4. MANAGEMENT OF UNDESCENDED TESTES
4.1. Background
The term cryptorchidism is most often used synonymously with undescended testes and is one of the most common congenital malformations of male neonates. The incidence varies and depends on gestational age, affecting 1.0-4.6% of full-term and 1.1-45% of preterm neonates. Following spontaneous descent within the first months of life, nearly 1.0% of all full-term male infants still have undescended testes at one year of age [64]. Cryptorchidism can occur bilaterally in up to 30% of cases [65]. In newborn cases with nonpalpable or undescended testes on both sides and any sign of disorders of sex development (DSDs), such as concomitant hypospadias, endocrinological and genetic evaluation is required to assess for any form of DSD (see Chapter 20, Disorders/differences of sex development) [66]. Children with abdominal wall defects such as gastroschisis and omphalocele were shown to have higher rates of cryptorchidism [67], with a higher atrophy rate overall [68]. Cryptorchidism is associated with an increased risk of impaired fertility and testicular cancer, not limited to congenital cases. Structural abnormalities are often found in the affected testicles, and they are typically smaller in size [69]. While congenital cryptorchidism increases the risk of infertility, ascending testes can also affect fertility potential, though to a lesser extent [70-73]. Histological changes in acquired cases mirror those seen in congenital cryptorchidism, highlighting the need for careful management to reduce fertility risks [70,71,73,74].
4.2. Classification
The most useful classification of undescended testes is distinguishing into palpable and nonpalpable testes, and clinical management is decided by the location and presence of the testes (see Figure 1). Approximately 80% of all undescended testes are palpable [75]. Acquired cryptorchidism is a condition where testes that were previously in a scrotal location ascend secondarily. This condition can be caused by entrapment after inguinal surgery or spontaneously referred to as ascending testes. The prevalence rates of ascending testis were found to range between 1 and 2% in a population study [76].
Palpable testes include true undescended testes in an inguinal or ectopic location. Nonpalpable testes include intra-abdominal, absent, and sometimes also some inguinal or ectopic testes. Some nonpalpable testes can become palpable under general anaesthesia.
Figure 1: Classification of undescended testes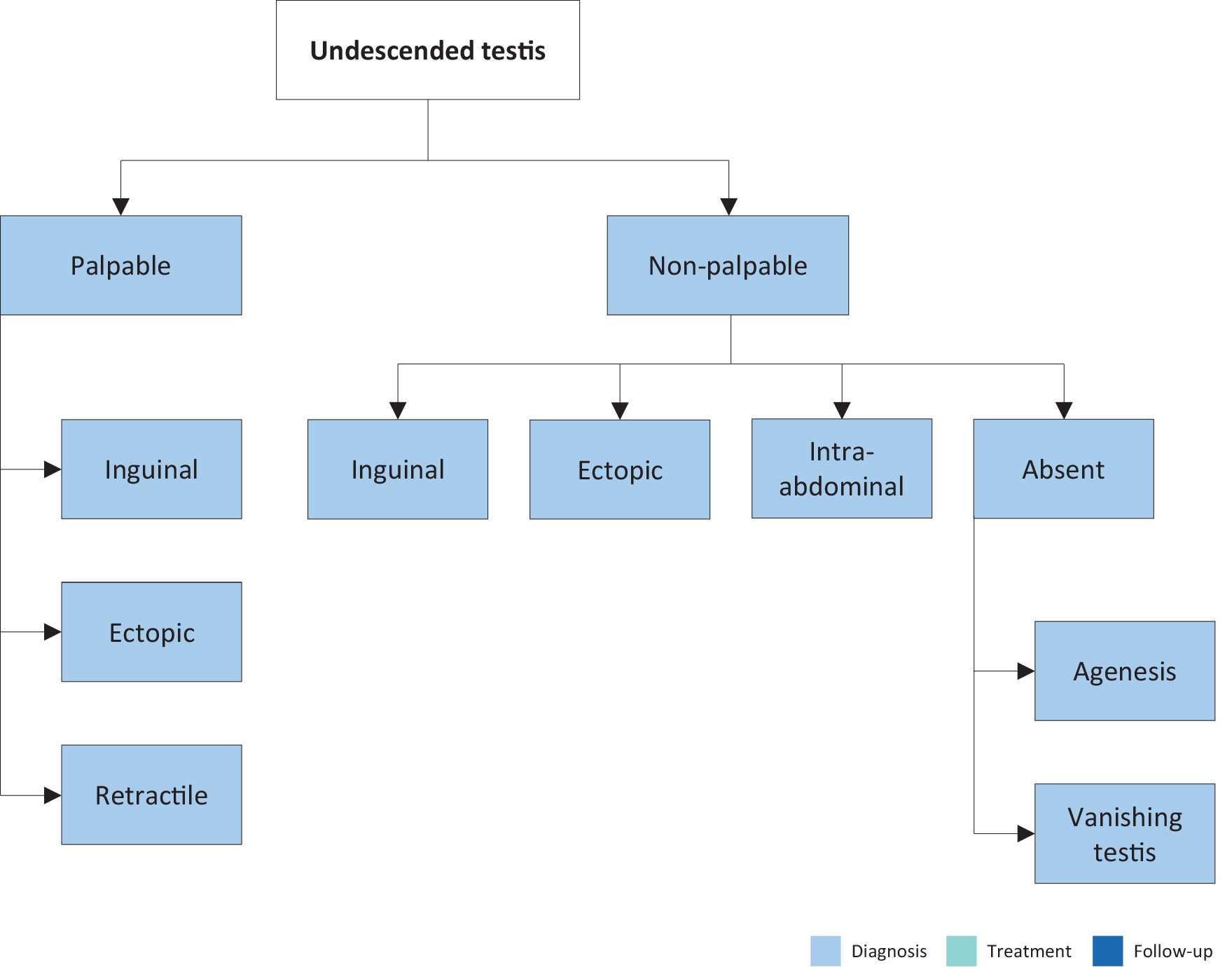
4.2.1. Palpable testes
Undescended testes
A true undescended testis is on its normal path of descent but is halted on its way down to the scrotum. Depending on the location, the testes may or may not be palpable, as in the case of testes arrested in the inguinal canal.
Ectopic testes
If the position of a testis is outside its normal path of descent and outside the scrotum, the testis is considered ectopic. The most common aberrant position is in the superficial inguinal pouch. In some case, an ectopic testis can be identified in a femoral, perineal, pubic, penile or even contralateral position. Usually, there is no possibility for an ectopic testis to descend spontaneously to the correct position. It therefore requires surgical intervention. In addition, an ectopic testis might not be palpable due to its position. Additionally, there are cases reported of patients born with supernumerary testes as a rare phenomenon [77].
Retractile testes
Retractile testes have completed their descent into a proper scrotal position but can be found again in a suprascrotal position along the path of their normal descent. This is due to an overactive cremasteric reflex [78]. Retractile testes can be easily manipulated down to the scrotum and remain there at least temporarily. They are typically normal in size and consistency. As retractile testes carry a higher risk of secondary ascent of up to 33%, yearly controls until puberty are recommended [79].
4.2.2. Nonpalpable testes
Among the 20% of nonpalpable testes, 50-60% are intra-abdominal, canalicular or peeping (changing location between high inguinal position and inside the internal inguinal ring). The remaining 20% are absent and 30% are atrophic or rudimentary.
Intra-abdominal testes
Intra-abdominal testes can be in different positions, with most of them being found close to the internal inguinal ring. However, possible locations include the kidney, anterior abdominal wall and retrovesical space. In the case of an open internal inguinal ring, the testis may be peeping into the inguinal canal.
Absent testes
Monorchidism can be identified in up to 4% of boys with undescended testes, and anorchidism (bilateral absence) in < 1%. Possible pathogenic mechanisms include testicular agenesis and atrophy after intrauterine torsion, which is referred to as ‘vanishing testes’ [80].
4.3. Diagnostic evaluation
History taking and physical examination are key in evaluating boys with undescended testes. Localisation studies using various imaging modalities are usually without any additional benefit.
4.3.1. History
Caregivers should be asked for maternal and paternal risk factors, including hormonal exposure and genetic or hormonal disorders. If the child has a history of previously descended testes, this might be suggestive of retractile testes or secondary testicular ascent [81]. A history of acute scrotal pain and swelling can be suggestive of a missed testicular torsion. Prior inguinal surgery is indicative of secondary undescended testes due to entrapment.
4.3.2. Physical examination
An undescended testis is pursued by carefully advancing the examining fingers along the inguinal canal towards the pubis region, perhaps with the help of lubricant. A possible inguinal testis can be felt to bounce under the fingers [82]. A nonpalpable testis in the supine position may become palpable with positional manoeuvres (e.g. frog-legged or cross-legged), which is a sign of a retractile testis. If no testis can be identified along the normal path of descent, possible ectopic locations must be considered.
In the event of unilateral nonpalpable testis, the contralateral testis must be examined. The size and location of the contralateral testis can have important prognostic implications. Any compensatory hypertrophy could suggest testicular absence or atrophy [83]. Nevertheless, this does not preclude surgical exploration, since the sign of compensatory hypertrophy is not specific enough [84-86].
In the case of bilateral nonpalpable testes or any evidence or sign of disorders/differences of sex development (DSDs) - such as genital ambiguity - or scrotal hyperpigmentation, further evaluation including endocrinological and genetic assessment becomes mandatory [87].
4.3.3. Imaging studies
Imaging studies cannot determine with certainty whether or not a testis is present [88]. Ultrasound (US) lacks the diagnostic sensitivity and specificity to detect the testis confidently or to establish the absence or presence of an intra-abdominal testis [89]. Consequently, the use of various imaging modalities for undescended testes, such as US or magnetic resonance imaging (MRI) [90], is limited and only recommended in specific and selected clinical scenarios (e.g. identification of Müllerian structures in cases with suspicion of DSDs) [89].
4.4. Management
Treatment should be started at the age of six months, since after that age, undescended testes rarely descend [91]. Any type of treatment leading to a scrotally positioned testis should be finished by 12 months, or 18 months at the latest, because histological examination of undescended testes at that age has already revealed a progressive loss of germ cells and Leydig cells [92]. The early timing of treatment is also driven by the final adult results on spermatogenesis and hormone production, as well as on the risk of tumour development [93].
4.4.1. Medical therapy
Unfortunately, most of the studies on hormonal treatment have been of poor quality, with heterogeneous and mixed patient populations, testis location, schedules and dosages of hormonal administration. Additionally, long-term data are almost completely lacking.
Short-term side effects of hormonal treatment include increased scrotal erythema and pigmentation and induction of pubic hair and penile growth. Some boys experience pain after intramuscular injection of human chorionic gonadotropin (hCG). All of these tend to regress after treatment cessation [94,95].
Although many systemic side effects are temporary and typically resolve within six months, there are documented impacts on the testes, such as interstitial bleeding and germ cell apoptosis, which have been linked to a reduction in testicular volume later in adulthood [96,97].
4.4.1.a. Medical therapy for testicular descent
Hormonal therapy using hCG or gonadotropin-releasing hormone (GnRH) is based on the hormonal dependence of testicular descent but has a limited success rate of only 20% [98]. However, it must be noted that nearly 20% of these descended testes have the risk of reascending later [99]. In general, success rates depend on testicular location. The higher the testis is located prior to therapy, the lower the success rate, suggesting that testicular position is an important determinant of success [94]. Some authors recommend combined hCG-GnRH treatment. Unfortunately, this type of treatment is poorly documented, and the treatment groups were diverse. Some studies reported successful descent in up to 38% of nonresponders to monotherapy [100]. A recent meta-analysis concludes that there is no consistent evidence to support hormone treatment alone in the management of undescended testes [69].
4.4.1.b. Medical therapy for fertility potential
Hormonal treatment may improve fertility indices [101,102] and therefore serve as an additional tool to orchidopexy. A longitudinal study on boys undergoing hormone therapy (hCG) with or without surgery showed that patients with hormone therapy had better sperm quality compared to patients undergoing surgery alone or no treatment at all [103]. There is no difference in treatment with GnRH before (neoadjuvant) or after (adjuvant) orchidopexy in terms of increasing fertility index, which may be a predictor for fertility later in life [104].
Identification of specific subgroups of boys with undescended testes who would benefit from using hormones is challenging. Since these important data on specific groups as well as additional support on the long-term effects are still lacking, the Nordic consensus does not recommend hormonal therapy [105].
As endocrine treatment may improve fertility index, the consensus of this Panel is to recommend endocrine treatment with GnRH analogues for boys with bilateral undescended testes to try to preserve fertility potential (LE: 4).
4.4.2. Surgical therapy
If a testis has not concluded its descent at the age of six months (corrected for gestational age), and because spontaneous testicular descent is unlikely to occur after that age, surgery should be performed within the subsequent year [93]. A systematic review and meta-analysis comparing outcomes of orchidopexy before and after one year of age found no difference in atrophy rate, however, early orchidopexy was associated with greater testicular volume and more spermatogonia per tubule (fertility index) as possible markers for improved fertility potential [106]. These findings underscore the importance of early orchidopexy between the ages of six and twelve months (18 months at the latest) [91]. But despite early and successful orchiopexy within the first year of life, up to 25% of boys with non-syndromic undescended testes may be at risk for infertility based on hormonal and histological data as a series on 333 boys showed [107]. This is especially true for bilateral cases, but in addition in approximately 5% of unilateral cases, reduced numbers of germ cells were detected in testicular biopsies as well [107].
4.4.2.a. Palpable testes
Surgery for palpable testes includes orchidofunicolysis and orchidopexy, either via an inguinal or scrotal approach, which are described in the following section.
4.4.2.a.1. Inguinal orchidopexy
Inguinal orchidopexy is a widely used technique with a high success rate of up to 92% (definition of success: correct scrotal position) [108]. Important steps include mobilisation of the testis and spermatic cord to the level of the internal inguinal ring, with dissection and division of all cremasteric fibres, to prevent secondary retraction and detachment of the gubernaculum testis. The patent processus vaginalis must be dissected off the spermatic cord proximally at the level of the internal ring, because an unidentified or inadequately repaired patent processus vaginalis is an important factor leading to failure of orchidopexy [109].
At this moment, the size of the testis can be measured and the connection of the epididymis to the testis can be judged and described in the protocol. Some boys have a significant dissociation between testis and epididymis, which is associated with a lower testicular volume [110]. Finally, the mobilised testicle must be placed in a subdartos pouch within the hemiscrotum without any tension. If the length achieved using the above-mentioned technique is still inadequate, the Prentiss manoeuvre, which consists of dividing the inferior epigastric vessels and transposing the spermatic cord medially to provide a straight course to the scrotum, might be an option [111]. With regard to fixation sutures, if required, they should be made between the tunica vaginalis and the dartos musculature [112]. A recent systematic review and meta-analysis showed no benefit of performing a transparenchymal fixation suture in standard orchidopexy [113]. Lymph drainage of a testis that has undergone surgery for orchidopexy may have changed from high retroperitoneal drainage to iliac and inguinal drainage, which might become important in the event of later malignancy [114].
4.4.2.a.2. Scrotal orchidopexy
Low-positioned, palpable undescended testis can be fixed through a scrotal incision including division of the gubernaculum. The processus vaginalis must be probed to check for patency [115]. Otherwise, fixation in the scrotum is carried out correspondingly to the inguinal approach. In up to 20% of cases, an inguinal incision will be required to correct an associated patent processus vaginalis [116]. Any testicular or epididymal appendages can be easily identified and removed. A systematic review shows that the overall success rates (definition of success rates: correct scrotal position) ranged from 88 to 100%, with rates of recurrence and postoperative testicular atrophy or hypotrophy < 1% [117]. Another systematic review and meta-analysis revealed similar outcome data regarding postoperative complications, including wound infection, testicular atrophy, testicular reascent and hernia for palpable low-positioned undescended testes. The only significant difference was the shorter operative time [118]. Overall, it can be concluded that scrotal orchidopexy appears to be a good surgical alternative for low lying testis [119-121].
4.4.2.b. Nonpalpable testes
For nonpalpable testes, surgery must clearly determine whether a testis is present or not [122]. If a testis is found, the decision must be made to bring the testis down to the scrotum or to remove it. An important step in surgery is a thorough re-examination once the boy is under general anaesthesia, since a previously non-palpable testis might be identifiable and subsequently change the surgical approach to standard inguinal orchidopexy, as described previously. The optimal approach to locate an intra-abdominal testis is diagnostic laparoscopy [123]. Subsequent removal or orchidolysis and orchidopexy can be carried out laparoscopically [124]. If an ipsilateral scrotal nubbin is suspected, and contralateral compensatory testicular hypertrophy is present, a scrotal incision with removal of the nubbin, thus confirming the vanishing testis, is an option avoiding the need for laparoscopy [125].
During laparoscopy for nonpalpable testes, possible anatomical findings include spermatic vessels entering the inguinal canal (40%), an intra-abdominal (40%) or peeping (10%) testis, or blind-ending spermatic vessels confirming vanishing testis (10%) [126].
In case of a vanishing testis, the procedure is finished once blind-ending spermatic vessels are clearly identified. If the vessels enter the inguinal canal, one may find an atrophic testis upon inguinal exploration or a healthy testis that needs to undergo standard orchidopexy [127]. If a nubbin is found during inguinal exploration, it should be resected [69,128]. A peeping testis can be placed down in the scrotum laparoscopically or by means of an inguinal incision [129]. Placement of an intra-abdominal testis can sometimes be a surgical challenge. Usually, testes lying high above the internal inguinal ring may not reach the scrotum with a single surgery [130]. Under such circumstances, a staged orchidopexy may be an option [131] (see Figure 2).
If a staged procedure is required, the most common techniques described in the literature are the Fowler-Stephens procedure and the Shehata technique for intra-abdominal testes [132]. Overall, laparoscopic orchidopexy for intraabdominal testes is associated with a slightly higher atrophy rate and rate of retraction compared to the conventional inguinal approach, especially if a staged approach is required [69].
The Fowler-Stephens technique involves proximal cutting and transection of the testicular vessels, with conservation of the collateral arterial blood supply, via the deferential artery and cremasteric vessels. A modification with low spermatic vessel ligation has gained popularity, allowing blood supply from the testicular artery to the deferential artery. An additional advantage is the position of the peritoneal incision, leading to a longer structure, to ease later scrotal placement [133]. Due to the nature of these approaches, the testis is at risk of hypotrophy or atrophy if the collateral blood supply is insufficient [134]. One- and two-stage FS have similar rates of retraction, whereas one-step procedures might have a higher rate of testicular atrophy [69]. The advantages of two-stage orchidopexy, with the second part usually done six months after the first, are to allow for development of collateral blood supply and to create greater testicular mobility [135]. In addition, preservation of the gubernaculum may also decrease the chance of testicular atrophy [136].
An alternative option to approach intra-abdominal testes is the Shehata technique. The first step involves spermatic vessel traction instead of vessel division [132]. A multicentre comparative study and a recent systematic review and meta-analysis show that both Fowler-Stephens orchidopexy and the traction technique are comparable in terms of postintervention testicular atrophy, testicular retraction/ascent [137,138]. However, the mean operative time is significantly less with FS technique in Stage I laparoscopic orchidopexy [139]. As several studies have validated the safety and similar success rate of these approaches, it is up to the surgeon’s preference and experience to decide which approach to use.
4.4.2.c. Complications of surgical therapy
Surgical complications are usually uncommon, including testicular atrophy. A systematic review revealed an overall atrophy rate for primary orchidopexy of 1.83%, 28.1% for a one-stage Fowler-Stephens procedure, and 8.2% for the two-stage approach [140]. Other rare complications include testicular ascent and vas deferens injury, in addition to local wound infection, dehiscence and haematoma.
4.4.2.d. Surgical therapy for undescended testes after puberty
The Panel consensus recommends orchidopexy in palpable undescended testes and orchiectomy in post-pubertal boys with an intraabdominal testis and a normal contralateral testis in a scrotal position. For adult recommendations, see the EAU Guidelines on Testicular Cancer [137,138].
Figure 2: Treatment of unilateral nonpalpable undescended testes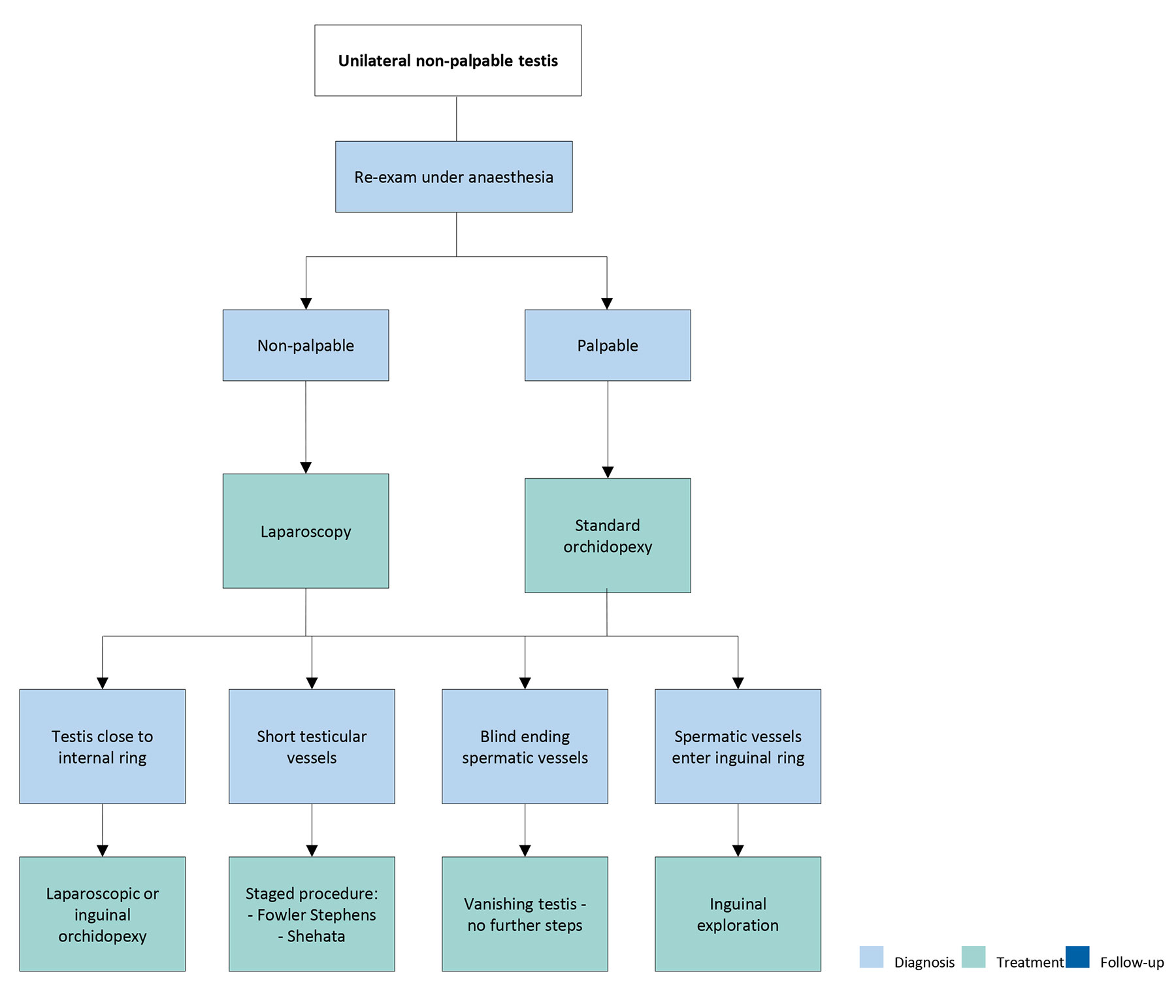
4.5. Undescended testes and fertility
The association of undescended testes with compromised fertility [143] is discussed extensively in the literature and seems to be a result of multiple factors, including germ cell loss, impaired germ cell maturation [144], Leydig cell diminution and testicular fibrosis [145].
Although boys with one undescended testis have a lower fertility rate, they have the same paternity rate as those with bilateral descended testes. Boys with bilateral undescended testes suffer both: lower fertility and paternity rates. Fertility rate is the number of offspring born per mating pair, individual or population, whereas paternity reflects the actual potential of fatherhood [146]. The age at which surgical intervention for an undescended testis occurs seems to be an important predictive factor for fertility later in life. Endocrinological studies revealed higher inhibin-B and lower follicle-stimulating hormone (FSH) levels in men who underwent orchidopexy at two years of age compared to individuals who had surgery later, which is indicative of a benefit of earlier orchidopexy [147,148]. In addition, others demonstrated a relation between undescended testes and increased loss of germ cells and Leydig cells, which is also suggestive of timely orchidopexy being a significant factor for fertility preservation [149]. Outcome studies for untreated bilateral undescended testes revealed that 100% are oligospermic and 75% azoospermic. Among those successfully treated for bilateral undescended testes, 75% remain oligospermic and 42% azoospermic [145].
In summary, early surgical correction of undescended testes is highly recommended before 12 months of age, and by 18 months at the latest, for preservation of fertility potential [92].
4.6. Undescended testes and malignancy
Boys who are treated for an undescended testis have an increased risk of developing testicular malignancy [141]. Screening and self-examination both during and after puberty is therefore recommended [150]. A Swedish study, with a cohort of nearly 17,000 men (56 developed a testicular tumour) who were treated surgically for undescended testes and followed for 210,000 person-years, showed that management of undescended testes before the onset of puberty decreased the risk of testicular cancer. The relative risk of testicular cancer among those who underwent orchidopexy before 13 years of age was 2.2 compared to the Swedish general population; this increased to 5.4 for those treated after 13 years of age [151]. A systematic review and meta-analysis of the literature have also concluded that prepubertal orchidopexy may reduce the risk of testicular cancer, and that early surgical intervention is indicated in boys with undescended testes [152].
4.7. Summary of evidence and recommendations for the management of undescended testes
| Summary of evidence | LE |
| A failed or delayed orchidopexy may increase the risk of testicular malignancy later in life. | 2a |
| Early treatment reduces the risks of impaired fertility and testicular cancer. | 2a |
| In unilateral undescended testis, fertility rate is reduced whereas paternity rate is not. | 1b |
| In bilateral undescended testes, both fertility and paternity rates are impaired. | 1b |
| The treatment of choice for undescended testis is surgical positioning into the scrotum. | 1b |
| The palpable testis is usually treated surgically using an inguinal approach. | 2b |
| The nonpalpable testis should be approached laparoscopically. | 2b |
| Recommendations | Strength rating |
| Do not offer medical or surgical treatment for retractile testes but undertake close follow-up on a regular basis until puberty. | Strong |
| Do not offer hormonal therapy in unilateral undescended testes for testicular descent only. | Strong |
| Offer endocrine treatment in cases of bilateral undescended testes to preserve future fertility potential. | Weak |
| Perform surgical orchidofunicolysis and orchidopexy before the age of 12 months, and by 18 months at the latest. | Strong |
| Perform an endocrinological workup in the setting of bilateral nonpalpable testes. | Strong |
| Perform an exam under anaesthesia and subsequent diagnostic laparoscopy to locate an intra-abdominal testicle. | Strong |
| Inform the patient/caregivers of the increased risk of malignancy with an undescended testis increasing with the age at orchidopexy. | Weak |