3. MALE HYPOGONADISM
3.1. Definition, epidemiology and classification of male hypogonadism
3.1.1. Definition
Male hypogonadism is a clinical syndrome which comprises of symptoms with or without signs and biochemical evidence of testosterone deficiency. Hypogonadism is associated with decreased testicular function and production of androgens and/or impaired sperm production [3]. This may be caused by impaired testicular function (hypergonadotropic hypogonadism or primary hypogonadism) or because of inadequate stimulation of the testes by the hypothalamic-pituitary axis (hypogonadotropic hypogonadism or secondary hypogonadism) (Table 3.1) or uncommonly by reduced ability of testosterone to stimulate the androgen receptor at the cellular level. Hypogonadism can adversely affect multiple organ functions and quality of life (QoL) [3,4]. This chapter specifically addresses the management of adult male hypogonadism also called late-onset hypogonadism (LOH). Some insights related to congenital or pre-pubertal hypogonadism are also provided.
3.1.2. Epidemiology
The prevalence of LOH increases with age, with the major causes being obesity, other comorbidities (e.g. diabetes) and overall poor health [5]. The incidence of hypogonadism has been reported to be between 12.3 and 11.7 cases per 1,000 people per year [6,7]. Ageing accounts for a low percentage of hypogonadism, as there is only a small gradual decline in testosterone, up to the age of 80 years, in healthy aging men [5]. In men aged 40-79 years, the incidence of symptomatic hypogonadism varies between 2.1 and 5.7% [6,8,9].
There is a high prevalence of LOH within specific populations, including patients with obesity, type 2 diabetes (T2DM), metabolic syndrome (MetS), cardiovascular diseases (CVD), chronic obstructive pulmonary disease (COPD), renal disease and cancer [9]. In particular, low testosterone levels are relatively common in men with T2DM [10,11] and in those with metabolic derangements.
Klinefelter syndrome, a trisomy associated with a 47,XXY karyotype, is the most prevalent genetic cause of primary hypogonadism, with a global prevalence of 1/500-1,000 live male births [12-15]. However, < 50% of individuals with Klinefelter syndrome are diagnosed during their lifetime [16].
3.1.3. Classification
Male hypogonadism can be classified according to the aetiology into primary hypogonadism or secondary hypogonadism (Table 3.1) [3,17]. A compensated or subclinical form of hypogonadism, characterised by normal testosterone serum levels and elevated luteinising hormone (LH) production, has also been reported [18]; the clinical significance of this condition is unclear [18-21].
Hypogonadism is generally classified into two broad categories: ‘Classical/Organic’ and ‘Functional,’ the latter often but incorrectly, referred to as LOH. Regardless of the underlying cause, the clinical manifestations of testosterone deficiency are common to all patients, although their severity and presentation may vary depending on factors such as age at onset.
Classical hypogonadism includes congenital or acquired diseases causing structural and/or irreversible impairment of the pituitary and/or testes. Functional hypogonadism is diagnosed on the absence of any recognised organic alterations in the HPG axis, and it is mainly a consequence of comorbidities, affecting the Hypothalamic-Pituitary-Testicular (HPT) axis and should be treated first by resolving or improving any underlying conditions (e.g. anorexia in younger male subjects). Late-onset hypogonadism represents an even broader clinical entity, including adult-onset forms of testosterone deficiency, which can have an organic or functional origin and can be primary or secondary [22]. Late-onset hypogonadism is often diagnosed when no identifiable classical cause of hypogonadism is present, and its prevalence increases with advancing age. By definition, LOH must comprise both persistent specific symptoms and biochemical evidence of testosterone deficiency [3,23].
Finally, hypogonadism can also result from several conditions leading to reduced sensitivity/insensitivity to testosterone and its metabolites [3].
The current Guidelines maintain a classification of primary and secondary hypogonadism, with special reference to LOH. The classification, based on the aetiology of hypogonadism, allows clinicians to adequately select appropriate treatment. In patients with secondary hypogonadism, both fertility and testosterone normalisation can be theoretically achieved with adequate treatment, whereas in primary hypogonadism only testosterone therapy can be considered, which eventually impairs fertility due to suppression of the HPT axis [3,17] (Table 3.1). It should also be recognised that symptoms and signs of hypogonadism can be similarly independent of the site of origin of the disease. Conversely, the age of onset of hypogonadism can influence the clinical phenotype [24]. Accordingly, for early onset, such as that occurring during foetal life, the clinical phenotype can span from an almost complete female phenotype (e.g. complete androgen insensitivity or enzymatic defects blocking androgen synthesis) to various defects in virilisation. In the case of a pre- or peripubertal appearance of hypogonadism due to a milder central (isolated hypogonadotropic hypogonadism [IHH]) or a peripheral defect (such as in Klinefelter syndrome), there may be delayed puberty with an overall eunuchoid phenotype. Finally, when hypogonadism develops after puberty, and especially with ageing, symptoms can be mild and often confused the with the ageing process [3,24].
Table 3.1: Classification of male hypogonadism
| Classification of male hypogonadism | |
| PRIMARY HYPOGONADISM (hypergonadotropic hypogonadism) | |
| Congenital or developmental disorders | |
| Common causes | Uncommon causes |
|
|
| Acquired disorders | |
| Drug-induced | Localised problems |
|
|
| Systemic diseases/conditions with hypothalamus/pituitary impact | |
|
|
| SECONDARY HYPOGONADISM (hypogonadotropic hypogonadism) | |
| Congenital or developmental disorders | |
| Common causes | Uncommon causes |
|
|
| Acquired disorders | |
| Drug-induced | Localised problems |
|
|
| Systemic diseases/conditions impacting the hypothalamus/pituitary | |
|
|
| ANDROGEN RESISTANCE/DECREASED TESTOSTERONE BIOACTIVITY | |
| Congenital or developmental disorders | |
| |
| Acquired disorders | |
| Drug-induced | Localised problems |
|
|
* Conditions acting at central and peripheral levels resulting in either primary or secondary hypogonadism.
1 Different autosomal translocation can cause rare cases of hypogonadism and infertility.
A brief discussion on the physiology of testosterone production can be found in Appendix 1, online supplementary evidence.
3.2. Comorbidities associated with male hypogonadism
3.2.1. Obesity
Low testosterone levels are common in men with obesity. Male hypogonadism is associated with a greater percentage of fat mass and a lower lean mass compared to men with adequate testosterone levels [25-27]. Low testosterone levels are strongly associated with increased visceral adiposity but also lead to lipid deposition in the liver and muscle and is associated with atherosclerosis [25,26].
3.2.2. Metabolic syndrome/type 2 diabetes
Hypogonadism is frequently associated with MetS or its related components, including central obesity, hyperglycaemia, insulin resistance, dyslipidaemia and arterial hypertension [28].
Several randomised controlled trials (RCTs) have demonstrated that testosterone therapy may improve insulin resistance and hyperglycaemia and lower total and low-density protein (LDL)-cholesterol [29-34]. Testosterone therapy in hypogonadal T2DM improved glycaemic control in some RCTs and registry trials; however, there is no conclusive evidence [30,35,36]. A large placebo-controlled RCT, including 1,007 patients with impaired glucose tolerance or newly diagnosed T2DM and total testosterone < 14nmol/L, showed that testosterone therapy for two years reduced the proportion of patients with T2DM regardless of a lifestyle programme [34]. Similarly, a registry study reported that testosterone therapy was associated in time with remission of T2DM [35]. High-density lipoprotein (HDL)-cholesterol may decrease, remain unchanged or increase with testosterone therapy.
Testosterone therapy in men with MetS and low testosterone has been shown to reduce mortality compared to untreated men [37,38], although no conclusive evidence is available.
Erectile dysfunction (ED) is common in men with MetS and T2DM (up to 70% of patients). The causes of ED are multi-factorial and 30% of men with ED have co-existing hypogonadism. Some evidence has suggested that ED is only found in men with T2DM and clearly reduced testosterone levels (< 8nmol/L or 2.31ng/mL) [39]. From a pathophysiological perspective, it has been reported that this is because ED is predominantly caused by vascular and neuropathic disease, and therefore not likely in men who do not have established vascular disease. Therefore, men presenting with ED should be screened for MetS. Likewise, patients with ED and diabetes may be offered testosterone measurement.
Placebo-controlled RCTs of testosterone therapy in T2DM have demonstrated improved sexual desire and satisfaction, but not erectile function (EF) [30,39]. Similar results were derived from a meta-analysis of published trials [40]. Accordingly, a large two-year RCT of testosterone undecanoate versus placebo showed that testosterone therapy significantly improved sexual function and ED in men with impaired glucose tolerance or newly diagnosed T2DM low testosterone (< 14nmol/L) [34].
Testosterone therapy has been associated with a reduced percentage of body fat and increase in lean body mass [41]. Data from a registry study have suggested that testosterone therapy with long-acting intramuscular testosterone undecanoate over a period of 11 years was associated with a substantial but gradual loss of weight, along with a reduction in waist circumference [42].
3.2.3. SARS-CoV-2/COVID-19
Data suggest that low circulating testosterone levels are more frequently associated with worse clinical outcomes in men with COVID-19 [43-50]. A cohort study, analysing two large academic health systems databases, including 723 men with a history of COVID-19, reported that men with hypogonadism had a higher risk of being hospitalised [51]. In addition, a meta-analysis suggested that reduced testosterone levels detected at hospital admission for COVID-19 are associated with a four- to five-fold increased risk of being admitted to the Intensive Care Unit or death, after adjustment for potential confounders [52].
Although no information on the role of testosterone therapy in the acute phase of the disease is currently available, data also showed that patients with hypogonadism under testosterone therapy had a reduced risk to be hospitalised after SARS-CoV-2 infection [51]. However, whether low testosterone directly contributes to worse COVID-19 outcomes is still under investigation. The possibility that low testosterone in the acute phase of COVID-19 infection represents an adaptive response mechanism to dampen non-essential activities not conducive to recovery (physical and sexual activities) by turning off testosterone-dependent functions, cannot be excluded [53,54]. Accordingly, a meta-analysis showed that secondary or mixed hypogonadism is more frequently observed in the acute phase of the infection [52].
Studies evaluating patients in the recovery phase of COVID-19 have documented either restored [55,56] or persistently low testosterone levels in the majority of cases [57]. A longitudinal evaluation study showed that during the recovery phase, a further improvement of testosterone levels can be observed up to 12 months after COVID-19. Male subjects who have recovered from COVID-19 should be accurately followed-up to exclude any long-term andrological consequences, such as impairment in sperm and testosterone production [52].
3.3. Late-onset hypogonadism
Testosterone production declines with ageing. The European Male Aging Study (EMAS) reported a 0.4% per annum (log hormone-age) decrease in total testosterone and a 1.3% per annum decline in free testosterone (fT) [5]. Late-onset hypogonadism is the term frequently used to describe this phenomenon and the detection of hypogonadism in adulthood. Evidence indicates that several associated diseases and chronic comorbidities can interfere with the HPG axis leading to development of primary hypogonadism or, more frequently, secondary hypogonadism in adulthood, thus significantly influencing the physiological age-dependent decline of testosterone. The combined data from three different waves of the Massachusetts Male Aging Study (MMAS)demonstrated that associated comorbidity and obesity significantly decreased, whereas smoking tended to increase, total, free and bio-available testosterone concentrations [58]. Data derived from the EMAS confirmed these findings [5,19]. Based upon these data and other evidence, the concept of functional and organic hypogonadism has been introduced (see above) [59]. The diagnosis of functional hypogonadism is based on the exclusion of a classical (organic) aetiology. The main causes suggested for functional hypogonadism are obesity, comorbidities and ageing, with the first two accounting for most cases. Inflammatory cytokines released in chronic inflammation and adipocytokines and E2 in obesity can suppress the HPG axis [60]. The role of ageing up to age 80 years seems relatively small [59,61]. Considering that suppression of HPG axis activity is functional and potentially reversible by empiric measures, such as weight loss, the need for testosterone therapy has been questioned [59]. The distinction between functional and organic hypogonadism remains a subject of ongoing debate, as any impairment of the HPT axis leading to symptomatic low testosterone levels is associated with a disorder, even in the absence of identifiable “pathological” causes.
3.3.1. Clinical diagnosis and evaluation
The mainstay of LOH diagnosis includes signs and symptoms consistent with hypogonadism (Table 3.2), coupled with biochemical evidence of low morning serum total testosterone levels on two or more occasions, measured with a reliable assay and in fasting conditions [62-64].
3.3.2. History taking
Specific symptoms associated with hypogonadism, including LOH, are shown in Table 3.2. These symptoms are not exclusively specific to hypogonadism and therefore must be interpreted with caution and always considered within the broader clinical and biochemical state. Several self-reported questionnaires or structural interviews have been developed for screening of hypogonadism. Although these case-history tools have demonstrated clinical utility in supporting the biochemical diagnosis of hypogonadism, or in the assessment of testosterone therapy outcomes, their specificity remains poor and they should not be used for systematic screening of men with hypogonadism [65]. Headache and/or visual disturbance may indicate a pituitary related disorder. History of surgical intervention for cryptorchidism or hypospadias must be considered as possible signs of congenital defects. Chronic and systemic comorbidities must be comprehensively investigated in every patient. Use of drugs that potentially interfere with the HPG axis should be excluded (Table 3.1). Acute diseases are associated with development of functional hypogonadism and determination of serum total testosterone levels should be avoided in these conditions; however, the role of testosterone in the case of acute illness remains to be clarified [43,47,52,66]. Fertility should always be discussed.
Table 3.2: Specific symptoms associated with late-onset hypogonadism
| Sexual symptoms | Physical symptoms | Psychological symptoms | |
| More specific |
|
|
|
| Less specific |
|
|
|
3.3.3. Physical examination
Since obesity is frequently associated with hypogonadism (mostly functional), the determination of body mass index (BMI) and the measurement of waist circumference are strongly recommended in all individuals [67]. Testicular and penile size, as well the presence of sexual secondary characteristics can provide useful information regarding overall androgen status. In addition, upper segment/lower segment ratio (n.v. > 0.92) and arm-span to height ratio (n.v. < 1.0) can be useful to identify an eunuchoid body shape, especially in subjects with pre-pubertal hypogonadism or delayed puberty. Finally, digital rectal examination (DRE) should be performed in all subjects to exclude prostate abnormalities before testosterone therapy (any type) or to support suspicion of hypogonadism (in case of reduced volume) [68].
3.3.4. Laboratory diagnostics
Available evidence clearly indicates that both total and calculated fT levels decrease within 30 minutes of oral glucose administration during an oral glucose tolerance test, irrespective of the degree of glucose tolerance [64]. Accordingly, it has been reported that measuring circulating testosterone levels under non-fasting conditions can result in an underestimation of total testosterone by more than 30% of its true value [63]. In addition, considering the circadian rhythm of testosterone secretion - with higher levels observed in the morning, a pattern that persists even in the aging population - 10:00 should be regarded as the latest acceptable time for morning sampling and subsequent testosterone measurement [62]. This limit may not be suitable for night-shift workers, as the diurnal rhythm of testosterone secretion is primarily driven by sleep patterns [69]. In these individuals, sampling should be performed under fasting conditions and after an adequate sleep period. Therefore, in the general male population, serum testosterone should be measured in the morning (between 07:00 and 10:00) and in the fasting state. A confirmatory measurement should always be undertaken in the case of a primary pathological value, and before starting any testosterone therapy.
Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) represents the most accurate method for sex steroid evaluation; however, standardised automated platform immuno-assays for total testosterone assessment demonstrate a good correlation with LC-MS/MS [70]. Available immuno-assays are not able to provide an accurate estimation of fT; therefore, direct fT evaluation with these methods is not recommended and should be avoided [71]. Equilibrium dialysis is the most accurate method for fT measurement [72]. Alternatively, fT can be derived from specific mathematical calculations taking into account serum SHBG and albumin levels [73] (http://www.issam.ch/freetesto.htm). It is clinically relevant to highlight how total testosterone values may change as a function of corresponding circulating SHBG levels. Table 3.3 lists the conditions most commonly associated with changes in circulating SHBG levels.
Although criticisms exist regarding its methodology, an internet-based survey across 27 European countries highlighted significant variability in laboratory practices, revealing widespread reliance on immuno-assays methods for total testosterone, limited LC-MS/MS adoption, and inconsistent pre-analytic protocols [74]. However, available normative data regulating commercial immuno-assays ranges is not sufficiently clear either for the used reference population or the quality of pre-analytic factors [75].
Therefore, close collaboration between clinicians managing men with hypogonadism and local laboratories is strongly advised, reaffirming the need for a rigorous technical measurement methodology to ensure accuracy and comparability across laboratories. These laboratories should adopt Centers for Disease Control and Prevention (CDC)-certified total testosterone thresholds through the Hormone Standardization Program (HoSt) [76]. However, for the diagnosis of male hypogonadism, it is recommended to use a widely recognised total testosterone threshold that is appropriately integrated with each patient’s clinical symptomatic profile. Meta-analyses have demonstrated that testosterone therapy is ineffective when baseline total testosterone levels exceed 12nmol/L (3.5ng/mL). In contrast, positive outcomes have been observed when total testosterone levels are below 12nmol/L, with the greatest benefits seen in symptomatic patients presenting with more severe hypogonadism (< 8nmol/L). Therefore, based on available outcome data, a threshold of 12nmol/L should be considered as a potential criterion for initiating testosterone therapy in the presence of hypogonadal symptoms [41,77].
Both cross-sectional [78] and longitudinal data [79] have clarified that the evaluation of fT can improve the accuracy of diagnosing symptomatic male hypogonadism, particularly in older men with obesity. In clinical conditions that may interfere with SHBG levels (Table 3.3), evaluation of fT should be considered to better estimate actual androgen levels (Figure 3.1). However, it should be recognised that single-nucleotide polymorphisms (SNPs) of SHBG gene are quite common in the general population, thus potentially affecting both SHBG circulating levels and its binding capacity. A large population-based survey showed that SHBG-related SNPs can affect total testosterone assessment but not fT when LC-MS/MS is used [80]. Hence, it is strongly recommended to perform SHBG measurement and fT calculation in all conditions characterised by altered SHBG levels (Table 3.3). In this setting, limited evidence indicates that a threshold of 220pmol/l (6,4ng/dl) for fT can be considered as a reliable threshold [8,78,79].
When pathological total testosterone levels are detected, the measurement of LH should be performed together with prolactin (PRL) to accurately identify the underlying condition and exclude potential organic causes (Figure 3.1). Follicle-stimulating (FSH) and LH hormones determination can further support the diagnosis of primary or secondary hypogonadism [21,81]. Due to its negative influence on libido, PRL can also be considered as first-line screening in patients with reduced sexual desire. In addition, contrast-enhanced pituitary magnetic resonance imaging (MRI) scanning, as well as other pituitary hormone evaluations, is required in the presence of specific symptoms, such as visual disturbances, headache and when hyperprolactinemia is confirmed [82,83]. Limited evidence suggests also performing pituitary MRI in the case of severe hypogonadism (< 6nmol/L, 1.75ng/mL) with inadequate gonadotropin levels (Figure 3.1) [82-84].
Table 3.3: Main factors associated with an increase or reduction of SHBG circulating levels
| SHBG increase | SHBG decrease |
|
|
3.3.5. Summary of evidence and recommendations for the diagnostic evaluation and screening oflate-onset hypogonadism
| Summary of evidence | LE |
| Sexual symptoms are the most specific symptoms associated with LOH. | 1a |
| Diagnosis of LOH should be based on specific signs and symptoms of androgen deficiency, together with consistently low serum testosterone levels. | 1a |
| Total testosterone 12nmol/L (3.5ng/mL) represents a reliable threshold to diagnose LOH. | 1a |
| Functional hypogonadism is a consequence of comorbidity/concomitant drugs, which can impair testosterone production in adulthood. The diagnosis of functional hypogonadism is a diagnosis of exclusion, after ruling out organic causes of hypogonadism. | 4 |
| Self-reported questionnaires and structural interviews have been developed for screening of hypogonadism, but their specificity remains poor. | 2a |
| Recommendations | Strength rating |
| Diagnostic evaluation | |
| Check for concomitant diseases, drugs and substances that can interfere with testosterone production/action. | Strong |
| Measure total testosterone (TT) in the morning (07:00 and 10:00) and in the fasting state, with a reliable laboratory assay. | Strong |
| Repeat TT on at least two separate occasions when < 12nmol/L and before starting testosterone therapy. | Strong |
| Use 12nmol/L TT (3.5ng/mL) as a reliable threshold to diagnose late-onset hypogonadism (LOH). | Strong |
| Measure sex hormone-binding globulin and free-testosterone calculation when indicated. | Strong |
| Analyse luteinising hormone and follicle-stimulating hormone serum levels to differentiate between the different types of hypogonadism. | Strong |
| Measure prolactin (PRL) levels if evidence of low sexual desire (or other suggestive signs/symptoms) and secondary hypogonadism is present. | Strong |
| Perform pituitary magnetic resonance imaging (MRI) in secondary hypogonadism, with elevated PRL or symptoms specific of a pituitary mass and/or presence of other anterior pituitary hormone deficiency. | Strong |
| Perform pituitary MRI in secondary severe hypogonadism (total testosterone < 6-nmol/L). | Weak |
| Screening | |
| Screen for LOH only in symptomatic men. | Strong |
| Do not use structured interviews and self-reported questionnaires for systematic screening for LOH as they have a low specificity. | Strong |
Figure 3.1: Diagnostic evaluation of Late-Onset Hypogonadism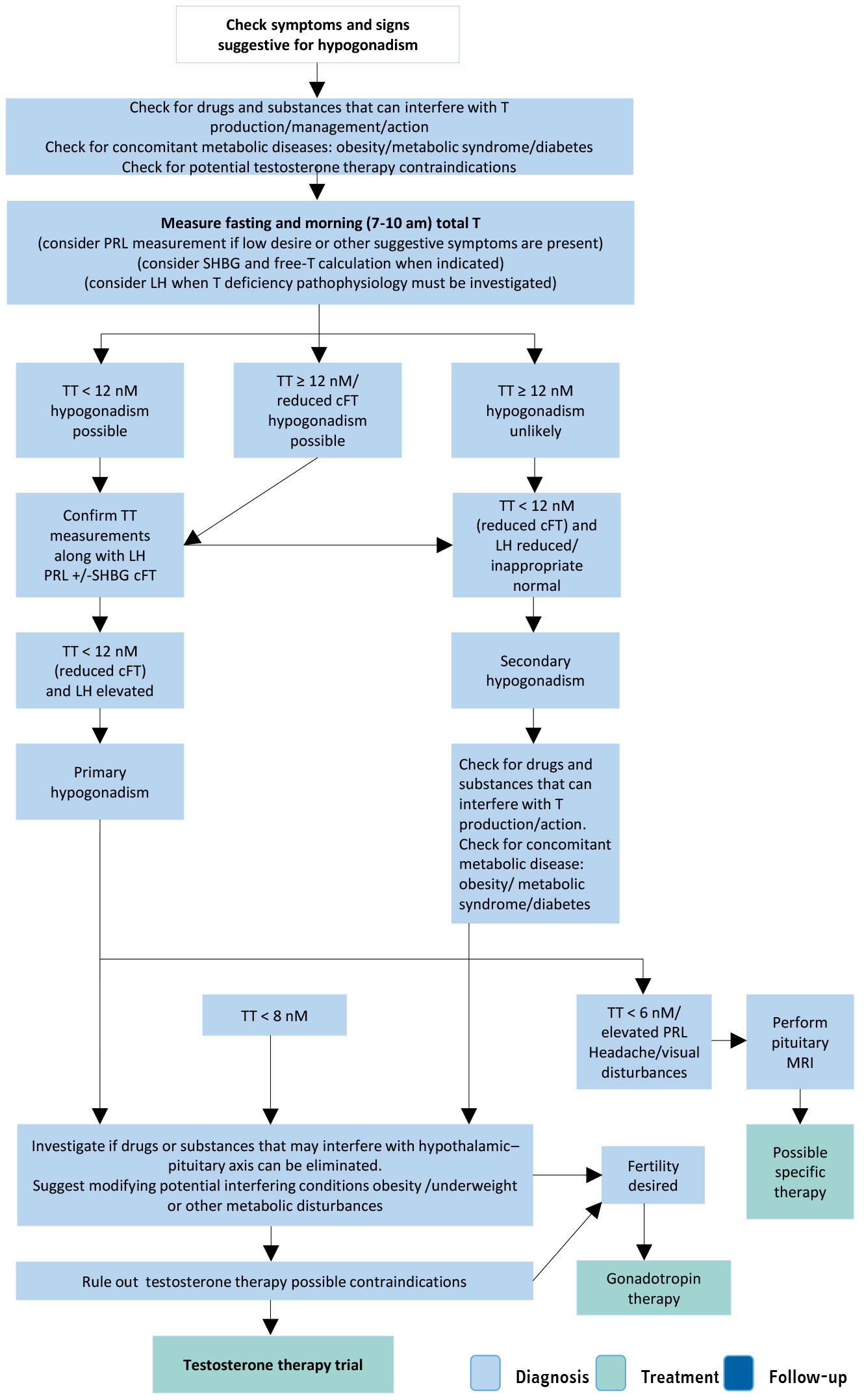 cFT = calculated free testosterone; LH = luteinising hormone; MRI = magnetic resonance imaging; PRL = prolactin; SHBG = sex hormone-binding globulin; TT = total testosterone.
cFT = calculated free testosterone; LH = luteinising hormone; MRI = magnetic resonance imaging; PRL = prolactin; SHBG = sex hormone-binding globulin; TT = total testosterone.
3.4. Treatment of classical and late-onset hypogonadism
3.4.1. Indications and contraindications for treatment of hypogonadism
Patients with symptomatic hypogonadism (total testosterone < 12nmol/L) without specific contraindications are suitable candidates to receive testosterone therapy (Table 3.4).
Absolute contraindications are untreated breast and prostate cancer (PCa). Similarly, conditions such as cardiovascular (CV) events as well as uncontrolled or poorly controlled congestive heart failure should be considered when prescribing testosterone therapy [85]. Conversely, severe lower urinary tract symptoms (LUTS), defined as an International Prostate Symptom Score (IPSS) > 19, represent a relative contraindication to testosterone therapy, as there is insufficient evidence regarding its long-term safety in these patients [70]. A positive family history for venous thromboembolism requires further analysis to exclude a condition of undiagnosed thrombophilia-hypofibrinolysis [86]. These patients need to be carefully counselled prior to testosterone therapy initiation. A haematocrit > 54% should require testosterone therapy withdrawal, reduction in dose, change of formulation and venesection, depending on the clinical situation, to avoid any potential cardiovascular complications. Lower baseline haematocrit (48-50%) should be carefully evaluated before testosterone therapy initiation to avoid pathological increases during treatment, especially in high-risk men, such as those with COPD or Obstructive Sleep Apnoea Syndrome (OSAS), as well as in natives living at high altitudes, since they may suffer from secondary excessive erythrocytosis [87]. Accordingly, the Framingham Heart Study showed that haematocrit > 48% represented a condition associated with increased risk of coronary artery disease and mortality, and was associated with CV disorders [88]. Testosterone therapy suppresses gonadotropin and endogenous testosterone secretion as well as spermatogenesis [89] ; therefore, testosterone therapy is contraindicated in individuals who desire fertility [90]. In this context, secondary hypogonadism is characterised by low or inappropriately normal gonadotropin levels, and the rationale is to substitute the gonadotropin deficiency with simultaneously FSH and LH analogues, if fertility is desired [91].
Table 3.4: Main contraindications of testosterone therapy
| Main contraindications of testosterone therapy | |
|---|---|
| Absolute contraindications | Locally advanced or metastatic prostate cancer Male breast cancer Men with an active desire to have children Haematocrit ≥ 54% Uncontrolled or poorly controlled congestive heart failure |
| Relative contraindications | IPSS score > 19 Baseline haematocrit 48-50% Familial history of venous thromboembolism |
3.4.2. Testosterone therapy outcomes
3.4.2.a. Sexual dysfunction
Sexual concerns are the main symptoms of hypogonadal patients [3,8,92,93]. A consistent body of evidence shows that testosterone therapy in men with hypogonadism (total testosterone < 12nmol/L) may have a beneficial effect on several aspects of sexual life; in contrast, there is no evidence of benefits in using testosterone therapy for treating sexual dysfunction in eugonadal men [77,94-96]. The beneficial effect on sexual function seems to be more related to testosterone level normalisation than the specific testosterone formulations used [96,97].
A meta-analysis of placebo-controlled RCTs showed that testosterone therapy significantly improves EF (as assessed by the EF domain score of the International Index of Erectile Function [IIEF]) and that patients with more severe hypogonadism (i.e. total testosterone < 8nmol/L) are more likely to achieve better improvement than patients with milder hypogonadism (i.e. total testosterone < 12nmol/L) [77]. Similar results were observed for sexual desire; however, the presence of metabolic comorbidity (such as diabetes and obesity) decreased the magnitude of these improvements. Testosterone therapy alone resulted in a clinically effective outcome only in patients with milder ED [77]. Similar results have also been confirmed in an update analysis [98]. Consistent with these findings, the non-inferiority TRAVERSE study (“Testosterone Replacement Therapy for Assessment of Long-term Vascular Events and Efficacy Response in Hypogonadal Men”) reported that, in middle-aged and older men with hypogonadism (total testosterone < 10.4nmol/L) with pre-existing CVD or at high CV risk, two years of testosterone gel therapy improved sexual activity, hypogonadal symptoms, and sexual desire, but did not enhance EF [99].
Other sexual function parameters, such as intercourse, orgasm and overall satisfaction, were all improved as compared with placebo [77,98]. Men with a comorbidity, such as T2DM, usually show modest improvements in terms of sexual function after testosterone therapy and may potentially require concomitant phosphodiesterase type 5 inhibitors (PDE5Is) to improve effectiveness [3,96,100]. A meta-analysis including 913 patients derived from eight RCTs suggested that combination therapy (testosterone and PDE5Is) was superior when compared to PDE5Is alone in improving EF [101]. The specific beneficial effect derived from the combined use of testosterone therapy and PDE5Is is unclear [94]. Similarly, there is a lack of evidence regarding the combined use of testosterone therapy with other pharmacological treatments for ED [3,96].
The Sexual Function Trial of the Testosterone Trials (TTrials) (one of the largest placebo-controlled trials on testosterone therapy) documented consistent improvements in 10 of 12 measures of sexual activities in older (≥ 65 years) men with hypogonadism, particularly in frequency of intercourse, masturbation and nocturnal erections (as measured by PDQ-Q4) [102]. The magnitude in improvement was shown to be proportional to the increase in serum total testosterone, fT and E2 levels; it was not possible to demonstrate a threshold level [103]. A study of 220 men with MetS, with or without T2DM, also found that sexual function improved in men who reported sexual problems, as reflected by improvements in IIEF scores, with specific increases in libido and sexual satisfaction [30].
3.4.2.b. Vitality and physical strength
The role of testosterone in stimulating muscle growth and strength is well established. Accordingly, androgenic-anabolic steroids have been used as performance-enhancing agents to increase physical performance in competitive sport [104]. In this regard, testosterone therapy in men with hypogonadism has been shown to increase muscle mass and reduce fat mass, with limited effects on final weight [41]. Despite this evidence, the role of testosterone therapy in older men with mobility limitations remains unclear. The 1999-2004 National Health and Nutrition Examination Survey [105] was unable to detect any association between overall circulating testosterone levels and the amount of physical activity. However, among non-obese men, those in the highest physical activity tertile were significantly less likely to have low or low-normal testosterone than those in the lowest tertile. Data from TTrials indicated that testosterone therapy did not substantially increase the fraction of men whose six-minute walking distance increased > 50m or the absolute increase in the distance walked by those enrolled in the physical function trial [102]. However, when the whole population of the TTrials was considered, a significant, although modest, positive effect on these two parameters was reported [102]. Similar data were derived from the Vitality Trial [102]. As support of the aforementioned considerations, a meta-analysis including 2,043 patients older than 60 years failed to show a significant improvement in muscle strength from testosterone therapy when compared to placebo [106].
3.4.2.c. Mood and cognition
Several observational studies have documented a relationship between depressive symptoms, reduced QoL and hypogonadism [107,108]. However, the specific relationship between hypogonadism and the incidence of depression is still unclear [108]. Only a few placebo-controlled RCTs have investigated the role of testosterone therapy in improving depressive symptoms. Data derived from TTrials showed that testosterone therapy improved mood and depressive symptoms, assessed as continuous measures using several instruments [102]. However, the final effect was small in magnitude. In line with this data, the largest meta-analysis of available studies, including 1,890 men with hypogonadism (baseline total testosterone < 12nmol/L or fT < 225pmol/L) from 27 RCTs, documented that the positive effect of testosterone therapy was particularly evident in patients with milder symptoms [109]. The BLAST study of testosterone therapy in T2DM reported that those men with depression were less likely to respond with regards to symptoms of sexual dysfunction compared to men without depression [36]. Findings from the TRAVERSE trial [85] showed that testosterone therapy alone did not appear to represent an effective treatment option for most men with clinical depressive disorders [110].
Robust data on the effect of testosterone therapy on QoL are limited. Although meta-analyses suggest a significant effect of testosterone therapy over placebo, the magnitude is low and the heterogeneity high, therefore reducing the scientific value of the effect [97,111].
The role of testosterone therapy in patients with cognitive impairment is even more uncertain. The TTrials evaluated the effect of testosterone therapy in 493 individuals with age-associated memory impairment to assess possible improvement of several aspects of cognitive function. However, results failed to demonstrate any beneficial effect of testosterone therapy in improving cognitive function [102]. Similarly, a meta-analysis involving 17 studies enrolling 1,438 patients with a mean age of 70.4 years and a mean follow-up of 45.6 weeks did not find any effect of testosterone therapy on cognitive domains [112].
3.4.2.d. Body composition and metabolic profile
Late-onset hypogonadism is associated with a greater percentage of fat mass and a lesser lean mass compared to testosterone-repleted men [113]. The major effect of low testosterone is to increase visceral adiposity, but it also leads to deposition of lipids in the liver and muscle and is associated with atherosclerosis [25]. Some published data have suggested that testosterone therapy reduces body fat percentage and increases lean mass [114]. Testosterone therapy has also been found to decrease waist circumference, body weight and BMI, with these effects more predominant after 12 months of treatment [114-116]. Over two years, the T4DM RCT reported that men receiving testosterone therapy in combination with a lifestyle programme had greater reductions in waist circumference and total and abdominal fat mass, as well as increases in total and arm muscle mass and strength in the non-dominant hand, compared with a lifestyle programme alone [34]. There was a trend toward a reduction in body weight, which approached but did not reach significance. The latter result is probably compounded by the increase in muscle mass as well as the decrease in fat mass. However, it should be recognised that the results of previous studies are mainly derived from registry and observational trials, which have important limitations due to the risk of selection bias for the non-random assignment of testosterone exposure. Accordingly, data derived from RCTs showed only an improvement of fat mass and lean mass of the same amount without any modifications in body weight [41]. A meta-analysis including 17 RCTs specifically investigated the role of testosterone therapy on several metabolic parameters in patients with T2DM and/or MetS [40]. In line with what was reported in the general population, testosterone therapy was associated with an improvement in body composition either in T2DM or MetS without any effects on body weight. Similarly positive effects were also observed on fasting glycemia and insulin resistance (HOMA index) whilst more conflicting data were obtained for HbA1c and lipid profile [40]. In line with these data, the results derived from the T4DM study showed that a combination of lifestyle and testosterone therapy can reduce the incidence of T2DM at follow-up [40]. However, a subgroup analysis of the TRAVERSE study reported that testosterone therapy did not improve glycaemic control in men with hypogonadism and prediabetes or diabetes [117]. The different characteristics of the populations as well as the lack of specific lifestyle program may partially explain the neutral effects observed in the TRAVERSE study.
3.4.2.e. Bone
Evidence suggests that bone mineralisation requires circulating sex steroids within the normal range [118]. The possible association between mild hypogonadism and osteopenia/osteoporosis is weak, whereas severe hypogonadism (total testosterone < 3.5nM) is frequently associated with bone loss and osteoporosis, independent of patient age [118]. Three independent meta-analyses showed a positive effect of testosterone therapy on bone mineral density, with the highest effect at the lumber level [119-121]. Interestingly, this latter meta-analysis has provided novel evidence that the role of testosterone on bone mineral density was even higher in patients with diabetes [121], who are themselves at a higher risk of hypogonadism and bone fracture [40,122,123]. Similarly, data derived from TTrials and the T4DM studies confirmed that testosterone therapy increased bone mineral density in ageing men with hypogonadism [102,124]. A subgroup analysis of the TRAVERSE study showed that the fracture incidence was numerically higher amongst men in the testosterone therapy group versus placebo; however, it should be noted that this was not the primary outcome of the study [125]. Furthermore, when the most clinically important fractures, such as bone and hip fractures, were considered, no difference between testosterone therapy and placebo was observed. Available data are insufficient to determine the effect of testosterone therapy alone on the risk of fractures [118]. The use of testosterone therapy as an adjunct to anti-resorptive treatment in patients with hypogonadism at high risk of fractures has not been established. Therefore, anti-resorptive therapy must be the first-choice treatment in men with hypogonadism at high risk for bone fractures. The combination of anti-resorptive treatment and testosterone therapy should be offered only in conjunction with hypogonadism-related symptoms.
3.4.2.f. Summary of evidence and recommendations for testosterone therapy outcome
| Summary of evidence | LE |
Testosterone therapy can improve: Milder forms of ED and libido in men with hypogonadism. Other sexual symptoms, including intercourse frequency, orgasm and overall satisfaction. Body composition and insulin resistance. Weight, waist circumference and lipid profile, but the evidence is conflicting. Mild depressive symptoms in men with hypogonadism. Bone mineral density, but information related to fracture risk is lacking. |
1a 1b 1a 3 1a 1a |
| Recommendations | Strength rating |
| Do not use testosterone therapy (TTh) in eugonadal men. | Strong |
| Do not use TTh for the treatment of male infertility and in males wishing to be fathers. | Strong |
| Use TTh as first-line treatment in hypogonadal patients with mild erectile dysfunction (ED). | Strong |
| Use a combination of phosphodiesterase type 5 inhibitors and TTh in more severe forms of ED associated with hypogonadism. | Weak |
| Use conventional medical therapies for severe depressive symptoms and osteoporosis. | Strong |
| Do not use TTh to reduce weight and enhance cardio-metabolic status. | Weak |
| Do not use TTh to improve cognition vitality and physical strength in ageing males. | Strong |
3.4.3. Choice of treatment
3.4.3.a. Lifestyle factors
Functional hypogonadism is frequently associated with obesity and metabolic disorders [126]. Therefore, weight loss and lifestyle changes should be the first approach for all men with hypogonadism that are overweight or obese. A previous meta-analysis documented that a low-calorie diet can revert obesity-associated secondary hypogonadism by increasing total testosterone and fT, reducing oestrogens and restoring normal gonadotropin circulating levels [127]. This was confirmed in an updated meta-analysis showing that an increase in testosterone is significantly associated with weight reduction [128]. Similar results can be obtained through physical activity, which is associated with the duration of scheduled exercise and weight loss [128]. However, it should be recognised that the increase in testosterone levels observed after a low-calorie diet and physical activity is small (1-2nmol) [127,128]. In addition, 60-86% of weight lost is regained after three years and 75-121% after five years [129]. Lifestyle changes represent an essential part of the management of obesity; however, some evidence suggests that when compared to lifestyle modifications alone, testosterone therapy-treated men with obesity benefit most from relief of their symptoms associated with testosterone deficiency, whereas those not treated did not benefit [91]. There is limited evidence to suggest that a combination of lifestyle interventions and testosterone therapy in symptomatic men with hypogonadism might result in better outcomes [113]. As described above, the T4DM study demonstrated that over two years of testosterone therapy with lifestyle intervention was superior to lifestyle intervention alone in reducing waist circumference as well as total and abdominal fat mass. There was no significant reduction in body weight when compared to lifestyle intervention alone [34].
3.4.3.b. Medical preparations
Several testosterone formulations are available (Table 3.5). Direct comparisons among different testosterone products are still lacking. Candidates for testosterone therapy should be adequately informed about the possible risks and benefits of all available testosterone preparations. The final choice should be based on the clinical situation, testosterone formulation availability, and patient needs and expectations [22,130].
3.4.3.b.1. Oral formulations
Since the 1970s, an oral formulation has been available in oleic acid and has subsequently been reformulated in a mixture of castor oil and propylene glycol laureate (TU caps) to allow storage at room temperature without degradation [22,130]. The main limitation is related to poor bioavailability, which is strongly dependent on dietary fat content [22,130]. The United States Food and Drug Administration (FDA) approved a new formulation of oral TU in a liquid-filled soft gel capsule, which improved oral availability [131]. Available evidence showed that TU capsule formulations can reach steady 24-hour average serum total testosterone levels in more than 80% of men with hypogonadism, resulting in a significant improvement of all sexual function domains at all time points when compared to baseline along with an excellent safety profile [131]. The FDA has approved a new oral formulation that contains Vitamin E, phytosterol esters, polyoxyl 40 hydrogenated castor oil, and propylene glycol monolaurate as carriers [131]. For all new oral TU formulations, a mild increase in arterial blood pressure has been reported. Hence, the FDA has required a black box warning that these drugs can induce a blood pressure increase [131].
Mesterolone is a 5α-dihydrotestosterone derivate available for oral administration. Along with dihydrotestosterone (DHT), mesterolone cannot be converted to oestrogens and can only be used for a limited period and specific indications, such as the presence of painful gynaecomastia. However, the lack of a full spectrum of testosterone bioactivity strongly limits its long-term use [22,130].
3.4.3.b.2. Parenteral formulations
Injectable testosterone preparations can be classified according to their half-lives (Table 3.5). Testosterone propionate is a short-term ester formulation requiring multiple fractionated doses (usually 50mg, every two to three days), thus representing a major limitation for its use [22,130]. Cypionate and enanthate-T esters are short-term formulations, requiring administration every two to four weeks. A formulation containing mixed testosterone esters (TU, isocaproate, phenyl propionate, propionate), which has the benefit of a steady release of testosterone into the circulation, is available in some countries. The use of these older formulations is associated with wide fluctuations in plasma testosterone concentrations and is often reported as unpleasant by patients and potentially resulting in adverse effects, such as polycythaemia [22,130,132]. A longer-lasting TU injectable formulation is widely available [22,130], with a good safety/benefit profile, allowing the maintenance of normal stable testosterone levels at a dose of 1,000mg initially every 12 weeks, following a six-week loading dose, but can be adjusted to a frequency of ten to 14 weeks dependent on the trough (pre-injection level) after three to five injections to maintain levels in the therapeutic range (usually > 12 and < 18nmol/L) [22,130,133].
3.4.3.b.3. Transdermal testosterone preparations
Among the available transdermal formulations, testosterone gels represent the most frequently used preparations. The gel is quickly absorbed by the stratum corneum, creating a reservoir within the subcutaneous tissues from where testosterone is continuously delivered for 24 hours, after a single daily application. These formulations have been shown to normalise serum testosterone levels with an excellent safety profile [22,130]. The introduction of specific devices and skin enhancers has resulted in better skin penetration of the drugs, thus reducing potential adverse effects. Local skin adverse effects are limited when compared to traditional testosterone patches, but they potentially allow transference of testosterone during close contact with the skin surface. The risk can be reduced by wearing clothing or by applying the gel on skin surfaces not usually touched (e.g. the inner thigh surface) [22,130]. To reduce the total amount of gel applied and residual quantities remaining on the skin, new formulations of testosterone gel have been introduced with a testosterone concentration of 1.62-2% [22,130]. Another transdermal testosterone formulation includes a topical, alcohol-based testosterone (2%) solution, which must be applied to the underarm once daily, using a metered dose applicator [22,130]. This testosterone formulation is not available in Europe. Testosterone levels should be monitored to optimise the testosterone dose. Blood collection is best taken at two to four hours after gel application to use the peak level of testosterone absorbed as a reference for adequate therapeutic levels. After application, levels of testosterone can vary and a repeat measurement may be indicated especially as sometimes, inadvertently, the skin over the venepuncture site can be contaminated by the gel, leading to falsely elevated results.
In some European countries, DHT is available as a hydroalcoholic 2.5% gel. It is rapidly absorbed, reaching a steady state in two to three days [22,130]. Similar to that reported for mesterolone, DHT is not aromatised but can be useful for treating particular conditions, such as gynaecomastia and microphallus [22,130].
3.4.3.b.4. Transmucosal formulations
A testosterone buccal system is still available in several countries. It consists of a sustained-release muco-adhesive buccal-testosterone-tablet requiring twice-daily application to the upper gums. The tablet does not dissolve completely in the mouth and must be removed after 12 hours. This formulation has been proven to restore testosterone levels within the physiological range with minimal or transient local problems, including gum oedema, blistering and gingivitis [22,130].
A gel for intranasal administration is available in some countries, including the United States of America (USA) and Canada. It requires administration two or three times daily using a specific metered-dose pump. The application is rapid, non-invasive, convenient, and avoids secondary transference observed with other topical products [22,130]. Preliminary results suggest that intranasal testosterone is associated with a lower Gn levels suppression and with a lower risk of haematocrit increases [134].
3.4.3.b.5. Subdermal depots
The implantation of testosterone pellets, available in a limited number of countries, represents the longest available testosterone formulation lasting from four to seven months. The procedure is invasive and may be unattractive to patients [22,130].
3.4.3.b.6. Anti-oestrogens
Anti-oestrogens, including selective oestrogen receptor modulators (SERMs) and aromatase inhibitors (AIs) have been suggested as off-label treatments to restore testosterone levels and fertility in men with functional secondary hypogonadism or idiopathic infertility. They work by preventing down-regulation of the HPG axis by oestrogens and for this reason are particularly useful in men with obesity and metabolic disorders [128,135]. In the latter case, the hypothesis is that the excess of adipose tissue leads to increased aromatase activity and oestrogens levels resulting in impairment of the HPG [126]. Due to their putative mechanism of action, they require an intact HPG axis and cannot work in primary hypogonadism or secondary hypogonadism due to organic damage of the HPG axis. Both types of SERMs, which bind oestrogen receptors with an agonist or antagonist effect depending upon the target tissue, and AIs, which prevent androgens from being converted into oestrogens by aromatase, have been used in clinical practice [22,130]. The evidence published so far is poor; all these products are off-label treatments and SERMs, due to their agonistic effect on venous vessels, could predispose men to the development of venous thromboembolism [22,130]. In this context, patients should be warned of the potential increased risk of venous thromboembolism, although data are lacking. Long-term use of these agents can lead to reduced bone density and development of osteoporosis, potentially increasing fracture risk.
3.4.3.b.7. Gonadotropins
Gonadotropin therapy should be considered the standard in men with secondary hypogonadism who desire paternity (Table 3.5) [22,130]. Recombinant human chorionic gonadotrophin (rhCG) and recombinant LH (rLH) formulations offer comparable effects to urinary-derived preparations [130]. According to a meta-analysis of the available evidence, human chorionic gonadotrophin (hCG) should be administered with FSH, since combined therapy results in better outcomes. Similar to rhCG, recombinant FSH (rFSH) offers comparable effects to urinary-derived preparations [133].
Table 3.5: Available preparations for hypogonadism treatment
| Formulation | Chemical structure | t ½ | Standard dosage | Advantages | Disadvantages |
| GONADOTROPINS | |||||
| Human chorionic gonadotrophin (hCG) | |||||
| Extractive | hCG purified from urine of pregnant women | NA | 1,000-2,000IU 3 times/week | Low cost | Multiple weekly administration |
| Recombinant | Human recombinant hCG | NA | No data in men | NA | NA |
| Luteotropic hormone (LH) | |||||
| Recombinant | Human recombinant LH | NA | No data in men | NA | NA |
| Follicle-stimulating hormone (FSH) | |||||
| Extractive | FSH purified from urine of pregnant women | NA | 75-150IU 3 times/week | Low cost | Multiple weekly administration |
| Recombinant | Human recombinant FSH | NA | 75-150IU 3 times/week | NA | Multiple weekly administration |
| TESTOSTERONE PREPARATIONS | |||||
| Oral | |||||
| Testosterone undecanoate | 17-α-hydroxylester | 4 hrs | 120-240mg 2 or 3 times daily | Reduction of liver involvement Oral convenience Modifiable dosage | Unpredictable absorption depending on dietary fat content Must be taken with meals |
Testosterone undecanoate self-emulsifying delivery system | 17-α-hydroxylester | 2-5 hrs | 100-237mg 2 times daily | Oral convenience Modifiable dosage Quick reversal | Gastrointestinal side effects Increase in blood pressure |
| Mesterolone | 1α-methyl-4, 5α-dihydro-testosterone | 12 hrs | 50-100mg 2 or 3 times daily | Oral convenience Modifiable dosage Useful in gynaecomastia | Not aromatisable |
| Parental | |||||
| Testosterone enanthate | 17-α-hydroxylester | 4-5 days | 250mg every 2-3 weeks | Low cost Short-acting preparation allowing drug withdrawal in case of adverse effects | Fluctuations in circulating testosterone levels Multiple injections Relative risk of polycythaemia |
| Testosterone cypionate | 17-α-hydroxylester | 8 days | 200mg every 2-3 weeks | ||
| Testosterone propionate | 17-α-hydroxylester | 20 hrs | 100mg every 2 days | ||
| Testosterone ester mixture* | 4-androsten-3-one- 17 beta-hydroxy-androst-4-en-3-one | 4-5 days | 250mg every 3 weeks | ||
| Testosterone undecanoate in castor oil | 17-α-hydroxylester | 34 days | 1,000mg every 10-14 weeks 750mg every 10 weeks | Steady-state testosterone level without fluctuation Long-lasting Less frequent administration | Pain at injection site Long-acting preparation not allowing rapid drug withdrawal in case of adverse effects |
| Surgical implants | Native testosterone | N/A | 4-6 200mg implants lasting up to 6 months | Long duration and constant serum testosterone level | Placement is invasive Risk of extrusion and site infections |
| TRANSDERMAL | |||||
| Testosterone patches | Native testosterone | 10 hrs | 50-100mg/day | Steady-state testosterone level without fluctuation | Skin irritation Daily administration |
| Testosterone gel 1-2% | Native testosterone | 6 hrs | 50-100mg/day | Possible transfer during intimate contact Daily administration | |
| Underarm testosterone (testosterone solution 2%) | Native testosterone | NA | 60-120mg/day | ||
| Dihydro-testosterone gel 2.5% | Native dihydro-testosterone | NA | 34-70mg/day | Steady-state testosterone level without fluctuation Useful in gynaecomastia | Possible transfer during intimate contact Daily administration Not aromatisable |
| TRANSMUCOSAL | |||||
| Testosterone buccal system | Native testosterone | 12 hrs | 60mg 3 times daily | Steady-state testosterone level without fluctuation | Possible oral irritation Twice-daily dosing Unpleasant taste |
| Testosterone nasal | Native testosterone | 6 hrs | 33mg 3 times daily | Nasal irritation Multiple daily administration | |
NA = not applicable.
* Testosterone ester mixture - propionate (30mg), phenylpropionate (60mg), isocaproate (60mg), decanoate (100mg).
3.4.3.c. Summary of evidence and recommendations for choice of treatment for late-onset hypogonadism
| Summary of evidence | LE |
| Weight loss obtained through a low-calorie diet and regular physical activity results in a small improvement in testosterone levels. | 1a |
| Testosterone gels and long-acting injectable TU represent testosterone preparations with the optimal safety profiles. | 1a |
| Gonadotropin treatment can be used to restore fertility in men with secondary hypogonadism. | 1a |
| Recommendations | Strength rating |
| Treat, when indicated, organic causes of hypogonadism (e.g. pituitary masses, hyperprolactinemia, etc.). | Strong |
| Improve lifestyle and reduce weight (e.g. obesity); withdraw, when possible, concomitant drugs that can impair testosterone production; treat comorbidities, before starting testosterone therapy. | Strong |
| Fully inform patients about expected benefits and adverse effects of any treatment option. Select the testosterone preparation in a joint decision-making process and fully inform patients of the risks and benefits. | Strong |
| Use testosterone gels rather than long-acting depot administration when starting initial treatment in high-risk men. | Weak |
3.5. Safety and follow-up in hypogonadism management
3.5.1. Hypogonadism and fertility issues
The aim of pharmacological management of hypogonadism is to increase testosterone levels to normal levels which resolve or improve symptoms of hypogonadism. The first choice is to administer exogenous testosterone. However, while exogenous testosterone has a beneficial effect on the clinical symptoms of hypogonadism, it inhibits gonadotropin secretion by the pituitary gland, resulting in impaired spermatogenesis and sperm cell maturation [136]. Therefore, testosterone therapy is contraindicated in men with hypogonadism seeking fertility treatment [90]. When secondary hypogonadism is present, gonadotropin therapy may maintain normal testosterone levels and restore sperm production [3].
3.5.2. Male breast cancer
Studies have clearly documented that breast cancer growth is significantly influenced by testosterone and/or by its conversion to E2 through different mechanisms and pathways [137]. Accordingly, the use of SERMs still represents an important therapeutic option in the management of this cancer [137]. No information is available on the role of testosterone therapy in patients successfully treated for male breast cancer; therefore, treated and active male breast cancer should be recognised as absolute contraindications for testosterone therapy.
3.5.3. Lower urinary tract symptoms/benign prostatic hyperplasia
A trial of 60 patients undergoing testosterone therapy for six months showed no significant differences on post-void residual urine and prostate volume, while storage symptoms as measured by IPSS significantly improved, despite an increase in prostate-specific antigen (PSA) level [138]. A larger pre-treatment prostate volume was a predictive factor of improvement in LUTS. Similarly, a placebo controlled RCT including 120 men with hypogonadism (total testosterone < 12nmol/L) with MetS and listed for benign prostatic hyperplasia (BPH) surgery, showed that testosterone therapy did not result in a difference in LUTS severity compared to placebo. Conversely, an improvement in ultrasound (US) markers of inflammation in the expression of several pro-inflammatory genes was found in the treatment active arm [139]. A long-term study of 428 men undergoing testosterone therapy for eight years demonstrated significant improvements in IPSS, no changes in maximum flow rate (Qmax) and residual urine volume, but also a significant increase in prostate volume [140]. Similar data from the Registry of Hypogonadism in Men (RHYME), including 999 patients with a follow-up of three years, did not demonstrate any significant difference in PSA levels or total IPSS in men undergoing testosterone therapy, compared to untreated patients [141]. Similar results were reported in an Italian registry (SIAMO-NOI), collecting data from 432 men with hypogonadism from 15 centres [142]. Meta-analyses have not found significant changes in LUTS between patients treated with testosterone or placebo [143-149]. According to the literature, there are no grounds to discourage testosterone therapy in hypogonadal patients with BPH/LUTS and there is evidence of limited benefit from androgen administration. The only concern is related to patients with severe LUTS (IPSS > 19), as they are usually excluded from RCTs; therefore, limiting the long-term safety data of testosterone therapy in this specific setting [68].
3.5.4. Prostate cancer (PCa)
A considerable number of observational studies have failed to demonstrate any association between circulating higher total testosterone levels and PCa [150]. In contrast, studies investigating the relationship between low levels of testosterone and risk of PCa have found that men with very low levels of fT have a reduced risk of developing low-to-intermediate-grade PCa, but have a non-significantly increased chance of developing high-grade PCa [150]. This peculiar pattern was also reported in trials such as the Health Professionals Follow-up Study, the Prostate Cancer Prevention Trial (PCPT) and the Reduction by Dutasteride of Prostate Cancer Events (REDUCE), with varying magnitudes of significance [151].
A meta-analysis, including 27 placebo-controlled RCTs, found no evidence of increased PSA levels following testosterone therapy for one year. The meta-analysis of 11 studies reporting on the occurrence of PCa found no evidence of increased risk of PCa. A one-year follow-up may be considered too short to draw firm conclusions on the risks of developing PCa. The analysis was restricted to studies with > one-year follow-up, but no significant changes in PSA levels nor increased risk of PCa were found [144]. After a median follow-up of five years across three independent registry studies involving more than 1,000 patients receiving testosterone therapy, the incidence of PCa consistently remained below the reported rate in the general population [152]. Similar findings were reported in a large observational study that included 10,311 men treated with testosterone therapy and 28,029 controls, with a median follow-up of 5.3 years [153]. The same study also showed that the risk of PCa was decreased for men in the highest tertile of testosterone therapy cumulative dose exposure as compared with controls [153]. The TRAVERSE trial found no difference in PCa incidence or high-grade PCa rates between the testosterone therapy and placebo groups after a mean follow-up of 33.0 months (standard deviation 12.1). However, the trial did report a significantly greater increase in total PSA from baseline in the testosterone group compared with the placebo group [85].
With regards to PCa survivors, safety in terms of the risk of recurrence and progression has not yet been established. Limited data are available in the literature, with most case series not providing sufficient data to draw definitive conclusions (e.g. insufficient follow-up, small samples, lack of control arms, heterogeneity in study population and treatment regimen, etc.) [154]. A meta-analysis derived from 13 studies including 608 patients, of whom 109 had a history of high-risk PCa, with follow-up of 1-189.3 months [155], suggested that testosterone therapy did not increase the risk of biochemical recurrence, but the available evidence is poor, limiting data interpretation [155]. In line with the latter study, a meta-analysis including 21 studies and 1,084 patients with a history of testosterone therapy after definitive local therapy for PCa showed a biochemical recurrence up to 2%, regardless of the PCa type. However, it should be recognised that the studies included and the criteria used for the definition of biochemical recurrence were quite heterogeneous. A further potential limitation was that 11 of the studies included in the analysis were available only as conference abstracts [156]. An RCT assessing the safety/benefit ratio of testosterone therapy in men with hypogonadism successfully treated with prostatectomy for non-aggressive PCa is ongoing [157].
In conclusion, literature does not support an increased risk of PCa in men with hypogonadism undergoing testosterone therapy. Although it is mandatory to avoid testosterone administration in men with advanced PCa, insufficient long-term prospective data on the safety of testosterone therapy in PCa survivors [156] should prompt caution in choosing to treat symptomatic men with hypogonadism in this setting. Specifically, patients should be fully counselled that the long-term effects of testosterone therapy in this setting are still unknown and requires further investigation. Due to the lack of strong evidence-based data on safety, the possible use of testosterone therapy in symptomatic men with hypogonadism previously treated for PCa should be fully discussed with patients and limited to low-risk individuals.
3.5.5. Cardiovascular disease
Evidence suggests that men with hypogonadism have an increased risk of CVD [158,159]. Whether or not LOH is a cause or a consequence of atherosclerosis has not been clearly determined. Late-onset hypogonadism is associated with CV risk factors, including central obesity, insulin resistance and hyperglycaemia, dyslipidaemia, pro-thrombotic tendency and chronic inflammatory state [159]. Atherosclerosis is a chronic inflammatory disease, that releases pro-inflammatory cytokines into the circulation, which are known to suppress testosterone release from the HPG axis. Evidence from RCTs of testosterone therapy in men with MetS and/or T2DM demonstrates some benefit in CV risk, including reduced central adiposity, insulin resistance, total cholesterol and LDL-cholesterol and suppression of circulating cytokines [29-31,36,159,160]. However, due to the equivocal nature of these studies, testosterone therapy cannot be recommended for use outside of treatment of specific symptoms.
Published data show that LOH is associated with an increase in all-cause and CVD-related mortality [7,161-164]. These studies are supported by a meta-analysis that concluded that hypogonadism is a risk factor for CV morbidity [148] and mortality [165]. Importantly, men with low testosterone when compared to eugonadal men with angiographically proven coronary disease have twice the risk of earlier death [159]. Longitudinal population studies have reported that men with testosterone in the upper quartile of the normal range have a reduced number of CV events compared to men with testosterone in the lower three quartiles [161]. Androgen deprivation therapy for PCa is linked to an increased risk of CVD and sudden death [166]. Conversely, two long-term epidemiological studies have reported reduced CV events in men with high normal serum testosterone levels [167,168]. Erectile dysfunction is independently associated with CVD and may be the first clinical presentation in men with atherosclerosis.
The knowledge that men with hypogonadism and/or ED may have underlying CVD should prompt individual assessment of their CV risk profile. Individual risk factors (e.g. lifestyle, diet, exercise, smoking, hypertension, diabetes and dyslipidaemia) should be assessed and treated in men with pre-existing CVD and in patients receiving androgen deprivation therapy. Cardiovascular risk reduction can be managed by primary care clinicians, but patients should be appropriately counselled by clinicians active in prescribing testosterone therapy [92]. If appropriate, patients should be referred to cardiologists for risk stratification and treatment of comorbidity.
No RCTs have provided a clear answer on whether testosterone therapy affects CV outcomes. The TTrial (n = 790) in older men [169], the TIMES2 (n = 220) [30] and, the BLAST studies in men with MetS and T2DM, and the pre-frail and frail study in elderly men, all of one year duration, and the T4DM two-year study, did not reveal any increase in Major Adverse Cardiovascular Events (MACE) [30,33,34,169,170]. Randomised controlled trials between three and 12 months in men with known heart disease treated with testosterone therapy have not found an increase in MACE, but have reported improvement in cardiac ischaemia, angina and functional exercise capacity [171-173]. A large cohort study (n=20,4857 men) found that neither transdermal gel or intramuscular testosterone was associated with an increased risk of composite CV outcome in men with or without prevalent CVD (mean follow up 4.3 years) [174]. The European Medicines Agency (EMA) has stated that "The Co-ordination Group for Mutual recognition and Decentralisation Procedures-Human (CMDh), a regulatory body representing EU Member States, has agreed by consensus that there is no consistent evidence of an increased risk of heart problems with testosterone therapy in men. However, the product information is to be updated in line with the most current available evidence on safety, and with warnings that the lack of testosterone should be confirmed by signs and symptoms and laboratory tests before treating men with these medicines" [175].
Data released from the TRAVERSE study confirm the findings of the EMA [85]. The latter is the first double-blind, placebo-controlled, noninferiority RCT with primary CV safety as an end point. The results showed that testosterone therapy was non-inferior to placebo with respect to the incidence of MACE. A mild higher incidence of atrial fibrillation, acute kidney injury, and pulmonary embolism was observed in the testosterone group [85]. The latter observations need to be confirmed, as previous available data do not support an increased risk of venous thromboembolism [86,176] or major arrhythmias [177] after testosterone therapy. Similarly, the long-term follow-up (median of 5.1 years since last injection) of the T4DM study showed no differences in self-reported rates of new diagnosis of CVD [178].
In conclusion, current available data from interventional studies suggest that there is no increased risk up to three years of testosterone therapy [179-183]. The currently published evidence has reported that testosterone therapy in men with diagnosed hypogonadism has neutral or beneficial actions on MACE in patients with normalised testosterone levels. The findings could be considered sufficiently reliable for at least a three-year course of testosterone therapy, after which no available study can exclude further or long-term CV events [184,185].
3.5.5.a. Cardiac failure
Testosterone therapy is contraindicated in men with severe chronic cardiac failure because fluid retention may lead to exacerbation of the condition. Some studies have shown that men with moderate chronic cardiac failure may benefit from low doses of testosterone, which achieves mid-normal range testosterone levels [172,186,187]. An interesting observation is that untreated hypogonadism increased the re-admission and mortality rate in men with heart failure [188]. If a decision is made to treat hypogonadism in men with chronic cardiac failure, it is essential that the patient is followed up carefully with clinical assessment and both testosterone and haematocrit measurements on a regular basis.
3.5.6. Erythrocytosis
An elevated haematocrit is the most common adverse effect of testosterone therapy. Stimulation of erythropoiesis is a normal biological action that enhances delivery of oxygen to testosterone-sensitive tissues (e.g. striated, smooth and cardiac muscle). Any elevation above the normal range for haematocrit usually becomes evident between three and 12 months after testosterone therapy initiation. However, polycythaemia can also occur after any subsequent increase in testosterone dose, switching from topical to parenteral administration, or the development of comorbidity that are linked to an increase in haematocrit (e.g. respiratory or haematological diseases).
There is no evidence that an increase in haematocrit up to and including 54% causes any adverse effects. If haematocrit exceeds 54%, there is a testosterone-independent but weak associated rise in CV events and mortality [88,189-191]. Any relationship is complex as these studies were based on patients with any cause of secondary polycythaemia, which included smoking and respiratory diseases. There have been no specific studies in men with only testosterone-induced erythrocytosis.
As detailed, the TRAVERSE study that included symptomatic men with hypogonadism, aged 45-80 years, who had pre-existing or high risk of CVD, showed a mild higher incidence of pulmonary embolism, a component of the adjudicated tertiary end-point of venous thromboembolic events, in the testosterone therapy than in the placebo group (0.9% vs. 0.5%) [85]. However, three previous large studies did not show any evidence that testosterone therapy is associated with an increased risk of venous thromboembolism [192,193]. Of those, one study showed that an increased risk peaked at six months after initiation of testosterone therapy, then declined over the subsequent period [194]. In one study, venous thromboembolism was reported in 42 cases, and 40 of these had a diagnosed underlying thrombophilia (including factor V Leiden deficiency, prothrombin mutations and homocysteinuria) [195]. A meta-analysis of RCTs of testosterone therapy reported that venous thromboembolism was frequently related to underlying undiagnosed thrombophilia-hypofibrinolysis disorders [86]. In an RCT of testosterone therapy in men with chronic stable angina, there were no adverse effects on coagulation by assessment of tissue plasminogen activator or plasminogen activator inhibitor-1 enzyme activity or fibrinogen levels [196]. Similarly, another meta-analysis and systematic review of RCTs found that testosterone therapy was not associated with an increased risk of venous thromboembolism [176]. With testosterone therapy, elevated haematocrit is more likely when baseline levels are near the upper limit of normal prior to initiation. Additional risk factors for increased haematocrit during therapy include smoking and pre-existing respiratory conditions. Elevated haematocrit occurs more frequently with parenteral formulations compared to topical preparations. In line with this, a large retrospective two-arm open registry comparing long-acting testosterone undecanoate with testosterone gels found that the injectable formulation was associated with a higher risk of haematocrit levels exceeding 50%, compared to gel therapy [197]. In men with pre-existing CVD, extra caution is advised with a definitive diagnosis of hypogonadism before initiating testosterone therapy and monitoring of testosterone as well as haematocrit during treatment.
In cases of elevated haematocrit without comorbidities, acute CV or venous thromboembolism events, management can include reducing the testosterone dose, switching to a different formulation, or - if haematocrit is markedly high - performing venesection (500mL), repeated if necessary. In most cases, discontinuation of testosterone therapy is not required.
3.5.7. Obstructive sleep apnoea
There is no evidence that testosterone therapy can result in onset or worsening of sleep apnoea. Combined therapy with Continuous Positive Airway Pressure (CPAP) and testosterone gel was more effective than CPAP alone in the treatment of obstructive sleep apnoea [198]. In one RCT, testosterone therapy in men with severe sleep apnoea reported a reduction in oxygen saturation index and nocturnal hypoxaemia after seven weeks of therapy compared to placebo, but this change was not evident after 18 weeks of treatment and there was no association with baseline testosterone levels [199].
3.5.8. Follow-up
Testosterone therapy alleviates symptoms and signs of hypogonadism in men in a specific time-dependent manner. The TTrials clearly showed that testosterone therapy improved sexual symptoms as early as three months after initiation [102]. Similar results have been derived from meta-analyses [86,94]. Hence, the first evaluation should be planned after three months of treatment. Further evaluation may be scheduled at six or 12 months, according to patient characteristics, as well as results of biochemical testing (see below). Patients at high risk of developing elevated haematocrit should be evaluated every three months during the first year of testosterone therapy and at least every six months thereafter. Accordingly, current guidelines suggest that haematocrit should be maintained below 45% in patients with polycythaemia vera to avoid thromboembolism risk [200]. Similarly, data derived using a multi-institutional database, including a large cohort of hypogonadal (total testosterone < 12nmol/L) men who received testosterone therapy and subsequently did (n = 5,887) or did not (n = 4,2784) develop polycythaemia (haematocrit > 52%), showed that men who had an increased haematocrit had a higher risk of MACE or venous thromboembolism mostly during the first year of therapy [201]. The risk was even higher when a haematocrit threshold of 54% was considered, whilst no risk was observed when a 50% threshold was applied [201]. Table 3.6 summarises the clinical and biochemical parameters that should be monitored during testosterone therapy.
Testosterone Trials were designed to maintain the serum testosterone concentration within the normal range for young men (280-873ng/dL or 9.6-30nmol/L) [102]. This approach resulted in a good benefit/risk ratio. A similar approach could be considered during follow-up. The correct timing for evaluation of testosterone levels varies according to the type of preparation used (Table 3.5). Testosterone is involved in the regulation of erythropoiesis [132] and prostate growth [68], hence evaluation of PSA and haematocrit should be mandatory before and during testosterone therapy. However, it is important to recognise that the risk of PCa in men aged < 40 years is low. Similarly, the mortality risk for PCa in men aged > 70 years has not been considered high enough to warrant monitoring in the general population [202]. Therefore, any screening for PCa through determination of PSA and DRE in men aged < 40 or > 70 years during testosterone therapy should be discussed with the patients.
Baseline and, at least, annually glyco-metabolic profile evaluation may be a reasonable consideration, particularly in the management of functional hypogonadism. Testosterone therapy may be beneficial for men with hypogonadism with low or moderate fracture risk [118]; therefore, dual energy X-ray absorptiometry (DEXA) bone scan may also be considered at baseline and 18-24 months following testosterone therapy, particularly in patients with more severe hypogonadism [118].
Digital rectal examination may detect prostate abnormalities that can be present even in men with normal PSA values. Hence, DRE is mandatory in all men at baseline and is recommended to be performed at least annually during testosterone therapy, as long as there is no significant increase in PSA velocity.
The decision to stop testosterone therapy or to perform prostate biopsy due to PSA increase or prostate abnormalities should be based on local PCa guidelines. There is a large consensus that any increase of haematocrit > 54% during testosterone therapy requires therapy withdrawal and phlebotomy to avoid potential adverse effects including venous thromboembolism and CVD, especially in high-risk individuals. In patients with lower risk of relevant clinical sequelae, the situation can be alternatively managed by reducing testosterone dose and switching formulation along with venesection. A positive family history of venous thromboembolism should be thoroughly investigated, and patients should be counselled regarding testosterone therapy to minimize the risk of thrombophilia and hypofibrinolysis [86]. Finally, caution should be exercised in men with pre-existing CVD or at higher risk of CVD [85].
Table 3.6: Clinical and biochemical parameters to be checked during testosterone therapy
| Parameters | Year 1 of treatment | After year 1 of treatment | ||||
| Baseline | 3 months | 6 months | 12 months | Annually | 18-24 months | |
| Clinical | ||||||
| Symptoms | X | X | X | X | X | |
| Body mass index | X | X | X | |||
| Waist circumference | X | X | X | X | ||
| Digital rectal examination | X | X | X | |||
| Blood pressure | X | X | X | X | ||
| Biochemistry | ||||||
| PSA (ng/mL) | X | X | X2 | X | X | |
| Haematocrit (%) | X | X | X1,2 | X | X | |
| Testosterone | X | X | X | X | ||
| Lipid and glycaemic profile | X | X | X | |||
| Instrumental | ||||||
| DEXA | X | X | ||||
1 Population with polycythaemia vera or at high risk of secondary polycythaemia (e.g. sleep apnea, morbid obesity, heavy smokers, chronic obstructive pulmonary disease).
2 Prostate cancer survivors.
DEXA = dual energy X-ray absorptiometry; PSA = prostate-specific antigen.
3.5.9. Summary of evidence and recommendations on safety and monitoring in testosterone therapy
| Summary of evidence | LE |
| Testosterone therapy is contraindicated in men with secondary hypogonadism who desire fertility. | 1a |
| Testosterone therapy is contraindicated in men with active PCa or breast cancer, as these patients are usually excluded from RCTs. | 1a |
| Testosterone therapy does not increase the risk of PCa, but long-term prospective follow-up data are required to validate this statement. | 1a |
| The effect of testosterone therapy in men with severe LUTS is limited, as these patients are usually excluded from RCTs. | 1a |
| There is no substantive evidence that testosterone therapy, when replaced to normal levels, results in the development of major adverse CV events. | 1a |
| There is no evidence of a relationship between testosterone therapy and mild, moderate or CPAP-treated severe sleep apnoea. | 1b |
| Recommendations | Strength rating |
| Fully counsel symptomatic hypogonadal men who have been surgically treated for localised prostate cancer (PCa) and who are currently without evidence of active disease considering testosterone therapy (TTh), emphasising the lack of sufficient safety data on long-term follow-up. | Weak |
| Restrict TTh to patients with a low risk of recurrent PCa after surgery*. Testosterone therapy should start after at least one year follow-up with prostate-specific antigen (PSA) level < 0.01ng/mL and no evidence of recurrence. | Weak |
| Advise patients that safety data on the use of TTh in males treated for breast cancer are unknown. | Strong |
| Advise patients on active surveillance or treated with non-surgical curative intent that safety data on the use of TTh are unclear. | Weak |
| Consider further diagnostic testing in PCa-naive patients if there is a significant rise or increase in PSA velocity or total PSA. | Strong |
| Evaluate total PSA in PCa survivors at three, six and 12 months during the first year of testosterone therapy, and annually thereafter. | Strong |
| Assess cardiovascular (CV) risk factors before commencing TTh. | Strong |
| Assess men with known cardiovascular disease (CVD) for CV symptoms before initiating TTh and monitor these men with close clinical assessment and evaluation during treatment. | Strong |
| Treat men with hypogonadism and pre-existing CVD, venous thromboembolism or chronic cardiac failure who require TTh with caution, by careful clinical monitoring and regular measurement of haematocrit (not exceeding 54%) and testosterone levels. | Weak |
| Exclude a family history of venous thromboembolism before starting TTh. | Strong |
| Monitor testosterone and haematocrit at three, six and 12 months after testosterone therapy initiation, and thereafter annually. A haematocrit > 54% requires TTh adjustment or withdrawal and venesection if required. Reintroduce testosterone therapy at a lower dose once the haematocrit has normalised and consider switching to topical testosterone preparations. | Strong |
| Evaluate patients with polycythaemia vera and those with a higher risk of developing elevated haematocrit every three months during the first year of testosterone therapy, and at least every six months thereafter. | Strong |
* For EAU risk groups for biochemical recurrence of localised or locally advanced PCa, see EAU Prostate Cancer Guidelines, 2026.