14. MONOSYMPTOMATIC NOCTURNAL ENURESIS - BEDWETTING
14.1. Epidemiology, aetiology and pathophysiology
Monosymptomatic nocturnal enuresis (NE), also known as bedwetting, is defined as an unvoluntary nocturnal incontinence. NE is a relatively frequent symptom in children: 5-10% at seven years of age and 1-2% in adolescents. There is also a gender difference in the incidence: two boys to one girl at any age [704]. With a spontaneous yearly resolution rate of 15% (at any age), NE is considered as a relatively benign condition [655,705]. Seven out of 100 seven-year-old bedwetting children will continue to wet their bed into adulthood. Nocturnal enuresis is considered primary when a child has not yet had a prolonged period of being dry (six months). The term ‘secondary NE’ is used when a child or adult begins wetting again after having stayed dry.
The aetiology of nocturnal enuresis mainly consists of three factors: a high arousal threshold, high nighttime urine production and/or a nighttime overactive bladder. The high arousal is the conditional factor. In a recent study in children with refractory monosymptomatic nocturnal enuresis, compared with children without bedwetting problems, polysomnography and ambulatory urodynamic monitoring were performed, it was found that the children with refractory nocturnal enuresis had a smaller nighttime bladder volume, more frequent nighttime detrusor contractions and arousal problems. The findings of this study have confirmed what is known about the pathophysiology of nocturnal enuresis, although in both studies, polysomnography and ambulatory urodynamic monitoring were not performed in the normal home setting [706].
Non-monosymptomatic NE is defined as the condition of NE in association with daytime lower urinary tract symptoms (LUTS, recurrent UTIs and/or bowel dysfunction) [705,707]. The presence of constipation has a negative association with bladder capacity [708].
Nocturnal enuresis has significant secondary stressful, emotional and social consequences for the child and their caregivers. A lower QoL has been reported for children with NE compared to controls, and NE can influence relationships with friends and family [709-713]. A study in six-year-old children with bedwetting problems has found a high level of shame, separation anxiety and fear of abandonment, and the author was also able to identify that the attitude of the parents contributes to these psychological problems [714]. Bedwetting children and their parents will often look for information and advice on social media, including YouTube channels, however, commercial companies can also influence this. The EAU therefore proposes a warrant for online professionalism standards (SoMe) [715,716]. Correct information regarding the pathophysiology of nocturnal enuresis and treatment is therefore advised from the age of six to seven years onwards, with consideration given to mental status, family expectations, social issues and cultural background. A clear hereditary factor is observed in NE. If none of the parents or their immediate relatives has suffered from bedwetting, the child has a 15% chance of wetting their bed. If one of the parents or their immediate relatives have suffered from bedwetting, the chance of bedwetting increases to 44%, and if both parents have a positive history, the chance increases to 77%. However, from a genetic point of view, enuresis is a complex and heterogeneous disorder. Loci have been described on chromosomes 12, 13 and 22 [707]. A genome-wide association study (GWAS) in a large Danish population-based cohort has identified genome-wide significant loci in chromosome 6 and 13 that are associated with nocturnal enuresis. In addition, the GWAS identified protein coding genes associated with impaired sleep, increased nighttime urine production and bladder dysfunction, but also a significant genetic overlap between nocturnal enuresis and ADHD [717].
A high arousal threshold is the most important pathophysiological factor in the aetiology of NE: the child does not wake up when the bladder is full. Children with NE are considered deep but poor sleepers due to high arousal thresholds and frequently disturbed sleep. A systematic review confirmed the fact that sleep has a central role in NE [718]. Full-night polysomnographic recordings support this hypothesis by demonstrating the disruption of children’s sleep microstructure [719]. In addition to the high arousal threshold, an imbalance is needed between night-time urine output and night-time bladder capacity [655,705,707]. Recently, attention has been given to the chronobiology of micturition, in which the existence of a circadian clock in kidney, brain and bladder is postulated [720]. ‘Micturition-desired awakenings’ is a new terminology that is suggested, and the authors of this study even found areas in the brain during functional MRI studies that could be responsible for waking up during sleep while being aware of feeling the urge to void [721].
It is unclear whether the age of start of toilet training or the prolonged use of diapers would have an influence on the incidence of nocturnal enuresis. One recent study was able to demonstrate that there is no influence - not even the age of becoming dry during the day had an influence. However, the same author performed a systematic review on the topic and found conflicting results, leaving as the hypothesis that the cultural influence may be important [722,723]. This hypothesis is in line with the findings of a large (5,433 children) study in Portugal, stating that developmental disorders and sociodemographic factors play a role in the timing to become dry at night [724].
A high incidence of comorbidity and correlation between nocturnal urine production and sleep disordered breathing, such as obstructive sleep apnoea, has been found and investigated [725]. Symptoms such as habitual snoring, apnoeas, excessive sweating at night and mouth breathing in the patient history or by means of sleep questionnaires, such as the BEARS questionnaire [726], can lead to the detection of sleep disorders and/or adenotonsillar hypertrophy. When present, a consultation with the ENT specialist can be considered [727]. A recent meta-analysis of studies looking into the effect of adenoidectomy and/or tonsillectomy in children with nocturnal enuresis and obstructive sleep apnoea has shown that the so-called improvement of incidence of bedwetting after the surgery is low, and probably lower than expected. This emphasises the fact that adenoidectomy and/or tonsillectomy should not be advised or performed to treat the bedwetting problem
[728,729].
Obesity and ADHD are associated with a higher incidence of NE and a lower efficacy for treatment [730,731]. The presence of allergic diseases has been recognised as a risk factor of NE and with a greater risk for more allergic episodes [732-734].
It is important to consider the child’s and family’s psychological status, because primary NE has been associated with psychopathology, such as attention deficit hyperactivity disorder (ADHD) and depressive symptoms [735-737]. In children with ADHD, symptoms of NE are more severe, and it is important to inform the child and the parents about a delayed success rate and higher relapse rate compared to children without ADHD [738].
The search for a central aetiology has inspired researchers to perform functional MRI investigations. Recent functional and structural MRI studies have identified alterations in brain connectivity in children with nocturnal enuresis compared to healthy controls. Zhang et al. reported differences in whole-brain functional connectivity density in 68 affected children versus 57 controls [739]. Zhong et al., found dysconnectivity between the salience network and default mode network in 33 children with enuresis compared to 33 controls, suggesting involvement of these networks in the pathophysiology [740]. In a related analysis, the same group observed abnormal resting-state functional connectivity of the hippocampus, proposing this region as a potential key node in brain-bladder control and cognitive processes linked to enuresis.
14.2. Diagnostic evaluation
The diagnosis is mainly obtained by history-taking. Focused questions to differentiate monosymptomatic versus non-monosymptomatic, primary versus secondary, comorbid factors, such as behavioural or psychological problems and sleep disorder breathing, should be asked. In addition, a two-day complete micturition and drinking diary, which records daytime bladder function and drinking habits, will further exclude comorbid factors such as LUTS and polydipsia [741].
Specific attention should be paid regarding bowel movements as irregular bowel movements can change the diagnosis from monosymptomatic NE to non-monosymptomatic NE. If constipation or faecal incontinence is found, which is reported in up to 20% of children with NE (37% occult constipation [742]), it should be treated simultaneously, and the family should be informed that constipation can negatively influence treatment outcomes [743-745].
The night-time urine production should be registered by weighing the nighttime diapers in the morning and adding the first morning voided volume [746]. The nighttime urine production should be recorded over a one-week period to diagnose an eventual differentiation between a high nighttime production (more than 130% of the age expected bladder capacity) versus a night-time OAB [741].
A physical examination should be performed, with special attention given to the child’s back (to exclude any neurological problem), the external genitalia and surrounding skin, as well as to the condition of the clothes (wet underwear or encopresis).
Urine analysis is indicated if there is a sudden onset of bedwetting, a suspicion or history of UTIs, or inexplicable polydipsia.
A uroflowmetry and US is indicated only if there is a history of previous urethral or bladder surgery and presence of daytime urinary symptoms. For further evaluation, see Chapter 13 on daytime LUT conditions.
At present, there is no clinical indication nor use for a functional MRI (fMRI) in the diagnosis of NE.
14.3. Management
As nocturnal enuresis is not an isolated phenomenon, management approaches should include a multifactorial approach. A phenotyping model suggested by Pereira et al. categorises NE into clinically significant phenotypic clusters. These clusters can be taken into account when developing a treatment plan [747]. Figure 8 shows the components and factors affecting nocturnal enuresis.
Before introducing any form of possible treatment, it is of utmost importance to explain the bedwetting condition to the child and the caregivers to demystify the problem. Parents should be encouraged to seek medical attention for their bedwetting children and be informed that it is known that the QoL of parents with a child with NE is negatively impaired. Medical providers assisting families with a child must be aware of this fact and guide parents by explaining that, key to treating a child with NE, is that the child is able to understand what is going on and to cooperate in the process [748].
Since the COVID-19 pandemic and the use of virtual contacts between doctors and patients, it has been shown that telemedicine is a good method of conducting close follow-up [749].
Figure 8: Components and factors contributing to nocturnal enuresis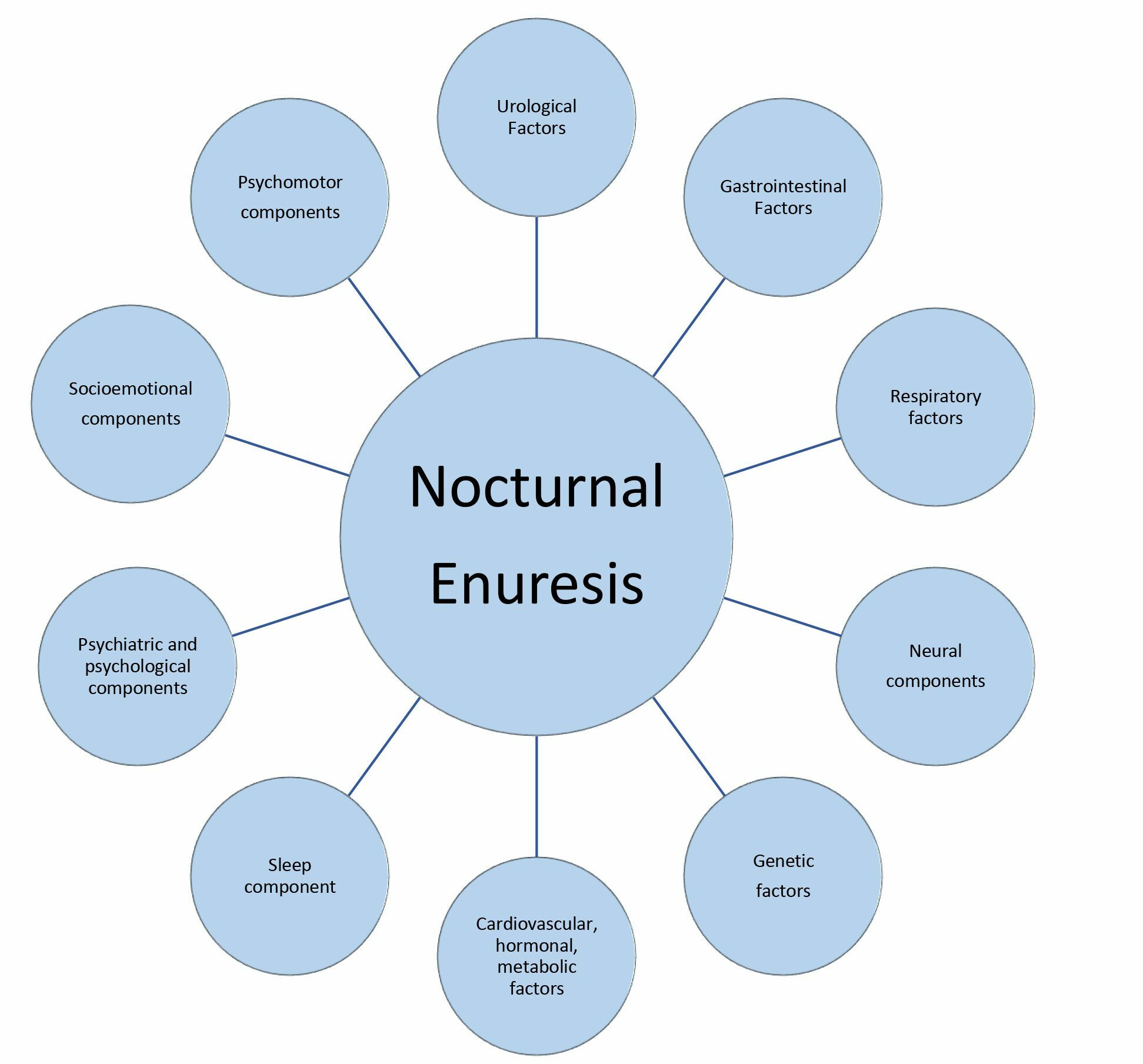 Adapted from Pereira et al., 2024 [747].
Adapted from Pereira et al., 2024 [747].
14.3.1. Supportive treatment measures
Initially, supportive measures including regular eating and drinking habits should be reviewed, stressing normal fluid intake during the day and reducing fluid intake in the hours before sleep. A reduction in caffeine intake should be encouraged [750].
Keeping a chart depicting wet and dry nights - also called basic bladder advice - has not been shown to be successful in the early treatment of NE [751]. To assure good sleep quality, specifically in children with NE, limiting the use of electronic devices before bedtime is also recommended [752].
Referral for psychological support should be advised and followed up for patients with NE and their families, particularly if the NE comorbid factor is developmental, ADHD or learning difficulties, family problems, and parental distress and possible punishment of the child are observed. Parental stress levels are higher compared to parents of non-NE children [753] and anger is found to be the most common parental reaction towards NE children [754]. This would explain why childhood traumas such as neglect and abuse are observed more often in children with NE [755]. Psychological interventions with parents of NE children were shown to significantly improve their coping mechanisms [756].
14.3.2. Wetting alarm treatment
The nocturnal alarm treatment relies on the use of a device that is activated by getting wet. The goal of this therapeutic approach is that the child wakes up from the alarm - which can be acoustic or tactile - either by itself or with the help of a caregiver. By repeatedly awakening the child when wet, the method of action aims to change a high arousal to a low arousal threshold, specifically when a status of full bladder is reached. In the most recent Cochrane review (even though the quality of the included studies was low), several studies have shown that alarm treatment reduces the number of wet nights per week. An alarm treatment has a higher complete response rate and a low relapse rate compared to no treatment at all [757]. In the event of relapse after initial success, one should actively investigate for OAB [758]. The recommended length of therapy with the alarm treatment continues to be uncertain, varying from 8-12 weeks (ICCS) to 16-20 weeks [759].
Regular follow-up will improve the success. It is of utmost importance that the child play an active role in the alarm treatment and is willing to understand the purpose of the treatment modality and persevere with it.
14.3.3. Medical treatment
If the child and the family would like to act on the high nighttime urine production and eventual nighttime OAB, the child should be able and willing to adjust their drinking habits and take either desmopressin or a combination of desmopressin and an anticholinergic drug.
Success rates of 70% can be obtained with desmopressin, either in tablet form (200-400μg) or as sublingual desmopressin oral lyophilisate (120-240μg). Nasal spray desmopressin is no longer recommended, due to the increased risk of overdose [760]. A rare side-effect is water intoxication, which can be prevented by adequate water intake. The dosage of 120ug has been shown to be effective and safe [761]. A structured titration increase of up to 240ug has been shown to be effective [762]. Predictive factors for success with desmopressin have been identified: older children, children with fewer wet nights, and children with high nighttime urine production [763]. Children that show a good response on low-dose desmopressin are more likely to show a complete response during the maintenance period [764]. When poor responses are seen on desmopressin, be aware of low compliance [765]. Relapse rates can be high after desmopressin discontinuation [655]. It is unclear whether structured withdrawal will result in lower relapse rates [766,767]. In the event of desmopressin-resistant treatment for NE, or if a suspicion exists for night-time OAB, combination of desmopressin with anticholinergics is safe and efficient, even after cessation of treatment [768-771]. Recent evidence suggests increased success rates when combination therapy of two agents (desmopressin and an anticholinergic) is offered at the very beginning of the therapeutic journey [772,773].
With nighttime OAB, a treatment failure to desmopressin can be explained because of bladder reservoir dysfunction [774]. There is no indication for monotherapy with an anticholinergic drug [775].
Newer studies suggest the use of fluoxetine as an emerging alternative [776,777].
Alarm and desmopressin treatment have comparable efficacy in achieving > 50% reduction in wet nights. Alarms offer superior treatment response (OR: 2.89, 95% CI: 1.38-6.04) and lower relapse rates (OR: 0.25, 95% CI: 0.12-0.50) in children [778]. Multimodal treatment can achieve a partial or full response in 80% of children [779].
14.3.4. Electrical neuromodulation
Several systematic reviews and randomised trials have documented potential benefits of electrical neural stimulation for NE. However, the quality of the studies included was low and different types of electrical neural stimulation, such as intra-anal stimulation and interferential current stimulation, have been included [780-783]. An RCT shows equivalent PTNS versus desmopressin effects on PMNE [784].
14.3.5. Complementary treatments:
A Cochrane review showed no benefit for treatments such as hypnosis, psychotherapy, acupuncture, chiropractic and medicinal herbs for the treatment of NE [785]. Newer studies, however, contribute potential merit to the acupuncture practice [786,787].
14.3.6. Conservative ‘wait-and-see’ approach
If the child and their family are unable to comply with a treatment, if the treatment options are not possible for the family situation, and if there is no social pressure, a ‘wait-and-see’ approach can be chosen [788]. However, in this approach, it is important to emphasise the fact that the child should wear diapers at night to ensure a normal quality of sleep [789]. The success rate of wait and see is 15% per year, independent of age. Figure 9 presents stepwise assessment and management options for NE.
Figure 9: A stepwise assessment and management options for NE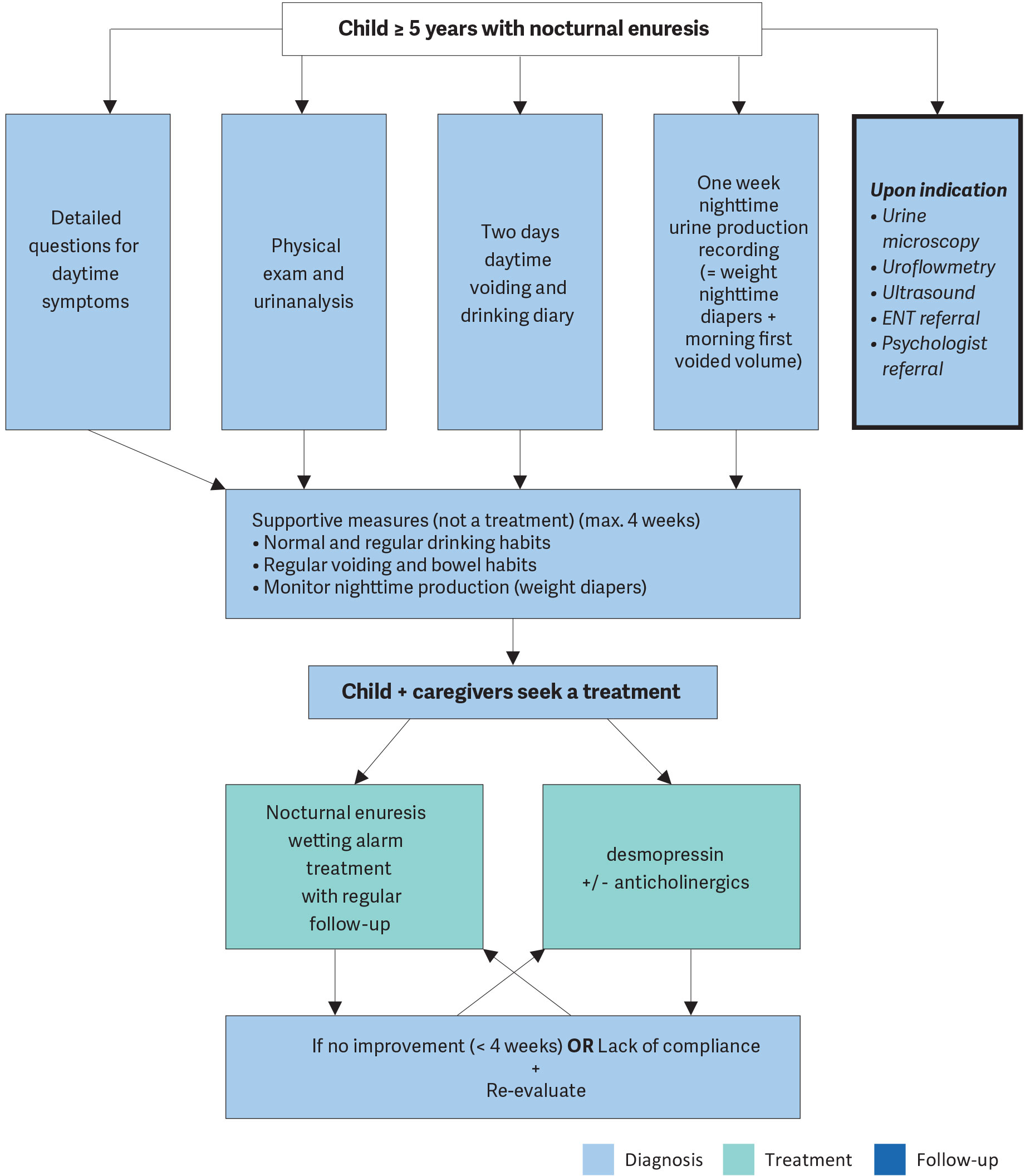 ENT = ear, nose and throat.
ENT = ear, nose and throat.
14.4. Summary of evidence and recommendations for the management of monosymptomatic enuresis
| Summary of evidence | LE |
| Chronobiology of micturition, in which the existence of a circadian clock has been proven in kidney, brain and bladder, and disturbances in this chronobiology play a major role in the pathophysiology of enuresis. | 1 |
| Aetiology of NE is multifactorial. | 1 |
| Recommendations | Strength rating |
| Do not treat children less than five years of age in whom spontaneous cure is likely, but rather inform the family about the involuntary nature, the high incidence of spontaneous resolution and the fact that punishment will not help to improve the condition. | Strong |
| Use micturition diaries or questionnaires to exclude daytime symptoms. | Strong |
| Perform a urine test to exclude the presence of infection or potential causes such as diabetes insipidus. | Strong |
| Offer supportive measures in conjunction with other treatment modalities, of which pharmacological and alarm treatment are the two most important. | Strong |
| Offer desmopressin in proven nighttime polyuria. | Strong |
| Offer alarm treatment in motivated and compliant families. | Strong |