10. PRIAPISM
Priapism is a persistent or prolonged erection in the absence of sexual stimulation that fails to subside. It can be divided into ischaemic, non-ischaemic and stuttering priapism. These Guidelines are based on three systematic reviews addressing the medical and surgical management of ischaemic and non-ischaemic priapism and the overall management of priapism related to sickle cell disease (SCD) [1392-1394].
10.1. Ischaemic (low-flow or veno-occlusive) priapism
10.1.1. Epidemiology, aetiology, pathophysiology and diagnosis
Ischaemic priapism is a persistent erection marked by rigidity of the corpora cavernosa and by little or no cavernous arterial inflow [1395]. Ischaemic priapism is the most common subtype of priapism, accounting for > 95% of all episodes [1395,1396]. In ischaemic priapism, there are time-dependent metabolic alterations within the corpus cavernosum progressively leading to hypoxia, hypercapnia, glucopenia and acidosis [1397,1398].
Ischaemic priapism that lasts beyond four hours is similar to a compartment syndrome and characterised by the development of ischaemia within the closed space of the corpora cavernosa, which severely compromises the cavernosal circulation. Emergency medical intervention is required to minimise irreversible consequences, such as smooth muscle necrosis, corporal fibrosis and the development of permanent ED [1399,1400]. The duration of ischaemic priapism represents the most significant predictor for irreversible consequences, including ED. A study showed that ED risk rose sharply beyond 20 hours [1401]. Interventions beyond 48-72 hours of onset may help to relieve the erection and pain but have little clinical benefit in preventing long-term ED [1402].
In most cases, no specific pathophysiological causes of ischaemic priapism can be identified [1395,1403], although the common aetiological factors include SCD [1404,1405], haematological dyscrasias, neoplastic syndromes and several pharmacological agents (e.g. intracavernosal PGE1 therapy) (Table 10.1). A Danish study concluded that haemolytic disorders, such as thalassemia or glucose-6-phosphate dehydrogenase deficiency, other than SCD, are not significant risk factors for priapism [1406]. Ischaemic priapism may occur (0.4-35%) after intracavernosal injection of erectogenic agents [1395,1399,1407-1409]. The risk is higher with papaverine-based combinations [1410], while the risk of priapism is < 1% following prostaglandin E1 injection [1411].
Second-generation antipsychotics (33.8%), other medications (11.3%) and alpha-adrenergic antagonists (8.8%) accounted for the greatest percentage of published drug-induced priapism cases [1412]. Isolated cases of priapism have been described in males who have taken PDE5Is [1395]. Data from the FDA Adverse Reporting System Public Dashboard showed that PDE5Is-induced priapism accounted for only 2.9% of drug-induced priapism. Most of these males also had other risk factors for priapism and it is unclear whether PDE5Is per se can cause ischaemic priapism [1395,1413]. Since most males who experience priapism following PDE5Is treatment have additional risk factors for ischaemic priapism, PDE5Is use is usually not regarded as a risk factor in itself. In terms of haemoglobinopathies, SCD is the most common cause of priapism in childhood, accounting for 63% of cases. It is the primary aetiology in 23% of adult cases [1411].
It has been reported that males with a history of priapism exhibit a higher incidence of CV and cerebrovascular disease. Specifically, they had a 24% increased risk of ischaemic heart disease, a 31% increased risk of cerebrovascular disease, and a 15% increased risk of arterial disease compared to males with ED in the years following a priapism episode. Moreover, individuals with recurrent episodes showed progressively higher rates of vascular comorbidities [1414].
Mechanisms of SCD-associated priapism may involve derangements of several signalling pathways in the penis [1415]. Contrary to traditional belief, maintenance of physiological testosterone levels does not cause priapism, but rather preserves penile homeostasis and promotes normal EF [1416,1417]. Testosterone deficiency is considered a controversial risk factor: it is prevalent in patients with SCD, but evidence indicates that it may not be a risk factor for priapism [1418].
Priapism resulting from metastatic or regional infiltration by tumour is rare and usually reflects an infiltrative process, more often involving the bladder and prostate as the primary cancer sites [1419]. In a large retrospective study including 412 males with ischaemic priapism, 11 (3.5%) had malignant priapism, of which seven cases were a consequence of local invasion, while the others were secondary to haematological malignancy [1420]. The conventional therapeutic recommendations for pharmacological treatment are unlikely to be effective, and all these males should have MRI of the penis and be offered supportive care and medical intervention for their primary cancer. In selected cases where palliative treatment options fail to control penile pain, a palliative penectomy can be considered.
Partial priapism, or idiopathic partial segmental thrombosis of the corpus cavemosum, is a rare condition. It is often classified as a subtype of priapism limited to a single crura without ischaemia, but rather a thrombus is present within the corpus cavernosum. Its aetiology is unknown, but bicycle riding, trauma, drug use, sexual intercourse, haematological diseases and α-blocker intake have all been associated with partial segmental thrombosis [1421]. The presence of a congenital web within the corpora is also a risk factor [1422].
Table 10.1: Aetiological factors for the development of priapism
| Aetiological factors for the development of priapism |
| Idiopathic |
| - |
| Haematological dyscrasias, vascular and other disorders |
|
| Infections (toxin-mediated) |
|
| Metabolic disorders |
|
| Neurogenic disorders |
|
| Neoplasms (metastatic or regional infiltration) |
|
| Medications |
|
10.1.1.a. Summary of evidence on the epidemiology, aetiology and pathophysiology of ischaemic priapism
| Summary of evidence | LE |
| Ischaemic priapism is the most common type, accounting for more than 95% of all cases. | 1b |
| Ischaemic priapism is identified as idiopathic in most patients, while SCD is the most common cause in childhood. | 1b |
| Ischaemic priapism occurs relatively often (about 5%) after intracavernous injections of papaverine-based combinations, while it is rare (< 1%) after prostaglandin E1 monotherapy. | 2a |
| Priapism is rare in males who have taken PDE5Is, with only sporadic cases reported. | 4 |
10.1.2. Diagnostic evaluation
10.1.2.a. History
Taking a comprehensive history is critical in priapism diagnosis and treatment [1395,1424]. The medical history must specifically enquire about SCD or any other haematological abnormality [1425,1426] and a history of pelvic, genital or perineal trauma. The sexual history must include the duration of the erection; the presence and degree of pain; prior drug treatment and recreational drug use; history of priapism and methods of treatment; and EF prior to the last priapism episode [1395]. The history can help to determine the underlying priapism subtype (Table 10.2). Ischaemic priapism is classically associated with progressive penile pain and the erection is rigid. Conversely, non-ischaemic priapism is often painless, and the erection often fluctuates in rigidity.
Table 10.2: Key findings in priapism (adapted from Broderick et al., [1395])
| Ischaemic priapism | Non-ischaemic priapism | |
| Corpora cavernosa fully rigid | Typical | Seldom |
| Penile pain | Typical | Seldom |
| Abnormal penile blood gas | Typical | Seldom |
| Haematological abnormalities | Sometimes | Seldom |
| Recent intracavernosal injection | Sometimes | Sometimes |
| Perineal trauma | Seldom | Typical |
10.1.2.b. Physical examination
In ischaemic priapism, the corpora are fully rigid and tender, but the glans penis is soft. The patient usually complains of severe pain. Pelvic examination may reveal an underlying pelvic or genitourinary malignancy [1420].
10.1.2.c. Laboratory testing
Laboratory testing should include a complete blood count, white blood cell count with blood cell differential, platelet count, and coagulation profile to assess anaemia and detect haematological abnormalities [1395,1424].
Aspiration of blood from the corpora cavernosa is compulsory as an entry level investigation. It usually reveals dark ischaemic blood. Blood gas analysis is essential to differentiate between ischaemic and non-ischaemic priapism (Table 10.3). Further laboratory testing should be directed by the history, clinical examination and laboratory findings. These may include specific tests (e.g. haemoglobin electrophoresis) for diagnosis of SCD or other haemoglobinopathies.
10.1.2.d. Penile imaging
Colour Doppler US of the penis and perineum is recommended after clinical diagnosis and can differentiate ischaemic from non-ischaemic priapism as an alternative or adjunct to blood gas analysis (Figure 10.1) [1427-1430]. Colour Doppler US can identify the presence of the fistula as a blush with 100% sensitivity and 73% specificity [1429,1431].
Penile US should be performed before corporal blood aspiration in ischaemic priapism to prevent aberrant blood flow which can mimic a non-ischaemic or reperfusion picture after intervention for low-flow priapism [1432].
Penile MRI can be used in the diagnostic evaluation of priapism and may be helpful in select cases of ischaemic priapism to assess the viability of the corpora cavernosa and the presence of penile fibrosis. In cases of refractory priapism or delayed presentation (> 48 hours), smooth muscle viability can be indirectly assessed. In a prospective study of 38 patients with ischaemic priapism, the sensitivity of MRI in predicting non-viable smooth muscle was 100% when correlated with corpus cavernosum biopsies [1432]. All patients with viable smooth muscle on MRI-maintained EF on clinical follow-up, with the non-viable group being offered early prosthesis.
Table 10.3: Typical blood gas values (adapted from Broderick et al., [1395])
| Source | pO2 (mmHg) | pCO2 (mmHg) | pH |
| Normal arterial blood (room air) (similar values are found in arterial priapism) | > 90 | < 40 | 7.40 |
| Normal mixed venous blood (room air) | 40 | 50 | 7.35 |
| Ischaemic priapism (first corporal aspirate) | < 30 | > 60 | < 7.25 |
pCO2 = partial pressure of carbon dioxide; pO2 = partial pressure of oxygen.
Figure 10.1: Differential diagnosis of priapism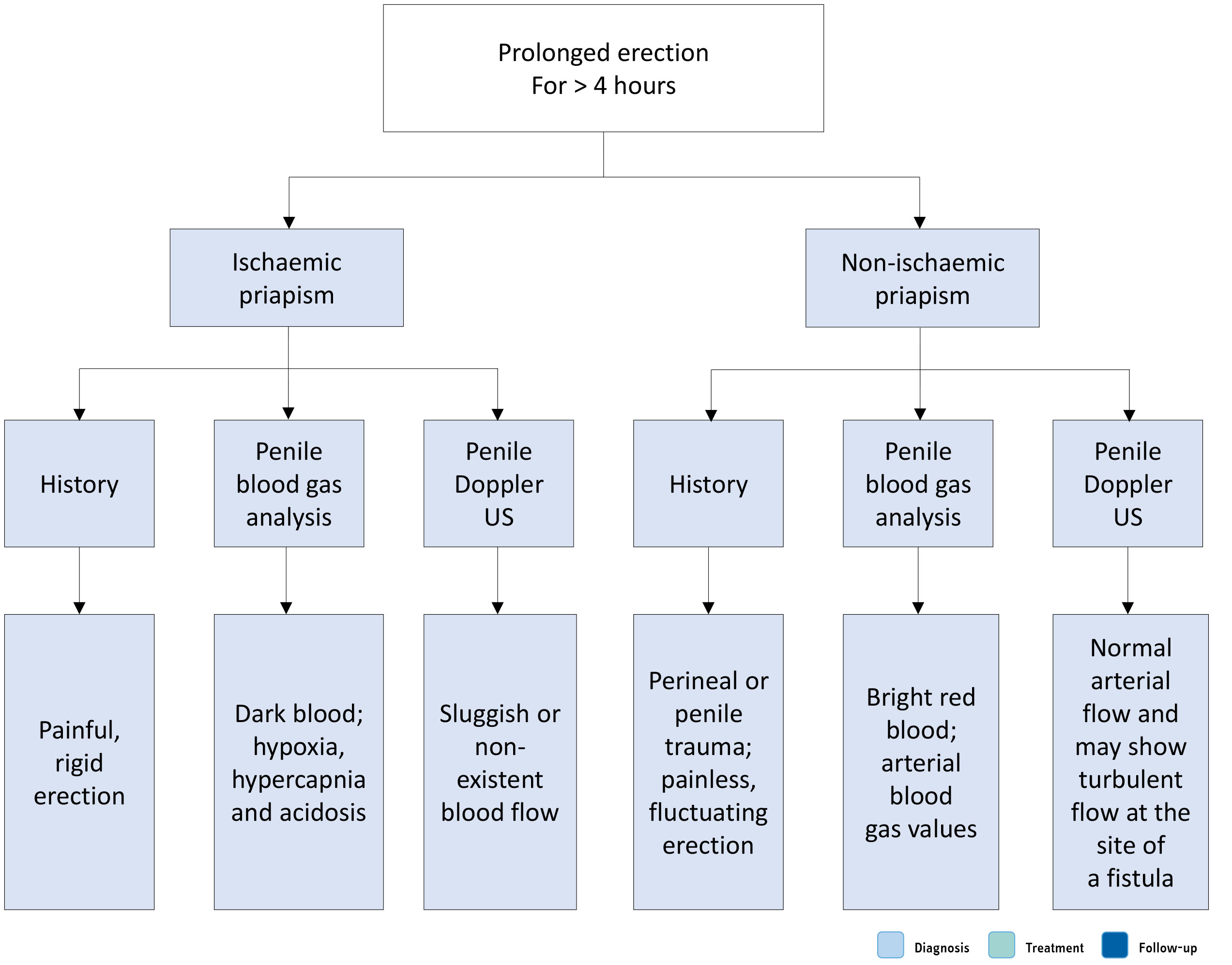 US = ultrasound.
US = ultrasound.
10.1.2.e. Summary of evidence and recommendations for the diagnosis of ischaemic priapism
| Summary of evidence | LE |
| Medical history, including the assessment of known haematological abnormalities (e.g. SCD), history of pelvic/perineal/genital trauma, prior drug treatment or recreational drug use, is essential to identify the possible aetiology and the type of priapism. | 3 |
| Blood gas analysis performed before blood aspiration from the corpora can differentiate between ischaemic and non-ischaemic priapism. Full blood count and haemoglobinopathy screen could reveal haematological alterations. | 3 |
| Penile colour Doppler US can differentiate between ischaemic and non-ischaemic priapism when performed before corporal blood aspiration. | 3 |
| Penile MRI can predict non-viable smooth muscle in patients with ischaemic priapism. | 3 |
| Recommendations | Strength rating |
| Take a comprehensive history to establish the diagnosis which can help to determine the priapism subtype. | Strong |
| Include a physical examination of the genitalia, perineum and abdomen in the diagnostic evaluation. | Strong |
| Include a full blood count, white blood cell count with blood cell differential, platelet count, and coagulation profile for laboratory testing. Perform further directed laboratory testing depending upon history and clinical and laboratory findings. Perform a complete evaluation of all possible causes of priapism in children. | Strong |
| Perform a haemoglobinopathy screen in patients with low-flow priapism who are at high risk of sickle cell disease or thalassemia. | Strong |
| In the emergency setting, analysis of blood gas parameters from aspirated corporal blood is essential to distinguish between ischaemic and non-ischaemic priapism. | Strong |
| Perform colour duplex ultrasound of the penis and perineum before aspiration to differentiate between ischaemic and non-ischaemic priapism. | Strong |
| Use magnetic resonance imaging of the penis in cases of prolonged ischaemic priapism or refractory priapism, and as an adjunct to predict smooth muscle viability. | Weak |
10.1.3. Disease management
Acute ischaemic priapism is a medical emergency. Urgent intervention is mandatory and should follow a stepwise approach. The aim of any treatment is to restore penile detumescence, without pain, to prevent corporal smooth muscle fibrosis and subsequent ED.
10.1.3.a. Medical management – first-line treatment
First-line medical treatments for ischaemic priapism lasting more than four hours are strongly recommended before any surgical treatment. Conversely, first-line treatments initiated beyond 48 hours, while relieving priapism, have little documented benefit on long-term EF preservation. This is likely a consequence of irreversible smooth muscle hypoxia and damage, which begin to develop approximately 48 hours after the onset of ischaemia [1399,1400,1402]. In a series of 50 patients with low-flow priapism who were successfully treated and followed-up for a mean of 66 months, those with priapism lasting for more than 48 hours had a significant risk of ED [1399].
Historically, several first-line treatments have been described including exercise, ejaculation, ice packs, cold baths and cold-water enemas [1395]. The evidence supporting the use of these conservative measures is limited, and their application may potentially worsen the condition in patients with SCD. Success rates achieved with conservative management alone are seldom reported. In a small case series, cold-water enemas were associated with detumescence in six of ten cases [1433]. In another study, 24.5% of 122 patients achieved detumescence following priapic episodes lasting for more than six hours by cooling of the penis and perineum, and walking upstairs [1434].
10.1.3.a.1. Penile anaesthesia/analgesia
Blood aspiration and intracavernous injection of a sympathomimetic agent can be performed without any anaesthesia; however, anaesthesia may be necessary when there is severe penile pain. Whilst anaesthesia may not alleviate the ischaemic pain, cutaneous anaesthesia facilitates subsequent therapies. The treatment options for penile anaesthesia/systemic analgesia include:
- dorsal nerve block;
- circumferential penile block;
- subcutaneous local penile shaft block; and
- oral conscious sedation (for paediatric patients).
10.1.3.a.2. Aspiration ± irrigation with 0.9% w/v saline solution
The first intervention for an episode of priapism lasting more than four hours consists of corporal blood aspiration to drain the stagnant blood from the corporal bodies, making it possible to relieve the compartment-syndrome-like condition within the corpus cavernosum. Blood aspiration may be performed with intracorporeal access either through the glans or via percutaneous needle access to the lateral aspect of the proximal penile shaft, using a 16 or 18G angio-catheter or butterfly needle. The needle must penetrate the skin, the subcutaneous tissue and the tunica albuginea to drain blood from the corpus cavernosum.
Some clinicians advocate using two angiocatheters or butterfly needles at the same time to accelerate drainage, as well as aspirating and irrigating simultaneously with a saline solution [1434]. Aspiration should be continued until bright red, oxygenated blood is aspirated.
Several case series have reported outcomes for first-line treatments. In most cases, aspiration and irrigation were combined with intracavernosal injection of sympathomimetic agents [1393], thus making it difficult to draw conclusions about the success rate of aspiration plus irrigation alone [1393]. Overall, case series and retrospective studies reported a success rate ranging from 0 to 100% of cases [1393]. In an RCT, 70 patients with ischaemic priapism lasting more than six hours, secondary to intracavernosal injection and were treated with aspiration plus saline irrigation at different temperatures [1434]. The study reported an 85% success rate, with the optimum results achieved using a 10°C saline infusion after blood aspiration.
There are insufficient data to determine if aspiration followed by saline intracorporeal irrigation is more effective than aspiration alone.
10.1.3.a.3. Aspiration ± irrigation with 0.9% w/v saline solution in combination with intracavernous injection of pharmacological agents
This combination is considered the standard of care for treatment of ischaemic priapism [1395,1435,1436]. Pharmacological agents include sympathomimetic drugs or α-adrenergic agonists. Intracavernous sympathomimetic agents include phenylephrine, etilefrine, ephedrine, epinephrine, norepinephrine and metaraminol with a resolution rate of up to 80% [1395,1435,1437-1444]. The use of intracavernous adrenaline injection alone has also been sporadically reported [1375]. It has been reported that the use of a sympathomimetic agent combined with prior intracavernosal aspiration or irrigation had a resolution ranging from 80 to 100% of cases as compared with 58% in those who had a sympathomimetic injection alone [1393,1436].
The potential TRAEs of intracavernous phenylephrine (and other sympathomimetic agents) include headache, dizziness, hypertension, reflex bradycardia, tachycardia and palpitations, and sporadic subarachnoid haemorrhage [376]. Monitoring of blood pressure and pulse should be performed during intracavernous administration of sympathomimetic agents. As intracavernous sympathomimetic agents can cause hypertension, the Panel is of the opinion that these agents are contraindicated in patients with malignant or poorly controlled hypertension, as there are case reports of significant CV and neurological complications following the use of these pharmacological agents for priapism [1438,1445,1446]. There are data suggesting that sympathomimetic agents cause a hypertensive crisis when administered with monoamine oxidase inhibitors; therefore, these medications should not be used concurrently [1447].
10.1.3.a.4. Intracavernosal and oral pharmacological agents
Pharmacological agents for the treatment of priapism are discussed in more detail in the following section. Table 10.4 summarises dosing and administration of these agents.
Phenylephrine
Phenylephrine is a selective α-1-adrenergic receptor agonist that has been observed in small case series to induce detumescence in priapism when administered as an intracavernosal injection, with few TRAEs [1443,1448]. Phenylephrine is the recommended adrenergic agonist drug of choice, due to its high selectivity for the α-1-adrenergic receptor without concomitant β-mediated inotropic and chronotropic cardiac effects [1437,1441,1442].
Phenylephrine has potential CV adverse effects [1395,1435,1437,1438,1441,1442,1449] and blood pressure and pulse must be monitored every 15 minutes for one hour after injection. This is particularly important in older males with pre-existing CVD. After injection, the puncture site should be compressed and the corpus cavernosum massaged to facilitate drug distribution.
Etilefrine
Etilefrine is also an adrenergic agonist that directly stimulates both α and ß adrenergic receptors [1436]. Most of the literature describing the use of etilefrine for treatment of priapism is related to males with SCD, but there are small retrospective case series that have reported its benefits for priapism secondary to iatrogenic causes [1450,1451]. Etilefrine is the second most widely used sympathomimetic agent [1438].
Methylene blue
Methylene blue is a guanylate cyclase inhibitor that may be a potential inhibitor of endothelial-mediated cavernous smooth muscle relaxation. Small retrospective case series have reported its successful use for treating short-term pharmacologically induced priapism [1452,1453]. Treatment-related adverse effects include a transient burning sensation and blue discolouration of the penis.
Adrenaline
Adrenaline produces both α-adrenergic receptor agonist and ß-adrenergic receptor activity. Intracavernosal adrenaline has been used in patients with ischaemic priapism due to an intracavernous injection of vasoactive agents. The limited literature [1454,1455] suggests that adrenaline can achieve detumescence in short-term priapism, with one small case series reporting a success rate of over 50% after a single injection, and an overall success rate of 95% after repeated injections [1454,1455].
β-2-agonist
Oral terbutaline is a β-2-agonist with minor β-1 effects and some α-agonist activity, although its mechanism of action is not yet fully understood [1456-1458]. The main use of terbutaline is for prevention of recurrent episodes of prolonged erection. Oral treatment with terbutaline was tested in three placebo-controlled RCTs [1457-1459], showing a success rate of 30 to 60% in patients with ischemic priapism associated with intracavernous injection of erectogenic agents. Terbutaline should be given cautiously in patients with coronary artery disease, increased intravascular fluid volume, oedema or hypokalaemia [1458]. In a single multi-centre prospective study, another β-2-agonist, salbutamol, has been reported to induce detumescence in 34% of cases of prolonged erection (more than three hours) after intracavernous injection of erectogenic agents [1460]. However, more robust data are needed to recommend oral salbutamol for the treatment of ischaemic priapism.
Table 10.4: Medical treatment of ischaemic priapism
| Drug | Dose/Instructions for use |
| Phenylephrine |
|
| Etilefrine |
|
| Methylene blue |
|
| Adrenaline |
|
| Terbutaline |
|
10.1.3.a.5. Management of priapism related to sickle cell disease
The results of a systematic review on the overall management of priapism related to SCD found that few studies were conducted exclusively on patients with SCD and studies on mixed populations usually did not report separate data on SCD patients [1394]. Clear and systematic reporting of patient characteristics, interventions and outcomes was lacking, and the length of follow-up, if reported, varied significantly among the studies. Overall, the quality of studies was deemed too poor to support high-quality, evidence-based recommendations.
Urgent intervention is essential for preservation of long-term sexual function [1461]. The general approach is similar to that described for other cases of ischaemic priapism and should be co-ordinated with a haematologist [1462-1464].
As with other haematological disorders, other therapeutic interventions may also need to be implemented [1462,1464,1465]. Specific measures for SCD-related priapism include intravenous hydration and narcotic analgesia while preparing the patient for aspiration and irrigation. Additionally, supplemental oxygen administration and alkalinisation with bicarbonate can be helpful [1415,1463].
Haemoglobin S percentage should be measured in all SCD patients with acute priapism. Exchange blood transfusion has also been proposed, with the aim of increasing tissue delivery of oxygen [1466]. The transfused blood should be sickle cell haemoglobin negative and Rh and Kell antigen matched [1467]. The evidence is inconclusive as to whether exchange transfusion itself helps to resolve priapism. A systematic review reported that the mean time to detumescence was 11 days with exchange transfusions compared to eight days with conventional treatment. Moreover, there were nine cases of ASPEN syndrome (association of SCD, priapism, exchange transfusion and neurological events) as a consequence of blood transfusion [1468].
A series of ten patients with SCD-related priapism reported that it was safe to perform exchange transfusion [1466]; however, several reports suggest that exchange transfusion may result in serious neurological sequelae [1468]. Therefore, routine use of exchange transfusion is not recommended as a primary treatment intervention in this group unless there is a risk of SCD-related symptoms. In patients who failed medical management, transfusion may be required to enable general anaesthesia to be safely administered prior to definitive surgery [1469-1471].
Crizanlizumab 5.0mg/kg IV infusion at week one, week three, and every four weeks thereafter has been reported to reduce veno-occlusive and priapism attacks in SCD patients up to 61% [1472].
10.1.3.b. Surgical management - second-line treatments
A number of clinical indicators suggest failure of first-line treatment, including continuing corporal rigidity, cavernosal acidosis, anoxia, severe glucopenia, absence of cavernosal artery inflow by penile colour duplex US, and elevated intracorporal pressure [1473].
Second-line intervention typically refers to surgical intervention in the form of penile shunt surgery and penile implant insertion for refractory or delayed ischaemic priapism and should only be considered when other medical management options have failed.
No definitive evidence defines the timing for surgical escalation after first-line therapy in ischemic priapism. A period of at least one hour of unsuccessful initial treatment may be considered before proceeding to surgery. Higher cumulative doses of phenylephrine and prolonged priapism duration have been associated with increased need for shunt procedures [1474], but prospective validation is lacking.
10.1.3.b.1. Penile shunt surgery
Penile shunt surgery aims to produce an outflow for ischaemic blood from the corpus cavernosum into the corpus spongiosal tissues, thereby allowing restoration of normal circulation within these structures. A shunt creates an opening in the tunica albuginea, with either the glans, corpus spongiosum, or a vein for blood drainage [1395,1435,1475].
The type of shunt procedure is chosen according to the surgeon’s preference and familiarity with the procedure. It is conventional practice for distal shunt procedures to be tried before considering proximal shunting.
It is important to assess the success of surgery by direct observation of penile rigidity or by repeated testing (e.g. cavernous blood gas testing) [1395,1435,1476,1477]. The use of penile colour US may not give appropriate information because of the hyperaemic (reperfusion) period that follows decompression after the ischaemic state [1478].
The recovery rates of EF in males undergoing shunt surgery following prolonged episodes of priapism are low and are directly related to the duration of priapism, preoperative erectile status and age [1476,1477,1479]. If ischaemic priapism resolves within 24 hours of onset, it has been reported that 78-100% of patients regain spontaneous functional erections (with or without PDE5Is use). In contrast, other studies have shown that priapism for more than 36-48 hours appears to result in both structural and functional effects on corporal smooth muscle, with poorer outcomes (ED > 90%) [1476,1480]. In general, shunt procedures undertaken after this time period (36-48 hours) may only serve to limit pain without any beneficial effects on EF and early PPI can be considered [1402,1481,1482].
Procedures for shunting require an incision through the tunica albuginea, which exposes collagen to coagulation factors in the penile blood and thereby activates the blood-clotting cascade. Perioperative anti-coagulation is advocated to facilitate resolution of the priapism. There was an 84% decrease in priapism recurrence in the shunt group that received peri-procedural anti-thrombotic treatment (325mg acetylsalicylic acid preoperatively; 5000IU intra-operative heparin; 81mg acetylsalicylic acid and 75mg clopidogrel postoperatively for five days) compared with the group that did not receive peri-procedural anti-thrombotic treatment after failed aspiration [1483].
Four categories of shunt procedures have been reported [1395,1436,1475,1481,1482,1484]. The limited data available does not allow one procedure to be recommended over another. However, distal shunts are less invasive and associated with lower rates of postoperative ED and are recommended as the first surgical intervention of choice (Appendix 6 online supplementary evidence Table S10.1).
Percutaneous distal (corpora-glanular) shunts
Winter’s procedure uses a Trucut biopsy needle to create a fistula between the glans penis and each corpus cavernosum [1395,1411,1436,1478,1485]. Postoperative sequelae are uncommon [1486]. Winter’s shunt is easy to perform, but has been reported as the least successful operation to create a distal shunt [1477]. This is because the diameter of the Trucut needle is only 1.6mm (14-18g) and therefore cannot accommodate the increased blood flow from post-ischaemic hyperaemia, resulting in poor drainage, increased intracavernous pressure, and consequent premature closure of the shunt [1478].
Ebbehoj’s technique involves making multiple tunical incision windows between the glans and each tip of the corpus cavernosum by means of a size 11 blade scalpel passed several times percutaneously [1395,1436,1478,1487,1488].
T-shunt involves performing a bilateral procedure using a scalpel with a size 10 blade inserted through the glans just lateral to the urethral meatus until it enters the tip of the corpus cavernosum. The blade is then rotated 90° away (to the lateral side) from the urethral meatus and withdrawn [1395,1436,1478,1489]. If unsuccessful, the procedure is repeated on the opposite side. The T-shunt can be followed by a tunnelling procedure using a size 8/10 Hegar dilator inserted through the glans and into the corpus cavernosum, which can also be performed using US guidance, mainly to avoid urethral injury [1489]. The entry sites in the glans are sutured following detumescence. Tunnelling with a 7mm metal sound or 7/8 Hegar dilator is necessary in patients with priapism duration > 48 hours. Tunnelling is a potentially attractive procedure as it combines the features of distal and proximal shunts with proximal drainage of the corpus cavernosum and may ameliorate the profibrotic effect of sludged blood retained in the corpus cavernosum [1479,1481,1489].
Open distal (corpora-glanular) shunts
Al-Ghorab’s procedure consists of an open bilateral excision of circular cone segments of the distal tunica albuginea via the glans penis, along with subsequent glans closure by running suture with absorbable material. A transverse incision on the glans may compromise arterial blood flow because distal deep dorsal arteries run longitudinally in the glans [1395,1436,1478,1490-1492].
Burnett’s technique (Snake manoeuvre) is a modification of the Al-Ghorab corpora-glanular shunt. It involves retrograde insertion of a 7/8 Hegar dilator into the distal end of each corpus cavernosum through the original Al-Ghorab glandular excision. After removal of the dilator from the corpus cavernosum, blood evacuation is facilitated by manual compression of the penis sequentially from a proximal to distal direction. After detumescence, the glans penis is closed as in the Al-Ghorab procedure [1395,1436,1478,1493-1495]. Reported complications include wound infection, penile skin necrosis and urethrocutaneous fistulae [1494].
Open proximal (corpora-glanular) shunts
Quackles’s technique uses a trans-scrotal or perineal approach; a proximal open shunt technique creates a communication between the corpus cavernosum and the corpus spongiosum. The most frequent complications include an unwanted urethro-cavernous fistula and urethral stricture or cavernositis [1395,1436,1475,1496]. The risk of urethral injury is less with a perineal approach to the bulb of the corpus spongiosum. Proximal shunts are more invasive and ED rates are documented to be higher [1473].
Peno-scrotal decompression
A proximal decompression technique with the aim to spare the glans with high success rates has been described. The technique is based upon opening of the proximal corpus cavernosum combined with proximal and distal tunnelling using a suction tip [1497]. In a cohort of 25 patients, 12 had undergone previous corpora-glanular shunt surgery. Recurrence was observed in two of 25 patients with unilateral peno-scrotal decompression. In the 15 patients who had follow-up data, 40% had ED. A 2025 systematic review by Mubarak et al., including 62 patients, reported PSD achieved detumescence in ~89%, with EF preservation in ~55%. Nearly half had failed prior distal shunts, supporting PSD as a secondary/salvage option rather than first-line therapy. While promising, evidence remains limited and prospective studies are required [1498]. A 2025 multicentre study by Basile et al., challenged this concept, reporting high detumescence (96%) and acceptable EF preservation, even when PSD was used as a primary intervention within 36 hours of priapism onset, suggesting it may represent an early-line alternative rather than solely a salvage procedure [1499]. While promising, evidence remains limited and prospective studies are required. Whilst representing a promising technique, PSD in cases of refractory priapism may further delay PPI with potential detrimental effects on surgical outcomes, including penile shortening and prosthetic infection.
Vein anastomoses/shunts
Grayhack’s procedure mobilises the saphenous vein below the junction of the femoral vein and anastomoses the vein end-to-side onto the corpus cavernosum. Venous shunts may be complicated by saphenofemoral thrombus formation and by pulmonary embolism [1395,1436,1500-1502].
10.1.3.b.2. Immediate penile prosthesis implantation
The studies pertaining to PPI surgery are principally retrospective non-randomised case series (Table S10.3 Appendix 6 online supplementary evidence). All the studies described priapism resolution rate, sexual function and surgical adverse events, although the follow-up period was variable [1392].
Refractory, therapy-resistant, acute ischaemic priapism or episodes lasting more than 48 hours usually result in complete ED, and possibly significant penile deformity in the long-term. In these cases, immediate PPI surgery is advocated [1482,1484,1503-1506].
Gadolinium-enhanced penile MRI [1432] and cavernosal smooth muscle biopsy have been used to diagnose smooth muscle necrosis (which, if present, would suggest that shunting is likely to fail) and may help in decision-making and patient counselling in cases of refractory or delayed presentation (> 48 hours) that may be considered for immediate PPI.
Early PPI is associated with lower infection rates (6-7% vs. 19-30%), penile shortening (3% vs. 40%) and revision rates (9% vs. 27%) compared to late insertion. General satisfaction rate for PPI is higher (96%) than for late implantation (60%) [1402,1507] (Table S10.4 Appendix 6 online supplementary evidence). Potential complications that could compromise immediate PPI include distal erosion and infection [1503,1505], along with a small rate of revision surgery [1503]. Early surgery also offers the opportunity to maintain penile length and girth and prevent penile curvature due to cavernosal fibrosis. The prosthesis can be exchanged for an inflatable prosthesis at a later date, which may allow upsizing of the implant cylinders [1508].
The decision on which type of implant to insert is dependent on patient suitability, surgeon experience, and availability and cost of the equipment. The immediate insertion of a malleable penile prosthesis is recommended to avoid the difficulty and complications of delayed prosthetic surgery in the presence of corporal fibrosis.
There are no randomised trials comparing the efficacy and complication rates of malleable and inflatable penile prostheses. Despite the higher infection rate in priapism patients compared to those with virgin prosthesis, immediate inflatable PPI may be undertaken in patients who are well motivated and counselled prior to the procedure [1482,1509]. In most cases, a semi-rigid implant is more suitable, as it is easier to implant and reduces operative time and, consequently, the risk of prosthetic infection. A further issue with immediate insertion of an inflatable penile prosthesis is that the patient must begin cycling the device immediately to avoid a fibrous capsule forming and contracting. Early cycling of an inflatable penile prosthesis prevents penile curvature and shortening [1402]. Early inflatable PPI appears to reduce complications and improve satisfaction compared with delayed procedures, although current evidence is mainly retrospective [1510].
There are no clear indications for immediate PPI in men with acute ischaemic priapism, although this can be considered in men with delayed or refractory priapism [1435,1484].
Relative indications include [1395]:
- Ischaemia that has been present for more than 48 hours.
- Failure of aspiration and sympathomimetic intracavernous injections in delayed priapism (> 48 hours).
- Corporal biopsy or MRI evidence of corporal smooth muscle necrosis [1395,1503].
- Failure of a shunting procedure; although, in delayed cases (> 48 hours), implantation might be considered ahead of shunt surgery.
- Refractory priapism in patients who have undergone shunting procedures.
The optimal time for PPI is within the first three weeks from the priapism episode [1402,1473,1511]. If shunt surgery has been performed, PPI can be further delayed to allow reduction of oedema, wound healing and risk of prosthetic infection. A vacuum device to avoid fibrosis and penile shortening may be used during this waiting period [1512].
10.1.3.b.3. Surgery for non-acute sequelae after ischaemic priapism
Structural changes may occur after ischaemic priapism including cavernosal tissue necrosis and fibrosis with consequent penile scarring, megalophallic deformities, penile shortening, and occasional penile loss [1475,1503,1513,1514]. Erectile dysfunction is also often observed [1395,1515]. These outcomes can occur despite apparently successful first- or second-line treatment in detumescence of the penis.
Penile prosthesis implantation is occasionally indicated in SCD patients with severe ED because other therapeutic options, such as PDE5Is and intracavernous injections, are avoided as they may provoke a further priapism event [1395,1435]. In severe corporal fibrosis, narrow-based prosthetic devices are preferable because they are easier to insert and need less dilatation [1503]. After severe priapism that has resulted in penile destruction with complicated deformities or even loss of penile tissue, it may be necessary to make changes to the surgical technique. Multiple corporotomies, corporal excavation, optical corporotomy-Shaeer technique, dilatation with Carrion-Rosello, Uramix or Mooreville cavernotomes, excision of scar tissue, and the use of a small-diameter prosthesis or penile reconstruction using grafts can be utilised if concomitant PPI is considered [1480,1516].
Figure 10.2: Management work-up of ischaemic priapism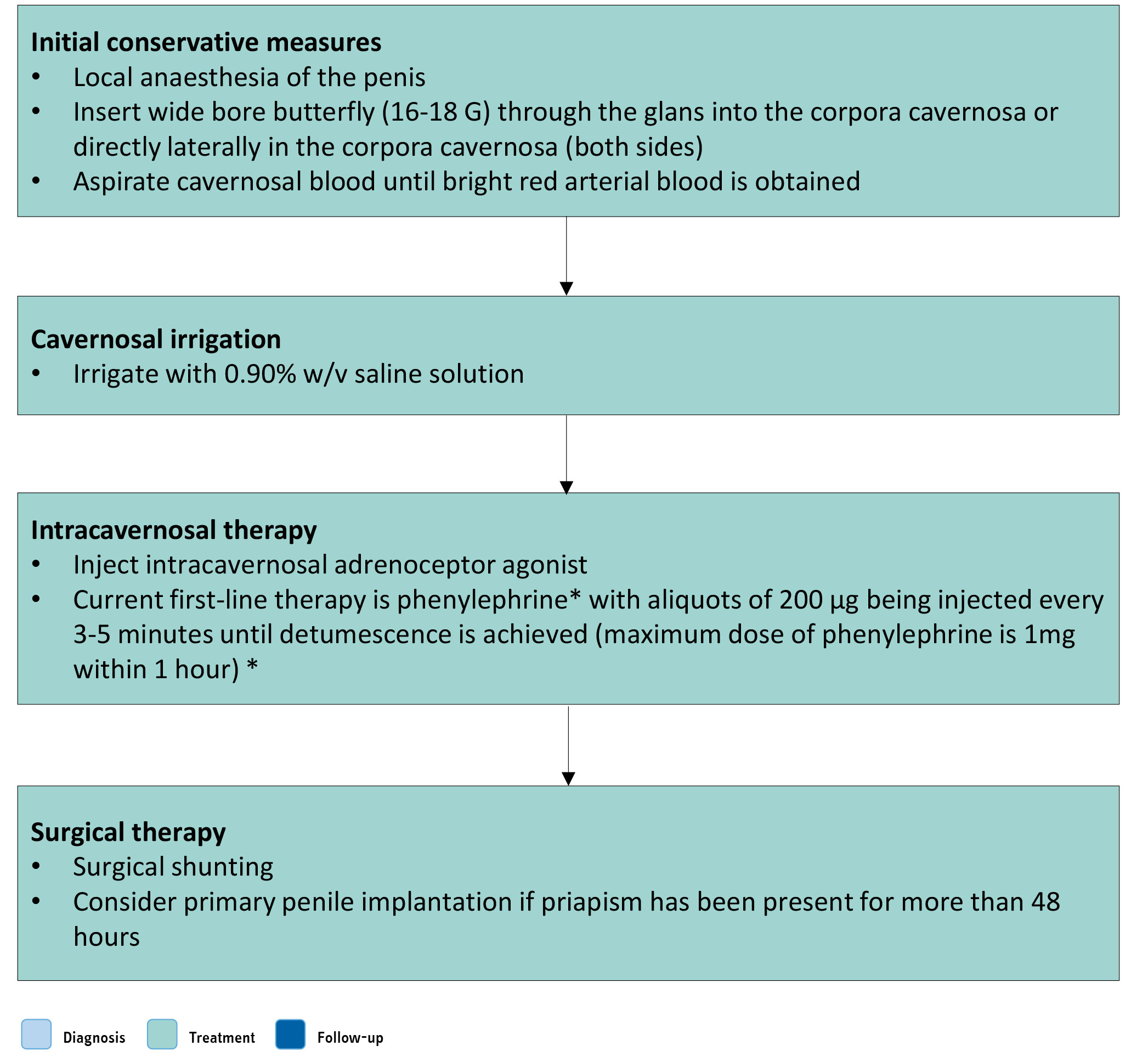 (*) Dose of phenylephrine should be reduced in children. It can result in significant hypertension and should be used with caution in men with cardiovascular disease. Monitoring of pulse and blood pressure is advisable in all patients during administration and for one hour afterwards. Its use is contraindicated in men with a history of cerebro-vascular disease and significant hypertension.
(*) Dose of phenylephrine should be reduced in children. It can result in significant hypertension and should be used with caution in men with cardiovascular disease. Monitoring of pulse and blood pressure is advisable in all patients during administration and for one hour afterwards. Its use is contraindicated in men with a history of cerebro-vascular disease and significant hypertension.
Figure 10.3: Surgical management of priapism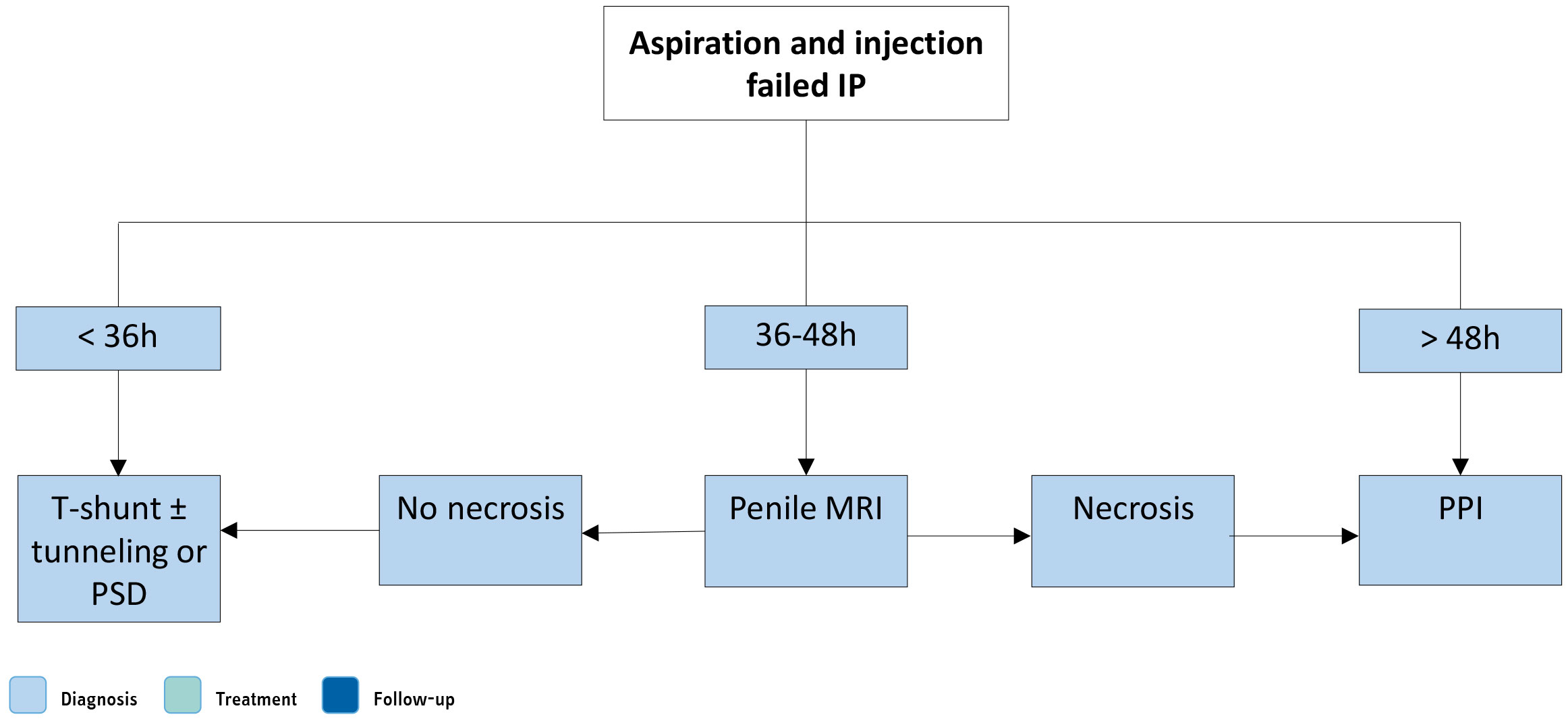 IP = ischaemic priapism; MRI = magnetic resonance imaging; PPI = penile prosthesis implantation.
IP = ischaemic priapism; MRI = magnetic resonance imaging; PPI = penile prosthesis implantation.
10.1.4. Summary of evidence and recommendations for treatment of ischaemic priapism
| Summary of evidence | LE |
| Ischaemic priapism is a medical emergency and immediate intervention is mandatory. | 2b |
| Erectile function preservation is directly related to the duration of ischaemic priapism, age and preoperative erectile status. | 2b |
| Medical treatment is variably effective in case of priapism lasting less than 48 hours. | 2b |
| Aspiration ± irrigation with 0.9% results in over 80% success rate when combined with intracavernous injection of sympathomimetic drugs. | 2b |
| Phenylephrine is the recommended drug due to its favourable safety profile in the CV system compared to other drugs. Phenylephrine is usually diluted in normal saline with a concentration of 100-500μg/mL and given in 200μg doses every three to five minutes directly into the corpus cavernosum. Maximum dosage is 1mg within one hour. Patient monitoring is highly recommended. | 2b |
| Oral terbutaline has a success rate in up to 60% of cases when priapism is associated with intracavernous injection of erectogenic agents. | 1b |
| Exchange transfusion in patients with priapism associated with SCD may result in serious neurological sequelae. | 2b |
| Shunt procedures are effective to resolve priapism and provide pain relief. No clear recommendation of the superiority of one type of shunt over another can be given. Distal shunts are less invasive and associated with lower rate of ED. | 2b |
| Peri- and postoperative anticoagulant prophylaxis (325mg acetylsalicylic acid preoperatively; 5,000IU heparin intra-operatively; and 81mg acetylsalicylic acid and 75mg clopidogrel five days postoperatively) may prevent priapism recurrence. | 3 |
| Erectile dysfunction is almost inevitable in prolonged cases or ischaemic priapism. Early PPI is associated with lower infection rates and complications compared to late implantation. | 2b |
| Recommendations | Strength rating |
| Start management of ischaemic priapism as early as possible (within four to six hours) and follow a stepwise approach. | Strong |
| Perform corpus cavernosum aspiration and washout until fresh red blood is obtained as first treatment step. | Strong |
| Replace blood aspiration with intracavernous injection of a sympathomimetic drug as the first step in priapism secondary to intracavernous injections of vasoactive agents. | Strong |
| Perform intracavernous injection of a sympathomimetic drug in priapism that persists despite aspiration. | Strong |
| Repeat aspiration and intracavernous injection of a sympathomimetic drug in cases that persist despite prior aspiration and intracavernous injection of a sympathomimetic drug, before considering surgical intervention. | Strong |
| Treat ischaemic priapism associated with sickle cell disease in the same fashion as idiopathic ischaemic priapism. Do not use exchange transfusion as a primary treatment. Provide other supportive measures but do not delay initial treatment to the penis. | Strong |
| Proceed to surgical treatment only when blood aspiration and intracavernous injection of sympathomimetic drugs have failed. | Strong |
| Perform distal shunt surgical procedures first and combine them with tunnelling if necessary. | Weak |
| Peno-scrotal decompression may be used as An alternative first-line option instead of distal shunting (with or without tunnelling). | Weak |
| Discuss penile prosthesis implantation in cases of delayed presentation (> 48 hours) and in cases refractory to injection therapy and distal shunting. | Weak |
| Delay implantation of a penile prosthesis if a shunt has been performed, to minimise the risk of infection and erosion of the implant. | Strong |
Decide on which type of implant to insert based on:
If a malleable penile prosthesis is implanted, it can be exchanged for an inflatable penile implant. | Strong |
10.2. Priapism in special situations
10.2.1. Stuttering (recurrent or intermittent) priapism
Stuttering priapism, also termed intermittent or recurrent priapism, is a distinct condition that is characterised by repetitive and painful episodes of prolonged erections. Erections are self-limiting with intervening periods of detumescence [1463,1517]. These are analogous to repeated episodes of ischaemic priapism. In stuttering priapism, the duration of the erections is generally shorter than in ischaemic priapism [1436]. The frequency and/or duration of these episodes are variable, and a single episode can sometimes progress into prolonged ischaemic priapism. Stuttering priapism needs to be differentiated from sleep-related painful erections as both are rare but distinct entities. A prospective cohort study found that although stuttering priapism and sleep-related painful erections share symptoms of painful nocturnal erections and poor sleep, they differ markedly in the characteristics, duration and resolution of nocturnal erections, as well as their penile Doppler US and polysomnographic features [1518].
Robust epidemiological studies of stuttering priapism are lacking [1519,1520]. Recurrent priapism episodes are common in males with SCD (42-64%) [1521,1522], while in adolescents and young men the incidence of priapism is 35%, of whom 72% have a history of stuttering priapism [1519].
The aetiology of stuttering priapism is similar to that of ischaemic priapism. Whilst SCD is the most common cause, idiopathic cases and cases due to a neurological disorder have been reported. Men with acute ischaemic priapism, particularly if prolonged (more than four hours), are at risk of developing stuttering priapism [1515].
Several studies have proposed alternative mechanisms for stuttering priapism, including inflammation, cellular adhesion, nitric oxide metabolism, vascular reactivity and coagulation [1395,1416,1463,1517,1523-1526]. Although debated, androgens have also been observed to have an association with priapism [1527]. One of the options for the treatment of stuttering priapism is to reduce serum testosterone levels to hypogonadal levels, which then suppresses androgen-associated mechanisms believed to be involved in triggering recurrent priapism.
10.2.1.a. Diagnostic evaluation
History, physical examination, laboratory testing and penile imaging follow the same principals as ischaemic priapism. In stuttering priapism there is a history of recurrent episodes of prolonged erections. These episodes may range from several daily to isolated incidents every few months, occurring either continuously or followed by incident-free periods of unknown duration, even months and/or years [1528]. The onset of the priapic episodes usually occurs during sleep and detumescence does not occur upon waking. These episodes can be painful and may be the reason that the patient first seeks medical attention. Erections are painful and the penis is rigid as in ischaemic priapism, but the duration of events is usually shorter. Between erections the penis is usually normal, but in some cases signs of fibrosis can be found. Rarely, the penis may become enlarged, a condition known as megalo-phallus.
Recommendations for the diagnosis of stuttering priapism are the same as those described in Section 10.1.2.e.
10.2.1.b. Disease management
The main goal in the management of stuttering priapism is the prevention of recurrence and the minimization of progression to prolonged ischaemic priapism unresponsive to conventional therapeutic options. In most cases, stuttering priapism may be managed pharmacologically. Acute episodes should be treated according to established protocols for ischaemic priapism, including corporal aspiration and irrigation, with or without the adjunctive use of intracavernous α-adrenergic agonists.
10.2.1.b.1.α-Adrenergic agonists
Studies of oral α-adrenergic agonists have suggested some prophylactic benefit for daily treatment with these agents [1529]. Adverse effects include tachycardia and palpitations. Pseudoephedrine is widely used as an oral decongestant and can be a first-line treatment option for stuttering priapism [1457]. Its effect on corporal smooth muscle is not fully understood. Etilefrine has been used successfully to prevent stuttering priapism caused by SCD. It is usually taken orally at doses of 5-10mg daily, with response rates of up to 72% [1530-1532]. In one RCT, placebo-controlled clinical study comparing medical prophylaxis with etilefrine and ephedrine, there was no difference in efficacy between the two drugs.
10.2.1.b.2. Hormonal manipulations of circulating testosterone
The aim of hormonal manipulation is to down-regulate circulating testosterone levels to suppress the action of androgens on penile erection [1415,1463,1533]. This can be achieved by GnRH agonists or antagonists, antiandrogens or oestrogens [1534,1535]. Potential adverse effects may include hot flushes, gynaecomastia, ED, loss of libido, and asthenia. All approaches have a similar efficacy profile, while the potential CV toxicity of oestrogens limits their clinical use. Alternative endocrine approaches that have been used with some success include 5-α-reductase inhibitors [1536,1537] and ketoconazole (an anti-fungal agent that reduces adrenal and testicular androgen production) [1533,1538].
The duration of hormonal treatment for effective suppression of recurrent priapism is problematic. It is not possible to draw any conclusions on the dose, duration of treatment and the efficacy. Caution is strongly advised when prescribing hormonal treatments to pre-pubertal boys and adolescents, and specialist advice from paediatric endocrinologists should be sought. Likewise, hormonal agents have a contraceptive effect and interfere with normal sexual maturation and spermatogenesis and affect fertility. Men who are trying with their partner to conceive should be comprehensively counselled before starting hormonal treatment. Sperm cryopreservation may be considered to mitigate any potential effects of anti-androgen therapy on fertility.
10.2.1.b.3. Digoxin
Digoxin is a cardiac glycoside and positive inotrope that is used to treat congestive heart failure. Digoxin regulates smooth muscle tone through several different pathways leading to penile detumescence [1415,1463,1539]. The use of maintenance digoxin doses (0.25-0.5mg/daily) in idiopathic stuttering priapism reduces the number of hospital visits and improves QoL [1463]. In a small, clinical, double-blind, placebo-controlled study, digoxin decreased sexual desire and excitement with a concomitant reduction in penile rigidity, regardless of any significant change in plasma levels of testosterone, oestrogens and LH [1539]. Adverse effects include decreased libido, anorexia, nausea, vomiting, confusion, blurred vision, headache, gynaecomastia, rash and arrhythmia.
10.2.1.b.4. Terbutaline
Terbutaline has been used to prevent stuttering priapism with detumescence rates of 36% in patients with alprostadil-induced priapism [1457]. The only RCT (n = 68) in patients with pharmacologically-induced priapism, demonstrated detumescence in 42% of the terbutaline-treated group compared to only 15% in the placebo-treated group [1458]. Adverse effects include nervousness, shakiness, drowsiness, palpitations, headache, dizziness, hot flushes, nausea and weakness.
10.2.1.b.5. Gabapentin
Gabapentin has anticonvulsant, antinociceptive and anxiolytic properties and is widely used as an analgesic and anti-epileptic agent. Its proposed mechanism of action is to inhibit voltage-gated calcium channels, which attenuates synaptic transmission [1533], and reduces testosterone and FSH levels [1540]. It is given at a dose of 400mg, four times daily, up to 2,400mg daily, until complete penile detumescence occurs, with subsequent maintenance administration of 300mg/daily [1541]. Adverse effects include anorgasmia and impaired EF.
10.2.1.b.6. Baclofen
Baclofen is a gamma-aminobutyric acid (GABA) derivative that acts as a muscle relaxant and anti-muscle spasm agent. It can inhibit penile erection and ejaculation through GABA activity and prevents recurrent reflexogenic erections or prolonged erections from neurological diseases [1532]. Oral baclofen has little efficacy, and it is not usually used in stuttering priapism, but intrathecal administration is more effective [1463,1542-1544]. Adverse effects include drowsiness, confusion, dizziness, weakness, fatigue, headache, hypotension and nausea.
10.2.1.b.7. Hydroxyurea
Hydroxyurea blocks the synthesis of deoxyribonucleic acid (DNA) by inhibiting ribonucleotide reductase, which has the effect of arresting cells in the S-phase [1533,1545]. Hydroxyurea is an established treatment for ameliorating SCD and improving life expectancy [1462,1546]. For patients with recurrent priapism, there is limited evidence to suggest a prophylactic role of hydroxyurea [1533,1545,1547]. Adverse effects include oligo-zoospermia and leg ulcers. It was found that sperm concentration, motility, and normal morphology significantly decreased on hydroxyurea therapy but recovered to prehydroxyurea levels three months after therapy cessation [1548].
10.2.1.b.8. Phosphodiesterase type 5 inhibitors
Low doses of PDE5Is have a paradoxical effect in alleviating and preventing stuttering priapism; mainly in patients with idiopathic and SCD-associated priapism [1415,1463,1524,1549-1553]. Randomised data show no added benefit of tadalafil over hydroxyurea alone, underscoring the need for further prospective trials [1548]. Therapy should be started when the penis is in its flaccid state and not during an acute episode. There is a delay of one week before treatment is effective. There are no reported impairments in male sexual function.
10.2.1.b.9. Intracavernosal injections
Some patients with stuttering priapism who have started on systemic treatment to prevent recurrence of unwanted erections, may not see therapeutic benefits immediately and temporarily require intracavernous self-injections at home with sympathomimetic agents [1415,1463]. The most commonly used drugs are phenylephrine and etilefrine [1395,1436,1520,1531].
Tissue plasminogen activator is a secreted serine protease that converts the pro-enzyme plasminogen to plasmin, which acts as a fibrinolytic enzyme. Limited clinical data have suggested that a single intracavernous injection of tissue plasminogen activator can successfully treat patients with recalcitrant priapism [1533,1554]. Mild bleeding is the most observed TRAE.
10.2.1.b.10. Penile prosthesis
Patients with medically refractory stuttering priapism require frequent visits to the emergency department and are always at risk of a major ischaemic episode, which can be mitigated with PPI [1480,1506,1555]. Penile prosthesis for preventing stuttering priapism should not be offered before medical treatment and PPI should be performed only in carefully selected patients as a last resort [1480,1506,1555]. In patients with permanent ED due to stuttering priapism, medical treatments for ED should be used cautiously because of the risk of inducing an ischaemic episode and PPI can be considered [1480,1556].
10.2.1.c. Summary of evidence and recommendations for treatment of stuttering priapism
| Summary of evidence | LE |
| The primary goal in the management of patients with stuttering priapism is prevention of future episodes, which can generally be achieved pharmacologically. | 2b |
| Hormonal therapy with GnRH agonists or antagonists or antiandrogens can reduce the risk of recurrent priapism episodes although it is associated with TRAEs (hot flushes, gynaecomastia, ED, loss of libido, asthenia and infertility). | 3 |
| Phosphodiesterase type 5 inhibitors have a paradoxical effect in alleviating and preventing stuttering priapism, mainly in patients with idiopathic and SCD-associated priapism. | 3 |
| The evidence on use of other systemic drugs (digoxin, α-adrenergic agonists, baclofen, gabapentin and terbutaline, hydroxyurea) is limited. | 3 |
| Recommendations | Strength rating |
| Manage each acute episode according to the recommendations for the treatment of ischaemic priapism (section 10.1.2.e). | Strong |
| Use hormonal therapies (mainly gonadotropin-receptor hormone agonists or antagonists) and/or anti-androgens for the prevention of future episodes in patients with frequent relapses. Do not use these before sexual maturation is reached. | Weak |
| Initiate treatment with phosphodiesterase type 5 inhibitors only when the penis is in its flaccid state. | Weak |
| Use digoxin, α-adrenergic agonists, baclofen, gabapentin or terbutaline only in patients with frequent and uncontrolled relapses. | Weak |
| Use intracavernous self-injections of sympathomimetic drugs at home for treatment of acute episodes on an interim basis until ischaemic priapism has been alleviated. | Weak |
10.2.1.d. Follow-up
Follow-up for stuttering priapism includes history and clinical examination to assess the efficacy of treatment in preventing or alleviating erectile events as well as assessing EF and penile fibrosis.
10.2.2. Priapism in children
The classification of priapism in children is similar to that in adults. In addition to ischaemic, stuttering and non-ischaemic priapism, a fourth type, neonatal priapism, is also described [1395,1557]. Priapism in children is considered rare as no data on its prevalence exist. Sickle cell disease is the major cause of priapism in children, followed by leukaemia (10%), trauma (10%), idiopathic causes (19%), and drugs (5%) [1558]. One study showed that 25% of children experienced SCD-related priapism in a pre-pubertal period [1559]. Another study revealed that 90% of men with SCD had their first priapism episode before age 20 years [1522]. A retrospective study found that ischaemic priapism in children can usually be managed with conservative measures such as oxygenation, intravenous fluids, and medical management. Surgical intervention with shunting procedures is rarely required [1560]. Ultimately, priapism in children should be evaluated and treated in a timely manner, as untreated ischaemic priapism may lead to ED and psychosexual disorders in adulthood [1561]. A multidisciplinary team approach should be utilised with specialist input from haematologists and paediatric endocrinologists.
10.3. Non-ischaemic (high-flow or arterial) priapism
Non-ischaemic priapism is a persistent erection caused by unregulated cavernous arterial inflow [1395]. According to aetiology, non-ischaemic priapism can be categorised into four types: traumatic, neurogenic, iatrogenic and idiopathic in origin.
10.3.1. Epidemiology, aetiology and pathophysiology
Epidemiological data on non-ischaemic priapism are almost exclusively derived from small case series [1395,1429,1470,1562-1564]. Non-ischaemic priapism is significantly less common than the ischaemic type, comprising only 5% of all priapism cases [1395]. The most frequent cause of non-ischaemic priapism is blunt perineal or penile trauma [1565]. The injury results in a laceration in the cavernosal artery or branches, leading to a fistula between the artery and the lacunar spaces of the sinusoidal space [1564]. The resultant increased blood flow leads to a persistent and prolonged erection [1566].
There is often a delay between the trauma and the development of the priapism that may be up to two to three weeks [1567]. This is suggested to reflect either spasm or ischaemic necrosis of the injured artery, with the fistula only developing as the spasm resolves or when the ischaemic segment “blows up”. The priapism typically occurs after a nocturnal erection or an erection related to sexual activity, resulting in the sudden increase of blood flow and pressure in the cavernous arteries [1568]. The patient typically reports an erection that is not fully rigid and is not associated with pain because the venous drainage is not compromised and the penile tissue does not become ischaemic [1569].
Non-ischaemic priapism can occur after acute spinal cord injury, presumably due to loss of sympathetic input leading to predominant parasympathetic input and increased arterial flow [1570]. It is also reported to occur following internal urethrotomy [1571], Nesbit procedure [1572], circumcision [1573], transrectal prostate biopsy [1574], and brachytherapy for PCa [1575]. Some cases have been described following shunting procedures performed for ischaemic priapism due to a lacerated cavernosal artery (conversion of low-flow to high-flow priapism) [1576-1578]. Although SCD is usually associated with ischaemic priapism, occasional cases of high-flow priapism have been reported; however, the pathophysiological mechanism remains unclear [1579]. Finally, metastatic malignancy to the penis can also rarely cause non-ischaemic priapism [1580,1581].
10.3.2. Diagnostic evaluation
10.3.2.a. History
A comprehensive history is mandatory in non-ischaemic priapism diagnosis and follows the same principles as described in section 10.1.2.1. Arterial priapism should be suspected when the patient reports a history of pelvic, perineal, or genital trauma; no penile pain (discomfort is possible); and a persistent, not fully rigid erection. The corpus cavernosum can become fully rigid with sexual stimulation, so sexual intercourse is usually not compromised. The onset of post-traumatic non-ischaemic priapism can be delayed by several hours to weeks following the initial injury [1395].
10.3.2.b. Physical examination
In non-ischaemic priapism, the corpora are tumescent but not fully rigid. Abdominal, penile and perineal examination may reveal evidence of trauma [1395]. Neurological examination is indicated if a neurogenic aetiology is suspected.
10.3.2.c. Laboratory testing
Laboratory testing should include a blood count with white blood cell differential and a coagulation profile to assess for anaemia and other haematological abnormalities. Blood aspiration from the corpus cavernosum shows bright red arterial blood in arterial priapism, while blood is dark in ischaemic priapism. Blood gas analysis is essential to differentiate between non-ischaemic and ischaemic priapism. Blood gas values in high-flow priapism show normal arterial blood [1395] (Table 10.3).
10.3.2.d. Penile imaging
Colour duplex US of the penis and perineum is recommended and can differentiate non-ischaemic from ischaemic priapism [1427-1429,1431]. Ultrasound must be performed without intracavernosal vasoactive drug injection [1582]. In non-ischaemic priapism, US helps to localise the fistula site and appears as a characteristic colour blush and turbulent high-velocity flow on Doppler analysis [1583]. Patients with non-ischaemic priapism have normal to high blood velocities in the cavernous arteries [1430,1584].
Selective pudendal arteriography can reveal a characteristic blush at the site of injury in arterial priapism [1585,1586]. However, due to its invasiveness, it should be reserved for the management of non-ischaemic priapism when embolisation is being considered [1395,1424].
The role of MRI in the diagnostic evaluation of priapism is controversial. Its role in non-ischaemic priapism is limited because the small penile vessels and fistulae cannot be easily demonstrated [1587].
10.3.2.e. Summary of evidence and recommendations for the diagnosis of non-ischaemic priapism
| Summary of evidence | LE |
| Non-ischemic priapism is less common than ischemic and is usually associated with blunt perineal or penile trauma leading to the development of intracavernosal fistula. | 2b |
| Medical history and blood gas analysis can differentiate between ischemic and non-ischemic priapism. | 2b |
| Blood aspiration from the corpora in case of non-ischemic priapism reveal bright red arterial blood with normal arterial gas values. | 2b |
| Penile duplex US can identify intracavernosal fistula responsible for non-ischemic priapism. | 2b |
| Recommendations | Strength rating |
| Take a comprehensive history to establish the diagnosis, which can help to determine the priapism subtype. | Strong |
| Include a physical examination of the genitalia, perineum and abdomen in the diagnostic evaluation. | Strong |
| Include a neurological examination if neurogenic non-ischaemic priapism is suspected. | Strong |
| Include complete blood count, with white blood cell differential, and coagulation profile for laboratory testing. | Strong |
| Analyse the blood gas parameters of blood aspirated from the penis to differentiate between ischaemic and non-ischaemic priapism. | Strong |
| Perform colour duplex ultrasound of the penis and perineum to differentiate between ischaemic and non-ischaemic priapism. | Strong |
| Perform selected pudendal arteriography when embolisation is planned for non-ischaemic priapism. | Strong |
10.3.3. Disease management
The conventional belief is that the management of non-ischaemic priapism is not an emergency because the corpus cavernosum does not contain ischaemic blood; however, data indicate that the duration of non-ischaemic priapism can also impact EF. In a case series consisting of six patients with high-flow priapism after median follow-up of 4.5 (2-12) weeks, all patients reported development of ED or distal penile flaccidity [1508]. The goal of treatment is closure of the fistula. Non-ischaemic priapism can be managed conservatively or by direct perineal compression. Failure of conservative treatment requires selective arterial embolisation [1588]. The optimal time interval between conservative treatment and arterial embolisation is under debate. Definitive management can be performed at the discretion of the treating physician and should be discussed with the patient so that they can understand the risks of treatment [1395,1424].
10.3.3.a. Conservative management
Conservative management may include applying ice to the perineum or perineal compression, which is typically US-guided. The fistula occasionally closes spontaneously. Even in cases where the fistula remains patent, intercourse is still possible [1429,1563,1589,1590]. Androgen deprivation therapy (e.g. leuprolide injections, bicalutamide and ketoconazole) has been reported in case series to enable closure of the fistula reducing spontaneous and sleep-related erections [1591]. Sexual dysfunction due to these treatments must be considered. Patients may develop ED or distal penile flaccidity while undergoing conservative treatment [1508].
Blood aspiration is not helpful for the treatment of arterial priapism, and the use of α-adrenergic antagonists is not recommended because of potential severe TRAEs (e.g. transfer of the drug into the systemic circulation).
10.3.3.b. Selective arterial embolisation
Selective arterial embolisation can be performed using temporary substances, such as autologous blood clot [1592-1595] and gel foam [1593-1597], or permanent substances, such as microcoils [1593,1596,1598,1599], ethylene-vinyl alcohol copolymer, and N-butyl-cyanoacrylate [1600]. It is assumed that temporary embolisation provides a decreased risk of ED, with the disadvantage of higher failure/recurrence rates because of artery embolisation using temporary materials. However, there is insufficient evidence to support this hypothesis. Success rates ranging between 61.7 and 83.3%, and ED rates from 0-33.3% after the first arterial embolisation, have been reported, suggesting that failure/recurrence may not be significantly higher with temporary embolisation materials, and preservation of EF may not be that different between the two modalities [1568]. Other potential complications of arterial embolisation include penile gangrene, gluteal ischaemia, cavernositis, and perineal abscess [1395,1601]. Repeated embolisation is a reasonable option for treating non-ischaemic priapism, both in terms of efficacy and safety [1568,1595].
10.3.3.c. Surgical management
Surgical ligation of the fistula is possible through a transcorporeal or inguinoscrotal approach, using intra-operative Doppler US. Surgery is technically challenging and associated with significant risks, particularly of ED [1602]. Surgery is rarely performed and should only be considered when there are contraindications for selective embolisation, if embolisation is unavailable, or repeated embolisation has failed. If the patient desires more definitive treatment and is not sexually active or has pre-existing ED, surgical intervention can be an appropriate option [1568]. Following treatment of non-ischaemic priapism, ED rates ranging from 0-50% have been reported, with surgical ligation having the highest reported rates [1568]. Patients can require PPI for ED in the long term [1480].
10.3.3.d. Summary of evidence and recommendations for the treatment of non-ischaemic priapism
| Summary of evidence | LE |
| Nonischaemic priapism can cause ED over time and early definitive management should be undertaken. | 3 |
| Conservative management applying ice to the perineum or site-specific perineal compression is an option in all cases. The use of androgen deprivation therapy may enable closure of the fistula reducing spontaneous and sleep-related erections. | 3 |
| Selective artery embolisation, using temporary or permanent substances, has high success rates. No definitive statement can be made on the best substance for embolisation in terms of sexual function preservation and success rate. | 3 |
| Repeated embolisation is a reasonable option for the treatment of non-ischaemic priapism. | 2b |
| Selective surgical ligation of the fistula is associated with high risk of ED. | 3 |
| Recommendations | Strength rating |
| Perform definitive management for non-ischaemic priapism at the discretion of the treating physician as it is not a medical emergency. | Weak |
| Manage non-ischaemic priapism conservatively with the use of site-specific perineal compression as the first step. Consider androgen deprivation therapy only in adults. | Weak |
| Perform selective arterial embolisation when conservative management has failed. | Strong |
| Perform the first selective arterial embolisation using temporary material. | Weak |
| Repeat selective arterial embolisation with temporary or permanent material for recurrent non-ischaemic priapism following selective arterial embolisation. | Weak |
| Reserve selective surgical ligation of a fistula as a final treatment option when repeated arterial embolisations have failed. | Weak |
10.3.3.e. High-flow priapism in children
Non-ischaemic priapism is a rare condition, especially in children. The embarrassment that children may have in speaking about it to their parents can lead to misdiagnosis and underestimating the prevalence of this condition [1603]. The aetiology, clinical presentation, diagnostic and therapeutic principles are comparable with those of arterial priapism in adults. However, some differentiating features should be noted.
Idiopathic non-ischaemic priapism can be found in a significant percentage of children [1604]. Perineal compression with the thumb may be a useful manoeuvre to distinguish ischaemic and non-ischaemic priapism, particularly in children, where it may result in immediate detumescence, followed by the return of the erection with the removal of compression [1568]. In children, conservative management using ice applied to the perineum or site-specific perineal compression may be successful [1605,1606]. Although reportedly successful, embolisation in children is technically challenging and requires treatment within a specialist paediatric vascular radiology department [1440,1607].
10.3.3.f. Follow-up
During conservative management of non-ischaemic priapism, physical examination and colour duplex US can be useful tools to assess treatment efficacy. Close follow-up using colour duplex US and MRI can help detect distal penile fibrosis and be beneficial in clinical decision-making to intervene with embolisation earlier [1508]. Follow-up after selective arterial embolisation should include clinical examination, colour duplex US, and EF assessment. If in doubt, repeat arteriography is required. The goals are to determine if the treatment was successful, identify signs of recurrence, and verify any anatomical and functional sequelae [1582].