), or long-term catheter use is minimal [684]. However, they may provoke allergic reactions [685] and carry a potential risk for migration [686] to proximal and distal lymph nodes [687]. Overall, post-procedural urinary retention rates range between 3 and 17% with rare need for temporary catheterisation, while postoperative UTIs ranged from 6 to 7% [682].
Practical considerations
Bulking agents have shown low cure rates but remain an option for men unfit for more invasive treatment options. They currently do not play a role in clinical practice. Further clinical trials are warranted.
| Summary of evidence | LE |
| There is very limited evidence that bulking agents are effective for the treatment of PP-SUI. | 2 |
5.6.5.b. Male slings
Male slings are used to treat mild-to-moderate PP-SUI [688]. However, the definitions of mild and moderate UI are unclear. Most studies define cure as ‘no pad use’ or ‘one security pad per 24 hours.’ Some authors used stricter criteria, such as ‘urine loss of less than 2g per 24-hour pad test’ [689].
5.6.5.b.1. Nonadjustable slings
Nonadjustable male slings are positioned under the urethra and fixed by means of a retropubic or transobturator approach. The tension is adjusted during the surgery and cannot be readjusted postoperatively. Synthetic slings restore continence in males either by urethral compression and/or by repositioning the bulb of urethra [690, 691].
Efficacy
An SR and meta-analysis involving 33 prospective cohorts and one RCT comparing sling to AUS reported that both options are effective in improving UI and QoL [692]. Following sling insertion, the overall cure rate was 60% (95% CI: 0.51-0.67) and 56% (95% CI: 0.44-0.68) for sling and AUS, respectively. The 24-hour pad use was -3.33 (95% CI: -4.33 to -2.34) and -3.75 (95% CI: -4.56 to -2.93) for slings and AUS, respectively. Similar findings were reported by a network meta-analysis that showed comparable efficacy between slings and AUS [693].
The MASTER trial, a non-inferiority RCT comparing the outcomes of continence surgery in men with bothersome urodynamic SUI, using a very strict definition of UI after prostate surgery reported that, at 12 months, incontinence rates were 87% for male sling versus 84.2% for AUS (95% CI: -11.6-4.6; PNI = 0.003), confirming non-inferiority [694]. The subgroup analysis suggested that male sling is inferior to AUS for men with greater incontinence at baseline (pad weight > 250g), however, the difference did not reach statistical significance.
For the repositioning sling (AdVance™ and AdVanceXP©), a mean subjective cure rate of 49% (8.6-73.7%) after mean follow-up of three years has been reported for 136 patients [688]. A prospective multicentre cohort study with 60-month follow-up in men with AdVanceXP demonstrated a constant continence outcome over time with a 57.6% cure rate, 25.4% improvement rate and 16.9% failure rate. These findings were verified in an additional study which reported cure rates of 66.7% and 71.7%, improvement rates of 26.5% and 24.4%, and failures rates of 6.9% and 13.3% at 24 and 48 months, respectively [695]. A retrospective comparative study showed that both options are safe and effective in the treatment of male SUI [696].
With the transobturator compressive I-Stop TOMS male sling, 38% of men were dry at 12 months, but this dropped to 23% and 15% after four and five years, respectively [697].
Tolerability and safety
An SR and meta-analysis of 1,170 men with SUI and male sling reported that the predictors of failure are prior radiation, severity of incontinence and previous surgeries [698]. Pelvic radiotherapy has also been reported in other studies as a negative prognostic factor [699]. A comparison among radiated versus nonradiated men who had AdVanceXP reported a greater degree of postoperative improvement in the nonradiated group (49.6% vs. 22.2%), as well as greater satisfaction rates (95% vs. 64%) [700]. The most common complication with male slings is pain and local superficial wound infection [701]. Chronic pain has been observed in 1.3% of men who had nonadjustable slings [701]. Postoperative transient voiding dysfunction occurred in 4.3-10.3%, mostly as de novo urgency or urinary retention, while erosions and chronic pain were uncommon (0-0.4%), as was explantation [688, 695, 702-704].
Practical considerations
Fixed male slings are considered safe and improve continence, but their efficacy is limited in men with severe incontinence or previous radiotherapy.
5.6.5.b.2. Adjustable slings in males
Mechanism of action
Adjustable slings in males are those for which the tension of the sling can be adjusted postoperatively. Three main systems have been used in men: Remeex® [705], Argus® [706] and ATOMS® [707].
Efficacy
One small RCT was carried out for adjustable slings in males [708]. Most studies consist of prospective or retrospective case series with variable follow-up and various definitions of success [705, 707-711]. An SR reported objective cure rates varying between 17 and 92% after adjustable sling implantation [701].
For the Remeex® system, only two studies with conflicting findings have been published. One study followed 19 patients for nearly seven years and reported 70% success, with no explants, infections or erosions [705]. The second study followed 14 patients for 25 months. Only 36% of patients were satisfied and multiple readjustments were needed. Mechanical failure was reported in 21% [709].
Data on the Argus® system has been reported for 404 men, but only a few series have reported on more than 40 patients, with the longest follow-up being 35-months. Success rates varied between 17-93%, with a mean of 73.0% reporting subjective cure [710,711]. A head-to-head comparison between the two Argus® systems reported similar efficacy outcomes at 44 months, but Argus T® was associated with a higher inguinal pain and explantation rate [712]. A small study of 22 men with PPI randomised to AdVance® or Argus T® reported superior 24-hour pad test results and of patient satisfaction levels for Argus T at 18-month follow-up [708].
An SR of the ATOMS® system reported the pooled evidence from 1,393 patients with 67% dryness, 90% improvement after adjustment and 16.4% complication rate [707]. The expulsion rate was 5.75%. Another SR and meta-analysis with 3,059 patients reported that ATOMS® was superior to ProACT® in mean dryness rate (68% vs. 55%), overall improvement (91% vs. 80%), satisfaction rate (87% vs. 56%), mean number of filing adjustments (2.4 vs. 3.5) and postoperative pad use per day (1.1 vs. 2.1) [713].
Tolerability and safety
The most frequent complications in adjustable male slings are pain, erosions and infections [701]. Pain at the implant site was usually only temporary, but chronic pain has been reported in 1.5% of men [710,711]. The number of implants requiring readjustment is reported between 8 and 38.6% [711, 714, 715]. Explantation (partial or total) rates range from 10 to 15.8% and the erosion rate is estimated at approximately 10% [698]. The most common reasons for explantation are device infection (4.1-8%), erosions (4-12%) and urethral perforations (2.7-16%). An SR reported a device explantation rate of 5% versus 25% and a major complication rate of 4.2% versus 10.4% for ATOMS® and ProACT®, respectively [713].
Practical considerations
There is no evidence that adjustability offers additional benefit as RCTs are lacking. Therefore, no recommendation can be made at this time. Explantation rate seems superior to fixed male sling based on external comparisons.
5.6.5.b.3. Autologous slings
The strategy of intraoperative placement of an autologous vas deferens sling below the vesicourethral anastomosis during RARP has been explored with the intention to improve early return of continence. Two comparative studies [716, 717] showed an advantage of sling versus no sling at one-month follow-up, and another study [718] showed an advantage of a six-branch versus a two-branch sling at one-month follow-up. However, a larger RCT (n = 195) showed that continence rate and near-continence rate were similar between groups at six months with 66% versus 65% and 86% versus 88%, respectively [719].
| Summary of evidence | LE |
| There is limited short-, medium- and long-term evidence that fixed transobturator male slings cure or improve PP-SUI in patients with mild-to-moderate SUI. | 1b |
| Men with severe incontinence, previous radiotherapy or transurethral surgery may have less benefit from fixed transobturator male slings. | 2 |
| There is limited evidence that adjustable male slings can cure or improve SUI in men. | 3 |
| There is no evidence that adjustability offers additional benefit over other types of slings. | 3 |
| Explantation rate of adjustable male slings seems higher than fixed male sling. | 4 |
| Recommendations | Strength rating |
| Offer nonadjustable transobturator slings to men with mild-to-moderate* post-prostatectomy stress urinary incontinence. | Weak |
| Inform men that severe incontinence, prior pelvic radiotherapy or transurethral surgery may worsen the outcome of nonadjustable male sling surgery. | Weak |
* The terms ‘mild’ and ‘moderate’” PPI remain undefined.
5.6.5.c. Compression devices in males
5.6.5.c.1. Artificial urinary sphincter
Mechanism of action
The AUS is the standard treatment for moderate-to-severe male SUI. The AMS 800 three-component system with inflatable cuff, control pump and pressure regulating balloon is the device with the longest follow-up and the greatest level of evidence [720]. The ZSI 375 is a two-component device, inflatable cuff and control pump, allowing an easier implantation process [721]. Other AUS devices have been launched, for example, the Victo, UroActive® and Br-SL-AS 904 systems, but robust evidence regarding their efficacy and safety is pending [722].
Efficacy
A meta-analysis of 33 cohort studies and one RCT reported significant improvement after AUS implantation in overall cure rates (56%) and reductions in pad use per 24-hours (-3.75) [692]. Several observational studies reported on functional outcomes after AMS 800. Social continence rates (zero to one pads used daily) ranged from 55 to 76.8% [723-725]. A 77.2% continence rate and 89.5% subjective satisfaction rate have been reported after a median follow-up of > 15 years in 57 men who had undergone AUS placement [726]. A prospective cohort study of 40 patients with a mean follow-up of 53 months showed that, of all urodynamic parameters, only low bladder compliance had a negative impact on outcome [727].
However, another retrospective study showed that no urodynamic factors adversely altered the outcome of AUS implantation [728].
Some recent multicentre studies have confirmed older statements that surgeon experience and higher surgical volume is associated with better outcomes and a lower revision rate after AUS implantation [729, 730].
The data regarding ZSI 375 are limited. A retrospective, non-randomised trial across several centres in Europe, reported an 84.4% success rate (19.3% dry rate and 65.1% improved zero to one pads per day) after 43 months [721]. A 72% success rate was reported at seven years’ follow-up for 45 patients who underwent placement of the ZSI 375 device in France [731].
A retrospective study of 168 patients receiving AUS (AMS 800 AUS system) placements carried out by a single surgeon from 2008 to 2016 at a high-volume academic institution evaluated the causes of AUS failure at a median follow-up from initial placement of 2.7 years. Overall, 63 patients (37.5%) experienced AUS failure requiring device explant/revision. Pressure-regulating balloon malfunction, cuff malfunction, pump malfunction, urethral atrophy, urethral erosion and infection occurred in 36.5%, 7.9%, 6.3%, 22.2%, 19.0% and 12.7% of all AUS failures, respectively. History of previous pelvic radiotherapy, urethral stricture, urethral sling placement and coronary artery disease were independent predictors of all-cause AUS failure [732].
A retrospective review of all cases of AUS implantation performed between 2005 and 2020 in 16 different French centres, including patients with primary implantation whose indication was moderate-to-severe SUI after radical prostatectomy (n = 417) or BPO surgery (n = 50) found similar social continence rates (zero or one pad per day) at three months between the groups (79% vs. 72%) [733].
Tolerability and safety
Artificial urinary sphincter complications include device infection/erosion (8.5%), mechanical failure (6.2%) and urethral atrophy (7.9%) [734]. In multivariate analysis, radiation therapy was independently associated with risk of urethral atrophy [725]. Urethral erosion is associated with previous irradiation and penoscrotal approach [735]. The reported revision rates at three years for any reason were 10-29.1% [723, 735-737]. The risk of urethral erosion after ZSI 375 AUS is 8.2-13.3% and risk of mechanical failure is 2.2-2.5% [721].
A retrospective database comparison found that significantly higher complication rates in patients have been reported to be significantly higher in patients undergoing AUS implantation due to UI following BPO surgery compared to patients with post-prostatectomy UI (8% vs. 18%; p = 0.044). The same was found for the Clavien-Dindo type 2 complication rate (20.6% vs. 44.4%; p = 0.026) [733].
A retrospective chart review of 1,020 patients who underwent AUS implantation in 16 centres showed an estimated five-year and ten-year overall revision-free survival of 60% and 40%, respectively. Reasons for revision were mechanical failures (27.6%), nonmechanical failures (56.5%) and other causes (15.9%). In multivariable analysis, larger cuff size was the only predictor of overall revisions (HR = 1.04 [1.01-1.07]; p = 0.01) and revision for nonmechanical failure (HR = 1.05 [1.02-1.09]; p = 0.004) [738].
A population-based retrospective cohort study including 8,475 men who underwent a first-ever AUS implantation for male SUI after prostate cancer or BPH treatment in France found reintervention-free survival rates of 71%, 57% and 40% at two, five and ten years, respectively. Reintervention-free survival was lower after BPH surgery, after radiotherapy combined with RP and in centres performing fewer implantations. Removal-free survival rates were 83%, 75% and 66% at two, five and ten years, respectively [739].
Practical considerations
Artificial urinary sphincter is efficacious and improves the QoL of men with PP-SUI. To minimise complications, it is advised to refer patients to specialised centres experienced in AUS implementation. Men considering insertion of an AUS should be fully informed that the success of the intervention relies on their ability to operate the pump. Treating physicians should bear in mind that operating the AUS may become difficult in men who develop cognitive impairment or lose manual dexterity. Artificial urinary sphincter has a limited lifespan and ‘maintenance’ reoperations are common in the long-term. These reinterventions should not be classified as complications [720].
The European prospective multicentre SATURN registry was developed to analyse surgical devices for male SUI. One-year self-reported cure rates (zero to one pads/die) by device were as follows: Advance-XP, 73%; other sling, 37%; ProACT®, 50%; AMS800, 76%; other AUS, 11%; and overall group, 68%. Overall, 132 patients had at least one revision (n = 122 for 110 patients with an AUS; n = 29 for 22 patients with a sling or ProACT®) [740].
5.6.5.c.2. Non-circumferential compression device (ProACT®)
Mechanism of action
The ProACT® system consists of two devices. Each device includes the balloon, the bilumen tubing and the volume-adjustment port. The devices are introduced by a trocar by means of two small perineal incisions and are placed under fluoroscopic guidance on each side of the bladder neck, close to the vesicourethral anastomotic site. The balloons can be filled and their volume can be adjusted postoperatively using a hypodermic needle injected through the intra-scrotal port.
Efficacy
An SR and meta-analysis of 19 studies including 1,264 patients reported a 60.2% dry rate, significant reduction in number of pads used per day (-3.1) and greatly improved QoL for ProACT® [741]. However, the level of heterogeneity among the included studies was high. A comparison between ATOMS® and ProACT® showed that the former is associated with higher improvement and satisfaction rates and fewer complications [713]. A quasi-randomised trial comparing ProACT® with bone-anchored male slings found that both resulted in similar improvements in SUI (68% vs. 65%, respectively) [742]. A questionnaire study showed that 50% of patients were still significantly bothered by persistent incontinence following ProACT® [743]. A subgroup analysis of radiotherapy patients reported worst outcomes as compared to patients not receiving radiotherapy (46% vs. 68% success rate) as well as a higher percentage of urethral erosion for ProACT® [744].
An updated SR and meta-analysis (18 studies involving 1,570 patients suffering from mild-to-moderate [60.7%] and severe [40.4%] post-RP SUI) showed an overall dryness rate (zero to one pads/day) of 55.1% at a mean follow-up of: 34.7 months [745].
Tolerability and safety
The most common intra-operative complication during ProACT® implantation is perforation of the bladder and/or urethra. A meta-analysis estimated a perforation rate of 5.3% [741]. The estimated overall revision rate is 22.2%, and the main causes are erosion (3.8%), device leaking (4.1%) and migration (6.5%) [741]. Other prospective series have shown that adverse events were frequent, leading to an explantation rate of 11-58% [742, 743, 746-748].
A meta-analysis reported a mean overall complication rate of 31.2%, an explantation rate of 26.5%, and a reoperation rate of 22.7% at a mean follow-up of 34.7 months [745].
Practical considerations
ProACT® has a satisfactory rate of success and appears to be a reasonable alternative for the treatment of male UI. However, it is associated with high complication rates.
| Summary of evidence | LE |
| Primary AUS implantation is effective for cure of SUI in men. | 1b |
| Prior pelvic radiotherapy is likely to have a negative impact on the outcomes of AUS implantation. | 3 |
| The non-circumferential compression device (ProACT®) is effective for treatment of PPI-SUI, however, it is associated with a high failure and complication rate leading to frequent explantation and particularly after pelvic radiation therapy. | 2b |
| The rate of explantation of the AUS due to infection or erosion remains high (up to 24% in some series). | 3 |
| Recommendations | Strength rating |
| Offer artificial urinary sphincter (AUS) to men with moderate-to-severe stress urinary incontinence. | Strong |
| Implantation of AUS or ProACT® for men should only be offered in expert centres. | Weak |
| Warn men receiving AUS or ProACT® that, although cure can be achieved there is a high risk of complications, mechanical failure and the need for explantation. | Strong |
| Do not offer non-circumferential compression device (ProACT®) to men who have had pelvic radiotherapy. | Weak |
5.6.6. Surgical treatment for urgency urinary incontinence
5.6.6.a. Bladder wall injection of botulinum toxin-A
Mechanism of action
The primary mechanism of action of BoNT-A is through the inhibition of neurotransmitter release from cholinergic neurons [749]. OnabotulinumtoxinA (onabotA; BOTOX®) 100 U is licenced in Europe to treat OAB with persistent or refractory non-neurogenic UUI in adults [750, 751].
Efficacy
An RCT of OAB-wet patients whose symptoms were not adequately managed with anticholinergics and who receive either bladder wall injections of onabotA (100 U) or saline reported a 50% reduction in UUI episodes/day, whilst the number of micturitions/day reduced by more than two in patients receiving onabotA [752]. A total of 22.9% of the patients in the onabotA arm were fully dry versus 6.5% in the saline arm.
An SR and meta-analysis comparing the efficacy of onabotA, mirabegron and anticholinergics in adults with idiopathic OAB reported that patients who received onabotA (100 U) achieved greater reduction in UUI episodes, micturition frequency and the highest odds of achieving dryness, as well as > 50% reduction from baseline UUI episodes per day [753].
A randomised, placebo-controlled pilot study assessing the effect of onabotA for the treatment of refractory OAB symptoms after prostatectomy reported significantly improved QoL and ICIQ scores and improvements in daily frequency in patients receiving onabotA compared to placebo [754]. A retrospective trial assessed onabotA efficacy in 65 non-obstructed men with refractory OAB and reported significant improvement in UDI-6 score (-4.2) and IIQ-7 (-6.0) scores compared to baseline [755].
In a retrospective, single-centre cohort study of 120 patients treated with onabotA for OAB, lower IC rates were reported in male patients with prior de-obstructive surgery compared with surgery-naïve patients (28.6% IC in the group without prior surgery, 7.5% in the TURP subgroup, and 4.2% in the radical prostatectomy subgroup) [756].
A phase IIIb trial of randomised solifenacin-naïve patients (10% males) with refractory OAB to onabotA, solifenacin or placebo showed that patients receiving onabotA had significantly greater changes in UUI episodes (-3.19) compared to solifenacin (-2.6) and placebo (-1.33) [757].
A network meta-analysis (male population range 9.8-40.2%) that compared onabotA to mirabegron demonstrated that onabotA was associated with improved outcomes in frequency episodes per day (-0.43; [-1.22-0.37]) and in UUI episodes per day (-0.46, [-1.46-0.53]) [758].
Tolerability and safety
Urinary retention and UTIs are the two most common adverse events after onabotA injection. Other reported adverse events include haematuria, dysuria and post-treatment pain [759]. Compared to mirabegron, onabotA is associated with higher risk for UTI and treatment emergent adverse events [758]. A retrospective analysis compared the use of IC after onabotA injection among men who had previous prostatectomy versus those without prior surgery [756]. A 7.5% catheterisation rate after TURP, 4.2% rate after radical prostatectomy and 28.6% rate in men without prior prostate surgery was reported.
Practical considerations
BoNT-A injection is a treatment option for men with refractory UUI. Despite the lack of a universally accepted injection protocol, gender-specific studies and absence of studies in BPO patients, BoNT-A seems superior to medical therapy. BoNT-A is associated with higher UTIs and urinary retention risk coupled with the need for repeated injections. A dedicated series in male population focused on treatment persistence has shown a high discontinuation rate (68.8%) [760]. Patients treated for OAB with onabotA treatment that have not undergone prior de-obstruction are more likely to develop retention and subsequent IC.
| Summary of evidence | LE |
| A single treatment session of onabotA (100 U) injected in the bladder wall is more effective than placebo at curing and improving UUI/OAB symptoms and QoL. | 2 |
| There is no evidence that repeated injections of onabotA have reduced efficacy, but discontinuation rates are high. | 3 |
| There is an increased risk of urinary retention and UTI with onabotA injections. | 2 |
| Recommendations | Strength rating |
| Offer bladder wall injections of onabotulinumtoxinA (100 U) to patients with overactive bladder/urgency urinary incontinence refractory to medical therapy. | Weak |
| Warn patients of the limited duration of response, risk of urinary tract infection and the possible prolonged need for intermittent catheterisation (ensure that they are willing and able to do so). | Strong |
5.6.6.b. Sacral neuromodulation (SNM)
Mechanism of action
Sacral neuromodulation (SNM) delivers low amplitude electrical impulses to the sacral nerve roots via an electrode implanted adjacent to the third sacral nerve root and connected to an attached pulse generator implanted in the buttock. Sacral neuromodulation work by modulating neural activity, thus stabilising bladder electrical activity through an unknown mechanism. It is a two-stage process: in the first stage, a percutaneous nerve evaluation lead or tined lead electrode is placed percutaneously near the S3 (or if not found, near the S4) root and linked to an external stimulator to assess the response. If symptoms reduced by at least 50%, patients are candidates for the second stage, which is the full implant.
Efficacy
Several trials assess the clinical effectiveness of SNM. All RCTs suffer from the limitation that patients and assessors cannot be blinded to the treatment allocation since all recruited subjects had to respond to a test phase before randomisation. In addition, the percentage of male population in these trials is approximately 10%. A meta-analysis compared the effectiveness of SNM to onabotA and reported no significant difference in successfully treated cases at six-months follow-up (RR 0.93; 95% CI: 0.63-1.39) [761].
Tolerability and safety
The main complications after SNM are pain at the implant site (13-42%), lead migration (4.0-21%), leg or back pain (3.0-18%) and wound infection (5.7-6.7%). Surgical revision is required in 29-33% of patients due to device malfunction, battery or device replacement or lead migration [762].
Practical considerations
SNM represents an alternative to onabotA in patients with refractory OAB, as it has shown good success rates and an acceptable safety profile.
| Summary of evidence | LE |
| Sacral nerve neuromodulation is effective after failed conservative treatment for OAB/UUI, but no sham controls have been used. | 2a |
| Recommendation | Strength rating |
| Offer sacral neuromodulation to patients who have urgency urinary incontinence refractory to medical therapy and are willing to undergo surgical treatment. | Weak |
5.6.6.c. Cystoplasty/urinary diversion
Mechanism of action
Augmentation cystoplasty involves the interposition of a detubularised segment of bowel into the bivalved bladder wall, aiming to increase bladder capacity and reduce OAB related symptoms. Urinary diversion remains a reconstructive option for patients with intractable UUI after multiple pelvic procedures, radiotherapy or pelvic pathology leading to irreversible sphincteric incompetence or fistula formation.
Efficacy
There are no RCTs comparing bladder augmentation to other treatments for patients with refractory OAB/UUI. In a large study with three-years follow-up, augmentation cystoplasty resulted in a postoperative continence rate of 93% in idiopathic DO patients, 78% in neurogenic overactivity and up to 90% when an AUS was implanted, respectively [763]. The largest case series of bladder augmentation in an idiopathic population included only women [764]. At an average follow up of 75.4 months only 53% were continent and satisfied with the surgery, whereas 25% had occasional leaks and 18% continued to have disabling UUI. A small prospective mixed gender trial reported high patient satisfaction rates with augmentation cystoplasty versus onabotA therapy [765]. A small study comparing ileal with colonic conduits concluded that there were no differences in the relative risks of upper urinary tract infection and uretero-intestinal stenosis [766]. However, there are no studies that have specifically examined these techniques in the treatment of intractable OAB/UUI [766]. Therefore, careful consideration on which operation is undertaken will depend on thorough preoperative counselling, access to stoma/continence nurses as well as patient factors to allow for fully informed patient choice.
Tolerability and safety
Cystoplasty and urinary diversion are major urological operations. The early postoperative complications include infection, bowel obstruction, bleeding and cardiorespiratory complications. Long-term complications include metabolic disturbances (hyperchloraemic metabolic acidosis if ileum is used), change in bowel habits, increased mucus production, stone formation, bladder perforation and rarely bladder cancer [767]. Following augmentation cystoplasty or diversion, the majority of patients will depend on self-catheterisation for bladder emptying. Patients with urinary conduit will depend on lifelong urine bags.
Practical considerations
Augmentation cystoplasty and urinary diversion represent realistic treatment options for men with refractory OAB. However, both options involve a major operation with a non-negligible long-term complication rate and a lifelong reliance on catheterisation or urine bags.
| Summary of evidence | LE |
| There is limited evidence of the effectiveness of augmentation cystoplasty and urinary diversion in treatment of idiopathic OAB. | 3 |
| The need to perform IC following augmentation cystoplasty is high. | 3 |
| Augmentation cystoplasty and urinary diversion are associated with high risks of short- and long-term complications. | 3 |
| There is no evidence comparing the efficacy or adverse effects of augmentation cystoplasty to urinary diversion. | 3 |
| Recommendations | Strength rating |
| Offer augmentation cystoplasty to patients with overactive bladder (OAB)/urgency urinary incontinence (UUI) who have failed all other treatment options and are able and willing to perform self-catheterisation. | Weak |
| Inform patients undergoing augmentation cystoplasty of the high risk of complications, the risk of having to perform intermittent catheterisation and the need for life-long surveillance. | Strong |
| Only offer urinary diversion to patients who have failed less invasive therapies for the treatment of OAB/UUI, who will accept a stoma. | Weak |
Figure 6: Evaluation and management of urinary incontinence in non-neurogenic MLUTS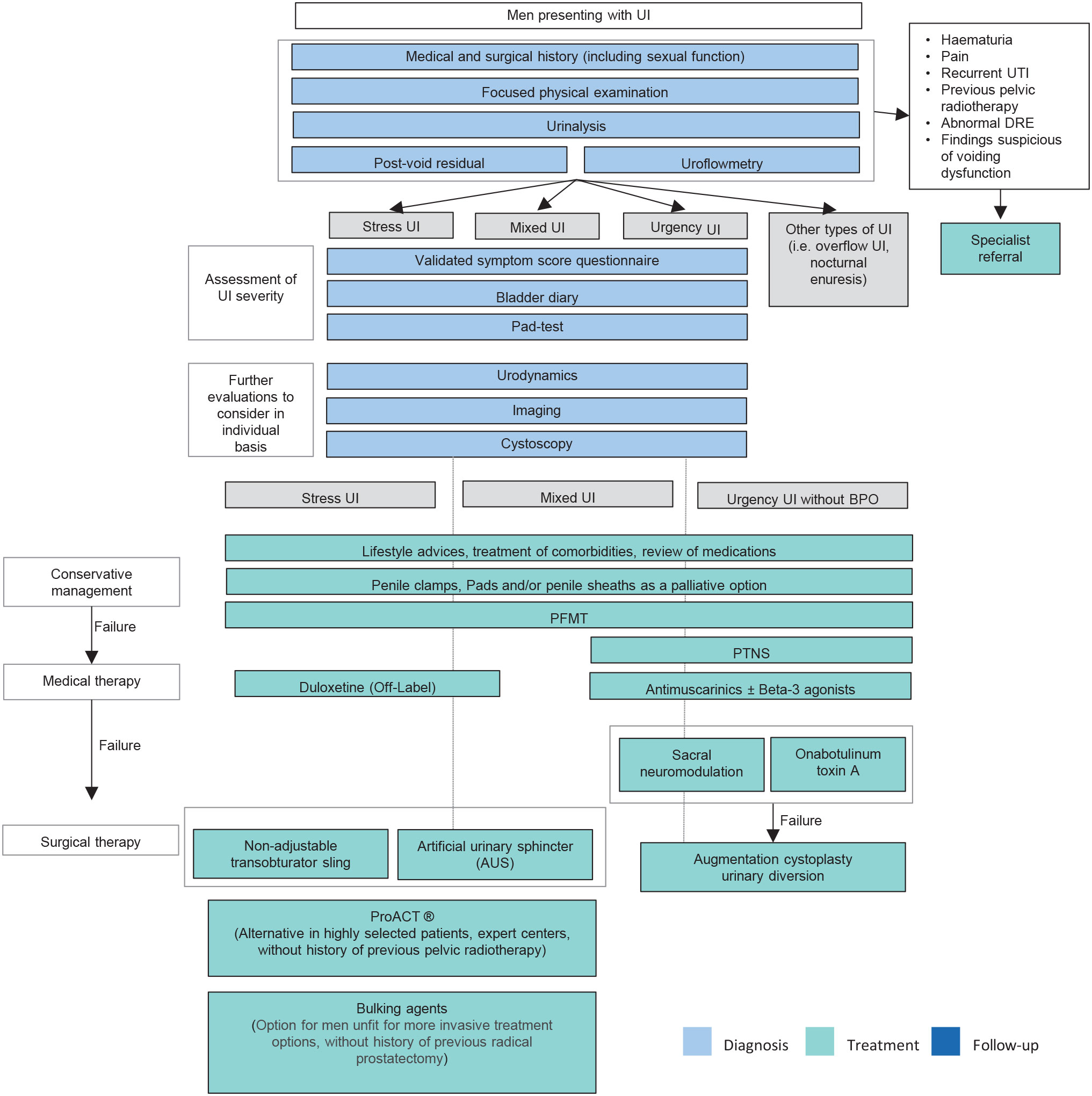 BPO = benign prostatic obstruction; DRE = digital rectal examination; LUTS = lower urinary tract symptoms; PFMT = pelvic floor muscle training; PTNS = posterior tibial nerve stimulation; UI = urinary incontinence;UTI = urinary tract infection.
BPO = benign prostatic obstruction; DRE = digital rectal examination; LUTS = lower urinary tract symptoms; PFMT = pelvic floor muscle training; PTNS = posterior tibial nerve stimulation; UI = urinary incontinence;UTI = urinary tract infection.
5.7. Management of underactive bladder
5.7.1. Epidemiology and pathophysiology
Various definitions of UAB and DU can be identified in the current literature. Detrusor underactivity appears as the most consistent concept, is based on UDS, and is defined by the International Continence Society as: ‘a contraction of reduced strength and/or duration, resulting in prolonged bladder emptying and/or failure to achieve complete bladder emptying within a normal time span’ [17]. Underactive bladder is a terminology that should be reserved for describing symptoms and clinical features related to DU. A tentative definition has been proposed as ‘a symptom complex suggestive of DU and usually characterized by prolonged urination time with or without a sensation of incomplete bladder emptying, usually with hesitancy, reduced sensation on filling, and a slow stream’ [768].
The prevalence of DU in the general population is unknown, because men with DU are either asymptomatic or have nonspecific LUTS. In clinical studies of men with non-neurogenic LUTS referred for VUDS, the prevalence of DU has been reported to be 10% [769,770] ranging up to 48% in the elderly (≥ 70 years) [771]. Detrusor underactivity is a chronic condition, but its natural history in untreated men has shown a plateau-like course with few symptomatic and urodynamic changes over time [772].
Healthy voluntary bladder muscle contraction requires a functional detrusor muscle, intact efferent and afferent innervation and integrated central neural control mechanisms. Dysfunction of any of these essential components can lead to DU, which may be the consequence of various pathological processes, suggesting a multifactorial pathophysiology.
Neurogenic
Neurogenic DU may be the consequence of peripheral or central nervous system disease. This etiology is covered in the EAU Guidelines on Neuro-Urology [773].
Myogenic
Several conditions can affect the myocytes or their extracellular matrix, resulting in attenuated detrusor contraction. Bladder outflow obstruction and diabetic cystopathy are common causes of myogenic DU. However, many aspects of the pathophysiology of myogenic DU that relate to BOO and diabetic cystopathy are mainly based on animal studies and the clinical picture in any individual patient is probably multifactorial in nature [774, 775]. Some SRs have addressed the role of chronic BOO on bladder dysfunction in humans. Aside from the impact on smooth muscle cells, multiple other consequences were identified including modifications on the extracellular matrix, and damage to the urothelium, suburothelium and bladder innervation. To date, BOO-related DU pathophysiology remains poorly understood [776].
Iatrogenic
Patients may experience DU following pelvic surgery (e.g. radical prostatectomy or extended rectal surgery) and/or radiation therapy [774]. Pharmacological treatments (e.g. drugs with anticholinergic effects or opioids) may also be involved in the impairment of detrusor contractility [774].
Idiopathic
Given the higher prevalence of DU in the elderly, it has been hypothesised that ageing would be a major contributor. However, available data do not provide strong evidence to support the assertion that detrusor contractile function declines with common ageing [774].
5.7.2. Diagnostic evaluation
5.7.2.a. Medical history and physical examination
A review of medical history, including comorbidities and current medications, can identify potential causes of UAB. The history should also include a thorough evaluation of LUTS, which should be classified into storage, voiding and post-micturition symptoms. There is no pivotal symptom to identify patients with UAB. The clinical presentation ranges from asymptomatic cases to symptomatic chronic urinary retention. Since UAB is a disorder of the voiding phase, voiding symptoms are the predominant symptoms, but they may be associated with storage symptoms, particularly in case of incomplete bladder emptying or other concomitant bladder dysfunctions [777].
Clinical diagnosis is even more difficult when patients have other conditions that may affect the presentation of LUTS, such as known or suspected BOO/BPO. In this setting, no validated tool is available to ascertain the respective roles of DU and BOO on voiding symptoms.
The Bristol group was the first to attempt to identify systematically which of the LUTS are most closely related to UAB [778]. Men with UAB reported a statistically significantly higher occurrence of decreased and/or interrupted urinary stream, hesitancy, feeling of incomplete bladder emptying, palpable bladder, feeling of incomplete bowel emptying, absent and/or decreased sensation and always straining to void compared with men with normal PFS. Several authors have proposed predictive models based on the patient’s LUTS to distinguish individuals with an UAB from patients with normal PFS or BOO [779, 780]. However, there is no conclusive evidence that one prognostic model is more accurate than another, and there is no published external validation of these models.
5.7.2.b. Questionnaires
There is no specific validated questionnaire for the diagnosis of UAB. Physicians can refer to validated questionnaires for MLUTS, but their clinical benefit to monitor symptom changes and treatment in patients with UAB is uncertain [781].
5.7.2.c. Uroflowmetry
Some authors have proposed to distinguish UAB from BOO based on uroflowmetry parameters, not only on Qmax, but on flow patterns and combinations of scores [782, 783]. The diagnostic accuracy of the developed models remains to be established, and external validation is lacking.
5.7.2.d. Ultrasound scan and post-void residual measurement
Ultrasound findings have been evaluated as noninvasive predictors of DU. In a single-centre prospective study including 143 adult males with LUTS, DWT ≤ 1.23mm and bladder capacity > 445mL was associated with urodynamically proven DU, with sensitivity and specificity of 42% and 100%, respectively [784].
Detrusor underactivity is often associated with prolonged bladder emptying time and/or PVR urine. However, while high PVR value have been associated with presence of DU, no consensus cut-off has been identified to diagnose DU and it is unlikely that one will ever exist [785].
5.7.2.e. Urodynamics
Underactive bladder is a clinical diagnosis based on sign and symptoms, and DU is a term that should be reserved for a urodynamic diagnosis [786]. Invasive UDS is the only widely accepted method for diagnosing DU [777]. However, current data do not allow for a consensus definition of the urodynamic criteria to be used for the diagnosis of DU. Three algorithms, which are all based on computer-urodynamic investigation and quantification of detrusor pressure during voiding, have been suggested to quantify detrusor power [785]:
- Griffiths’ Watt factor: quantification of detrusor power with a formula consisting of detrusor pressure during voiding, contraction speed, and BV at each point of micturition, expressed as W/m2. Detrusor power varies during voiding, single calculations are usually offered on urodynamic evaluation sheets, for example, maximum detrusor power (Wmax) or detrusor power at maximum flow (WQmax). However, it remains controversial which of the calculations and what threshold value should be used. Expert opinion suggested using a Wmax threshold value of 7.0W/m2 [785].
- Schafer’s detrusor-adjusted mean PURR factor (DAMPF): detrusor power can be quantified as very weak, weak, normal or strong if linearised passive urethral resistance (linPURR) is drawn into the Schafer nomogram. The length of linPURR determines detrusor strength.
- Bladder contractility index: quantification of detrusor power/contractility can be derived from Schafer’s linPURR lines and calculated by the formula: BCI = pdetQmax + 5Qmax. BCI>150 describes strong contractility, 100-150 normal contractility, and < 100 weak contractility. The BCI is currently the most widely used in the literature and clinical trials.
None of these models are validated and their concordance for the diagnosis of DU is uncertain [787, 788], preventing a consensus to be reached on the optimal method for diagnosing DU. Moreover, detrusor contraction strength is only one aspect of voiding efficiency, and future models will need to encompass several aspects of assessing detrusor contraction (e.g. strength, durability) as well as how the bladder empties.
5.7.3. Conservative management
In general, the treatment of UAB should focus on symptom relief, avoiding complications (such as infections) and improving or at least maintaining QoL. It involves a pragmatic approach ensuring timely bladder drainage by trying to improve bladder contraction and/or decrease urethral resistance [785].
5.7.3.a. Behavioural interventions
There are no randomised studies or large, high-quality studies available investigating the effect of behavioural interventions in male patients with UAB. The main goal is to reduce bothersome symptoms and prevent complications of incomplete bladder emptying (such as recurrent UTI) while avoiding invasive treatments. Asymptomatic patients at risk of UAB should be informed of this (like diabetes mellitus or after pelvic surgery/radiotherapy), because diagnosis is delayed due to loss in bladder sensation and enlarged bladder capacity. In patients with sensory impairment, timed or scheduled voiding schemes can be recommended. In patients with bothersome frequency, double or triple voiding, as well as Valsalva or the Credé manoeuver can reduce PVR and may improve their symptoms, however, no clinical trial has proven efficacy or harm of these measures in the non-neurogenic male population.
A descriptive cohort investigated male patients with DU started on conservative treatment (not further defined) and the need for intermittent catheterization (IC) after five years. They concluded that male patients with non-neurological DU can remain stable without the need to initiate IC. No urodynamic risk factors for IC were detected [789].
5.7.3.b. Pelvic floor muscle relaxation training with biofeedback
Successful voiding is initiated by relaxation of the pelvic floor and urinary sphincter. A failure of at least one of these mechanisms can result in voiding dysfunction, as well as inhibition of detrusor contraction, which is called the guarding reflex [790]. Therefore, physiotherapy with pelvic floor muscle relaxation is usually a first line therapy for voiding dysfunction, but no RCT in male adults investigates its effect on UAB. Evidence is derived mostly from paediatric studies. One RCT in children compared the effect of standard urotherapy (diet, adapted fluid intake and timed voiding + toilet training) with animated biofeedback therapy versus urotherapy alone in children with proven DU. Improvement was seen in PVR, recurrent UTI and urodynamic parameters, such as flow curve and maximum flow rate, in both treatment groups, but improvement was significantly better at all stages in children receiving biofeedback [791].
5.7.3.c. Intermittent catheterisation
In patients with persistently elevated PVR, IC is the preferred method for complete and timely bladder drainage. No data exist on the maximum accepted PVR, but after 300ml, the risk of UTIs increases [792]. Intermittent catheterisation is preferred over indwelling catheters, as it might reduce the risk of complications, such as UTI, bacterial colonisation, bladder stones and overflow incontinence. However, this technique demands a certain level of manual dexterity and cognition if performed by individuals themselves or it can be performed by a caregiver or second person, for example, a partner. More details are provided in Section 5.6.3.a.4.
5.7.3.d. Indwelling catheters
Indwelling catheters should be avoided in the long-term unless other treatment options are not indicated, and the patient is unwilling or unable to perform IC, and there is no one else that can perform it for them. In this case, suprapubic catheters are preferred over urethral catheters due to the risk of traumatic hypospadias [636]. For further details, see Section 5.6.3.a.4.
5.7.3.e. Intravesical electrical stimulation
Intravesical electrotherapy (IVES) is an electrical stimulation technique that stimulates the A-delta mechanoreceptor afferents thereby reinforcing bladder contractions. Therapy consists of daily sessions of stimulation with 10-15 sessions considered as a trial period. Afferent circuits should be intact together with a healthy detrusor muscle. An RCT in a mixed-gender population with neurogenic predominant UAB, reported improvement at four weeks after IVES compared to sham in PVR (-97ml vs. -10.5ml, p<0.01), in BVE ( +13.3 vs. 0.0, p<0.01) and in Qmax (+1.3 vs. +0.2, p<0.04) [793]. One small retrospective study with a mixed population (11 women and 5 men) showed some improvement in voiding after IVES in two-third of the study population [794]. Other evidence is mainly derived from small studies in children [777].
5.7.3.f. Extracorporeal shockwave therapy
Extracorporeal shockwave therapy (ESWT) improves neovascularisation and tissue regeneration and theoretically could improve detrusor contractility. A small placebo-controlled RCT reported significant improvements in PVR and UAB-Q scores at week 4 but not at week 12 of follow-up [795,796].
| Summary of evidence | LE |
| Behavioural interventions can be attempted in men with UAB and high PVR. Avoid increasing intrabdominal pressure with Valsalva or Credé in those with poorly compliant bladders. | 3 |
| Pelvic floor muscle relaxation techniques can help with voiding dysfunction. | 4 |
| Intermittent catheterisation is first-line therapy in men with UAB and high PVR over 300ml. | 3 |
| Indwelling catheters (urethral or suprapubic) are associated with a range of complications as well as an enhanced risk of UTI. | 3 |
| If indwelling catheters are the treatment choice, the suprapubic route is favoured over urethral due to risk of traumatic hypospadias. | 3 |
| Recommendations | Strength rating |
| Initiate intermittent catheterisation if there is a risk of upper tract damage or PVR is > 300mL. | Weak |
| Offer indwelling transurethral catheterisation or suprapubic cystostomy only when other modalities for urinary drainage have failed or are unsuitable. | Weak |
5.7.4. Pharmacological management
5.7.4.a. Parasympathomimetics
For the mechanism of action of parasympathomimetics, see Section 5.2.3.
An SR and meta-analysis were performed in both male and female patients with UAB, evaluating 12 RCTs on the use of both subgroups (muscarinic agonists and acetylcholinesterase inhibitors) of parasympathomimetics [797, 798]. The meta-analysis showed a small benefit in some patients with (post-procedure) urinary retention with no increase in adverse events, but without improvement of PVR. However, the results of this SR are confounded by the low number of small studies, weak data, high heterogeneity and a very short-term follow-up. Therefore, based upon the available literature, no strong evidence-based conclusions can be drawn on the role of parasympathomimetic as an effective pharmaceutical treatment for UAB.
5.7.4.b. Alpha-adrenergic blockers
One alternative to improve bladder emptying and micturition is by reducing outflow resistance in patients with UAB. Although there is a lack of high-quality RCTs, some evidence exists that lowering outflow resistance improves voiding functions and bothersome symptoms in men with UAB. A single-blind prospective RCT investigated 119 patients with UAB treating them with a cholinergic drug, an alpha-adrenergic blocker or both. They showed both a significant improvement in symptoms as well as PVR and flow rate in patients treated with combination therapy compared to monotherapy [799]. A study evaluated the effects of tadalafil (a PDE5 inhibitor) and silodosin on voiding function in male patients with non-neurogenic DU. After propensity score matching, they showed improvement of both QoL and UDS parameters and voiding parameters in both subgroups [779]. Overall, the clinical rigor required to provide evidence‐based support for the use of this class of medications in treating UAB is still lacking.
5.7.4.c. Prostaglandins
Prostaglandins are involved in the modulation of bladder function. There are five subtypes, of which prostaglandins E2 and F2a appear to be predominant in stimulating detrusor contractions. A Cochrane review analysing three RCTs using intravesical instillation of PGE2 and PGF2a suggests a reduction of postoperative retention after catheter removal [800]. However, due to methodological limitations of the included trials, the use in clinical practice remains uncertain. One placebo-controlled trial investigated the combination of intravesical PGE2 with bethanechol chloride in 19 patients with UAB [801]. Although they showed a reduction in PVR compared to placebo, clinical relevance is questioned. Overall, the efficacy of the prostaglandin agents in treating UAB is not established.
5.7.4.d. Other drugs
As previously stated, treatment with PDE5 inhibitors (such as tadalafil) showed improvement of both QoL and UDS parameters and voiding parameters in male patients with UAB [779]. For more information on PDE5s, please see Section 5.2.5.
| Summary of evidence | LE |
| Evidence on the effect of parasympathomimetics on clinical or urodynamic parameters of UAB is lacking. Possible serious adverse effects should be considered before administration. | 4 |
| There is limited evidence about effectiveness of alpha-adrenergic blockers in men with UAB. | 4 |
| The efficacy of the prostaglandin agents in treating UAB is not established. | 4 |
| Recommendations | Strength rating |
| Do not routinely recommend parasympathomimetics for treatment of men with underactive bladder. | Strong |
| Offer alpha-adrenergic blockers before more-invasive techniques. | Weak |
5.7.5. Surgical treatment for underactive bladder
Surgical options for male patients with non-neurogenic UAB/DU include benign prostatic surgery and SNM [170, 802-808].
5.7.5.a. Benign prostatic surgery in patients with UAB
An SR evaluated the outcomes of BPS for BPE in men with preoperative DU or acontractile detrusor [170]. Mean total IPSS variation following surgery was reported to range from -3 to -19.5 points. A > 3 points improvement in terms of total IPSS score was evident in 14 studies included into the SR [170]. Mean QoL variation ranged from -0.9 to -3 points [170]. Mean Qmax improvement ranged from +1.4 to +11.7mL/s. Mean PVR improvement ranged from -16.5 to -736mL [170].
Direct comparisons between patients with DU versus without DU provided conflicting results [170]. A study found that postoperative outcomes one month after photoselective laser vaporization prostatectomy (PVP) were worse in patients with DU compared to patients without DU [809]. In men with preoperative DU, mean total IPSS score improved from 12.0 to 15.2, mean Qmax improved from 4.8ml/sec. to 8.2ml/sec., and mean PVR improved from 918.3mL to 325.9mL. In patients without preoperative DU mean total IPSS improved from 17.5 to 9.7, mean Qmax improved from 6.9mL/sec. to 17.6mL/sec., and mean PVR improved from 225.0mL to 99.2mL [805]. A statistically significant difference (p ≤ 0.05) was observed in terms of mean IPSS, mean Qmax, and mean PVR when comparing patients with and without preoperative DU at one month follow-up [805].
In a study, the outcomes of BPO patients with and without DU after HoLEP were assessed at 6-month follow-up: there were no differences with regards to perioperative complications. However, Qmax (30.52 vs. 23.26mL/s) and total IPSS (6.66 vs. 5.42) were significantly better improved in patients without DU than in those with DU. Recatherisation rate was significantly higher in the DU group (10.7 vs. 5.8%, p = 0.022) [810].
Similarly, another study found that patients with DU had smaller decrease in terms of median total IPSS (-6.5 vs. -11) and a smaller increase in the Qmax (+3.5mL/s vs. +8.2mL/s) after PVP than did those without DU [804]. Statistical analysis found a significant differences between the two groups at follow-up evaluations only for Qmax (p = 0.023).
A study from a prospective cohort on 158 men with DU (Group 1: 41 patients BCI < 100 and BOOI < 40; Group 2: 77 patients: BCI < 100 and BOOI ≥ 40) versus non-DU patients (BCI ≥ 100 and BOOI ≥ 40) showed a similar decrease for IPSS in all groups, however, the postoperative Qmax at 12 month was lowest in DU group 1, when compared to group 2 and the non-DU patients (group 1: Qmax 12 months, mL/s 10.1 ± 4.32, group 2: 12.9 ± 7.51, group 3: 14.6 ± 6.95), as well as greater postoperative PVR > 80mL (5.1% in group one, 7.3% in group 2 and 33.9% in group 3) [112].
Other authors failed to find differences in terms of symptom improvement and success rates between patients with and without preoperative DU [170]. Mean BCI Index improvement was reported to range from 4 to 44.6 [170].
A retrospective study found that 81.7% of patients with DU or acontractile detrusor undergoing TURP or TUIP achieved a satisfactory treatment outcome defined as improved QoL and voiding efficiency of > 50% [806]. Among these patients, mean BCI significantly increased from 16.4 to 61.0 and 69.4% of them had recovery of detrusor function within three months [806].
A prospective case series found that 78% of patients with DU or acontractile detrusor and concurrent BPO undergoing HoLEP exhibited significant return of bladder contractility, determined by the presence of a sustained, volitional detrusor contraction at six-months follow-up [807]. Of note, 94.7% of patients were voiding spontaneously with acceptable PVR volume, whereas 5.3% required continued IC postoperatively [807].
A retrospective study investigated the effect of DU on the efficacy of TURP by comparing the subjective and objective parameters preoperatively and three months postoperatively between severe (BCI < 82), mild (82 ≤ BCI < 100), and absent DU (BCI ≥ 100) in two obstruction groups (20 ≤ BOOI < 40 and BOOI ≥ 40) [808]. Successful improvement after the operation was defined according to established criteria [32]. In patients with preoperative BOOI ≥ 40 the successful improvement rates for the IPSS, IPSS-Storage, IPSS-Voiding, QoL and free Qmax were 60.6%, 54.5%, 54.5%, 63.6% and 66.6% in patients with severe DU. The same figures in patients with BOOI ≥ 40 and mild DU were: 87.7%, 63.2%, 94.7%, 75.4% and 75.4%, respectively. Finally, in patients with BOOI ≥ 40 and absent DU the successful improvement rates were 94.1%, 82.8%, 95.6%, 90.6% and 71.4%, respectively.
In the 20 ≤ BOOI < 40 group, the successful improvement rates for the IPSS, IPSS-Storage, IPSS-Voiding, QoL and free Qmax in the severe DU patients were only 38.2%, 38.2%, 44.1%, 41.2% and 38.2%, respectively. The same figures in patients with mild DU were: 83.3%, 66.7%, 83.3%, 75.0% and 41.7%, respectively. Finally, in patients with absent DU the successful improvement rates were 90.9%, 90.9%, 90.9%, 90.9% and 81.8%, respectively.
Therefore, patients with varying degrees of BPO can benefit from TURP, but for patients with severe DU in the 20 ≤ BOOI < 40 group, TURP should be carefully considered only after adequate counselling [808].
The few long-term data available seem to reveal poor durability of benefits [170].
Factors influencing the surgical outcomes have been investigated: older age, lack of obstruction, concomitant DO, lower detrusor contractility and use of TURP or PVP instead of HoLEP were associated with worse outcomes [170].
5.7.5.b. Sacral neuromodulation
Sacral neuromodulation has been reported to improve idiopathic urinary retention in women in long-term studies [811]. However, only scarce evidence exists in men with DU or acontractile detrusor. A multicentric, retrospective case series reported the outcomes of SNM in adults with a urodynamic‐based diagnosis of DU and symptom duration of more than six months who failed to respond to first and second‐line treatment options [803]. Patients with cognitive disabilities, BOO and those who had any contraindication for SNM were excluded. Overall, 35 males were included. Of these, 51.4% responded to the first stage and were candidates for the full implant. Overall, 72% of male patients had favourable responses after the full implantation. Voided volume, PVR, and the median Qmax improved in both sexes [803]. In men who underwent full implantation voided volume improved from 75.0mL to 300.0mL, PVR improved from 350.0mL to 90.0mL, and mean Qmax improved from 7.0mL/sec. to 16.0mL/sec. Evidence from another retrospective study suggests that residual detrusor contractility is more likely to respond to a trial of SNM compared to detrusor acontractility [812].
| Summary of evidence | LE |
| In patients with DU or acontractile detrusor and concomitant BPO, de-obstruction procedures are associated with improvements in BCI mean total IPSS, mean maximum urinary flow and mean PVR. | 3 |
| Older age, lack of BOO, concomitant DO, lower bladder contractility and use of transurethral resection of the prostate or photovaporisation instead of laser enucleation of the prostate are associated with worse postoperative outcomes after de-obstruction procedures. | 3 |
| Sacral neuromodulation provides statistically significant improvement in terms of voided volume, PVR, and median maximum flow rate in men with refractory DU and no BPO. | 3 |
| Recommendations | Strength rating |
| Counsel patients with evidence of detrusor underactivity (DU) or acontractile detrusor and concomitant benign prostatic enlargement about the potential subjective and objective benefits of benign prostatic surgery. | Weak |
| Offer men with DU and no benign prostatic obstruction, test phase sacral neuromodulation. | Weak |
5.7.6. Follow-up
The natural history and clinical evolution at long-term follow-up of men with UAB is not well documented. A small retrospective cohort evaluated recovery of detrusor contraction one year after (medical or surgical) treatment through videourodynamic studies [813]. In this small cohort, bladder contractility recovery was seen in 43.9% of patients and an optimal bladder compliance cut-off value of < 80mL/cmH2O was predictive of better recovery. The interval between follow-up visits depends on patient characteristics, treatments given and the frequency of urinary complications. Two studies looked at the natural evolution of detrusor contractility with or without TURP for BOO [109, 772]. Although retrospective, they showed that detrusor contractility does not get worse with persisting BOO nor that it improves after performing a TURP. The underactive detrusor seems to remain underactive, but at least it does not deteriorate with time.
Figure 7: Management of detrusor underactivity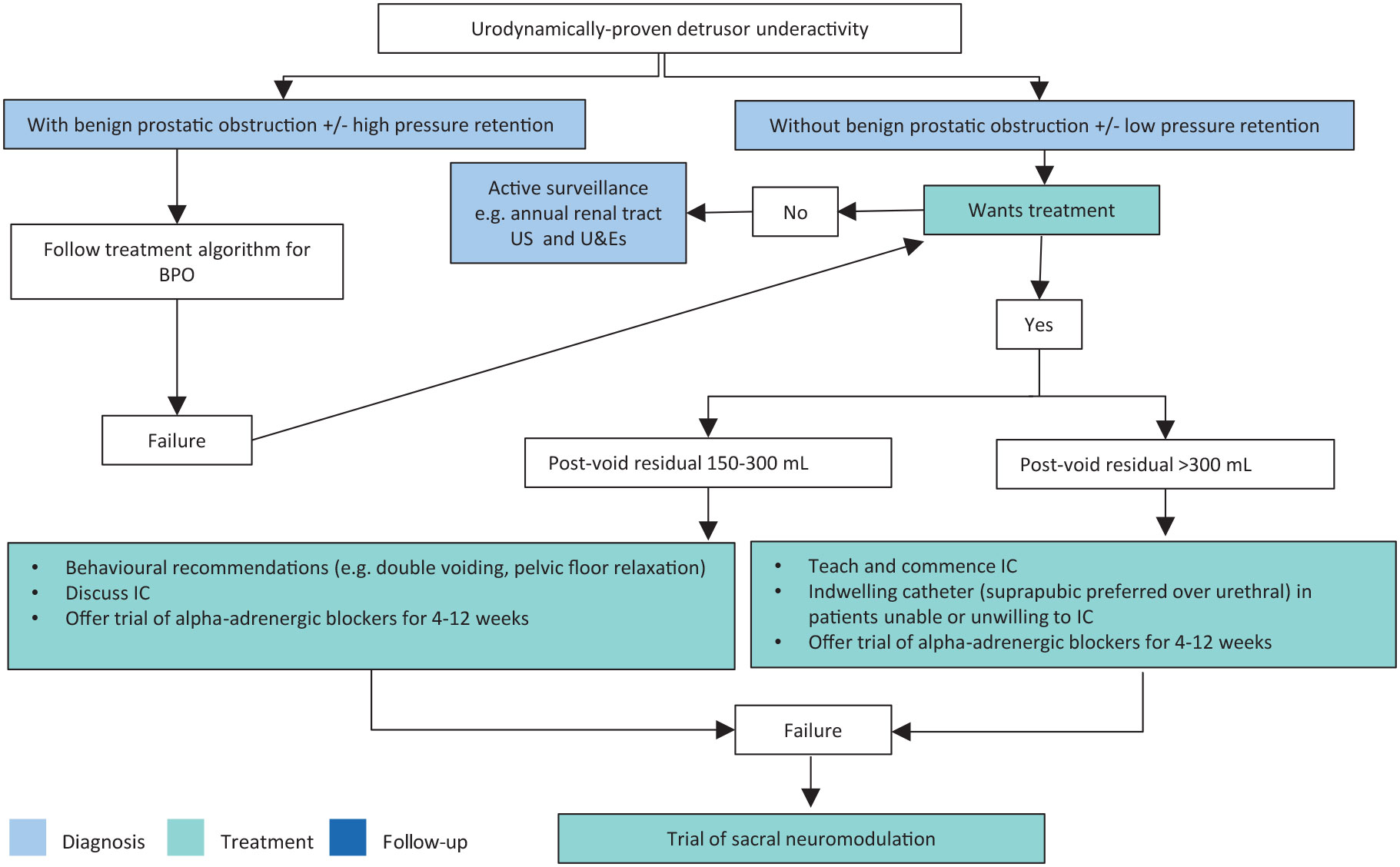 Inform patients of the benefits and harms of each treatment, and of the lack of solid evidence of their efficacy prior to surgery.
Inform patients of the benefits and harms of each treatment, and of the lack of solid evidence of their efficacy prior to surgery.
Initial assessment follows the EAU MLUTS Guidelines algorithm.
BPO = benign prostatic obstruction; IC = intermittent catheterisation; SNM = sacral neuromodulation; U&Es = urea and electrolytes; US = ultrasound.
5.8. Voiding dysfunction in young men
Up to 20% of men aged < 50 years report at least moderate LUTS [814, 815]. Primary bladder neck obstruction (PBNO) (defined as the inability of the bladder neck smooth muscle to open adequately during voiding, with a subsequent increase of detrusor pressure to try to overcome the resistance of the bladder neck and allow urine to flow) and DV (a condition characterised by an intermittent and/or fluctuating flow due to inadequate or variable relaxation generally of the sphincters during voiding in neurologically normal men) represent the two most common voiding disorders in men aged 18-50 years, leading to functional BOO and non-neurogenic LUTS [816]. Although the exact prevalence of PBNO and DV in the general population is largely unknown, epidemiological studies involving VUDS in selected populations of men aged 18-50 years with non-neurogenic LUTS showed a prevalence of PBNO and DV up to 54% and 43%, respectively [814-818].
5.8.1. Diagnostic evaluation
5.8.1.a. Medical history
Most patients with PBNO and DV complain of long-lasting LUTS, with a mean duration of symptoms before diagnosis reaching up to 53.8 months and nearly 88% of patients complaining of symptoms for > one year [814-818]. Although being disorders of the voiding phase, and voiding symptoms are more frequently reported in some series, there is no pivotal symptom, and the spectrum of LUTS may include storage, voiding and postmicturition symptoms [814-818]. Chronic pelvic pain is evident in up to 24.5% of PBNO patients, while recurrent UTI is observed in 28% of cases [819, 820].
5.8.1.b. Questionnaires
Currently, there is no specific validated questionnaire for the assessment of patients with PBNO and DV. Therefore, physicians can refer to a validated questionnaire for MLUTS, such as the IPSS, the ICIQ-MLUTS or the American Urological Association (AUA) questionnaire.
5.8.1.b.1. Physical examination
Pelvic examination, in addition to conventional physical examination, helps in evaluating anatomical and functional abnormalities of the pelvic-perineal area. This involves external visual inspection/palpation of the perineal/perianal regions and internal digital palpation of the perianal musculature to identify muscular tone abnormalities [821].
5.8.1.c. Uroflowmetry and PVR measurement
Although a Qmax of < 15mL/s has frequently been observed in patients diagnosed with PBNO and DV, this finding is not specific [770,814]. A clinical trial failed to find statistically significant differences in terms of Qmax between patients with PBNO, DV and BPO (12.0, 11.8 and 9.8mL/s, respectively) [770]. Similarly, although an elevated PVR is frequently observed, it lacks specificity [770].
5.8.1.d. Ultrasound scan
Although young men with PBNO and DV tend to have a prostate volume of < 30cm3, the value of this finding in predicting PBNO and DV from other causes of LUTS in men remains controversial [770, 814, 822].
5.8.1.e. VUDS and electromyography
The definitive diagnosis of PBNO and DV formally relies on the findings from in-depth functional assessments, with VUDS representing the gold standard approach [814]. The VUDS criteria for PBNO are the followings: high-pressure/low-flow voiding pattern, radiographic evidence of obstruction at the bladder neck, relaxation of the striated sphincter, and no evidence of distal obstruction. Currently, however, there are no commonly accepted urodynamic threshold parameters in this subset of patients [603,814]. Although electromyography (EMG) demonstrates relaxation of the striated sphincter concomitant with the detrusor contractions during micturition or an attempt at micturition in PBNO patients, the presence of increased EMG activity should not in and of itself exclude the diagnosis of PBNO due to false-positive results that can occur with surface EMG, which assesses pelvic floor activity rather than sphincter activity [819, 823].
The criteria adopted to diagnose DV include the following: electrical activity of the external sphincter during voiding detected on EMG in the absence of abdominal straining; brief and intermittent closing of the membranous urethra during voiding detected on fluoroscopy; and intermittent increases and decreases of flow in an undulating fashion detected on uroflowmetry performed in a private setting [814, 824].
A study showed a significant association between the extent of bladder neck opening impairment observed on VCUG and obstruction and contraction urodynamic parameters, but no association with the severity of urinary symptoms [820]. In details, the detrusor PdetQmax, BOOI and BCI were significantly higher in patients with absence of bladder neck opening compared to those with incomplete bladder neck opening [820].
However, VUDS can be expensive, time consuming, unpleasant for the patient and often unavailable in everyday clinical practice [820, 825]. Additionally, some patients fail to void during the study due to psychological and physical discomfort [814, 820]. Evaluation of data separately collected by standard UDS and VCUG has been considered a reliable alternative approach by some authors [820], although it is difficult to correlate anatomy with physiology at various points in time.
5.8.1.f. Magnetic resonance imaging
Preliminary data exist regarding the value of MRI in the diagnosis of PBNO. Reported accuracies of MRI and MR voiding cystourethrography in identifying PBNO of 87% and 100%, respectively [825].
5.8.1.g. Urethrocystoscopy
Some authors consider urethrocystoscopy a useful test to rule out urethral strictures or enlarged prostate in selected cases. However, the role of urethrocystoscopy in the diagnosis of PBNO and DV remains controversial [814].
| Summary of evidence | LE |
| Most patients with PBNO and DV complain of long-lasting LUTS, with a mean duration of symptoms before diagnosis reaching up to several years. | 4 |
| Videourodynamics represents the gold standard approach for the diagnosis of PBNO and DV. | 4 |
| Evaluation of data separately collected by standard UDS and VCUG has been considered a reliable alternative approach by some authors. | 4 |
| Electromyography demonstrates relaxation of the striated sphincter concomitant with the detrusor contractions during micturition or an attempt at micturition in PBNO patients and electrical activity of the external sphincter during voiding in DV patients. | 4 |
| Recommendations | Strength rating |
| Suspect primary bladder neck obstruction (PBNO)/dysfunctional voiding (DV) in young men complaining of long-lasting LUTS. | Weak |
| Perform videourodynamics (VUDS) studies (or standard urodynamic studies with voiding cysto-urethrography if VUDS studies are unavailable) ± electromyography in young men with suspected PBNO or DV as initial diagnostic assessment or after failed first-line therapy. | Weak |
5.8.2. Treatment
The goals of therapies in young men with LUTS secondary to PBNO and DV include improvement of symptoms and QoL, relief of obstruction, prevention of disease-related complications, and preservation of sexual/reproductive health [814].
5.8.2.a. Conservative treatment
5.8.2.a.1. Observation
Observation has been proposed as an option for PBNO patients with minimal symptom bother and without clinical or urodynamic evidence of upper or LUT decompensation [814]. Available evidence in men aged 18-50 years only derives from one study describing the outcomes of no treatment in nine PBNO patients unresponsive to α1-blockers refusing surgery demonstrating not statistically significant variations in terms of the mean AUA-6 score, mean Qmax and mean PVR at a mean follow-up of 36 months [826].
5.8.2.a.2. Behavioural modifications plus biofeedback
Behavioural modifications (instructions to relax the pelvic floor muscles during voiding) plus biofeedback were assessed in one study involving 43 patients with DV unresponsive to alpha blockers [824]. Treatment success, defined as a decrease of symptoms of at least 50% in two consecutive visits, was observed in 35 patients (83%) at three months [824].
Individual treatment programs administered by pelvic floor physical therapists typically include an initial phase when patients are familiarised with the anatomy and physiology of the pelvic organs and muscles, and subsequent phases when verbal guidance and feedback through palpation of the pelvic floor muscles and biofeedback measurements were used to teach how to correctly contract and relax the pelvic floor muscles [827].
5.8.2.b. Pharmacological treatment
5.8.2.b.1. Alpha-blockers
Alpha-blockers are commonly prescribed as first-line treatment in patients with PBNO. A meta-analysis found a mean total IPSS improvement ranging from -1 to -12 points (pooled estimate at three months: -7.0 [95% CI: -8.3 to -5.8; p < 0.000]), a mean IPSS QoL sub-score improvement ranging from -1.5 to -1.9 points (pooled estimate at three months: -1.7 points [95% CI: -2.0 to -1.3; p < 0.000]), a mean Qmax variation ranging from 3.7 to 11.1mL/s (pooled estimate at three months: 4.0mL/s [95% CI: 3.1-4.9; p < 0.001]), a mean PVR variation ranging from to 26.9 to -70mL (pooled estimate at 3 months: -31.1mL [95% CI: -36.3 to -25.9; p < 0.001) [814]. Two studies evaluated BOOI variation following treatment with α1-blockers. One reported a median BOOI improvement of 21 points, while another reported a mean BOOI improvement of 34.5 points [828, 829]. A major drawback of α1-blockers is the considerable discontinuation rate, ranging from 4.7% to 55%, mainly due to unsuccessful outcomes or adverse events [814]. A higher baseline urodynamic obstruction grade and younger age have been identified as predictors of α1-blockers failure [814]. Unsuccessful outcomes were reported in a percentage of patients ranging from 23% to 52% [814]. Systemic adverse events were reported by 0-16.1% of patients, with giddiness being the most frequent adverse event. The incidence of retrograde ejaculation ranged from 47% to 50% [814].
5.8.2.c. Surgical treatment
Surgery typically involves BNI and is mostly performed in patients failing therapy with α1-blockers [814]. It aims to relieve the obstruction by interrupting muscular fibres of the internal sphincter located at the bladder neck [814]. Outcomes of surgery have been assessed in PBNO patients. Surgical procedures include bladder neck incision (single or bilateral), TURP and/or transurethral resection of the bladder neck. Globally, a meta-analysis found a mean total IPSS improvement ranging from -8.8 to -23.3 points (pooled estimate at three months: -11.2 points [95% CI: -16.12 to -6.33; p < 0.0001]); a mean IPSS QoL subscore improvement ranging from -1.9 to -2.2 points; a mean Qmax improvement ranging from 4.2 to 18.5mL/s (pooled estimate at three months: 6.9mL/s [95% CI: 1.4-12.5; p = 0.014]); and a mean PVR variation ranging from to 7.6 to -156 mL [1]. One study evaluated PdetQmax variation after treatment by reporting an improvement in terms of the mean PdetQmax by 68cmH2O [16]. An unsuccessful outcome was reported in 0-15.3% of patients [814]. Overall, EjD rates ranged from 0 to 88.8% (pooled estimate following BNI: 3.0% [95% CI: 1-6%]) [814]. Results stratified according to the type of BNI revealed pooled EjD rates of 0% (95% CI: 0-1%) and 9% (95% CI: 2-21%) following single and bilateral BNI, respectively (p = 0.128) [814]. A retrospective analysis was recently conducted on 67 males < 40 years with PBNO therapies with ABs or surgery (BNI or TUIP). Both groups showed significant improvements, but the surgical group demonstrated superior outcomes. At six months, the surgical group had a greater reduction in IPSS (11.9 ± 4.40 vs. 4.38 ± 3.73; p < 0.001) and greater improvements in Qmax (+5.59 ± 4.08mL/s vs. +2.54 ± 3.85mL/s; p < 0.001) [830].
5.8.2.d. OnabotulinumtoxinA (BoNTA) injection therapy
A single study assessed the outcomes of a transurethral bladder neck injection of BoNTA in 30 patients with PBNO after unsuccessful α1-blockers therapy [831]. The mean total IPSS varied from 21.9 at baseline to 7.8, 10.3, 16.6 and 19.9 at two-, six-, nine- and 12-months follow-up, respectively. The mean Qmax varied from 7.8mL/s at baseline to 16.9, 15.5, 12.7 and 8.6mL/s at two-, six-, nine- and 12-month follow-up, respectively. None of the patients reported EjD. Erectile dysfunction was not assessed [831]. Currently, this procedure should be considered as experimental.
5.8.2.e. Intermittent catheterisation
Intermittent catheterisation remains a further option in patients with both PBNO and DV refractory to first-line therapy [814].
5.8.2.f. Sacral neurostimulation
Some authors consider SNM an exciting new treatment for refractory voiding disorders, including DV [814]. Currently, this procedure should be considered as experimental, but having a trial phase using percutaneous nerve evaluation is likely the only method that could confirm whether a permanent implant would be suitable.
| Summary of evidence | LE |
| Alpha-blockers provide a mean total IPSS improvement ranging from -1 to -12 points (pooled estimate at three months: -7.0); a mean IPSS QoL subscore improvement ranging from -1.5 to -1.9 points (pooled estimate at three months: -1.7 points); a mean Qmax variation ranging from 3.7 to 11.1mL/ s (pooled estimate at three months: 4.0mL/s); and a mean PVR variation ranging from to 26.9 to -70mL (pooled estimate at three months: -31.1mL) in young men with PBNO. | 3 |
| Behavioural modifications (instructions to relax the pelvic floor muscles during voiding) plus biofeedback provide a decrease of symptoms of at least 50% in two consecutive visits in 83% of patients with DV at three months. | 4 |
| Bladder neck incision provide a mean total IPSS improvement ranging from -8.8 to -23.3 points (pooled estimate at three months: -11.2 points); a mean IPSS QoL sub-score improvement ranging from -1.9 to 2.2 points; a mean Qmax improvement ranging from 4.2 to 18.5mL/s (pooled estimate at three months: 6.9mL/s); and a mean PVR variation ranging from to 7.6 to -156mL in young men with PBNO. | 3 |
| Ejaculation dysfunction rates following BNI range from 0% to 88.8% (pooled estimate: 3.0%). | 3 |
| Recommendations | Strength rating |
| Consider alpha blockers as first-line treatment in young men with primary bladder neck obstruction (PBNO). | Weak |
| Consider behavioural modifications plus biofeedback as first-line therapy in young men with dysfunctional voiding (DV). | Weak |
| Consider bladder neck incision or intermittent catheterisation (IC) in well-informed young men with PBNO unresponsive to alpha blockers. | Weak |
| Consider IC in young men with DV unresponsive to behavioural modifications plus biofeedback. | Weak |