21. CONGENITAL LOWER URINARY TRACT OBSTRUCTION
Introduction
The term congenital lower urinary tract obstruction (CLUTO) is used to refer to intrauterine dilatation of the bladder and/or the upper urinary tract. During pregnancy, the diagnosis is usually based on ultrasound examinations. There is a broad spectrum of conditions that could cause an intrauterine dilatation of the urinary tract. Congenital lower urinary tract obstruction is most commonly the result of posterior urethral valves (PUV) in approximately 60% of cases. Postpartum diagnosis, however, comprises any number of anatomical and functional disorders/anomalies/malformations causing dilatation, e.g. anterior urethral valves, urethral atresia/stenosis, prune belly syndrome, dilating VUR, cloacal malformation, prolapsing ureterocele, megacystis-microcolon-intestinal hypoperistalsis or megacystis-megaureter syndrome [1228].
Due to the heterogeneity and the rare spectrum of clinical manifestations of CLUTO, referral of such cases is recommended to a tertiary centre with multidisciplinary expertise in prenatal and postnatal management of obstructive uropathies [1228].
Megacystis
In the first trimester, foetal megacystis is defined as a bladder with a longitudinal diameter ≥ 7mm. A longitudinal (craniocaudal) diameter of 7-12mm in the first trimester is usually transient, disappearing in approximately 90% of cases during the second trimester. A measurement > 15mm, however, indicates that CLUTO is unlikely to resolve [1228]. In the second and third trimester, megacystis is defined by an enlarged bladder failing to empty during an extended US examination lasting at least 40 minutes. Two-thirds of cases are secondary to CLUTO, and the remainder are associated with genetic syndromes, developmental or chromosomal abnormalities, including anorectal malformations, of which 14% were normal or had an isolated urological abnormality (e.g. VUR, duplex system) [1228,1229]. In a systematic review, ≥ 45% of megacystis cases were shown to be associated with oligohydramnios and 15% had chromosomal abnormalities [1230]. Final diagnoses included PUV (57%), urethral atresia/stenosis (7%), prune belly syndrome (4%), megacystis-microcolon-intestinal hypoperistalsis syndrome (MMIHS) (1%), cloacal abnormality (0.7%), and undefined pathologies (36.5%) [1230].
The prognosis of the foetus depends on the underlying pathology, the timing of diagnosis, the presence of oligo/anhydramnios, and bladder volume.
Posterior urethral valves
21.1. Epidemiology and pathophysiology
Posterior urethral valves are one of the few life-threatening congenital anomalies of the urinary tract found during the neonatal period. The risk for chronic kidney disease (CKD) is estimated to reach 32% and 20% for end-stage renal disease (ESRD), as reported in a systemic review [1231]. Up to 17% of paediatric ESRD can be attributed to PUV [1232]. An incidence of PUV of 1 in 7,000-8,000 live births has been estimated [1233,1234].
The kidneys start to produce urine at approximately the tenth week of antenatal life. The intrauterine obstruction leads to a decreased urine output, which could result in oligo- or anhydramnios. Amniotic fluid is necessary for normal development of the lungs, and its absence may lead to pulmonary hypoplasia.
An obstruction at the level of the urethra affects the entire urinary tract to varying degrees:
- The prostatic urethra is distended, and the ejaculatory ducts may be dilated due to urinary reflux.
- The bladder neck is hypertrophied and rigid.
- The hypertrophied bladder may occasionally have multiple diverticula.
- Nearly all valve patients have dilatation of the upper urinary tract. This may be due to the valve itself and the high pressure in the bladder, or due to obstruction of the ureterovesical junction by the hypertrophied bladder.
- If there is secondary VUR, the affected kidney functions poorly in most cases.
Secondary VUR is observed in at least 50% of patients with PUV [1235]. It is generally accepted that unilateral high-grade VUR associated with ipsilateral renal dysplasia acts as a ‘pressure pop-off valve,’ which would protect the contralateral kidney, leading to a better prognosis [1236]. Other types of pop-off mechanisms include large-bladder diverticula, urinary extravasation with or without urinary ascites, and a patent urachus [1237]. Protective effects of pop-off phenomena on renal function remain equivocal, however, as long-term outcomes from different studies have been discrepant, with some studies showing a protective effect [1238,1239], while others have shown no protective benefit [1240-1242]. A possible explanation for such discrepancy could relate to differences in defining the exact nature of what constitutes a pop-off mechanism.
21.2. Classification systems of the urethral valves
Until today, the original classification by Hugh Hampton Young is the most commonly used classification [1243], which described three categories: type I, type II and type III. However, today, only type I and type III are found to be obstructive. Hampton Young’s descriptions of type I and III are as follows:
Type I (90-95%)
In the most common type, there is a ridge lying on the floor of the urethra, continuous with the verumontanum, which takes an anterior course and divides into two fork-like processes in the region of the bulbomembranous junction [1243].
Type III
Has been found at various levels of the posterior urethra and apparently bears no relation to the verumontanum. This obstruction attaches to the entire circumference of the urethra, with a small opening in the centre [1233].
21.3. Diagnostic evaluation
During prenatal US screening, hydroureteronephrosis and a distended bladder are suspicious signs of a urethral valve. A thick-walled bladder appears to better predict PUV than a dilated posterior urethra (‘keyhole’ sign) [1244]. However, differentiation between obstructive and nonobstructive aetiologies on prenatal US is challenging, as both have a similar sonographic appearance [1245]. In the presence of increased echogenicity of the kidney, dilatation of the urinary tract and oligohydramnios, the diagnosis of a PUV should strongly be considered. Prenatal US is adequate in most cases (90%) [1246]. However, in some circumstances, when technical ultrasound conditions are poor, foetal MRI may provide additional information [1247].
Postnatally, creatinine, blood urea nitrogen and electrolytes should be monitored closely during the first few days. Initial management involving a multidisciplinary team including a paediatric nephrologist. The clinician must be aware of a noteworthy association between PUV and undescended testicles (UDT) and/or inguinal hernia [1248]. Undescended testicles occurred in 12-17 % of PUV, which is consistent with a tenfold increase [1249].
A VCUG (including lateral views of the urethra during the voiding phase without a catheter in situ) is recommended to assess the presence of a PUV. This study is essential whenever there is a question of an infravesical obstruction, as the urethral anatomy is well-outlined during voiding. The extent of posterior urethral deformity - as expressed by the posterior urethral height: width ratio - appears to correlate positively with nadir creatinine at twelve months of age [1250]. Nuclear renography with split renal function is important in assessing contributory renal function and/or scarring (DMSA or MAG3).
21.4. Management
21.4.1. Prenatal treatment
Most cases of PUV are suspected prenatally [1233,1251,1252]. The potential for spontaneous resolution of bladder enlargement, and the timing of renal imaging are the main obstacles for prenatal intervention. As renal dysplasia is irreversible, it is important to identify those foetuses with good renal function [1253].
Prenatal interventions aim to restore amniotic fluid volume and attenuate the risk of pulmonary hypoplasia or further renal damage [1254]. The decision for prenatal intervention can be based on a staging system that is composed of renal ultrasonographic findings, amnion amount and foetal urine biochemistry [1252]. Early intervention (before the age of 16 weeks of gestation) may be beneficial for renal function. However, making the correct diagnosis and the detection of other severe comorbidities is extremely difficult currently [1255]. Later interventions are mostly of benefit for lung development, but not for renal function [1256]. There are, however, emerging reports of interventions as early as the end of first trimester, with results pointing to a potential preservation of long-term renal function. These reports are still preliminary. The techniques used are intricate and can be associated with a higher risk of foetal demise in these very frail and tiny patients [1257-1259].
Foetal urine samples before 23 weeks of gestation (ß2-microglobulin, sodium, chloride and calcium) may be helpful to distinguish between those who could benefit from intrauterine therapy and those in whom the outcome is most likely to be compromised [1260].
In a small study, normal biochemistry - a sodium level below 100mmol/L, a chloride value of < 90mmol/L, calcium < 8mg/dL, and ß2-microglobulin < 6mg/L obtained in the first foetal urine sample or biochemistry that improves between two sequential samplings, the latter scenario prompting foetal intervention - was associated with high foetal survival and normal renal function at five years [1261]. The status of amnion fluid, the appearance of the kidneys, as well as the foetal urine biochemistry may be helpful in counselling. Proteomic analysis of foetal urine using a signature peptide expressed in foetuses who go on to develop ESRD by the age of two years may show promise in assessing CLUTO. This should be considered as experimental.
The placing of a vesicoamniotic shunt (VAS) is a prenatal treatment designed to restore amniotic fluid cycling. A complication rate of 21-59% is reported, with dislocation of the shunt being the most common [1254]. The PLUTO trial (randomised study) failed to show a long-term benefit on renal function by placing a VAS [1262]. A meta-analysis on interventions for CLUTO reported that VAS resulted in a higher perinatal survival rate than conservative management (57.1% vs. 38.8%), with no significant differences in six-to-twelve-month survival, two-year survival or postnatal renal function [1263]. Foetal cystoscopy with laser ablation has a high complication rate without evidence for the effectiveness of these interventions [1264]. To avoid the severe complication of the laser ablation, balloon dilation has been tried [1265]. The number of patients and designs of these studies are insufficient to yield any recommendations. Parental information is very important and the natural history of CLUTO, including the postnatal outcomes with or without prenatal treatment, as well as the uncertainties and/or controversies regarding CLUTO diagnosis and treatment, should be discussed [1254].
21.4.2. Postnatal treatment
Bladder drainage
Following delivery, the bladder should be drained transurethrally or suprapubically. This catheter drainage tube can then be used to perform a VCUG to confirm the diagnosis.
Valve ablation
When the neonate’s medical situation has stabilised, the next step is to perform an endoscopic valve ablation, provided the urethra is accessible with available equipment. In cases in which the urethra is too small, urinary diversion should be maintained until valve ablation can be performed. Small paediatric cystoscopes and resectoscopes are available either to incise, ablate or to resect the valve at the 5, 7 and/or 12 o’clock positions, depending on the surgeon’s preference. It is important to avoid extensive electrocoagulation, as the most common complication of this procedure is stricture formation. Two studies demonstrated a lower urethral stricture rate using the cold knife compared to diathermy [1266,1267]. Currently, no strong evidence exists to support the use of laser ablation of PUVs. Preliminary studies on the use of Holmium YAG and Thulium lasers, show that laser fulguration is safe and effective [1268]. Within the three months following initial treatment, effectiveness of the treatment should be demonstrated either by clinical improvement (ultrasound and renal function), control VCUG or a re-look cystoscopy, depending on the clinical course [1269-1271].
Bladder neck incision
Bladder neck incision has been suggested as a means of managing secondary bladder neck obstruction [1272]. There is no current evidence which demonstrates that bladder neck incision has a role in preventing reintervention or rehospitalisation rates and therefore bladder neck incision cannot be recommended as a routine management option.
Vesicostomy
A vesicostomy is indicated if the child is too small to undergo endoscopic surgery, has failed endoscopic valve ablation, or has shown no clinical improvement following valve ablation. This is an alternative to prolonged catheter drainage and has been shown to stabilise/improve the upper tracts in up to 90% of cases [1273]. The most prevalent vesicostomy procedure in children was described by Blocksom and modified by Duckett [1274]. Key technical points are to ensure an adequate mobilisation of the bladder dome to enable a tension-free anastomosis with the fascia and skin. Common complication following vesicostomy are stomal stenosis, mucosal prolapse, peristomal dermatitis, and bladder calculi. The risk of prolapse is usually due to an extensive bladder mobilisation and due to too inferior placement of the stoma on the abdominal wall, allowing the posterior bladder wall to evert through the stoma.
High diversion
In cases in which bladder drainage is insufficient to prevent recurrent infections of the upper tract, improve renal function and/or a decrease upper tract dilatation, high urinary diversion should be considered. The choice of urinary diversion depends on the surgeon’s preference for high loop ureterostomy, ring ureterostomy, end ureterostomy or pyelostomy, with each technique having its advantages and disadvantages [1275-1277]. Diversion can delay progression to end stage renal failure [1278].
Vesicoureteric reflux is very common in PUV patients (up to 72%) and is described bilaterally in up to 32% [1279]. A recent prospective observational study identified high-grade VUR as an independent risk factor for developing febrile UTIs, particularly in the first nine months of life. Therefore, antibiotic prophylaxis should be considered in such patients [1280]. Moreover, circumcision can be discussed to further reduce the risk of UTIs [1280]. In the above-mentioned multicentre, randomised, controlled trial, after two years of follow-up, this study demonstrated a statistically significant effect of circumcision as an adjunct to antibiotic prophylaxis in preventing febrile UTIs. The hazard ratio for developing a febrile UTI in the group with antibiotics alone was 10.3 (95% CI: 1.3-82.5) compared with the combined prophylactic antibiotics and circumcision group.
Early administration of oxybutynin may improve bladder function as shown in one study with eighteen patients [1281] and enhances resolution of hydronephrosis and VUR, as shown in a randomised controlled study of 49 patients. However, oxybutynin treatment had no discernible effect on renal function or risk of UTI [1272]. High-grade VUR is usually associated with a poorly functioning kidney, however, early removal of a nonfunctioning renal unit in an asymptomatic patient appears to be unnecessary. Deterioration of renal function without an anatomical obstruction and higher urine output (polyuria) may lead to an overdistension of the bladder during the night. Drainage of the bladder during the night by a catheter may be beneficial for the hydronephrosis, as well as for renal function [1282,1283]. Patients with high daytime post-void residual urine volumes may benefit from clean intermittent catheterisation (CIC) [1284]. In those who do not want, or are not able, to perform CIC via the urethra, the placement of a catheterisable channel is a good alternative [1285].
Clean intermittent catheterisation has been shown to delay the onset of dialysis in patients with chronic kidney disease, progressing to ESRD, and has also resulted in significantly better ten-year graft survival rates in transplanted patients [1286,1287].
21.5. Follow-up
Several prognostic factors for the prediction of future renal function have been described. Various serum nadir creatinine levels are specified in the literature (0.85mg/dl to 1.2mg/dl [μmol/L]) [1250,1288-1291]. Renal parenchymal quantity (total renal parenchymal area) and quality (corticomedullary differentiation and renal echogenicity) on initial postnatal US also have prognostic value [1292].
Life-long monitoring of these patients is mandatory, as bladder dysfunction (‘valve bladder’) is common, and the delay in day- and nighttime continence is a significant problem [1293]. Urodynamic studies play an important role in the management of patients with valve bladder, especially in those with suspicion of bladder dysfunction [1294,1295], however, there is no consensus as to optimal timing or frequency of such studies. Poor bladder sensation and compliance, detrusor overactivity and polyuria (especially at night) and their combination are responsible for bladder dysfunction. In those with bladder overactivity, anticholinergic therapy can improve bladder function. In patients with poor bladder emptying, α-blockers can be used to reduce the post-void residual [1296,1297].
Chronic kidney disease develops in up to 65% of PUV patients and approximately 20% of these progress towards ESRD [1298]. Renal transplantation in these patients can be performed safely and effectively [1299,1300]. Deterioration of the graft function is mainly related to lower urinary tract dysfunction (LUTD) [1299]. Figure 14 provides an assessment and treatment algorithm.
Limited data is available pertaining to sexual function and fertility in patients with PUV. Long-term studies have demonstrated normal erectile function and fertility potential [1301,1302]. However, a negative influence of the individual patient’s fertility must be taken into account, as these patients have a higher risk for bilateral cryptorchidism, recurrent epididymitis and ESRD [1301].
Figure 14: An algorithm on the assessment, management and follow-up of newborns with possible PUV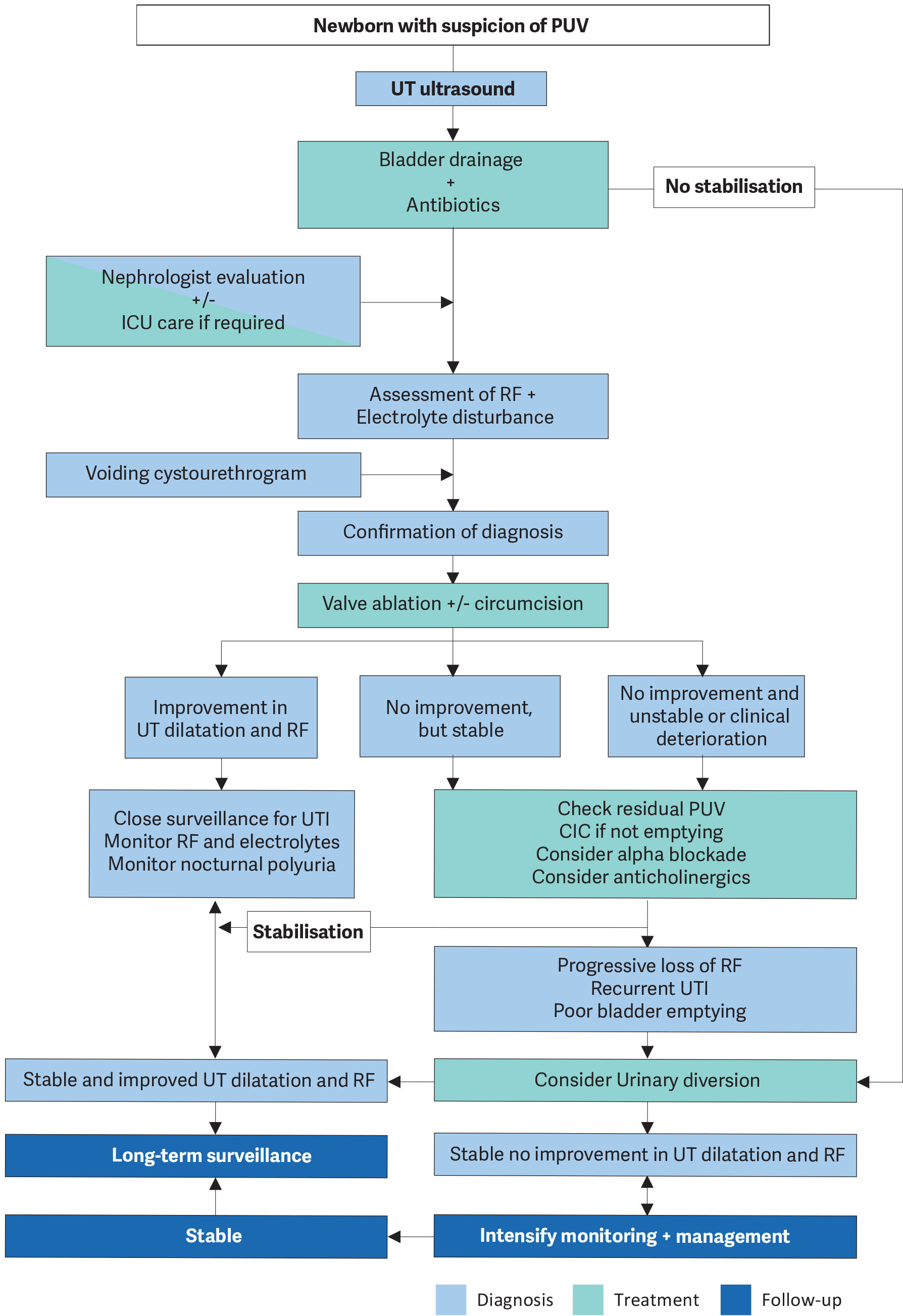 CIC = clean intermittent catheterisation; OAB = overactive bladder; PUV = posterior urethral valve; RF = renal function; UT = urinary tract; UUT = upper urinary tract; VCUG = voiding cystourethrogram.
CIC = clean intermittent catheterisation; OAB = overactive bladder; PUV = posterior urethral valve; RF = renal function; UT = urinary tract; UUT = upper urinary tract; VCUG = voiding cystourethrogram.
Anterior urethral valve
Anterior urethral valve (AUV) is a semilunar or iris-like band of tissue on ventral aspect of urethra. It can be isolated or can be seen in association with anterior urethral diverticulum. The aetiology of isolated AUV is speculated to be secondary to congenital urethral obstruction, malunion of glandular and penile urethra, congenital cystic dilatation of periurethral glands, or ruptured distal lip of a syringocele [1303]. AUV can be present in the bulbous urethra, the penoscrotal junction and penile urethra. Patients may present with poor urinary stream, penile ballooning, UTI or haematuria. Firlit classified anterior urethral valves depending on the presence of diverticulum and dilatation of urethra and upper tract [1304]. The diagnosis is based on a VCUG, with possible findings of dilated or elongated posterior urethra, dilatation of the anterior urethra, thickened trabeculated bladder, hypertrophied bladder neck, VUR and urethral diverticula. In doubtful cases, retrograde urethrography may be helpful showing linear filling defect along the ventral wall, or it may show a dilated urethra ending in a smooth bulge or an abrupt change in the calibre of the dilated urethra on VCUG [1305].
Treatment is mainly by means of endoscopic valve ablation. In selected patients, temporary diversion may be considered until the child is big enough for endoscopy. Open surgery is reserved for patients with very large diverticulum and defective spongiosum. Renal failure may develop in 22%, the risk being highest in patients with pretreatment azotaemia, VUR and UTI [1306].
Anterior urethral diverticulum
Common postnatal presenting features of anterior urethral diverticulum (AUD) are compressible ventral penile swelling, urinary postmicturition dribble, voiding difficulty, poor stream, and recurrent UTIs [1307-1309]. Diagnosis is by VCUG with or without a retrograde urethrogram. In small AUD, endoscopic cutting or deroofing of distal lip of the diverticulum can be sufficient treatment. Larger diverticulum requires excision with subsequent two-layered urethroplasty, or marsupialisation with staged urethroplasty. In cases of urosepsis and obstructive uropathy, a suprapubic catheter or temporary urinary diversion (vesicostomy or proximal cutaneous urethrostomy) may be indicated prior to definitive surgical management [1310,1311]. Anatomically, AUVs have normal corpus spongiosum development, whereas AUDs do not [1311].
Syringocele
Cowper’s glands are two bulbourethral glands that are located within the urogenital diaphragm which open into the urethra 1-2cm distal to the sphincter. Syringocele is the cystic dilatation of these glands. The aetiology can be congenital or acquired (trauma or infection). Syringocele has been classified as simple, imperforate, perforate and ruptured [1312]. A simpler grouping is suggested as to merge simple, perforated and ruptured syringocele into ‘open syringocele’ and imperforate to ‘closed syringocele.’ Closed syringoceles cause obstructive symptoms while open syringoceles act as a diverticulum and cause post-void dribbling and, in some case, obstruction [1313]. Depending on the syringocele type, patients can present with post-void dribbling, urethral discharge, UTI, perineal pain, haematuria, obstructive voiding symptoms, dysuria, or retention. Diagnosis is based on antegrade and/or retrograde urethrogram, which shows a cystic defect distal to prostate. If such studies are inconclusive, US and/or MRI may be used. Asymptomatic syringoceles can be managed conservatively. Endoscopic deroofing with various energy sources (cold knife, electrocautery and holmium laser) in both obstructing and non-obstructing syringoceles is an effective method of marsupialisation [1314]. In cases in which endoscopic approach is not feasible, open correction may be considered.
Cobb’s collar
Cobb’s collar is a congenital membranous stricture of the bulbar urethra. Unlike congenital obstructive posterior urethral membrane (COPUM), Cobb’s collar is independent of the veru montanum and external sphincter and is believed to represent a persistence of part of the urogenital membrane [1315]. Voiding cystourethrogram shows narrowing in the proximal bulbar urethra with folds extending proximally, a dilated posterior urethra, prominent bladder neck and other findings of infravesical obstruction. Treatment with endoscopic cold-knife incision showed lower recurrence rates than electrocautery [1316].
Urethral atresia/hypoplasia
Male urethral atresia is congenital, complete obstruction of the urethra caused by a membrane that is usually located at the distal end of the prostatic urethra. The urethra distal to this point is usually hypoplastic, presumably from lack of foetal voiding [1317]. Urethral atresia is associated with bladder distention, VUR, hydronephrosis and renal dysplasia [1318]. Most cases reported have the phenotypic characteristics of the prune belly syndrome. Antenatal intervention may be beneficial in terms of foetal survival [1319]. Although progressive augmentation by dilating the urethra anterior (PADUA) procedure was described as a treatment modality, the majority of cases require some form of supravesical diversion [1317,1318].
Posterior urethral polyps (PUP)
Although PUP does not cause antenatal hydronephrosis, it could cause obstruction later in life. Posterior urethral polyps is a polypoid, pedunculated, fibroepithelial lesion arising in the posterior urethra proximal to the veru montanum. The PUP lies on the floor of the urethra with its tip reaching into the bladder neck, and obstruction occurs due to distal displacement of the polyp during micturition [1320]. Patients complain of dysuria, haematuria and obstructive symptoms such as poor urinary stream and intermittent retention episodes. Diagnosis can be suspected by VCUG and/or US but is confirmed during cystourethroscopy. Treatment is usually by endoscopic resection of the polyp. The course of the disease is benign, and no recurrences have been reported in the literature [1321,1322].
21.6. Summary of evidence and recommendations for the management of posterior urethral valves
| Summary of evidence | LE |
| Posterior urethral valves are one of the few life-threatening congenital anomalies of the urinary tract. | 1b |
| Antenatal therapy could be considered based on ultrasound findings, foetal urine biochemistry, amniotic fluid level and chromosomal status. | 4 |
| Serum creatinine nadir above 0.85mg/dL is correlated with a poor prognosis. | 2a |
| Neonatal circumcision as an adjunct to antibiotic prophylaxis in PUV patients significantly decreases the risk of developing febrile UTIs during the first two years of life. | 1b |
| Early pharmacological management with oxybutynin may improve bladder function. | 1b |
| Despite optimal treatment, 20% of patients will develop end stage renal disease. | 2a |
| Renal transplantation in these patients is safe and effective if the bladder function is managed. | 3 |
| Recommendations | Strength rating |
| Drain the bladder in newborns with a suspected diagnosis of infravesical obstruction and place on antibiotic prophylaxis. | Strong |
| Perform a voiding cystourethrography in patients in whom a diagnosis of posterior urethral valves (PUV) is suspected. | Strong |
| Attempt endoscopic valve ablation after bladder drainage and stabilisation of the child. | Strong |
| Consider neonatal circumcision as an adjunct to antibiotic prophylaxis to decrease the risk of urinary tract infection in those with a PUV, particularly in the presence of high-grade vesicoureteral reflux. | Strong |
| Offer prolonged urinary diversion (suprapubic/transurethral) for bladder drainage if the child is too small for valve ablation. | Strong |
| Use serum creatinine nadir as a prognostic marker. | Strong |
| Assess split renal function by dimercaptosuccinic acid scan or mercaptoacetyltriglycine clearance. | Strong |
| Consider high urinary diversion if bladder drainage is insufficient to drain the upper urinary tract, or in the absence of clinical improvement. | Strong |
| Monitor and manage bladder and renal function lifelong. | Strong |