7. DISEASE MANAGEMENT
All patients with suspicion of UTUC based on radiology, cystoscopy and urine cytology should be discussed in a multidisciplinary team prior to diagnostic ureteroscopy and the initiation of treatment [162]. This is supported by population-based data reporting increased use of invasive diagnostic modalities in hospitals with a lower caseload [161].
7.1. Low-risk disease
7.1.1. General considerations on kidney-sparing surgery
Kidney-sparing surgery for low-risk UTUC reduces the morbidity associated with RNU (e.g. loss of kidney function) without compromising oncological outcomes [163]. In low-risk cancers, kidney-sparing surgery is the preferred approach, because survival is similar to that after RNU [163,164]. This option should therefore be discussed with all patients with low-risk UTUC, irrespective of the status of the contralateral kidney, as part of a shared decision-making process. Recommendations for kidney-sparing management of UTUC are listed in section 7.1.7.
7.1.2. Endoscopic ablation with ureteroscopy
Endoscopic tumour ablation should be considered in patients with low-risk UTUC [165,166]. Patients should be informed of the need and be willing and able to comply with an early second-look URS [167] and stringent surveillance, because one in two patients develop ipsilateral upper tract recurrence within two years [168]. Additionally, complete tumour resection or destruction is necessary [167]. Nevertheless, a risk of disease downstaging or progression remains with endoscopic management due to the suboptimal performance of imaging and biopsy for risk stratification and tumour biology [169]. A systematic review confirmed the comparable survival outcomes of endoscopic treatment to those of RNU, but at the cost of higher local recurrence rates and need for repeated interventions. In addition, the evidence on renal preservation after endoscopic treatment is inconsistent and the long-term benefits are not assured, possibly due to the cumulative effects of repeated procedures and imaging during follow-up [170].
The use of flexible ureteroscopes with an external diameter < 9F is useful in the endoscopic management of UTUC [171].
Ablation is preferably performed with lasers; however, data regarding optimal laser type or settings are lacking. Although there is no evidence on optimal laser settings, excessive power should be avoided, as high power settings will generate heat and cause thermal injury of normal tissue [172].
Second-look URS after initial endoscopic treatment is recommended to ensure complete tumour resection and evaluate residual disease. Second-look URS should be performed within eight weeks following initial endoscopic treatment to assess for residual tumours or recurrence [167]. Other studies reported that up to nearly 50% of patients showed residual or recurrent disease during the second-look procedure, emphasising the value of early follow-up [173]. Therefore, early second-look URS plays a crucial role in optimising the outcomes of conservative treatment in UTUC by ensuring thorough tumour control.
7.1.3. Percutaneous antegrade access
Percutaneous antegrade management can be considered for low-risk UTUC in the renal pelvis [165,174]. This can also be offered for low-risk tumours that are inaccessible or difficult to manage by means of flexible retrograde URS, for example, in patients with urinary diversions. This approach is being used less in the wake of improved endoscopic possibilities with smaller sized ureteroscopes [166,174]. Although rare, the risk of tumour seeding remains with percutaneous antegrade access [174].
7.1.4. Ureteral resection
Segmental or distal ureterectomy and ureteral resection with adequate margins - ideally based on frozen section analysis - provides sufficient pathological specimens for staging and grading while preserving the ipsilateral kidney. Further direct anastomoses using either an end-to-end technique or ureteroneocystostomy are usually performed, but ileal-ureteral substitution or renal autotransplantation are also technically feasible, depending on the length of ureter removed [175,176]. Segmental resection of the proximal two-thirds of ureter is associated with higher failure rates than for the distal ureter [177,178]. Distal ureterectomy with ureteroneocystostomy for tumours in the distal ureter is reported with a low cumulative incidence of ipsilateral upper tract recurrence (0-18%) [179-181] compared to 25-85% after endourologic kidney sparing [170].
7.1.5. Chemoablation
A single-arm phase III trial including 71 patients with biopsy-proven low-grade UTUC measuring less than 15mm showed that the use of mitomycin-containing reverse thermal gel (UGN-101) instillations (six-weekly induction) in a chemoablation setting by means of a retrograde catheter to the renal pelvis and calyces was associated with a complete response (CR) rate in a total of 41 patients (58%) [182]. The most frequently reported all-cause adverse events (AEs) were ureteric stenosis in 31 (44%), urinary tract infection in 23 (32%), haematuria in 22 (31%), flank pain in 21 (30%), nausea in 17 (24%) and 19/31 (61%) reported ureteric stenosis requiring treatment. Among patients with CR, 29/41 (71%) received at least one maintenance instillation (median of six), and 23/41 (56%) remained disease free at one year [182]. Long-term outcomes remain uncertain [183].
7.1.6. Adjuvant instillations
7.1.6.a. Upper urinary tract
The antegrade instillation of BCG or mitomycin C in the upper urinary tract by means of percutaneous nephrostomy after complete tumour eradication has been studied for CIS after kidney-sparing management [184,185]. Retrograde instillation through a single-J open-ended ureteric stent is also used [186]. Before both the antegrade and retrograde approach, a nephroureterogram must rule out ureteric obstruction or leakage, asses that there is no infection and ensure a low-pressure system to avoid pyelovenous backflow during instillation/perfusion. The reflux obtained from a double-J stent has been used, but this approach is suboptimal because the drug often does not reach the renal pelvis [187,188].
A systematic review and meta-analysis assessing the oncologic outcomes of patients with papillary (Ta-T1) UTUC or CIS of the upper urinary tract treated with kidney-sparing surgery and adjuvant endocavitary therapies (i.e. chemotherapeutic agents and/or BCG) did not find any difference between the method of drug administration (antegrade vs. retrograde vs. combined approach) in terms of recurrence, progression, CSS and OS. However, all included studies were underpowered and highly heterogeneous. Furthermore, the recurrence rates following adjuvant instillations are comparable to those reported in the literature in untreated patients, questioning their efficacy [189]. The analyses were based on retrospective small studies suffering from publication and reporting bias.
Further evidence suggests that early single-adjuvant intracavitary upper-tract instillation of mitomycin C in patients with low-grade UTUC might reduce the risk of local recurrence [190]. Limited complications related to the instillations were reported, and the need for a retrograde pyelography before instillations are commenced to exclude contrast extravasation was confirmed.
7.1.6.b. Bladder
No data is currently available to support the use of bladder instillation of chemotherapy after kidney-sparing surgery, as available randomised controlled trials (RCTs) included only patients who received RNU.
7.1.7. Recommendations for kidney-sparing management of localised low-risk upper urinary tract urothelial carcinoma
| Recommendations | Strength rating |
| Offer kidney-sparing management as primary treatment option to patients with low-risk tumours. | Strong |
| Discuss both endoscopic management and distal ureterectomy in low-risk tumours of the distal ureter based on tumour characteristics and shared decision-making with the patient. | Strong |
| Perform second-look ureteroscopy within eight weeks following initial endoscopic management. | Weak |
7.2. Localised high-risk disease
7.2.1. Local treatments
7.2.1.a. Radical nephroureterectomy
7.2.1.a.1. Surgical approach
Although the open approach has long been the standard [13], both laparoscopic and robot-assisted RNU can be used to treat high-risk UTUC, providing perioperative benefits such as decreased risk of complication and shorter hospital stay [191,192]. In addition, equivalent oncological outcomes have generally been reported between the three procedures [191-193], except for a higher risk of intravesical recurrence after minimally invasive RNU [194]. It is worth noting that, although laparoscopic RNU was historically purported to provide inferior oncological outcomes [195] in locally advanced UTUC, this was not confirmed with the use of robotic RNU [194].
A meta-analysis of six retrospective comparative studies showed that the use of a retroperitoneal versus transperitoneal route at the time of laparoscopic RNU provides similar perioperative and oncological outcomes, except for a longer operative time and shorter recovery time to bowel function in the retroperitoneal group [196]. Similarly, retroperitoneal robotic RNU is safe and feasible to perform [197].
Regardless of the approach, RNU must be performed according to oncological principles to prevent tumour seeding:
- Perform en bloc removal of the kidney, ureter and bladder cuff.
- Avoid entering the urinary tract, except when performing a bladder cuff excision and only after prior clipping of the ureter and complete drainage of the bladder [198].
7.2.1.a.2. Bladder cuff management
Resection of the distal ureter and its orifice is performed because there is a considerable risk of tumour recurrence in this area and in the bladder [32,177,199]. Several techniques have been described, including the transvesical, extravesical and endoscopic approaches with similar oncological outcomes regarding CSS. Uncertainty remains regarding their impact on intravesical recurrence [200,201], with the endoscopic approach potentially affecting a higher risk of recurrence [201]. Several other techniques have been proposed to simplify distal ureter resection, including the pluck technique, stripping, transurethral resection of the intramural ureter, and intussusception, but none of these techniques have convincingly been shown to be equal to complete bladder cuff excision [33,201].
7.2.1.a.3. Lymph node dissection
No high-level evidence is available to support the routine use of LND for upper tract tumours. However, template-based LND and its completeness may reduce the risk of local recurrence and improve CSS [202]. This could have a greater impact than the number of removed LNs [203]. Even in clinically [204] and pathologically [205] node-negative patients, LND may improve survival. Moreover, clinical tumour staging is inaccurate preoperatively. Provided the patient meets the strong criteria for high-risk disease (Figure 6.1), template-based LND could be offered to those who are scheduled for RNU, particularly given the low risk of major postoperative complications [206]. The templates for LND vary according to primary tumour location [202,207,208].
7.2.1.b. Kidney-sparing surgery
7.2.1.b.1. Elective indications
Distal ureterectomy
Distal ureterectomy - particularly with adequate surgical margins based on frozen section analysis - followed by ureteroneocystostomy may achieve equivalent long-term oncological outcomes and improved renal preservation as compared to RNU for high-risk UTUC located in the distal ureter [163,164,209,210]. However, the risk of local- and intravesical recurrence is significantly higher following distal ureterectomy, emphasising the necessity of patient selection and close postoperative surveillance [211].
Two retrospective studies [212-215] including mainly high-risk patients, and one prospective study [216] including only high-risk patients, showed ipsilateral upper tract recurrence in 16-28% of cases after segmental ureterectomy, highlighting the need to weigh this risk against performing RNU on an individual patient basis. This procedure can be performed with concomitant LND.
Ureterorenoscopy with laser ablation or segmental ureterectomy
Patients with high-risk UTUC that harbour more favourable features (predominantly “weak” high-risk criteria, low-grade disease and no infiltrative features at imaging), cannot be systematically considered as an indication for RNU [217,218]. Alternatively, the use of ureterorenoscopy with laser ablation or segmental ureterectomy may be proposed on a case-by-case basis if feasible.
7.2.1.b.2. Imperative indications
Ureterorenoscopy with laser ablation or segmental ureterectomy can be considered on a case-by-case basis for patients with high-risk UTUC and imperative kidney-sparing indications. This includes situations such as solitary kidney, bilateral UTUC, and even those harbouring high-grade disease and/or infiltrative features, but only in the presence of severe chronic kidney disease or any other comorbidity compromising the use of RNU. However, there is a greater risk of progression after kidney-sparing surgery for high- versus low-risk UTUC, with a direct impact on survival [163].
7.2.2. Perioperative treatments
7.2.2.a. Neoadjuvant systemic treatments
7.2.2.a.1. Chemotherapy
The primary advantage of neoadjuvant chemotherapy (NAC) is the ability to give cisplatin-based regimens when patients still have maximal renal function. Several retrospective studies evaluating the role of NAC have shown evidence of pathological downstaging and CR rates at RNU [219-223] with a direct impact on OS [224]. Moreover, NAC has been shown to result in lower disease-recurrence and mortality rates compared to RNU alone, without compromising the use of definitive surgical treatment with a potential OS benefit [222,225-227].
No RCTs have been published yet, but prospective data from phase II trials showed that NAC based on cisplatin combination therapy was associated with a 14-19% pathological CR rate in high-grade and/or cT2-T4N0M0 UTUC [228,229]. In addition, final pathological stage was < ypT1 in more than 60% of included patients with acceptable toxicity profile. In a systematic review and meta-analysis comprising more than 800 patients, NAC has shown a pathologic partial response of 43% and a downstaging in 33% of patients, resulting in an OS and CSS benefit compared with RNU alone [230]. A further systematic review and meta-analysis included 21 trials involving 14,117 patients with UTUC, comprising 1,983 patients who received NAC. Among those treated with NAC, 10% achieved a pathological CR and 42% experienced pathological downstaging; however, no survival benefit was demonstrated [231]. It is important to note that these findings are not conclusive, as the evidence is limited by significant bias and heterogeneity of the available data. If there is residual ≥ ypT2 and/or ypN+ disease after NAC and subsequent RNU, patients are likely to have a poorer prognosis [232]. Adjuvant immunotherapy should be discussed.
7.2.2.a.2. Immunotherapy
Only a small phase II study including ten patients with high-risk UTUC evaluated the efficacy of pembrolizumab in the neoadjuvant setting [233]. However, no pathological response was observed, and one treatment-related death was reported. Therefore, no evidence is currently available to support the use of neoadjuvant immunotherapy for high-risk UTUC.
7.2.2.a.3. Chemoimmunotherapy
The efficacy of the neoadjuvant combination of durvalumab with gemcitabine plus cisplatin (cohort 1) or carboplatin (cohort 2) before RNU has been evaluated in a negative phase II study showing pathological CR rates of 13% in those who received cisplatin and 5% in those who received carboplatin. These were both below the prespecified statistical hypotheses, although a final pathological stage < pT1 was found in 63% and 47% of patients included in cohort 1 and 2, respectively [234].
7.2.2.b. Perioperative intravesical instillations
7.2.2.b.1. Adjuvant intravesical instillations
The rate of bladder recurrence after RNU for UTUC is approximately 30% [32,201]. Two prospective randomised trials [235,236] and two meta-analyses [237,238] have demonstrated that a single postoperative dose of intravesical chemotherapy (mitomycin C, pirarubicin) two to ten days after surgery reduces the risk of bladder tumour recurrence within the first years post-RNU in patients without a history of BC. Prior to instillation, a cystogram can be considered in case of concerns about drug extravasation. All studies showed a very low risk of AEs.
Based on current evidence, it is unlikely that additional instillations beyond one perioperative instillation of chemotherapy further substantially reduce the risk of intravesical recurrence [239]. Management is outlined in Figures 7.1 and 7.2. One low-level evidence study suggested that bladder irrigation might reduce the risk of bladder recurrence after RNU [240].
No data are currently available to support the use of bladder instillation of chemotherapy after kidney-sparing surgery, because available RCTs included only patients who received RNU.
7.2.2.b.2. Other perioperative intravesical instillations
Intravesical chemotherapy has been administered safely at the time of RNU prior to bladder cuff opening, removing the need for a postoperative cystogram but with low-level data for efficacy [241].
Given worldwide low compliance with the use of adjuvant bladder instillations after RNU, its efficacy has more recently been tested in the preoperative setting. A single bladder instillation of mitomycin C within three hours before RNU or segmental ureterectomy, combined with saline irrigation, provided a two-year intravesical recurrence rate of 24%, which was above the pre-specified threshold of 20% in a prospective study [242]. Nonetheless, a significant reduction in the risk of intravesical recurrence was observed in the subgroup of patients who did not receive diagnostic ureterorenoscopy before radical surgery using an historical cohort as a comparative group.
7.2.2.c. Adjuvant treatments
7.2.2.c.1. Systemic chemotherapy
The POUT phase III multicentre prospective RCT (n = 261) evaluating the benefit of four cycles of adjuvant (AC) gemcitabine-platinum combination chemotherapy initiated within 90 days after RNU versus surveillance reported a significant improvement in DFS in patients with pT2-pT4, N (any) or positive (pT any, N1-3) M0 UTUC (three year DFS 71% vs. 50%; five year DFS 63% vs. 46%; HR: 0.54; 95% CI: 0.36-0.79; three- and five-year metastasis-free survival 19% improvement HR: 0.55; 95% CI: 0.36-0.77) [243]. Patients were stratified to gemcitabine/cisplatin or gemcitabine/carboplatin chemotherapy based on glomerular filtration rate (GFR) alone, with benefit seen irrespective of chemotherapy type. There was a non-significant trend towards improved OS (12% at three years), but because the study had met its primary endpoint of three-year DFS, the study closed early, leaving it underpowered for the secondary endpoint of OS. An updated analysis showed five-year DFS of 62% versus 45% (HR: 0.55; 95% CI: 0.38-0.80; p = 0.001) and mean restricted survival time was 18 months longer in the chemotherapy arm. Five-year OS was 66% versus 57% with univariate HR 0.68 (95% CI: 0.46; p = 0.49). Treatment effect was consistent across chemotherapy regimens (carboplatin or cisplatin) and disease stage [244]. The main potential limitation of using adjuvant chemotherapy is the concern that renal function may deteriorate after RNU, precluding cisplatin use in patients who could benefit from this [245,246]. A review of perioperative predictors of decline in renal function after RNU showed three-month GFR levels of approximately 50mL/min [247]. With split dose and hydration, cisplatin may be considered in patients with a GFR down to 45mL/min. In addition, carboplatin can be used to GFR of 30mL/min. or above. Table 2 outlines the eligibility criteria for platinum chemotherapy.
In a retrospective study, histological subtypes of UTUC exhibited different survival rates and adjuvant chemotherapy was only associated with an OS benefit in patients with pure UC [248]. However, whilst histological subtypes of UTUC exhibit different survival rates in retrospective studies, adjuvant chemotherapy should be considered whenever UC is the dominant pathology.
Table 2: Definitions of platinum-eligibility for systemic treatment of urothelial carcinoma [2]
| Platinum eligible | Platinum ineligible | |
| Cisplatin eligible | Carboplatin* eligible | |
| ECOG PS 0-1 and GFR > 50-60mL/min. and audiometric hearing loss Grade < 2 and peripheral neuropathy Grade < 2 and cardiac insufficiency NYHA class < III | ECOG PS 2 or GFR 30-60mL/min. or not fulfilling other cisplatin-eligibility criteria | Any of the following:
|
* Carboplatin is not indicated for neoadjuvant treatment.
ECOG PS = Eastern Cooperative Oncology Group performance status; GFR = glomerular filtration rate; NYHA = New York Heart Association.
7.2.2.c.2. Systemic immunotherapy
In a phase III, multicentre, double-blind RCT involving patients with high-risk muscle-invasive UC who had undergone radical surgery (pT3, pT4a or pN+), adjuvant nivolumab improved DFS compared to placebo in the intention-to-treat population (20.8 vs. 10.8 months) and among patients with a programmed death-ligand 1 (PD-L1) expression level of 1% or more [249]. The patient population predominantly consisted of BC patients post RC, with an additional smaller cohort of patients with UTUC post RNU (approx. 25%). The median RFS outside the urothelial tract in the entire intention-to-treat population was 22.9 months for nivolumab and 13.7 months for placebo. Treatment-related AEs > grade 3 occurred in 17.9% of the nivolumab group and 7.2% of the placebo group. On subgroup analysis, patients with UTUC included in this study did not seem to benefit from adjuvant nivolumab, which requires further follow-up and analysis. Nevertheless, the European Medicines Agency (EMA) approved nivolumab as monotherapy for the adjuvant treatment of patients with muscle-invasive UC and tumour cell PD-L1 expression > 1% who are at high risk of recurrence after radical surgery and who decline or are unfit for adjuvant chemotherapy [250]. A further study of 702 patients with UC treated with either RC or RNU, and with persistent high-risk features, were randomised to receive either adjuvant pembrolizumab or observation [251]. The DFS was significantly longer with pembrolizumab (29.6 months vs. 14.2 months); however, the number of patients with UTUC (25% of overall population) in the study was small and on subgroup analyses did not seem to benefit from adjuvant pembrolizumab [251].
A network meta-analysis suggests superior oncological benefit for adjuvant platinum-based chemotherapy over immune checkpoint inhibitors (CPIs) in patients treated with radical surgery for UTUC [252].
7.2.2.c.3. Radiotherapy
Adjuvant radiation therapy has been suggested to control locoregional disease after surgical removal. The data remains controversial and insufficient for conclusions [253-256]. Moreover, its added value to chemotherapy remains questionable [255].
7.2.3. Summary of evidence and recommendations for the management of high-risk non-metastatic upper urinary tract urothelial carcinoma
| Summary of evidence | LE |
| Radical nephroureterectomy is the standard treatment for high-risk UTUC, regardless of tumour location. | 2a |
| Open, laparoscopic and robotic approaches have similar oncological outcomes. | 2a |
| Failure to completely remove the bladder cuff increases the risk of BC recurrence. | 3 |
| Template-based LND may improve survival in muscle-invasive UTUC. | 3 |
| Postoperative platinum-based adjuvant chemotherapy improves DFS. | 1b |
| Single postoperative intravesical instillation of chemotherapy lowers the BC recurrence rate. | 1b |
| Recommendations | Strength rating |
| Discuss all patients with suspicion of upper urinary tract urothelial carcinoma (UTUC) on imaging in a multidisciplinary team meeting. | Strong |
| Perform radical nephroureterectomy (RNU) in patients with high-risk non-metastatic UTUC. | Strong |
| Use an open, laparoscopic or robotic approach to perform RNU in patients with high-risk non-metastatic UTUC. | Weak |
| Perform a template-based lymphadenectomy in patients with high-risk non-metastatic UTUC. | Weak |
| Offer adjuvant platinum-based chemotherapy after RNU to eligible patients with pT2-T4 and/or pN+ disease. | Strong |
| Deliver a postoperative bladder instillation of chemotherapy to lower the intravesical recurrence rate in patients without a history of bladder cancer. | Strong |
| Discuss adjuvant nivolumab with programmed death-ligand 1 (PD-L1) positive patients unfit for, or who declined, platinum-based adjuvant chemotherapy for ≥ pT3 and/or pN+ disease after previous RNU alone or ≥ ypT2 and/or ypN+ disease after previous neoadjuvant chemotherapy followed by RNU. | Weak |
| Discuss adjuvant pembrolizumab with patients unfit for, or who declined, platinum-based adjuvant chemotherapy for ≥ pT3 and/or pN+ and/or positive margin disease after previous RNU alone or ≥ ypT2 and/or ypN+ and/or positive margin disease after previous neoadjuvant chemotherapy followed by RNU. | Weak |
| Offer distal ureterectomy to selected patients with high-risk tumours limited to the distal ureter. | Weak |
| Discuss kidney-sparing management of high-risk patients with imperative indication, on a case-by-case basis, in a shared decision-making process with the patient, despite the higher risk of disease progression. | Strong |
Figure 7.1: Proposed flowchart for the management of upper urinary tract urothelial carcinoma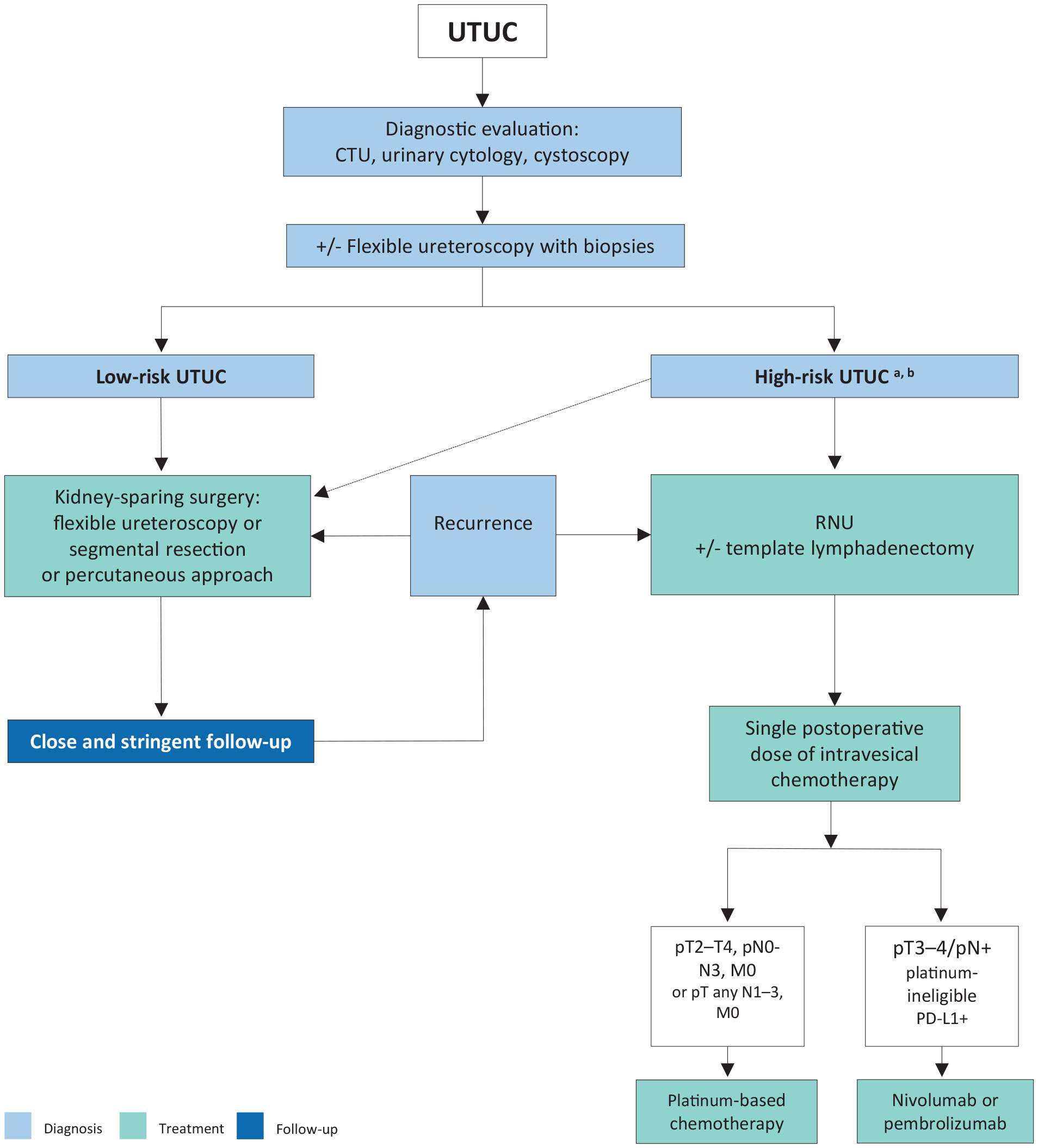 a: In patients with solitary kidney, consider a more conservative approach.
a: In patients with solitary kidney, consider a more conservative approach.
b: In low-grade patients without invasive features, consider a more conservative approach.CTU = computed tomography urography; PD-L1 = programmed death-ligand 1; RNU = radical nephroureterectomy; UTUC = upper urinary tract urothelial carcinoma.
Figure 7.2: Surgical treatment according to location and risk status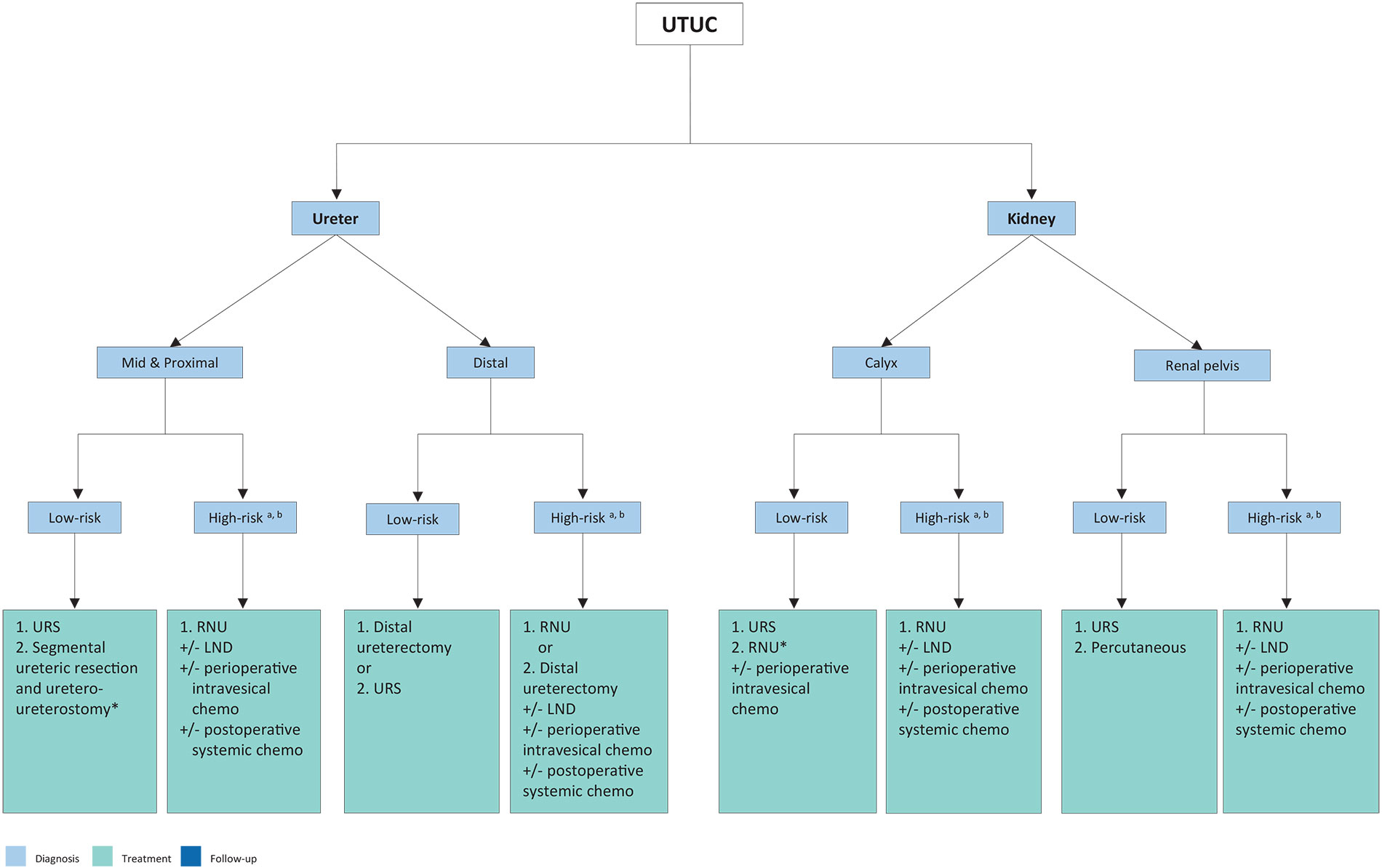 a: In patients with solitary kidney, consider a more conservative approach.
a: In patients with solitary kidney, consider a more conservative approach.
b: In low-grade patients without invasive features, consider a more conservative approach.
1 = first treatment option; 2 = secondary treatment option.
*If not amendable to endoscopic management.
LND = lymph node dissection; RNU = radical nephroureterectomy; URS = ureteroscopy; UTUC = upper urinary tract urothelial carcinoma.
7.3. Metastatic disease
7.3.1. Clinical locoregional lymph node metastases
Patients with clinical evidence of positive locoregional LNs which are surgically resectable fall in between localised and metastatic disease, and their treatment remains an area of debate. There are a number of options that could be considered based on published clinical trial data, but first-line treatment relies on systemic therapy. Induction platinum-based chemotherapy or enfortumab vedotin with pembrolizumab (EV + P) combination could be considered for these patients [257,258]. Radical nephroureterectomy with template-based LND can be discussed in a multidisciplinary team for patients responding to initial systemic therapy [259]. A systematic review and meta-analysis of 15 studies showed that induction chemotherapy followed by RNU is associated with greater odds of pathological downstaging (response rate = 3.06; 95% CI: 2.48-3.77) and appears to prolong OS compared with RNU plus AC. However, the analysis is limited by the heterogeneity of the studies, fewer adjuvant trials, and differences in how nodal status was reported in the studies (e.g. type of nodal surgery) [259]. In addition, no data are available on the use of RNU combined with EV + P in these patients.
In patients whose cancer is stable or progresses, maintenance avelumab or second-line treatment can be offered, similar to distant metastatic disease. Unresectable cN+ patients should be treated as distant metastatic patients.
7.3.2. Distant metastases
7.3.2.a. Systemic treatments - first-line setting
7.3.2.a.1. Enfortumab vedotin plus pembrolizumab combination therapy
For more than 23 years, despite multiple attempts with new agents and/or combinations of treatments, platinum-based chemotherapy remained the standard of care for previously untreated advanced or metastatic UC. In October 2023, the landscape changed dramatically with the EV302 phase III randomised multicentre study. This compared the nectin-4–directed antibody–drug conjugate EV + P with platinum-based combination chemotherapy (gemcitabine-cisplatin or gemcitabine-carboplatin. See Table 2 for the definition of cisplatin eligibility).
The study showed significant improvement in both PFS (HR: 0.45; 95% CI: 0.38-0.54) and OS (HR: 0.47; 95% CI: 0.38-0.58) with a response rate of 68% (vs. 44%) and a CR of 29%. Overall survival benefit was seen across subgroups regardless of cisplatin eligibility. The most common grade 3 or above treatment-related AEs of special interest included skin reactions (15.5%), peripheral neuropathy (6.8%) and hyperglycaemia (6.1%). The proportion of UTUC patients in this study was 25% and preplanned subgroup analysis showed benefit irrespective of tumour location [260].
Sequencing of treatment after EV + P is currently unclear and later-line treatments will depend on what agents the patient has previously received (Figure 7.3).
7.3.2.a.2. Patients ineligible for enfortumab vedotin plus pembrolizumab and fit for cisplatin-based combination chemotherapy
Upper tract UC and urothelial BC both respond to systemic platinum-based chemotherapy. Eligibility for platinum-based chemotherapy in the metastatic setting is based on the same criteria outlined in Table 2. A retrospective analysis of three RCTs showed that primary tumour location in the lower or upper urinary tract had no impact on progression-free survival (PFS) or OS in patients with locally advanced or metastatic UC treated with platinum-based combination chemotherapy [261]. Therefore, cisplatin-containing combination chemotherapy is the standard treatment for advanced or metastatic UTUC in patients who are ineligible for EV + P [2]. A number of cisplatin-containing chemotherapy regimens have proven efficacy, although gemcitabine and cisplatin are the most widely used. The use of cisplatin-based chemotherapy is widely considered in patients with eGFR > 45mL/min [261].
The efficacy of immunotherapy using programmed death 1 (PD-1) or PD-L1 inhibitors has been evaluated in the first-line setting for the treatment of cisplatin/carboplatin-fit patients with metastatic UC, including those with UTUC [262]. First-line immune CPIs or the combination of platinum-based chemotherapy with immune CPIs have not previously resulted in positive significant survival advantages and were thus not previously recommended [263-265]. These studies included both cisplatin and carboplatin combinations.
A phase III RCT in advanced/metastatic UC has now shown an OS benefit from the addition of nivolumab to chemotherapy (gemcitabine-cisplatin). Median OS was improved (21.7 months vs. 18.9 months; HR: 0.78; 95% CI: 0.63-0.96) as well as median PFS (7.9 months vs. 7.6 months; HR: 0.72; 95% CI: 0.59-0.88). Objective response rates were 57.6% compared with 43.1% for chemotherapy alone [266]. Although there is no subgroup analysis based on tumour position in this study, 12.6% of patients had UTUC.
7.3.2.a.3. Patients ineligible for enfortumab vedotin plus pembrolizumab and unfit for cisplatin-based combination chemotherapy
Carboplatin-based chemotherapy is recommended in patients unfit for cisplatin [2]. Carboplatin with gemcitabine is the preferred regimen [267], irrespective of PD-L1 status. In a critical re-analysis of RCTs comparing OS after cisplatin versus carboplatin-based regimens in advanced UC, cisplatin conferred a minor OS benefit compared to carboplatin [268].
7.3.2.a.4. Maintenance therapy after first-line platinum-based chemotherapy
Maintenance avelumab is recommended in patients with complete/partial response or stable disease after four to six cycles of platinum-based chemotherapy, given in the first line setting only. Data from a phase III RCT showed that the use of maintenance avelumab therapy after four to six cycles of gemcitabine plus cisplatin or carboplatin (started within ten weeks of completion of first-line platinum-based chemotherapy) significantly prolonged OS as compared to best supportive care alone in those patients with advanced or metastatic UC who did not experience disease progression during, or responded to, first-line chemotherapy (HR: 0.69; 95% CI: 0.56-0.86) [269,270]. An increase in median OS from 14 to 21 months was observed with avelumab. Although no subgroup analysis based on tumour location was available in this study, almost 30% of the included patients had UTUC. Similarly, in a phase II study comprising 108 patients with metastatic UC achieving at least stable disease on first-line platinum-based chemotherapy, maintenance pembrolizumab improved PFS compared to placebo (5.4 vs. 3.0 months) [271].
7.3.2.a.5. Patients unfit for any combination therapy
Pembrolizumab or atezolizumab are alternative choices for patients who are PD-L1 positive and not eligible or fit for platinum-based chemotherapy. In a single-arm phase II trial (n = 370) of cisplatin-ineligible UC, pembrolizumab monotherapy was associated with an objective response rate of 26% in 69 metastatic UTUC patients [272]. In the overall cohort, a PD-L1 expression of 10% was associated with a greater response rate to pembrolizumab. Treatment-related toxicity was in line with previous studies. In a single-arm phase II trial (n = 119) of cisplatin-ineligible UC, atezolizumab monotherapy was associated with an objective response rate of 39% in 33 (28%) metastatic UTUC patients [273]. Median OS in the overall cohort was 15.9 months and treatment-related toxicity was in line with previous studies [264].
7.3.2.b. Systemic treatments - later-line setting
Subsequent treatments depend on the type of treatment given in the first-line setting.
7.3.2.b.1. Platinum-based chemotherapy
Platinum-based chemotherapy should be the second-line treatment of choice if not received in the first-line setting. No data support the use of maintenance avelumab outside of the first-line setting. In addition, patients in this category are likely to have already received a CPI in the first-line setting, either in combination with enfortumab vedotin (EV) or as monotherapy.
7.3.2.b.2. Immunotherapy
A phase III RCT including 542 patients who received prior platinum-based chemotherapy for advanced UC showed that pembrolizumab decreased the risk of death compared to second-line chemotherapy (the investigator’s choice of paclitaxel, docetaxel or vinflunine); median OS: 10.3 months for pembrolizumab and 7.4 months for chemotherapy (HR: 0.73; 95% CI: 0.59-0.91) [274]. Responses were more frequent and durable for pembrolizumab compared to chemotherapy (21% vs. 11%). In the UTUC subgroup (n = 75/13.8%), the OS benefit seemed larger (50%).
The IMVigor211 trial explored atezolizumab in PD-L1-positive tumours in patients with tumours that relapsed after platinum-based chemotherapy. The results of the trial failed to show a significant OS advantage of atezolizumab compared to second-line chemotherapy [275].
Other immunotherapies, such as nivolumab [276], avelumab [277,278] and durvalumab [279], have shown objective response rates ranging from 17.8% [279] to 19.6% [276], and median OS ranging from 7.7 months to 18.2 months, in patients with platinum-resistant metastatic UC. These results were obtained from single-arm phase I or II trials only, and the number of UTUC patients included in these studies was only specified for avelumab (n = 7/15.9%) without any subgroup analysis based on primary tumour location [278].
The immunotherapy combination of nivolumab plus ipilimumab has shown significant antitumour activity with objective response rate up to 38% in a phase I/II multicentre trial including 78 patients with metastatic UC experiencing disease progression after platinum-based chemotherapy [280]. Although UTUC patients were included in this trial, no subgroup analysis was available. A real-world study of 235 patients with UTUC treated with pembrolizumab after progression on first-line platinum-based chemotherapy confirmed objective response rate of 32% [281]. Other immunotherapy combinations may be effective in the second-line setting, but data are currently limited [282].
7.3.2.b.3. Novel agents
Fibroblast growth factor receptors inhibition
Erdafitinib, a pan-FGFR tyrosine kinase inhibitor of FGFR 1-4, was associated with a 40% radiological response rate according to the Response Evaluation Criteria in Solid Tumours (RECIST) phase II trial of 99 patients with locally advanced or metastatic UC who progressed after first-line chemotherapy and harboured a FGFR DNA genomic alterations (FGFR 2/3 fusions or FGFR 3 mutations) [136]. This study included 23 UTUC patients with visceral metastases showing a 43% radiological response rate. The subsequent phase III Thor trial randomised 266 patients with advanced UC who had similar mutations and had experienced disease progression after 1-2 lines of previous treatment, to treatment with either erdafitinib or investigators choice of chemotherapy (vinflunine or docetaxel). Significant improvements in median OS (4.3 months; HR: 0.64; 95% CI: 0.47-0.88), PFS (2.9 months; HR: 0.58; 95% CI: 0.44-0.78) and a 36% risk reduction in death were observed. In this study, 33.5% of patients had UTUC [283]. A potentially greater impact of FGFR 3 targeting agents is anticipated, because the rate of activating alterations of FGFR 3 is higher in UTUC than in BC [284]. UTUC patients should be tested for FGFR alterations (FGFR 2/3 mutations or FGFR 3 fusions) prior to erdafitinib treatment. Early testing for FGFR 2/3 alterations, mutations and deletions should be considered for patients presenting with advanced/metastatic UTUC
(Section 5.7).
Antibody drug conjugates
A phase II study enrolled 89 patients (43% of whom had UTUC) with cisplatin-unfit metastatic UC experiencing disease progression after therapy with PD-1 or PD-L1 inhibitors. All patients received the antibody-drug conjugate EV. The objective radiological response rate (RECIST) was 52%, of which 20% of patients achieved CR [285]. In a phase III trial of EV for the treatment of patients with locally advanced or metastatic UC who had previously received platinum-containing chemotherapy and had disease progression during or after treatment with a PD-1 or PD-L1 inhibitor, EV significantly prolonged survival as compared to standard chemotherapy (median OS 12.88 vs. 8.97 months) [286].
In an open-label phase II trial, a total of 108 patients with metastatic UC who progressed after platinum-based chemotherapy and CPIs were treated with the antibody-drug conjugate sacituzumab govitecan. The objective radiological response rate was 27%, with median duration of response of 7.2 months, median PFS of 5.4 months and median OS of 10.9 months. However, the proportion of patients with UTUC was not mentioned in the publication [287].
A preplanned subgroup analysis from the phase III RANGE trial assessed the impact on outcomes and safety of ramucirumab added to docetaxel after disease progression on both platinum-based chemotherapy and immune CPIs [288]. Median PFS was 3.15 months on ramucirumab/docetaxel versus 2.73 months on placebo/docetaxel (HR: 0.786; 95% CI: 0.404-1.528; p = 0.4877). This trend for ramucirumab benefit occurred despite the ramucirumab arm having a higher percentage of patients with poorer prognosis. However, these findings need confirmation by further studies, because this analysis is limited by patient numbers and an imbalance in the treatment arms.
7.3.2.c. Surgery
7.3.2.c.1. Radical nephroureterectomy
Data regarding RNU in the metastatic setting are lacking with evidence mainly limited to retrospective observational studies [289-291].
Although evidence remains very limited, RNU may be associated with CSS [290,292-294] and OS benefit in selected patients, especially those fit enough to receive cisplatin-based chemotherapy [289,290]. It is noteworthy that these benefits may be limited to those patients with only one metastatic site [290]. Nonetheless, given the high risk of bias of the observational studies addressing RNU for metastatic UTUC, indications for RNU in this setting should mainly be reserved for palliative patients, aimed at controlling symptomatic disease [24,295].
7.3.2.c.2. Metastasectomy
There is no UTUC-specific study supporting the role of metastasectomy in patients with advanced disease. Reports suggesting that resection of metastatic lesions could be safe and oncologically beneficial in selected patients should be interpreted with caution [296-300]. In the absence of data from RCTs, patients should be evaluated on an individual basis and the decision to perform a metastasectomy (surgically) should be made following a shared decision-making process with the patient.
7.3.3. Summary of evidence and recommendations for the treatment of metastatic upper urinary tract urothelial carcinoma
| Summary of evidence | LE |
| Enfortumab vedotin plus pembrolizumab offers an OS benefit compared to gemcitabine-cisplatin in the first-line setting. | 1b |
| Cisplatin-based combination chemotherapy can improve median survival. | 2 |
| Cisplatin-containing combination chemotherapy is the standard of care in advanced or metastatic patients fit enough to tolerate cisplatin and who are ineligible for EV + P. | 1b |
| Cisplatin-containing combination chemotherapy in combination with nivolumab offers a survival advantage compared with chemotherapy alone in the first-line setting. | 1b |
| Carboplatin-based combination chemotherapy offers a survival benefit in cisplatin unfit patients. | 1b |
| Non-platinum combination chemotherapy has not been tested against standard chemotherapy in patients who are fit or unfit for cisplatin combination chemotherapy. | 4 |
| Maintenance avelumab is associated with an OS advantage compared with best supportive care in patients who did not have disease progression after four to six cycles of gemcitabine plus either cisplatin or carboplatin. | 1b |
| Pembrolizumab, a PD-1 inhibitor, has been approved for patients who have experienced disease progression during or after previous platinum-based chemotherapy and did not receive previous immune therapy, based on the results of a phase III trial. | 1b |
| Nivolumab, a PD-1 inhibitor, has been approved for patients who have experienced disease progression during or after previous platinum-based chemotherapy and did not receive previous immune therapy, based on the results of a phase II trial. | 2a |
| Pembrolizumab, a PD-1 inhibitor, has been approved for patients with advanced or metastatic UC unfit for platinum-based first-line chemotherapy, based on the results of a phase II trial, but use of pembrolizumab is restricted to PD-L1 positive patients. | 2a |
| Atezolizumab, a PD-L1 inhibitor, has been approved for patients with advanced or metastatic UC unfit for platinum-based first-line chemotherapy, based on the results of a phase II trial, but use of atezolizumab is restricted to PD-L1 positive patients. | 2a |
| Erdafitinib was associated with improved OS in platinum-refractory patients with locally advanced or metastatic UC and FGFR DNA genomic alterations (FGFR 2/3 mutations or FGFR 3 fusions). | 1b |
| Enfortumab vedotin was associated with OS benefit in patients who had previously received platinum-containing chemotherapy and experienced disease progression during or after treatment with a PD-1 or PD-L1 inhibitor. | 1b |
| Palliative RNU can improve quality of life by controlling symptomatic disease. | 3 |
| Radical nephroureterectomy could confer a survival benefit in highly selected patients with metastatic UC, for example, after response to platinum-based combination chemotherapy with limited metastatic burden. | 4 |
| Recommendations | Strength rating |
| Offer EV + P as first-line treatment to patients with advanced/metastatic disease. | Strong |
| First-line treatment for platinum-eligible patients who are unsuitable/ineligible for EV + P | |
| Offer platinum combination chemotherapy to platinum-eligible patients. | Strong |
| Offer cisplatin-based chemotherapy with gemcitabine-cisplatin plus nivolumab in cisplatin-eligible patients. | Weak |
| Offer cisplatin-based chemotherapy with gemcitabine/cisplatin or HD-MVAC to cisplatin-eligible patients. | Strong |
| Offer gemcitabine/carboplatin chemotherapy to cisplatin-ineligible patients. | Strong |
| Offer maintenance avelumab to patients who did not have disease progression after four to six cycles of platinum-based combination chemotherapy. | Strong |
| First-line treatment in patients ineligible for any combination therapy | |
| Offer CPIs pembrolizumab or atezolizumab to patients with PD-L1 positive tumours. | Weak |
| Later lines of treatment | |
| Offer platinum-based combination chemotherapy as second-line treatment of choice if not received in the first-line setting. | Strong |
| Offer CPI (pembrolizumab) to patients with disease progression during or after platinum-based combination chemotherapy for metastatic disease who did not receive maintenance avelumab. | Strong |
| Offer EV to patients previously treated with platinum-containing chemotherapy and who had disease progression during or after treatment with a PD-1 or PD-L1 inhibitor. | Strong |
Offer erdafitinib as an alternative subsequent-line therapy to patients:
| Strong |
| Only offer vinflunine to patients with metastatic disease as second-line treatment if immunotherapy or combination chemotherapy is not feasible. Alternatively, offer vinflunine as third- or subsequent-line treatment. | Strong |
| Offer RNU as a palliative treatment to symptomatic patients with resectable locally advanced tumours. | Weak |
CPI = checkpoint inhibitors; DNA = deoxyribonucleic acid; EV = enfortumab vedotin; EV + P = enfortumab vedotin plus pembrolizumab; FGFR = fibroblast growth factor receptors; HD-MVAC = high-dose intensity methotrexate, vinblastine, adriamycin plus cisplatin; PD-1 = programmed death-1; PD-L1 = programmed death-ligand 1; RNU = radical nephroureterectomy.
Figure 7.3 Flowchart for the management of metastatic upper urinary tract urothelial carcinoma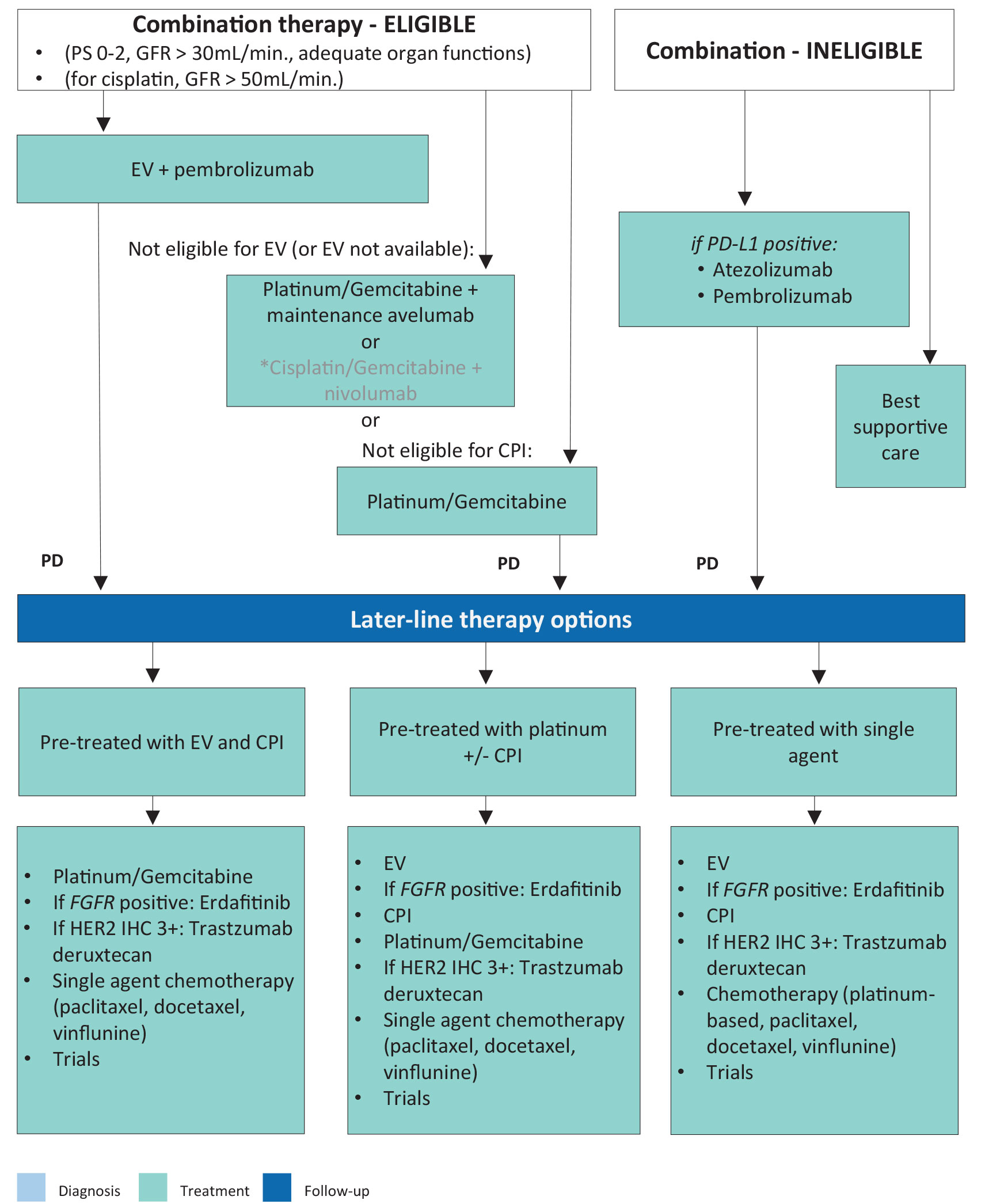 *In view of lack of subgroup analysis data for upper urinary tract urothelial carcinoma.
*In view of lack of subgroup analysis data for upper urinary tract urothelial carcinoma.
CPI = checkpoint inhibitor; EV = enfortumab vedotin; FGFR = fibroblast growth factor receptor; GFR = glomerular filtration rate; PD = programmed death; PD-L1 = programmed death-ligand 1; PS = performance status.