6. DISORDERS OF EJACULATION
6.1. Introduction
Ejaculation is a complex physiological process that comprises emission and expulsion processes and is mediated by interwoven neurological, hormonal and anatomical pathways [584,585]. Any interference with those pathways may cause a wide range of ejaculatory disorders. The spectrum of ejaculation disorders includes PE, retarded or DE, anejaculation, painful ejaculation, RE, anorgasmia and haemospermia.
6.2. Premature ejaculation
6.2.1. Epidemiology
Historically, the main problem in assessing the prevalence of PE has been the lack of a universally recognised definition at the time that surveys were conducted [586]. See Section 4.2 for a comprehensive discussion of the epidemiology of PE.
6.2.2. Pathophysiology and risk factors
The aetiology of PE is relatively unknown, with limited data to support suggested biological and psychological hypotheses, including anxiety [587-590], penile hypersensitivity [591-598] and 5-hydroxytryptamine (HT) receptor dysfunction [599-604]. The classification of PE into four subtypes [214] has contributed to a better delineation of lifelong, acquired, variable and subjective PE [605-607]. It has been hypothesised that the pathophysiology of lifelong PE is mediated by a complex interplay of central and peripheral serotonergic, dopaminergic, oxytocinergic, endocrinological, genetic and epigenetic factors [608]. Acquired PE may occur due to psychological problems, such as sexual performance anxiety, psychological or relationship problems, and/or comorbidity, including ED, prostatitis, hyperthyroidism and poor sleep quality [609-612]. Variable PE is considered to be a normal variation of sexual function, whereas subjective PE can stem from cultural or abnormal psychological constructs [214].
A significant proportion of males with ED also experience PE [218,613-616]. High levels of performance anxiety related to ED may worsen PE, with a risk of misdiagnosing PE instead of the underlying ED. According to the National Health and Social Life Survey (NHSLS), the prevalence of PE is not affected by age [210], unlike ED which increases with age. Premature ejaculation is not affected by marital or income status [210,617]. However, PE is more common in black and hispanic males, as well as males from regions where an Islamic background is common [210,618-621]. The prevalence may be higher in males with a lower educational level [210,218]. Other reported risk factors for PE include genetic predisposition [604,622-625], poor overall health status and obesity [210], prostate inflammation [626-630], hyperthyroidism [609], low prolactin levels [631], high testosterone levels [632], vitamin D and B12 deficiency [633,634], diabetes [635,636], MetS [637,638], lack of physical activity [639], emotional problems and stress [210,640,641], depressive symptoms [641], and traumatic sexual experiences [210,218].
6.2.3. Impact of PE on quality of life
Men with PE are more likely to report low satisfaction with their sexual relationship, low satisfaction with sexual intercourse, difficulty relaxing during intercourse, and less frequent intercourse [642-644]. Premature ejaculation can have a detrimental effect on self-confidence and the relationship with a partner, and may sometimes cause mental distress, anxiety, embarrassment and depression [642,645,646]. Premature ejaculation may also affect the partner’s sexual functioning, and their satisfaction with the sexual relationship decreases with increasing severity of the patient’s condition [647-649]. Despite the possible serious psychological and QoL consequences of PE, few men seek treatment [211,218,615,650-652].
6.2.4. Classification
There is still little consensus about the definition and classification of PE [653]. It is now universally accepted that 'premature ejaculation' is a broad term that includes several concepts belonging to the common category of PE. The International Classification of Diseases 11th Revision (ICD-11) renamed PE as Early Ejaculation [654], with the following definition: “Male early ejaculation is characterized by ejaculation that occurs prior to or within a very short duration of the initiation of vaginal penetration or other relevant sexual stimulation, with no or little perceived control over ejaculation. The pattern of early ejaculation has occurred episodically or persistently over a period of at least several months and is associated with clinically significant distress.”
This definition includes four categories: male early ejaculation; lifelong generalised and situational; acquired generalised and situational; and unspecified. The Diagnostic and Statistical Manual of Mental Disorders V (DSM-V) [220] and the International Society for Sexual Medicine (ISSM) [655] published definitions for lifelong and acquired PE. These definitions are overlapping, with three shared factors (1. Time to ejaculation assessed by IELT; 2. Perceived control; 3. Distress, bother, frustration, interpersonal difficulty related to the ejaculatory dysfunction), resulting in a multidimensional diagnosis [655].
Two more PE syndromes have been proposed [606]:
- ‘Variable PE’ is characterised by inconsistent and irregular early ejaculations, representing a normal variation in sexual performance.
- ‘Subjective PE’ is characterised by subjective perception of consistent or inconsistent rapid ejaculation during intercourse, while ejaculation latency time is in the normal range or can even last longer. It should not be regarded as a symptom or manifestation of true medical pathology [656].
6.2.5. Diagnostic evaluation
Diagnosis of PE is based on the patient’s medical and sexual history [657-660]. History should classify PE as lifelong or acquired and determine whether PE is situational (under specific circumstances or with a specific partner) or consistent. Special attention should be given to the duration time of ejaculation, degree of sexual stimulus, impact on sexual activity and QoL, and drug use or abuse. It is also important to distinguish PE from ED. Many patients with ED develop secondary PE caused by the anxiety associated with difficulty in attaining and maintaining an erection [613,661]. Furthermore, some patients are unaware that loss of erection after ejaculation is normal and may erroneously complain of ED, while the actual problem is PE [615].
6.2.5.a. Intravaginal ejaculatory latency time (IELT)
Although it has been suggested as an objective diagnostic criterion and treatment outcome measure [662,663], the use of IELT alone is not sufficient to define PE, as there is significant overlap between men with and without PE [664,665]. Moreover, some men may experience PE in their non-coital sexual activities (e.g. during masturbation, oral sex or anal intercourse); thus, measuring IELT will not be suitable for their assessment [666]. Although PE is apparently less prevalent and less bothersome among MSM [667], many of these patients may also suffer from PE and IELT cannot be applied [667-669]. Although some studies demonstrated that MSM report longer ejaculation latency time compared to heterosexual men [667], others failed to demonstrate such a difference [670].
In everyday clinical practice, self-estimated IELT is sufficient [671]. Self-estimated and stopwatch-measured IELT are interchangeable and correctly assign PE status with 80% sensitivity and 80% specificity [672,673].
Measurement of IELT with a calibrated stopwatch is mandatory in clinical trials. For any drug treatment study of PE, Waldinger et al. suggested using geometric mean instead of arithmetic mean IELT because the distributed IELT data are skewed. Otherwise, any treatment-related ejaculation delay may be overestimated, if the arithmetic mean IELT is used instead of the geometric mean IELT [674].
6.2.5.b. Premature ejaculation assessment questionnaires
The need to objectively assess PE has led to the development of several questionnaires based on using patient-reported outcome measures (PROMs). Only two questionnaires can discriminate between patients who have PE and those who do not:
- Premature Ejaculation Diagnostic Tool (PEDT): A five-item questionnaire based on focus groups and interviews from the USA, Germany, and Spain assesses control, frequency, minimal stimulation, distress and interpersonal difficulty [675]. A total score of > 11 suggests a diagnosis of PE, 9 or 10 suggests a probable diagnosis, and < 8 indicates a low likelihood of PE.
- Arabic Index of Premature Ejaculation (AIPE): A seven-item questionnaire developed in Saudi Arabia assesses sexual desire, hard erections for sufficient intercourse, time to ejaculation, control, satisfaction of the patient and partner, and anxiety or depression [676]. A cut-off score of 30 (range 7-35) discriminates PE diagnosis best. Severity of PE is classified as severe (score: 7-13), moderate (score: 14-19), mild-to-moderate (score: 20-25) and mild (score: 26-30).
Other questionnaires used to characterise PE and determine treatment effects include the Premature Ejaculation Profile (PEP) [665], Index of Premature Ejaculation (IPE) [677], and Male Sexual Health Questionnaire (MSHQ) - Ejaculatory Dysfunction [678]. Currently, their role is optional in everyday clinical practice. The masturbatory PEDT has also been proposed [679], due to fact that PE patients report longer IELTs and lesser bother/distress during masturbation than partnered sex [666]. However, further validation studies are required before the routine use of this questionnaire in this population.
6.2.5.c. Physical examination and investigations
Physical examination may be part of the initial assessment of men with PE. It may include a focused examination of the urological, endocrine and neurological systems to identify underlying medical conditions associated with PE or other sexual dysfunctions, such as endocrinopathy, PD, urethritis or prostatitis. Laboratory or physiological testing should be directed by specific findings from history or physical examination and is not routinely recommended [659].
6.2.5.d. Summary of evidence and recommendations for the diagnostic evaluation of PE
| Summary of evidence | LE |
| A comprehensive medical history and a thorough physical examination can serve as valuable tools for clinicians in identifying the underlying medical factors contributing to PE. | 3 |
| PE can negatively impact self-confidence, strain partner relationships, and potentially lead to emotional distress, anxiety, shame, and depression. | 2a |
| Several questionnaires can be used for the diagnosis of PE (PEDT, AIPE) and for assessing the therapeutic outcomes of PE interventions (PEP). | 2b |
| Although relying on IELT is inadequate for characterising PE, self-reported IELT proves satisfactory in routine clinical contexts. | 3 |
| Recommendations | Strength rating |
| Perform the diagnosis and classification of premature ejaculation (PE) based on medical and sexual history, which should include assessment of intravaginal ejaculatory latency time (self-estimated), perceived control, distress, and interpersonal difficulty due to the ejaculatory dysfunction. | Strong |
| Use patient-reported outcomes in daily clinical practice. | Weak |
| Include physical examination in the initial assessment of PE to identify anatomical abnormalities that may be associated with PE or other sexual dysfunctions, particularly erectile dysfunction. | Strong |
| Do not perform routine laboratory or physiological tests. They should only be directed by specific findings from history or physical examination. | Strong |
6.2.6. Disease management
Before commencing any treatment, it is essential to define the subtype of PE and discuss patient’s expectations thoroughly.
Pharmacotherapy must be considered the first-line treatment for patients with lifelong PE, whereas treating the underlying cause (e.g. ED, prostatitis, LUTS, anxiety and hyperthyroidism) must be the initial goal for patients with acquired PE [659].
Dapoxetine (30 and 60mg) is the first on-demand oral pharmacological agent approved for lifelong and acquired PE in many countries, except for the USA [573]. The metered-dose aerosol spray of lidocaine (150mg/mL) and prilocaine (50mg/mL) combination is the first topical formulation to be officially approved for on-demand treatment of lifelong PE by the EMA in the European Union [680]. All other medications used in PE are off-label indications [681]. In this context, daily or on-demand use of selective serotonin re-uptake inhibitors (SSRIs), clomipramine and on-demand topical anaesthetic agents have consistently shown efficacy in PE [682-685]. The long-term outcomes of pharmacological treatments are unknown.
Various behavioural techniques may be beneficial in treating variable and subjective PE [686]. Psychotherapy can also be considered for PE patients who are uncomfortable with pharmacological therapy or in combination with pharmacological therapy [687-689]. However, there is weak and inconsistent evidence regarding the effectiveness of these psychosexual interventions, and their long-term outcomes in PE are unknown [690].
An evidence-based analysis of all current treatment modalities was performed. Levels of evidence and grades of recommendation are provided, and a treatment algorithm is presented (Figure 6.1).
Figure 6.1: Management of premature ejaculation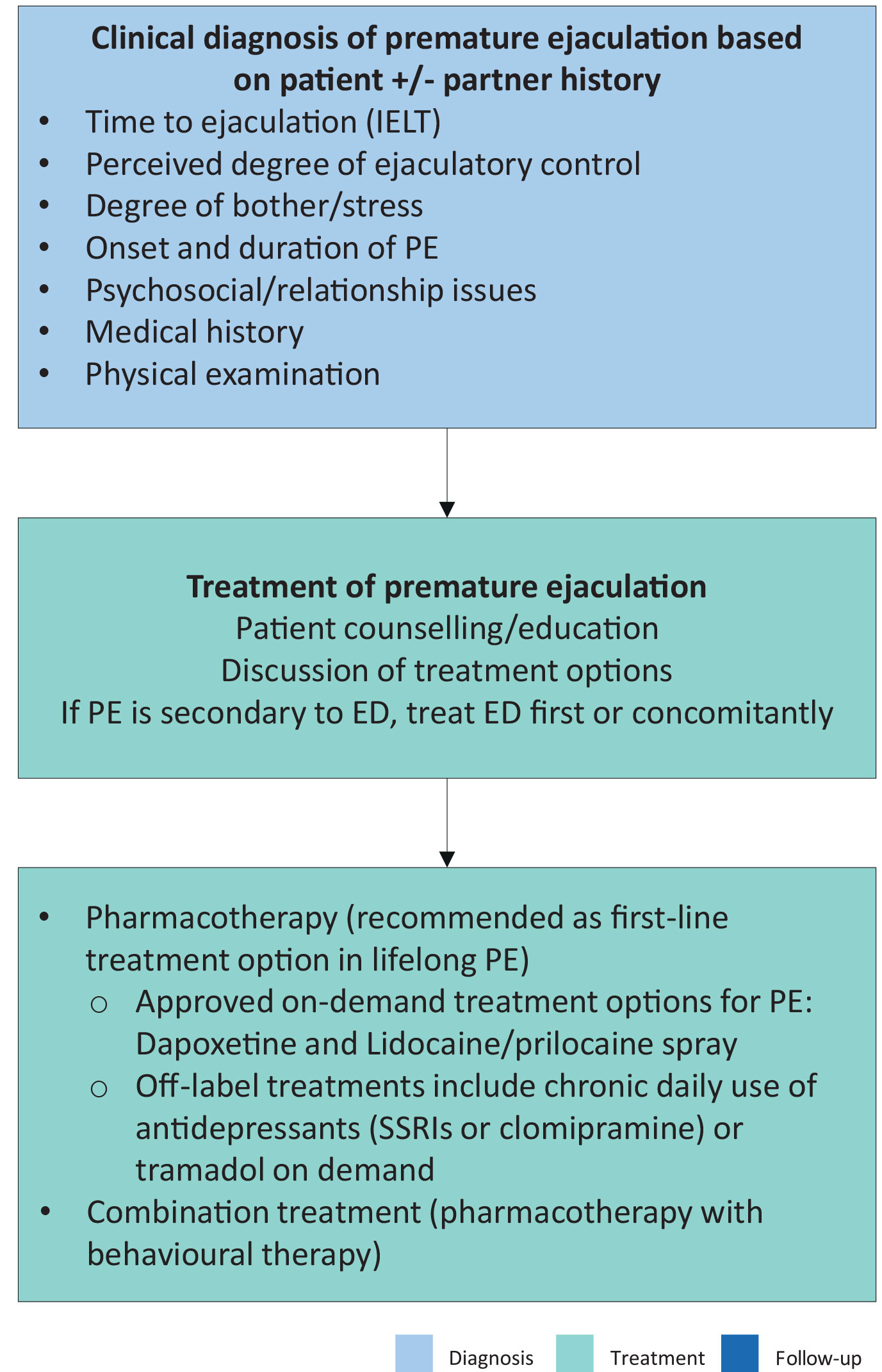 ED = erectile dysfunction; IELT = intravaginal ejaculatory latency time; PE = premature ejaculation; SSRI = selective serotonin receptor inhibitor.
ED = erectile dysfunction; IELT = intravaginal ejaculatory latency time; PE = premature ejaculation; SSRI = selective serotonin receptor inhibitor.
Adapted from Lue et al., 2004.
6.2.6.a. Psychological aspects and intervention
Psychosexual interventions, whether behavioural, cognitive, or focused on the couple, are aimed at teaching techniques to control/delay ejaculation, gaining confidence in sexual performance, reducing anxiety, and promoting communication and problem-solving within the couple [686]. Interventions with a focus on sexual education or acceptance may be positive as well [691]. Psychosexual interventions alone for PE lack empirical support. Evidence suggests that start-stop exercises, combined with psychoeducation and mindfulness techniques improve PE symptoms, as well as PE-associated distress, anxiety and depression [692]. The potential benefits of mindfulness have been reported [693]. Behavioural and biofeedback therapy may be most effective when used to ‘add value’ to medical interventions [694]. Smartphone-delivered psychological intervention, aimed at improving behavioural skills for ejaculatory delay and sexual self-confidence, has positive effects, supporting e-health in the context of PE [695].
Figure 6.2: Key aspects for psychosexual evaluation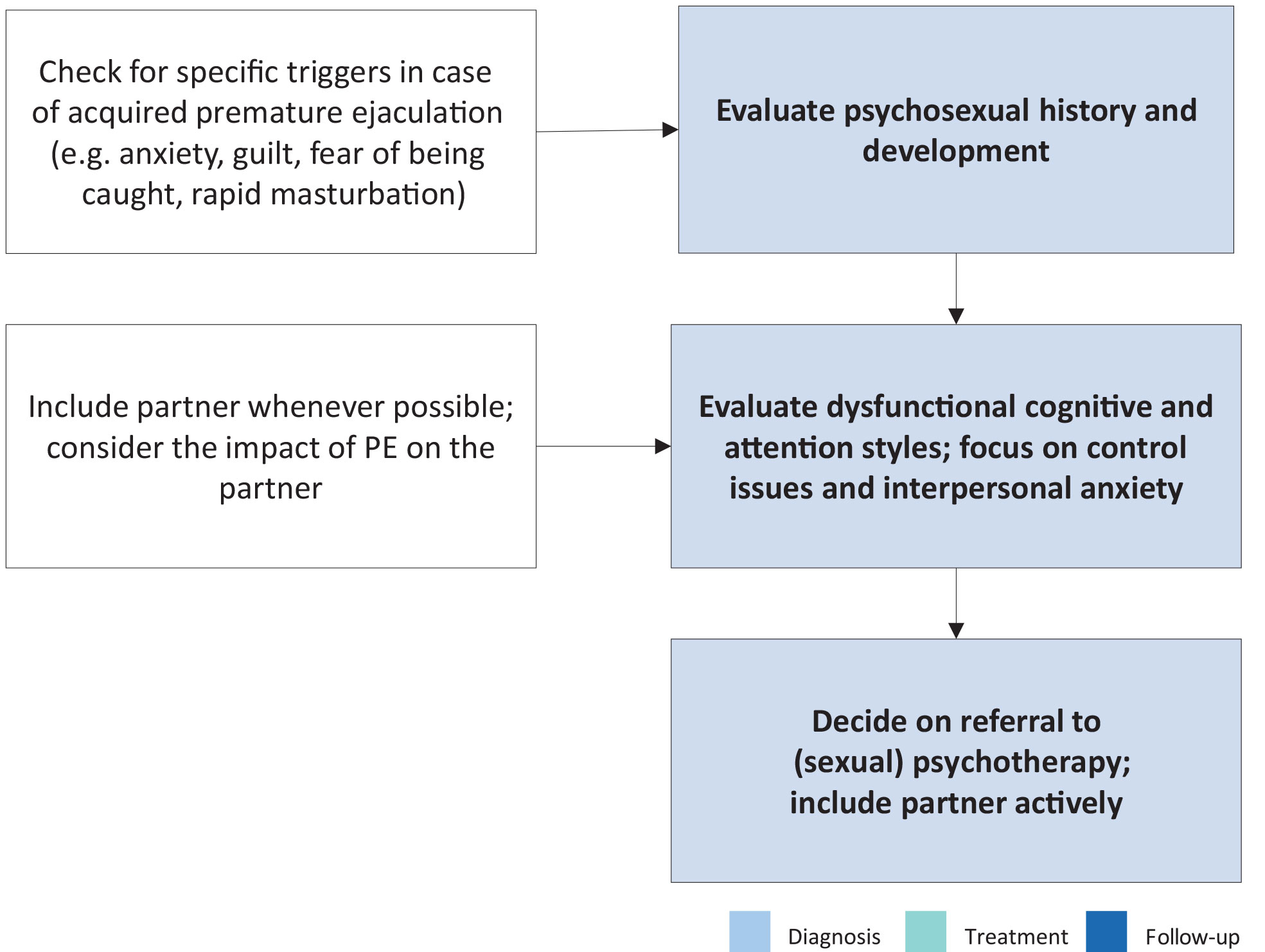
6.2.6.a.1. Summary of evidence and recommendations for the assessment and treatment (psychosexual approach) of PE
| Summary of evidence | LE |
| The incorporation of psychosexual approach, alongside psychoeducational guidance and mindfulness techniques, ameliorate symptoms of PE and alleviate the associated distress, anxiety and depression. | 2b |
| The combination of psychosexual approaches and pharmacological treatments yields superior outcomes compared to pharmacological interventions alone. | 3 |
| Recommendations for assessment | Strength rating |
| Assess sexual history and psychosexual development. | Strong |
| Assess anxiety and interpersonal anxiety; focus on control issues. | Strong |
| Include the partner if available; check for the impact of premature ejaculation on the partner. | Strong |
| Recommendations for treatment (psychosexual approach) | |
| Use behavioural, cognitive and/or couple therapy approaches in combination with pharmacotherapy. Discuss the use of mindfulness exercises. | Weak |
6.2.6.b. Pharmacotherapy
6.2.6.b.1. Dapoxetine
Dapoxetine hydrochloride is a short-acting SSRI with a pharmacokinetic profile suitable for on-demand treatment of PE [696]. It has a rapid Tmax (1.3 hours) and a short half-life (95% clearance rate after 24 hours) [697,698]. It is approved for on-demand treatment of PE in European countries and elsewhere, but not in the USA. Both available doses of dapoxetine (30mg and 60mg) have shown 2.5- and 3.0-fold increases, respectively, in IELT overall, rising to 3.4- and 4.3-fold in patients with a baseline average IELT < 30 seconds [699].
In RCTs, dapoxetine, 30mg or 60mg 1-2 hours before intercourse was effective at improving IELT and increasing ejaculatory control, decreasing distress and increasing satisfaction [699]. Dapoxetine has shown a similar efficacy profile in men with lifelong and acquired PE [699,700]. Treatment-related adverse effects were dose-dependent and included nausea, diarrhoea, thirst, headache and dizziness [700]. Treatment-emergent adverse events (TEAEs) were responsible for study discontinuation in 4% (30mg) and 10% (60mg) of subjects [671]. With abrupt dapoxetine cessation, there was no indication of an increased risk of suicidal ideation or suicide attempts, and little indication of withdrawal symptoms [699,700]. Dapoxetine is safer than formal anti-depressant compounds used for treatment of PE [701].
A low rate (0.1%) of vasovagal syncope was reported in phase III studies [702]. According to the summary of product characteristics, vital orthostatic signs (blood pressure and heart rate) must be measured prior to starting dapoxetine, and dose titration must be considered [703]. The EMA assessment report for dapoxetine concluded that the potential increased risk for syncope had been proven manageable with adequate risk minimisation measures [703]. No cases of syncope were observed in a post-marketing observational study, which identified patients at risk for the orthostatic reaction using the patient’s medical history and orthostatic testing [704].
Many patients and physicians may prefer using dapoxetine in combination with a PDE5I to extend the time until ejaculation and minimise the risk of ED due to dapoxetine treatment. Phase I studies of dapoxetine have confirmed that it has no pharmacokinetic interactions with PDE5Is (i.e. tadalafil 20mg and sildenafil 100mg) [705]. When dapoxetine is co-administered with PDE5Is, it is well tolerated, with a safety profile consistent with previous phase III studies of dapoxetine alone [706]. An RCT, including PE patients without ED, demonstrated that a combination of dapoxetine with sildenafil could significantly improve IELT values and PROMs compared with dapoxetine alone or sildenafil alone, with tolerable adverse events [707]. The efficacy and safety of dapoxetine/sildenafil combination tablets for the treatment of PE have also been reported [708].
The discontinuation rates of dapoxetine seem moderate to high [709]. The cumulative discontinuation rates increase over time, reaching 90% at two years after initiation of therapy. The reasons for the high discontinuation rate are cost (29.9%), disappointment that PE was not curable and the on-demand nature of the drug (25%), adverse effects (11.6%), perceived poor efficacy (9.8%), a search for other treatment options (5.5%), and unknown (18.3%) [710]. Similarly, it was confirmed that many patients on dapoxetine treatment spontaneously discontinued treatment, while this rate was reported at 50% for other SSRIs and 28.8% for paroxetine, respectively [711]. In a Chinese cohort study, 13.6% of the patients discontinued dapoxetine due to lack of efficacy (62%), adverse effects (24%), and low frequency of sexual intercourse (14%) [712].
6.2.6.b.2. Off-label use of antidepressants
Selective SSRIs are used to treat mood disorders but can delay ejaculation and therefore have been widely used ‘off-label’ for PE since the 1990s [713-715]. Commonly used SSRIs include continuous intake of citalopram, fluoxetine, fluvoxamine, paroxetine and sertraline, all of which have similar efficacy, whereas paroxetine exerts the most substantial ejaculation delay [662,716,717]. In a double-blind, placebo-controlled trial, a novel 5-HT1A receptor antagonist, GSK958108, was reported to significantly delay ejaculation [718].
Clomipramine, the most serotoninergic tricyclic antidepressant, was first reported in 1977 as an effective PE treatment [719,720]. In an RCT, on-demand use of clomipramine 15mg two to six hours before sexual intercourse was found to be associated with an IELT fold change and significant improvements in PROMs in the treatment group as compared with the placebo group (4.66 ± 5.64 vs. 2.80 ± 2.19; P < 0.05) [721,722]. The most reported TEAEs were nausea in 15.7% and dizziness in 4.9% of men [721,722]. Furthermore, two RCTs demonstrate that daily duloxetine 30mg significantly reduces PE severity and distress with acceptable tolerability [723,724]. Further studies are needed to confirm its long-term efficacy and safety before widespread recommendation.
Several meta-analyses suggest SSRIs may increase the geometric mean IELT by 2.6-13.2-fold [725]. Paroxetine is superior to fluoxetine, clomipramine and sertraline [726,727]. Sertraline is superior to fluoxetine, whereas the efficacy of clomipramine is not significantly different from that of fluoxetine and sertraline. Paroxetine was evaluated in doses of 20-40mg, sertraline 25-200mg, fluoxetine 10-60mg and clomipramine 25-50mg [725-728].
Ejaculation delay may start a few days after drug intake, but it is more evident after one to two weeks, as receptor desensitisation requires time to occur. Although efficacy may be maintained for several years, tachyphylaxis (decreased response to a drug following chronic administration) may occur after six to 12 months [719]. Common TEAEs of SSRIs include fatigue, drowsiness, yawning, nausea, vomiting, dry mouth, diarrhoea and perspiration; TEAEs are usually mild and gradually improve after two to three weeks of treatment [719,729]. Decreased libido, anorgasmia, anejaculation and ED have also been reported.
Due to the risk of suicidal ideation or suicide attempts, caution is recommended in prescribing SSRIs to young adolescents aged ≤ 18 years with PE, and to men with PE and a comorbid depressive disorder, particularly when associated with suicidal ideation. Patients should be advised to avoid sudden cessation or rapid dose reduction of daily-dosed SSRIs, which may be related to SSRI withdrawal syndrome [671]. Moreover, PE patients trying to conceive should avoid using these medications because of their detrimental effects on sperm cells [730-733].
6.2.6.b.3. Topical anaesthetic agents
The use of local anaesthetics to delay ejaculation is the oldest form of pharmacological therapy for PE [734]. Several trials [594,735,736] support the hypothesis that topical desensitising agents reduce the sensitivity of the glans penis, thereby delaying ejaculatory latency, but without adversely affecting the sensation of ejaculation. Meta-analyses have confirmed the efficacy and safety of these agents for the treatment of PE [737]. In a meta-analysis, the efficacy of local anaesthetics was best among the other treatment options including SSRIs, dapoxetine 30 and 60mg, PDE5Is and tramadol for < 8 weeks of therapy [737].
6.2.6.b.3.a. Lidocaine/prilocaine cream
Lidocaine/prilocaine creams can significantly increase the stopwatch-measured IELT from 1-2 minutes to 6-9 minutes [738,739]. Although no significant TEAEs have been reported, topical anaesthetics are contraindicated in patients or partners with an allergy to any ingredient in the product. These anaesthetic creams/gels may be transferred to the partner, resulting in vaginal numbness. Therefore, patients are advised to use a condom after applying the cream to their penis. Alternatively, the penis can be washed to clean off any residual active compound prior to sexual intercourse. Since these chemicals may be associated with cytotoxic effects on fresh human sperm cells, couples seeking parenthood should not use topical lidocaine/prilocaine-containing substances [740].
6.2.6.b.3.b. Lidocaine/prilocaine spray
The eutectic lidocaine/prilocaine spray is a metered-dose aerosol spray containing purely base forms of lidocaine (150mg/mL) and prilocaine (50mg/mL) that has been approved by the EMA for treatment of lifelong PE [741]. Compared to topical creams, the metered-dose spray delivery system has been proven to deposit the drug in a dose-controlled, concentrated film covering the glans penis, maximising neural blockage and minimising the onset of numbness [742], without absorption through the penile shaft skin [743].
Several studies have demonstrated the efficacy of lidocaine/prilocaine spray in improving both IELT and PROMs with three sprays administered five minutes before sexual intercourse [744,745]. Published data showed that lidocaine/prilocaine spray increases IELT over time up to 6.3-fold over three months, with a month-by-month improvement during the course of the treatment in long-term studies [746]. A low incidence of local TEAEs in both patients and partners has been reported, including genital hypoaesthesia (4.5% and 1.0% in men and female partners, respectively) ED (4.4%), and vulvovaginal burning sensation (3.9%), but is unlikely to be associated with systemic TEAEs [747,748]. Lidocaine-only sprays are also available and effective in the treatment of PE [749,750].
6.2.6.b.4. Tramadol
Tramadol is a centrally acting analgesic agent that combines opioid receptor activation and serotonin and noradrenaline re-uptake inhibition. Tramadol is a mild-opioid receptor agonist, but it also displays antagonistic properties on transporters of noradrenaline and 5-HT [751]. This mechanism of action distinguishes tramadol from other opioids, including morphine. Tramadol is readily absorbed after oral administration and has an elimination half-life of five to seven hours.
Several clinical trials evaluated the efficacy and safety of tramadol ODT (62 and 89mg) and tramadol HCI in the treatment of PE [752]. Up to 2.5-fold increases in the median IELT have been reported among patients who received on demand tramadol treatment [753,754].
Adverse effects were reported at doses used for analgesic purposes (≤ 400mg daily) and included constipation, sedation and dry mouth. In May 2009, the FDA released a warning letter about tramadol’s potential to cause addiction and difficulty in breathing [755]. The tolerability during the 12-week study period in men with PE was acceptable [756]. Several other studies have also reported that tramadol exhibits a significant dose-related efficacy along with potential TRAEs in the treatment of PE [753,754].
The Guidelines Panel considers tramadol a possible alternative to established first line therapies in men with PE. Nevertheless, the use of tramadol should be approached with caution due to the limited evidence available on the long term safety of this agent in this specific context.
6.2.6.b.5. Phosphodiesterase type 5 inhibitors
Although IELT was not significantly improved, sildenafil increased confidence, the perception of ejaculatory control, and overall sexual satisfaction, and reduced anxiety and the refractory time to achieve a second erection after ejaculation [757,758]. Several open-label studies have shown that a combination of PDE5Is and SSRIs is superior to SSRI monotherapy, which has also been confirmed by a Bayesian network meta-analysis [737,759].
6.2.6.b.6. Other drugs
In addition to the aforementioned drugs, there is continuous research into other treatment options. Considering the abundant α1a-adrenergic receptors in seminal vesicles and the prostate and the role of the sympathetic system in ejaculation physiology, the efficacy of selective α-blockers in the treatment of PE has been assessed [760-762]. A study demonstrated that wake-promoting agent modafinil may be effective in delaying ejaculation and improving PROMs [763]. Decreasing penile sensitivity with glans penis augmentation using hyaluronic acid (HA) for the treatment of PE was initially proposed by Korean researchers in 2004 [764]. Since then, it has gained popularity mainly in Asian countries [765,766]. Randomised controlled studies demonstrated that HA glans injections were safe, with a modest but significant increase in IELT, along with improvements in PROMs [765-769]. No serious TEAEs were reported. However, this procedure may result in serious complications and more safety studies must be conducted before recommending this treatment to PE patients [770]. Selective dorsal neurectomy has also been suggested for the treatment of PE, mainly by Asian researchers [771-777]. However, considering the irreversible nature of these procedures, more safety data are warranted.
Considering the importance of central oxytocin receptors in the ejaculation reflex, several researchers have assessed the efficacy and safety of oxytocin receptor antagonists in the treatment of PE [778]. Epelsiban [779] and cligosiban [780-783] have been found to be safe and mildly effective in delaying ejaculation, but further controlled trials are needed [782,783]. Delayed ejaculation was associated with the use of pregabalin, a new generation of gapapentinoid, as a side effect. On-demand oral pregabalin 150mg was found to increase the IELTs of patients 2.45 ± 1.43-fold. Treatment-emergent side effects (blurred vision, dizziness, vomiting) were minimal and did not lead to drug discontinuation [784].
The role of other proposed treatment modalities for the treatment of PE, such as penis-root masturbation [785], vibrator-assisted start-stop exercises [692], transcutaneous functional electric stimulation [786,787], transcutaneous posterior tibial nerve stimulation [788,789], bulbospongiosus botulinum-toxin A injection [790,791], acupuncture [792-794], and practicing yoga [795] need more evidence to be considered in the clinical setting.
6.2.7. Summary of evidence and recommendations for the treatment of PE
| Summary of evidence | LE |
| Pharmacotherapy includes either dapoxetine on-demand (an oral short-acting SSRI) and eutectic lidocaine/prilocaine spray (a topical desensitising agent), which are the only approved treatments for PE, or other off-label antidepressants (daily/on-demand SSRIs and clomipramine). | 1a |
| Both on-demand dapoxetine treatment and daily SSRI treatment improve IELT values significantly. | 1a |
| Both on-demand dapoxetine treatment and daily SSRI treatment have generally tolerable side effects when used for treatment of PE. | 1a |
| Daily/on-demand clomipramine treatments improve IELT values significantly and have generally tolerable side effects when used for treatment of PE. | 1a |
| Cream and spray forms of lidocaine/prilocaine improve IELT values significantly and have a safe profile. | 1b |
| Tramadol is effective in the treatment of PE, but the evidence is still inadequate for its long-term safety profile including addiction potential. | 1a |
| Combination of PDE5Is and SSRIs overtakes SSRI monotherapy in effectiveness. | 1a |
| Hyaluronic acid injections are effective in decreasing penile sensitivity. | 2b |
| Recommendations | Strength rating |
| Treat erectile dysfunction (ED), other sexual dysfunction or genitourinary infection (e.g. prostatitis) first. | Strong |
| Use either dapoxetine or a lidocaine/prilocaine spray as first-line treatments for lifelong premature ejaculation (PE). | Strong |
| Use off-label oral treatment with daily selective serotonin re-uptake inhibitor (SSRIs) or daily/on-demand clomipramine as a viable alternative for second-line treatments. | Strong |
| Use off-label tramadol with caution as a viable on-demand third-line treatment alternative to on-demand/daily antidepressants (SSRIs or clomipramine). | Strong |
| Use phosphodiesterase type 5 inhibitors alone or in combination with other therapies in patients with PE (without ED). | Strong |
| Use psychological/behavioural therapies in combination with pharmacological treatment in the management of acquired PE. | Weak |
| Use hyaluronic acid injection with caution as a treatment option for PE compared to other more established treatment modalities. | Weak |
| Do not perform dorsal neurectomy as more safety data are warranted. | Weak |
6.3. Delayed ejaculation
6.3.1. Definition and classification
The American Psychiatric Association (APA) defines DE as requiring one of two symptoms: marked delay, infrequency or absence of ejaculation on 75-100% of occasions that persists for at least six months and causes personal distress [220]. A multinational study in 2,679 men with and without self-reported DE during partnered sex confirmed significant differences in EF, percentage of episodes ending in ejaculation, pleasure/satisfaction and bother/distress. All parameters intensified in men with DE during partnered sex. Anxiety/depression was reported significantly more in the DE group. Bisexual orientation showed a higher percentage of DE, compared to hetero- or homosexual groups [796].
Similar to PE, there are distinctions among lifelong, acquired and situational DE [220]. Males with self-reported lifelong DE are younger, report greater DE symptomatology, less likely to have a medical issue or medication that can cause DE, and are more likely to masturbate for anxiety/distress reduction than for pleasure as compared with males with acquired DE [797]. Although the evidence is limited, the prevalence of lifelong and acquired DE is estimated at around 1% and 4%, respectively [221].
6.3.2. Pathophysiology and risk factors
The aetiology of DE can be psychological, organic (e.g. incomplete spinal cord lesion or iatrogenic penile nerve damage), or pharmacological (e.g. SSRIs, antihypertensive drugs or antipsychotics) [798,799] (Table 6.1). Other factors that may play a role in the aetiology of DE include tactile sensitivity and tissue atrophy [691]. A population analysis using a USA claims database reported a DE prevalence and incidence of respectively 0.02% and 0.01% in 2019, both of which increased over time. In the year before a new DE diagnosis, men were prescribed SSRIs (12%), tamsulosin (7.2%) and finasteride (2.3%). Associated comorbidities were ED (22.3%), anxiety disorders (18.1%) and depression (16.9%) [800].
Although low testosterone level has been considered a risk factor in the past [632,801], more contemporary studies have not confirmed any association between ejaculation times and serum testosterone levels [802,803]. Atypical masturbation technique, masturbation and lack of desire for stimuli are also proposed risk factors for DE [804-806].
Table 6.1: Aetiological causes of delayed ejaculation and anejaculation [796, 807-809]
| Ageing men | Degeneration of penile afferent nerves inhibited ejaculation |
| Congenital | Mullerian duct cyst Wolfian duct abnormalities Prune belly syndrome Imperforate anus Genetic abnormalities |
| Anatomic causes | Transurethral resection of prostate Bladder neck incision Circumcision Ejaculatory duct obstruction (can be congenital or acquired) |
| Neurogenic causes | Diabetic autonomic neuropathy Multiple sclerosis Spinal cord injury Radical prostatectomy Proctocolectomy Bilateral sympathectomy Abdominal aortic aneurysmectomy Para-aortic lymphadenectomy |
| Infective/Inflammation | Urethritis Genitourinary tuberculosis Schistosomiasis Prostatitis Orchitis |
| Endocrine | Hypogonadism Hypothyroidism Prolactin disorders Disorders of lipid metabolism |
| Medication | Antihypertensives; thiazide diuretics Alpha-adrenergic blockers Antipsychotics and antidepressants Alcohol Antiandrogens Ganglion blockers |
| Psychological | Anxiety Psychoses Acute psychological distress Relationship distress Psychosexual skill deficit Disconnect between arousal and sexual situations Masturbation style |
6.3.3. Investigation and treatment
Patients should have a full medical and sexual history performed along with a detailed physical examination when evaluating for DE. Understanding the details of the ejaculatory response, sensation, frequency, and sexual activity/techniques; cultural context and history of the disorder; quality of the sexual response cycle (desire, arousal, ejaculation, orgasm, and refractory period); partner’s assessment of the disorder and if the partner suffers from any sexual dysfunction themselves; and the overall satisfaction of the sexual relationship are all important to garner during history-taking [660].
6.3.3.a. Psychological aspects and intervention
There is scarce literature on the psychological aspects relating to DE, as well as on empirical evidence regarding psychological treatment efficacy. Studies on psychological aspects have revealed that men with DE show a strong need to control their sexual experiences. Delayed ejaculation is associated with difficulties surrendering to sexual pleasure during sex - i.e. the sense of letting go [810] - which denotes an underlying psychological mechanism influencing the reaching of orgasm [811]. As for psychological treatments, these may include, but are not limited to: increased genital-specific stimulation; sexual education; role-playing on their own and in front of their partner; retraining masturbatory practices; anxiety reduction on ejaculation and performance; and re-calibrating the mismatch of sexual fantasies with arousal (such as with pornography use and fantasy stimulation compared to reality). Masturbation techniques that are either solo or partnered can be considered practice for the 'real performance' which can eventually result in greater psychosexual arousal and orgasm for both parties [806]. Although masturbation with fantasy can be harmful when not associated with appropriate sexual arousal and context, fantasy can be supportive if it allows blockage of critical thoughts that may prevent orgasm and ejaculation. Techniques geared towards reducing of anxiety are important skills that can help overcome performance anxiety, as anxiety can often interrupt the natural EF through orgasmic progression. Referral to a sexual therapist, psychologist or psychiatrist is appropriate and often warranted. A 2024 RCT concluded that sexual therapy and vacuum negative-pressure massage was more effective than sexual therapy or instruction alone in increasing pleasure and achieving intravaginal ejaculation [812].
6.3.3.b. Pharmacotherapy
Several pharmacological agents, including cabergoline, bupropion, alpha-1-adrenergic agonists (pseudoephedrine, midodrine, imipramine and ephedrine), buspirone, oxytocin, testosterone, bethanechol, yohimbine, amantadine, cyproheptadine and apomorphine have been used to treat DE with varied success [691]. Unfortunately, there is no FDA- or EMA-approved medications to treat DE, as most of the cited research is based on case-cohort studies that were not randomised, blinded or placebo-controlled. Many drugs have been used as primary treatments and/or antidotes to other medications that can cause DE. A survey of sexual health providers demonstrated an overall treatment success of 40%, with most providers commonly using cabergoline, bupropion or oxytocin [813]. However, this survey measured the anecdotal results of practitioners. There was no proven efficacy or superiority of any drug due to a lack of placebo-controlled, randomised, blinded, comparative trials [807]. In addition to pharmacotherapy, penile vibratory stimulation (PVS) is also used as an adjunct therapy for DE [814]. Combination therapy of midodrine and PVS increased autonomic stimulation in 158 men with spinal cord injury led to ejaculation in almost 65% of the patients [815].
| Summary of evidence | LE |
| Delayed ejaculation can be caused by several aetiologies, including congenital, anatomic, neurogenic, infective, hormonal, drug-related and psychological. | 3 |
| There is not enough evidence to support a definitive treatment for DE. | 3 |
6.4. Anejaculation
6.4.1. Definition and classification
Anejaculation involves the complete absence of antegrade or retrograde ejaculation. It is caused by the failure of semen emission from the seminal vesicles, prostate and ejaculatory ducts into the urethra [816]. True anejaculation is usually associated with a normal orgasmic sensation and is always associated with central or peripheral nervous system dysfunction or with drugs [817].
6.4.2. Pathophysiology and risk factors
Generally, anejaculation shares similar aetiological factors with DE and RE (Table 6.1).
6.4.3. Investigation and treatment
Drug treatment for anejaculation caused by lymphadenectomy (LND) and neuropathy, or psychosexual therapy for anorgasmia, is not effective. In all these cases, and in men who have a spinal cord injury, PVS (i.e. application of a vibrator to the penis) is the first-line therapy. In anejaculation, PVS evokes the ejaculation reflex [818], which requires an intact lumbosacral spinal cord segment. If the quality of semen is poor or ejaculation is retrograde, the couple may enter an in vitro fertilisation (IVF) program whenever fathering is desired. If PVS has failed, electro-ejaculation can be the therapy of choice [819]. Other sperm-retrieval techniques may be used when electro-ejaculation fails or cannot be carried out [820]. Anejaculation following either retroperitoneal surgery for testicular cancer or total mesorectal excision can be prevented using unilateral LND or autonomic nerve preservation [821], respectively.
6.5. Painful ejaculation
6.5.1. Definition and classification
Painful ejaculation is a condition in which a patient feels mild discomfort to severe pain during or after ejaculation. The pain can involve the penis, scrotum and perineum [822].
6.5.2. Pathophysiology and risk factors
Many medical conditions can result in painful ejaculation, but it can also be an idiopathic problem. Initial reports demonstrated possible associations of painful ejaculation with calculi in the seminal vesicles [823], sexual neurasthenia [824], sexually transmitted diseases (STIs) [822,825], inflammation of the prostate [241,826], PCa [827,828], BPH [239], prostate surgery [829,830], pelvic radiation [831], herniorrhaphy [832], and antidepressants [833-835]. Further case reports have suggested that mercury toxicity or ciguatera toxin fish poisoning may result in painful ejaculation [836,837]. Psychological issues may also be the cause of painful ejaculation, especially if the patient does not experience this problem during masturbation [838].
6.5.3. Investigation and treatment
Treating painful ejaculation must be tailored to the underlying cause, if detected. Psychotherapy or relationship counselling, withdrawal of suspected agents (drugs, toxins or radiation) [833,834,839] or the prescription of appropriate medical treatment (antibiotics, α-blockers or anti-inflammatory agents) may ameliorate painful ejaculation. Behavioural therapy, muscle relaxants, antidepressant treatment, anticonvulsant drugs and/or opioids, and pelvic floor exercises may be implemented if no underlying cause can be identified [840,841].
6.5.3.a. Surgical intervention
If medical treatments fail, surgical operations such as TURP, transurethral resection of the ejaculatory duct (TURED), and neurolysis of the pudendal nerve have been suggested [842,843]. However, there is no strong supporting evidence that surgical therapy improves painful ejaculation and it must be used with caution.
6.6. Retrograde ejaculation
6.6.1. Definition and classification
Retrograde ejaculation is the total, or sometimes partial, absence of antegrade ejaculation, due to semen passing backwards through the bladder neck into the bladder. Patients may experience a normal or decreased orgasmic sensation. The causes of RE can be divided into neurogenic, pharmacological, urethral or bladder neck incompetence [822].
6.6.2. Pathophysiology and risk factors
The process of ejaculation requires complex co-ordination and interplay between the epididymis, vas deferens, prostate, seminal vesicles, bladder neck and bulbourethral glands [844]. Upon ejaculation, sperm are rapidly conveyed along the vas deferens and into the urethra via the ejaculatory ducts. From there, the semen progresses in an antegrade fashion, partly maintained by coaptation of the bladder neck and rhythmic contractions of the periurethral muscles, co-ordinated by a centrally mediated reflex [844]. Closure of the bladder neck and seminal emission is initiated via the sympathetic nervous system from the lumbar sympathetic ganglia and subsequently hypogastric nerve. Prostatic and seminal vesicle secretion, as well as contraction of the bulbocavernosal, ischiocavernosal and pelvic floor muscles are initiated by the S 2-4 parasympathetic nervous system via the pelvic nerve [844].
Any factor that disrupts this reflex and inhibits contraction of the bladder neck (internal vesical sphincter) may lead to retrograde passage of semen into the bladder. These can be broadly categorised as neurogenic, anatomic (urethral/bladder neck), pharmacological and endocrinal causes of RE (Table 6.2).
Table 6.2: Aetiology of retrograde ejaculation [269, 822, 845]
| Aetiology of retrograde ejaculation | |
| Neurogenic | Spinal cord injury Cauda equina lesions Multiple sclerosis Autonomic neuropathy Retroperitoneal lymphadenectomy Sympathectomy or aortoiliac surgery Anterior lumbar surgery Prostate, colorectal and anal surgery Parkinson´s disease Diabetes mellitus Psychological/behavioural |
| Anatomic | |
| Urethral | Ectopic ureterocele Urethral stricture Urethral valves or verumontaneum hyperplasia Congenital dopamine β-hydroxylase deficiency |
| Bladder neck incompetence | Congenital defects/dysfunction of hemitrigone Bladder neck resection (transurethral resection of the prostate) Prostatectomy |
| Pharmacological | Antihypertensives, thiazide diuretics α-1-Adrenoceptor antagonists Antipsychotics and antidepressants |
| Endocrine | Hypothyroidism Hypogonadism Hyperprolactinaemia |
6.6.3. Disease management
6.6.3.a. Pharmacological
Sympathomimetics stimulate the release of noradrenaline and activate α- and β-adrenergic receptors, resulting in closure of the internal urethral sphincter, restoring the antegrade flow of semen. The most common sympathomimetics are synephrine, pseudoephedrine hydrochloride, ephedrine, phenylpropanolamine and midodrine [846]. As time progresses, their effect diminishes [847]. Many studies published about the efficacy of sympathomimetics in the treatment of RE suffer from small sample size, with some represented by case reports.
An RCT randomised patients to receive one of four α-adrenergic agents (dextroamphetamine, ephedrine, phenylpropanolamine or pseudoephedrine) with or without histamine. The patients suffered from failure of ejaculation following retroperitoneal LND. They found that four days of treatment prior to ejaculation was the most effective and that all the adrenergic agonists restored antegrade ejaculation [846]. In a systematic review, the efficacy of this group of medications was found to be 28% [224]. The adverse effects of sympathomimetics include dryness of mucous membranes and hypertension.
Antimuscarinic therapy has been reported as a treatment option, either alone or in combination with sympathomimetics. Agents described in the literature include brompheniramine maleate and imipramine. The reported efficacy of antimuscarinics as monotherapy is approximately 22%, increasing to 39% when used in combination with sympathomimetics [224]. Although combination therapy appears to offer greater effectiveness, the limited sample sizes currently available preclude robust statistical analysis.
6.6.3.b. Management of infertility
Infertility has been the major concern of patients with RE. Beyond standard sperm-retrieval techniques, such as testicular sperm aspiration/extraction (TESA/TESE), three different methods of sperm acquisition have been identified for managing infertility in patients with RE. These include: i) centrifugation and resuspension of post-ejaculatory urine specimens; ii) Hotchkiss (or modified Hotchkiss) technique; and iii) ejaculation on a full bladder.
1. Centrifugation and resuspension. To improve the ambient conditions for the sperm, the patient is asked to increase their fluid intake or take sodium bicarbonate to dilute or alkalise the urine, respectively. Afterwards, a post-orgasmic urine sample is collected by introducing a catheter or spontaneous voiding. This sample is then centrifuged and suspended in a medium. The types of suspension fluids are heterogeneous and can include bovine serum albumin, human serum albumin, Earle’s/Hank’s balanced salt solution and the patient’s urine. The resultant modified sperm mixture can then be used in assisted reproductive techniques. A systematic review of studies in couples in which the male partner had RE found a 15% pregnancy rate per cycle (0-100%) [224].
2. Hotchkiss method. The Hotchkiss method involves emptying the bladder prior to ejaculation, using a catheter, and then washing out and instilling a small quantity of Lactated Ringers to improve the ambient condition of the bladder. The patient then ejaculates and semen is retrieved by catheterisation or voiding [848]. Modified Hotchkiss methods involve variance in the instillation medium. Pregnancy rates were 24% per cycle (0-100%) [224].
3. Ejaculation on a full bladder. The patient is encouraged to ejaculate on a full bladder and semen is suspended in Baker’s Buffer. The pregnancy rate in two studies, which included only five patients, have described results using this technique [849,850].
6.7. Anorgasmia
6.7.1. Definition and classification
Anorgasmia is the perceived absence of orgasm and can give rise to anejaculation. Regardless of the presence of ejaculation, anorgasmia can be a lifelong (primary) or acquired (secondary) disorder [221].
6.7.2. Pathophysiology and risk factors
Primary anorgasmia starts from a man’s first sexual intercourse and lasts throughout his life, while secondary anorgasmia patients should have a normal period before the problem starts [851]. Substance abuse, obesity and some non-specific psychological aspects, such as anxiety and fear, are considered risk factors for anorgasmia. Only a few studies have described anorgasmia alone and generally, it has been considered a symptom linked to ejaculatory disorders, especially with DE, and therefore, they are believed to share the same risk factors. However, psychological factors are considered responsible for 90% of anorgasmia problems [852]. The causes of delayed orgasm and anorgasmia are shown in Table 6.3 [851].
Table 6.3: Causes of delayed orgasm and anorgasmia
| Causes of delayed orgasm and anorgasmia | |
| Endocrine: | Testosterone deficiency Hypothyroidism |
| Medications: | Antidepressants Antipsychotics Opioids |
| Psychosexual causes | |
| Hyperstimulation | |
| Penile sensation loss | |
6.7.3. Disease management
The psychological/behavioural strategies for anorgasmia are similar to those for DE. The patient and their partner should be examined physically and psychosexually in detail, including determining the onset of anorgasmia, medication and disease history, penile sensitivity and psychological issues. Adjunctive laboratory tests can also be used to rule out organic causes, such as testosterone, prolactin and TSH levels. Patients who have loss of penile sensitivity require further investigations [851].
6.7.3.a. Psychological/behavioural strategies
Lifestyle modifications may be advised and include changes in masturbation habits, measures aimed at improving intimacy, and reduction of alcohol consumption. Several psychotherapeutic interventions, either alone or in combination, have been described, including modification of arousal techniques, reduction of sexual anxiety, role-playing of exaggerated orgasm, and increased genital stimulation [811,853]. Due to heterogeneity of study design, interventions and outcomes, reliable success rates cannot be determined from the current literature. Repetitive transcranial magnetic stimulation (rTMS) is a non-invasive neuromodulation technique widely used in the management of various psychiatric and neurological conditions. Its potential role in the treatment of anejaculation has been evaluated in an RCT. Both high-frequency and low-frequency rTMS were associated with significantly higher rates of intravaginal ejaculation at eight weeks compared with sham stimulation (4 [12.9%], 17 [54.8%], and 0 patients, respectively) [854]. The available evidence is preliminary, originating from a single-centre study with a limited sample size. Further adequately powered, multicentre RCTs are required before rTMS can be recommended in routine clinical practice.
6.7.3.b. Pharmacotherapy
Several drugs have been reported to reverse anorgasmia, including cyproheptadine, yohimbine, buspirone, amantadine and oxytocin [855-860]. These reports are generally from case-cohort studies and drugs have limited efficacy and significant adverse effect profiles. Current evidence is not strong enough to recommend drugs to treat anorgasmia.
6.7.3.c. Management of infertility
If patients fail the treatment methods mentioned above, penile vibratory stimulation, electro-ejaculation or TESE are options for sperm retrieval in anorgasmia cases [851].
6.8. Haemospermia
6.8.1. Definition and classification
Haemospermia is defined as the appearance of blood in the ejaculate. Although it is often regarded as a symptom of minor significance, blood in the ejaculate causes anxiety in many men and may indicate underlying pathology [244,861].
6.8.2. Pathophysiology and risk factors
Several causes of haemospermia have been acknowledged and can be classified into the following subcategories: idiopathic, congenital malformations, inflammatory conditions, obstruction, malignancies, vascular abnormalities, iatrogenic/trauma and systemic causes (Table 6.4) [862].
Table 6.4: Pathology associated with haemospermia [862-867]
| Category | Causes |
| Congenital | Seminal vesicle (SV) or ejaculatory duct cysts |
| Inflammatory | Urethritis, prostatitis, epididymitis, tuberculosis, cytomegalovirus, HIV, schistosomiasis, hydatid, condyloma of urethra and meatus, urinary tract infections |
| Obstruction | Prostatic, SV and ejaculatory duct calculi, post-inflammatory, seminal vesicle diverticula/cyst, urethral stricture, utricle cyst, BPH |
| Tumours | Prostate, bladder, SV, urethra, testis, epididymis, melanoma |
| Vascular | Prostatic varices, prostatic telangiectasia, haemangioma, posterior urethral veins, excessive sex or masturbation |
| Trauma/iatrogenic | Perineum, testis, instrumentation, post-haemorrhoid injection, prostate biopsy, vaso-venous fistula, stereotactic radio-therapy |
| Systemic | Hypertension, haemophilia, purpura, scurvy, bleeding disorders, chronic liver disease, renovascular disease, leukaemia, lymphoma, cirrhosis, amyloidosis |
| Idiopathic | - |
The risk of any malignancy in patients presenting with haemospermia is approximately 3.5% (0-13.1%) [864,868,869]. In a study in which 342 patients with haemospermia were included, the most relevant aetiology for haemospermia was inflammation/infection (49.4%), while genitourinary cancers (i.e. prostate and testis) only accounted for 3.2% of the cases [870].
6.8.3. Investigations
As with other clinical conditions, a systematic clinical history and assessment is undertaken to help identify the cause of haemospermia. Although the differential diagnosis is extensive, most cases are caused by infections or other inflammatory processes [244].
The basic examination of haemospermia should start with a thorough symptom-specific and systemic clinical history. The first step is to understand if the patient has true haemospermia. Pseudo-haemospermia may occur because of haematuria or even suction of a partner’s blood into the urethra during copulation [822,871,872]. A sexual history should be taken to identify those whose haemospermia may be a consequence of a STI. Recent foreign travel to areas affected by schistosomiasis or tuberculosis should also be considered. The possibility of co-existing systemic diseases such as hypertension, liver disease and coagulopathy should be investigated along with systemic features of malignancy such as weight loss, loss of appetite or bone pain. Examination of the patient should include measurement of blood pressure, as there have been several case reports suggesting an association between uncontrolled hypertension and haemospermia [873,874].
Most authors who propose an investigative baseline agree on the initial diagnostic tests, but there is no consensus in this regard [862,863,868,871]. Urinalysis should be performed, and urine sent for microscopy, culture, and sensitivity testing. If tuberculosis or schistosomiasis is the suspected cause, the semen or prostatic secretions should be sent for analysis. A full sexually-transmitted disease screen, including first-void urine as well as serum and genitourinary samples, should be tested for chlamydia, ureaplasma and herpes simplex virus. Using this strategy, it may be possible to find an infectious agent in cases that would have been labelled as idiopathic haemospermia [875].
Serum PSA should be taken in men aged > 40 years who have been appropriately counselled [245,869]. Blood work, including a full blood count, liver function tests and a clotting screen should be taken to identify systemic diseases. The question of whether further investigation is warranted depends on clinician judgment, patient age and an assessment of risk factors [862]. Digital rectal examination should also be performed, and the meatus re-examined after DRE for bloody discharge [876]. Detection of a palpable nodule in the prostate is important because an association between haemospermia and PCa has been postulated, although not completely proven.
Magnetic resonance imaging is increasingly used as a definitive means to investigate haemospermia. The multiplanar ability of MRI to accurately represent structural changes in the prostate, seminal vesicles, ampulla of vas deferens, and ejaculatory ducts has enabled the technique to be particularly useful in determining the origin of midline or paramedian prostatic cysts and in determining optimal surgical management [877]. The addition of an endorectal coil can improve diagnostic accuracy for identifying the site and possible causes of
haemorrhage [878].
Cystoscopy has been included in most suggested investigative protocols in patients with high-risk features (refractory to conservative treatment with persistent haemospermia). It can provide valuable information, as it allows direct visualisation of the main structures in the urinary tract that can be attributed to causes of haemospermia, such as polyps, urethritis, prostatic cysts, foreign bodies, calcifications and vascular abnormalities [879,880].
With the advancement of optics, the ability to create ureteroscopes of diameters small enough to allow insertion into the ejaculatory duct and seminal vesicles has been made possible [881]. In a prospective study, 106 patients with prolonged haemospermia underwent transrectal US (TRUS) and seminal vesiculoscopy. With both methods combined, the diagnosis was made in 87.7% of patients. When compared head-to-head, the diagnostic yield for TRUS versus seminal vesiculoscopy was 45.3% and 74.5%, respectively (P < 0.001) [882]. Transutricular seminal vesiculoscopy, performed under real-time TRUS guidance, has been proposed as a diagnostic and therapeutic approach. In a cohort study of 281 patients with persistent haematospermia, transutricular seminal vesiculoscopy was successfully performed in 96.8% of cases and resulted in symptom alleviation or resolution in 244 patients (89.7%) [883]. Transutricular seminal vesiculoscopy appears to be a safe and minimally invasive procedure and can be performed as a day case surgery under caudal block anaesthesia [884].
Melanospermia is a consequence of malignant melanoma involving the genitourinary tract and is a rare condition that has been described in two case reports [885,886]. Chromatography of the semen sample can be used to distinguish the two by identifying the presence of melanin if needed.
6.8.4. Disease management
Conservative management is generally the primary treatment option when the patients are aged < 40 years and have a single episode of haemospermia. The primary goal of treatment is to exclude malignant conditions like PCa and bladder cancer and treat any other underlying cause. If no pathology is found, the patient can be reassured [244,862].
Middle-aged patients with recurrent haemospermia warrant more aggressive intervention. Appropriate antibiotic therapy should be prescribed to patients who have urogenital infections or STIs. Urethral or prostate varices or angiodysplastic vessels can be fulgurated, whereas cysts, either of the seminal vesicles or prostatic urethra, can be aspirated transrectally [244]. Ejaculatory duct obstruction is managed by transurethral incision at the duct opening [887,888]. Systemic conditions should be treated appropriately [868,872,889,890].
Defining a management algorithm for haemospermia is based on the patient age and degree of haemospermia. Patients often find blood in the ejaculate alarming, and investigations should be aimed at excluding a serious, despite infrequent, underlying cause (e.g. cancer), while at the same time preventing over-investigation and alleviating patient anxiety. The literature describes a multitude of causes for haemospermia, although many of these are not commonly found after investigation. Men may be stratified into higher-risk groups according to several factors including age > 40 years, recurrent or persistent haemospermia, the actual risk for PCa (e.g. positive family history), and concurrent haematuria. Based upon the literature, a management algorithm is proposed (Figure 6.3) [868,872,889,890].
Figure 6.3: Management of haemospermia [868, 872, 889, 890]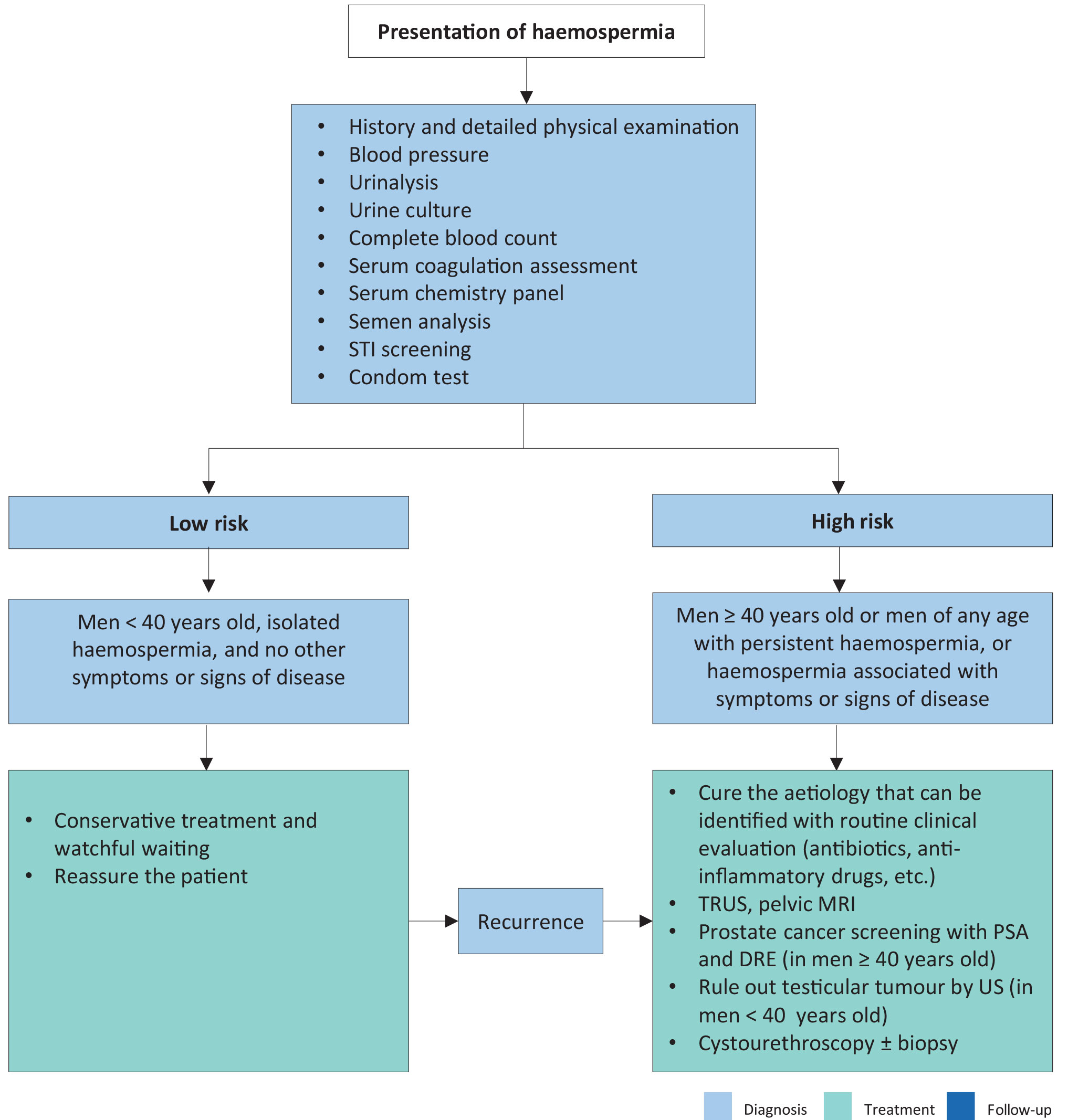 DRE = digital rectal examination; MRI = magnetic resonance imaging; PSA = prostate-specific antigen; STI = sexually transmitted infections; TRUS = transrectal ultrasonography; US = ultrasonography.
DRE = digital rectal examination; MRI = magnetic resonance imaging; PSA = prostate-specific antigen; STI = sexually transmitted infections; TRUS = transrectal ultrasonography; US = ultrasonography.
6.8.5. Summary of evidence and recommendations for the investigation and management of haemospermia
| Summary of evidence | LE |
| While haemospermia has traditionally been attributed to benign causes, it is a potential indicator warranting thorough diagnostic evaluation and, if necessary, targeted treatment. | 3 |
| The principal objective of treatment is to rule out malignancies while addressing any other underlying causes as well. | 3 |
| Recommendations | Strength rating |
| Perform a full medical and sexual history with detailed physical examination. | Strong |
| Use risk-stratification system to manage the disease systematically. | Weak |